Abstract
Objective:
Glyceraldehyde-derived advanced glycation end products contribute to vascular inflammation in diabetes. However, what advanced glycation end product structure could evoke inflammatory reactions remains unknown. We examined whether and how methylglyoxal-derived hydroimidazolone 1, one of the advanced glycation end products formed from glyceraldehyde, elicits inflammatory reactions in human umbilical vein endothelial cells.
Materials and methods:
Glyceraldehyde-advanced glycation end products-aptamer was prepared using a systemic evolution of ligands by exponential enrichment. The binding affinities of methylglyoxal-derived hydroimidazolone 1 to receptor for advanced glycation end products or advanced glycation end product-aptamer were measured with a quartz crystal microbalance. Intracellular reactive oxygen species generation and THP-1 cell adhesion were evaluated using fluorescent probes. Gene expression was analysed by reverse transcription polymerase chain reaction.
Results:
Methylglyoxal-derived hydroimidazolone 1 bound to receptor for advanced glycation end products and advanced glycation end product-aptamer with a dissociation constant (Kd) of 56.7 µM and 1.51 mM, respectively. Methylglyoxal-derived hydroimidazolone 1 at 100 µg/mL significantly increased reactive oxygen species generation in human umbilical vein endothelial cells, which were attenuated by anti-receptor for advanced glycation end products antibody or advanced glycation end product-aptamer. In all, 100 µg/mL methylglyoxal-derived hydroimidazolone 1 significantly increased receptor for advanced glycation end products and intercellular adhesion molecule-1 messenger RNA levels in, and THP-1 cell adhesion to, human umbilical vein endothelial cells, all of which were blocked by anti-receptor for advanced glycation end products antibody.
Conclusion:
Our present results indicate that methylglyoxal-derived hydroimidazolone 1 evokes inflammatory reactions in human umbilical vein endothelial cells via receptor for advanced glycation end products, although apparently limited to supraphysiological levels of methylglyoxal-derived hydroimidazolone 1. Methylglyoxal-derived hydroimidazolone 1 is a distinct advanced glycation end product structure that could mediate harmful effects of methylglyoxal and glyceraldehyde-mediated glycation processes.
Keywords
Introduction
The prevalence of atherosclerotic cardiovascular disease (CVD) is extremely high in diabetic patients, and approximately one half of diabetic patients die from cardiovascular events in developed countries. 1 Two large clinical studies have shown that early blood glucose control can produce sustained risk reduction of CVD and death in both type 1 and type 2 diabetic patients. 2 These observations have suggested the involvement of phenomenon called ‘the metabolic memory’ in the pathogenesis of CVD in diabetes.2,3 Therefore, it is conceivable that cumulative diabetic exposure could continue to cause endothelial cell damage, an initial step for atherosclerosis even after achieving good glycemic control.2,3 Although various biochemical pathways are activated under diabetic conditions, advanced glycation end products (AGEs) are considered to play a central role in the phenomenon ‘the metabolic memory’ because (1) their formation and accumulation could reflect cumulative diabetic exposure and (2) AGEs once formed are not easily degraded and remain in the vessels for a long time. 2 Furthermore, an increasing body of evidence has suggested the pathological role of AGEs in CVD in diabetes.3–5 Indeed, we have previously shown that glyceraldehyde-derived AGEs (glyceraldehyde-AGEs) evoke oxidative stress generation and inflammatory reactions in endothelial cells and vessels of diabetic animals through an interaction with cell surface receptor RAGE (receptor for advanced glycation end products), and circulating levels of glyceraldehyde-AGEs are associated with endothelial cell damage and vascular inflammation in patients with both type 1 and type 2 diabetes.3,6–8 However, what structurally distinct AGEs could elicit oxidative stress generation and inflammatory reactions remain unknown. In this study, we examined whether and how methylglyoxal-derived hydroimidazolone 1 (MG-H1), one of the AGEs formed from glyceraldehyde in the process of non-enzymatic glycation, 9 evokes oxidative stress generation and inflammatory reactions in human umbilical vein endothelial cells (HUVECs).
Materials and methods
MG-H1 was purchased from PolyPeptide Laboratories (Strasbourg, France). Preparation and section of DNA-aptamer raised against glyceraldehyde-AGEs (AGE-aptamer) were performed using systemic evolution of ligands by exponential enrichment as described previously; sequence of AGE-aptamer was 5′-TgTAgcccgAgTATcATTcTccATcgcccccAgATAcAAG-3′, where phosphorothioate nucleotides are indicated as capital letters. 3 Neutralizing rabbit polyclonal antibody directed against RAGE (RAGE-Ab), which recognizes the amino acid residues 167–180 of human RAGE, was prepared as described previously. 3 The binding affinities of MG-H1 to extracellular domain of RAGE (vRAGE) or AGE-aptamer were measured using a sensitive 27-MHz quartz crystal microbalance (QCM) (Affinix Q; Initium Inc., Tokyo, Japan). 3 In brief, recombinant vRAGE or AGE-aptamer was immobilized on a QCM surface. After adding MG-H1 to a reaction vessel, the time courses of the frequency decreases of bound MG-H1 on the QCM surface were monitored.
HUVECs were maintained in endothelial basal medium supplemented with 2% foetal bovine serum, 0.4% bovine brain extracts, 10 ng/mL human epidermal growth factor and 1 µg/mL hydrocortisone according to the supplier’s recommendation (Lonza Group Ltd., Basel, Switzerland). Then, HUVECs were cultured in a medium lacking epidermal growth factor and hydrocortisone (assay medium) and treated with the indicated concentrations of MG-H1 in the presence or absence of 5 µg/mL RAGE-Ab for 4 h [reactive oxygen species (ROS) generation assay and real-time reverse transcription polymerase chain reaction (RT-PCR) analysis] or 24 h (cell adhesion assay). In the cell experiments of AGE-aptamer, assay medium was pre-incubated with or without 100 µg/mL MG-H1 or 1 µM AGE-aptamer for 2 h, and then HUVECs were treated with the medium for 4 h. ROS generation was evaluated by CellRox oxidative stress reagents (Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer’s recommendation. Gene expression of RAGE and intercellular adhesion molecule-1 (ICAM-1) was evaluated by RT-PCR analyses; IDs of primers for RAGE, ICAM-1 and 18S gene were Hs00542592_g1, Hs00164932_m1 and Hs03003631_g1, respectively. 10 THP-1 cell adhesion to HUVECs were assayed with a fluorescent probe as reported previously. 10
All values were presented as mean ± standard error. One-way analysis of variance (ANOVA) followed by Games-Howell for Figure 1(c) or Student’s t-test for Figure 1(d) to (g) was performed for statistical comparisons; p < 0.05 was considered significant.
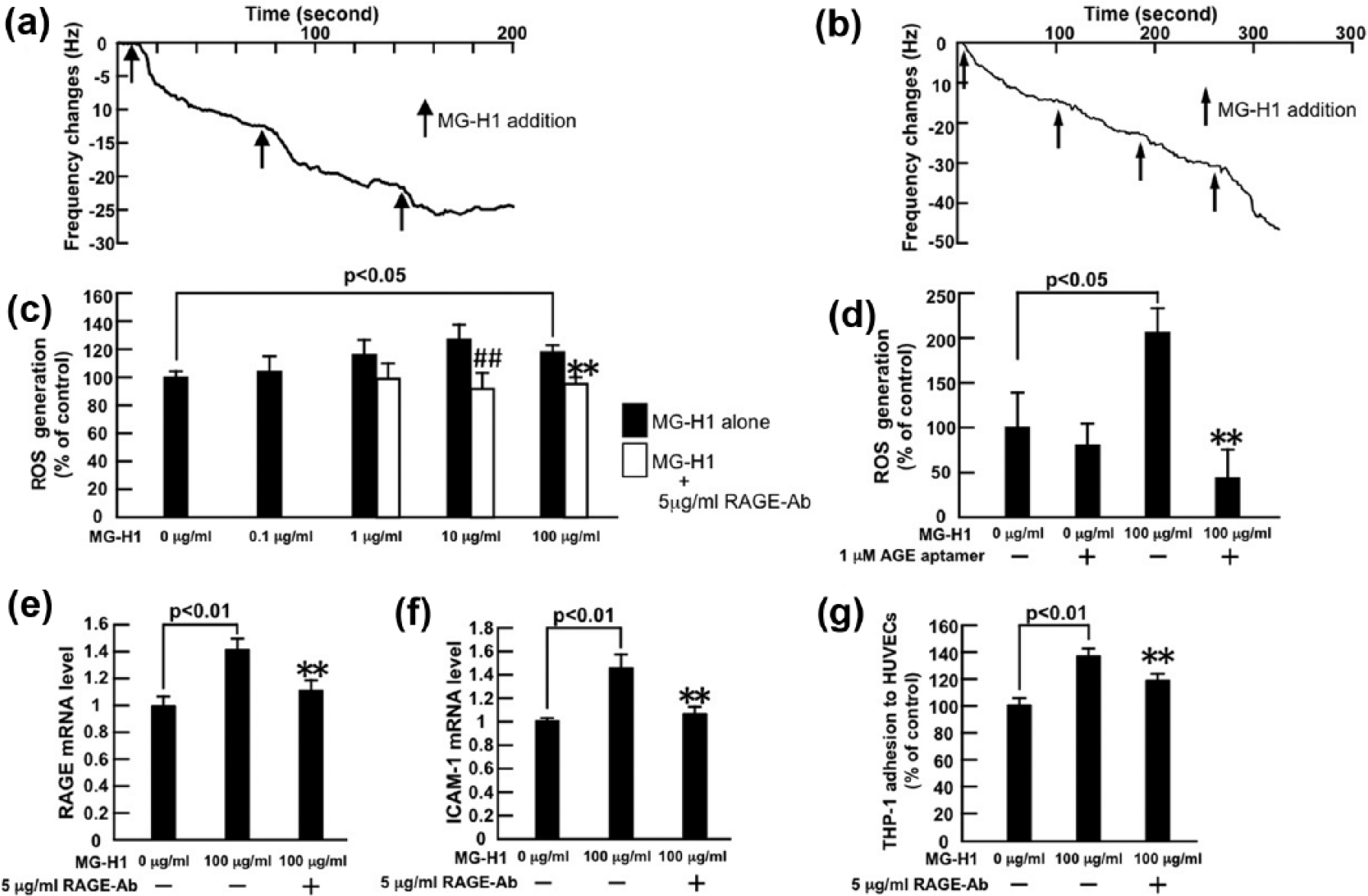
(a, b) Binding affinities of MG-H1 to vRAGE or AGE-aptamer were measured using a sensitive 27-MHz QCM. N = 3 per group. (c, d) Effects of MG-H1 on ROS generation, (e, f) RAGE and ICAM-1 mRNA levels in, and (g) THP-1 cell adhesion to, HUVECs. HUVECs were treated with the indicated concentrations of MG-H1 in the presence or absence of 5 µg/mL RAGE-Ab or 1 µM AGE-aptamer for (c–f) 4 h or (g) 24 h. (c, d) Then, ROS generation was evaluated by fluorescent intensity. (c) N = 6 per group. (d) N = 8 per group. (e, f) Total RNAs were transcribed and amplified by real-time PCR. Data were normalized by the intensity of 18S mRNA-derived signals and then related to the control values without MG-H1. N = 8 per group. (g) THP-1 cell adhesion to HUVECs was measured. N = 6 per group. ## and **, p < 0.01 compared to the values with 10 µg/mL and 100 µg/mL MG-H1 alone, respectively.
Results
In a QCM system, the resonance frequency decreases in proportional to binding amount of molecules on oscillating quartz crystal. 3 As shown in Figure 1(a) and (b), MG-H1 bound to vRAGE and AGE-aptamer with a dissociation constant (Kd) of 5.67 ± 0.82 × 10−5 M and 1.51 ± 1.13 × 10−3 M, respectively.
As shown in Figure 1(c), MG-H1 increased ROS generation in HUVECs in a saturation-type manner; 5 µg/mL RAGE-Ab significantly inhibited the increase in ROS generation in 10 or 100 µg/mL MG-H1-exposed HUVECs. Although 1 µM AGE-aptamer alone did not affect ROS generation in HUVECs, it significantly reduced ROS generation by HUVECs exposed to 100 µg/mL MG-H1 (Figure 1(d)).
MG-H1 at 100 µg/mL significantly up-regulated RAGE and ICAN-1 gene expression in, and stimulated THP-1 cell adhesion to, HUVECs, all of which were attenuated by 5 µg/mL RAGE-Ab (Figure 1(e) to (g)). We have confirmed that 5 µg/mL normal rabbit IgG does not affect inflammatory gene expression in, or THP-1 cell adhesion to, HUVECs. 10
Conclusion
In this study, we found for the first time that neutralizing RAGE-Ab significantly attenuated the MG-H1-evoked ROS generation and inflammatory gene expression in, and THP-1 macrophage cell adhesion to, HUVECs. We also found here that MG-H1 actually bound to vRAGE immobilized on a QCM surface with a Kd value of 5.67 × 10−5 M, and RAGE-Ab inhibited the up-regulation of RAGE messenger RNA (mRNA) levels in MG-H1-exposed HUVECs. We have previously shown that glyceraldehyde-AGEs stimulate RAGE and ICAM-1 gene expression in, and THP-1 cell adhesion to, HUVECs, all of which were suppressed by the treatment with RAGE-Ab or anti-oxidants.2,10 Therefore, our present observations suggest that MG-H1 may evoke inflammatory reactions in HUVECs through ROS production via the interaction with RAGE, whose gene expression is amplified by RAGE ligand MG-H1, thus forming a positive feedback loop between MG-H1-induced superoxide generation and RAGE gene expression. However, a control with overexpression of superoxide dismutase is required to justify this speculation. Furthermore, it should be noted that plasma-free MG-H1 concentration in normal healthy controls and patients with stage 5 chronic kidney disease is 0.12 and 4.8 µM, respectively. 11 So, MG-H1 concentration used here [10 and 100 µg/mL MG-H1 (44 and 440 µM)] are not physiologically levels, and it may only evoke oxidative stress and inflammatory reactions in HUVECs at supraphysiological concentrations. However, Xue et al. 12 reported that binding affinity of MG-H1 to RAGE was 1000-fold stronger than ours with a fluorescence titration method, which can evaluate the Kd for MG-H1 binding to RAGE in a reaction solution with high molecular flexibility.
In this study, we also found that although binding affinity of AGE-aptamer to MG-H1 was very weak, it inhibited the ROS generation by MG-H1-exposed HUVECs. We have recently found that the same AGE-aptamer not only inhibits glyceraldehyde-AGE-induced inflammatory reactions in HUVECs but also attenuates vascular injury in diabetic animals by suppressing the binding of glyceraldehyde-AGEs to RAGE.13,14 These findings suggest that MG-H1 is a distinct glyceraldehyde-related AGE structure that could mediate oxidative stress and inflammatory reactions in HUVECs. MG-H1 is mainly derived from methylglyoxal in vivo.15,16 However, given that MG-H1 is also formed in the process of non-enzymatic glycation of N-acetylarginine by glyceraldehyde in vitro 9 and that circulating glyceraldehyde-AGEs is one of the biomarkers associated with endothelial cell damage and vascular inflammation in both type 1 and type 2 diabetic patients,3,6–8 MG-H1 may be a target AGE structure for inhibiting vascular damage in patients with diabetes. Further clinical study is needed to clarify the relevance of blockade of the MG-H1-RAGE axis as a novel therapeutic approach to vascular complications in diabetes.
Footnotes
Acknowledgements
S.Y. conceptualized and designed the study; acquired, analysed and interpreted data; and drafted the manuscript; he takes responsibility for the integrity of the data and the accuracy of the data analysis. Y.I., T.M., N.N., A.S., and Y.H. acquired, analysed, and interpreted data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported, in part, by Grants-in-Aid for Scientific Research B (grant number 22390111) (S.Y.) from the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
