Abstract
Aim:
To evaluate an additional rapid-acting insulin bolus on postprandial lipaemia, inflammation and pro-coagulation following high-carbohydrate high-fat feeding in people with type 1 diabetes.
Methods:
A total of 10 males with type 1 diabetes [HbA1c 52.5 ± 5.9 mmol/mol (7.0% ± 0.5%)] underwent three conditions: (1) a low-fat (LF) meal with normal bolus insulin, (2), a high-fat (HF) meal with normal bolus insulin and (3) a high-fat meal with normal bolus insulin with an additional 30% insulin bolus administered 3-h post-meal (HFA). Meals had identical carbohydrate and protein content and bolus insulin dose determined by carbohydrate-counting. Blood was sampled periodically for 6-h post-meal and analysed for triglyceride, non-esterified-fatty acids, apolipoprotein B48, glucagon, tumour necrosis factor alpha, fibrinogen, human tissue factor activity and plasminogen activator inhibitor-1. Continuous glucose monitoring captured interstitial glucose responses.
Results:
Triglyceride concentrations following LF remained similar to baseline, whereas triglyceride levels following HF were significantly greater throughout the 6-h observation period. The additional insulin bolus (HFA) normalised triglyceride similarly to low fat 3–6 h following the meal. HF was associated with late postprandial elevations in tumour necrosis factor alpha, whereas LF and HFA was not. Fibrinogen, plasminogen activator inhibitor-1 and tissue factor pathway levels were similar between conditions.
Conclusion:
Additional bolus insulin 3 h following a high-carbohydrate high-fat meal prevents late rises in postprandial triglycerides and tumour necrosis factor alpha, thus improving cardiovascular risk profile.
Introduction
Structured education provided to patients with type 1 diabetes for managing mealtime insulin dose focuses on the carbohdyrate-counting method,1,2 whereby people calculate the dose of insulin administered at mealtimes based on the total carbohdyrate content of that meal. 3 While this has been demonstrated as an effective strategy for HbA1c reduction, 3 typical eating patterns consist of the consumption of mixed-macronutrient meals, 4 and in reality many people with type 1 diabetes still struggle to maintain postprandial euglycaemia. 5 This is particularly the case for individuals treated with modern insulin analogue injections, as this method of insulin delivery is associated with less mealtime insulin dose flexibility compared to continuous subcutaneous insulin infusion therapy (CSII). 6
The addition of fat to a carbohydrate-based meal has been shown to cause postprandial hyperglycaemia, and increase insulin requirements late into the postprandial period.7,8 In clinical practice, people with type 1 diabetes are often reluctant to administer an additional injection of bolus insulin either at mealtime or late into the postprandial period for fear of hypoglycaemia or because increasing injection frequency is considered to be burdensome.6,9 Recently, we have showed that when consuming a carbohydrate-based meal with a high-fat (HF) content, adopting the carbohydrate-counting method for insulin dose adjustments at mealtime followed by the administration of an additional insulin bolus late into the postprandial period is important for the normalisation of glycaemia. 7 Specifically, compared to the carbohydrate-counting method alone, administration of additional bolus insulin 3 h later resulted in a 23% reduction in blood glucose area under the curve (AUC). 7 Importantly, this method did not cause hypoglycaemia whereas simply increasing the amount of rapid-acting insulin dose administered at mealtime did. 7
Insulin has an important role not only in the control of postprandial glucose excursions but also in the regulation of postprandial lipaemia. 10 Excessive increases in both glycaemia and lipaemia can create a pro-inflammatory and pro-coagulant milieu,11–15 and are collectively and independently associated with cardiovascular disease (CVD) and early mortality.16,17 Considering the substantial pre-existing risk of CVD-associated early mortality in this cohort,18,19 and the potential for this to be heightened by exaggerated postprandial lipaemia,20–24 optimising mealtime insulin dosage is important for cardiovascular risk management, not just normalisation of glycaemia per se. However, the influence of administering additional insulin late into the postprandial period on metabolic or cardiovascular risk factors in patients with type 1 diabetes treated with basal-bolus injection therapy has never been assessed. In this study, we manipulated rapid-acting insulin injection dosage and timing in response to a high-carbohydrate HF meal feeding to test the hypothesis that an additional but delayed rapid-acting insulin bolus is required to normalise postprandial lipaemia and the associated metabolic, inflammatory and pro-coagulant response.
Methods
Patients
The study population consisted of 10 male type 1 diabetes patients [mean ± standard deviation (SD); age: 26 ± 4 years, body mass index (BMI): 25.4 ± 1.6 kg m2, duration of diabetes: 17 ± 5 years, age at diagnosis: 9 ± 4 years; HbA1c: 52.5 ± 5.9 mmol/mol (7.0% ± 0.5%)]. Patients were eligible for inclusion if they were aged between 18 and 50 years, with a duration of diabetes greater than 5 years on enrolment, treated on basal-bolus insulin regimen and were familiar with carbohydrate-counting and using a stable insulin-to-carbohydrate ratio. Patients were treated on a stable basal-bolus insulin analogue regimen consisting of either insulin glargine (n = 8) or detemir (n = 2) and fast-acting insulin analogue as part (n = 10), for a minimum of 12 months. Patients were free of diabetes-related complications and were receiving no additional medication other than insulin. All patients had received structured education in carbohydrate-counting as part of their diabetes care. This study received approval by the local National Health Service Research Ethics Committee (R&D Ref: 7241). All patients who participated provided written informed consent. Eligible patients underwent randomisation by computer programme to determine the sequence of three crossover conditions.
Pre-laboratory phase
Patients arrived at the laboratory after an overnight fast (>10 h) having replicated their diet in the previous 48 h (assessed using weighed dietary recording sheets). Participants were fitted with a real-time continuous glucose monitor (CGM; Paradigm Veo, Medtronic Diabetes, Northridge, CA) as described previously7,11,25–27 to aid in the maintenance of normal glycaemia during the pre-laboratory period. Additionally, patients were instructed to maintain their normal insulin regimen, with basal insulin dose standardised (dose, injection site, time of injection) across conditions. Patients were also given a pedometer (Omron Healthcare Europe B.V., The Netherlands), which they were instructed to wear over the course of 24 h prior to experimental visits. Patients were required to avoid strenuous activity in the previous 48 h and maintain similar activity patterns between visits.
Main experimental visits
In a randomised and counter-balanced fashion, participants attended three separate morning-time (~07:00 AM) laboratory-based visits, each interspersed by 7 days. Upon arrival to the laboratory, patients assumed a seated and rested position while a 20-gauge cannula (Vasofix, B. Braun, Melsungen AG, Melsungen, Germany) was inserted into the antecubital vein of their non-dominant arm; resting, fasted, venous blood samples were then collected prior to experimental testing.
Each experimental visit involved the consumption of meals matched for carbohydrate and protein content, but differing in (1) fat content and (2) rapid-acting insulin bolus dose and timing. The low-fat (LF) condition involved administration of rapid-acting insulin according to individual patient carbohydrate-counting requirements (dose per 10 g: 1.1 ± 0.8 IU) prior to the consumption of a low-fat meal (Table 1). The HF condition involved administration of rapid-acting insulin according to the individual patient carbohydrate-counting requirements (as administered in LF); however, the meal contained an additional 50 g of fat to constitute a HF meal (Table 1). The high-fat split (HFA) condition involved the administration of rapid-acting insulin according to individual carbohydrate-counting requirements (as administered in LF and HF) prior to the consumption of a HF meal, and an additional 30% of rapid-acting insulin administered at 180 min post-ingestion (Table 1). The aim of this was such that the additional units of insulin would enter the circulation to coincide with the occurrence of peak postprandial lipaemia. 28
Experimental meal composition and accompanying insulin administration.
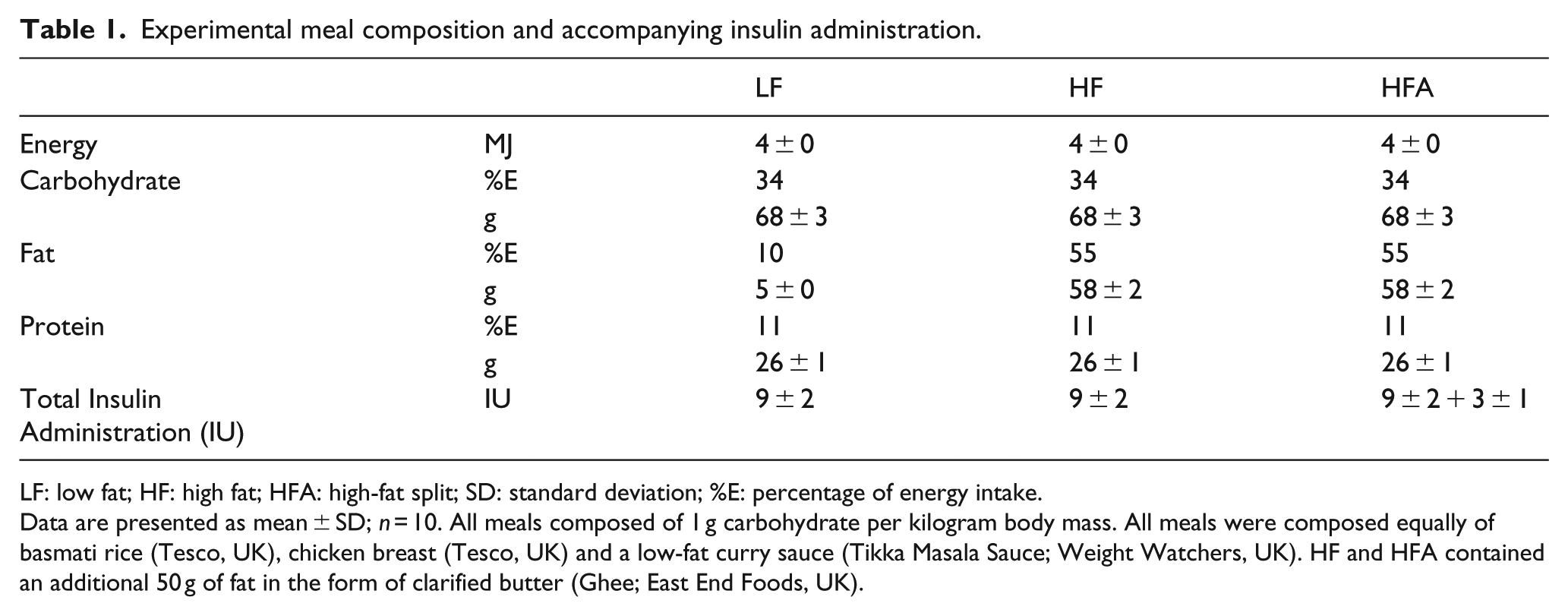
LF: low fat; HF: high fat; HFA: high-fat split; SD: standard deviation; %E: percentage of energy intake.
Data are presented as mean ± SD; n = 10. All meals composed of 1 g carbohydrate per kilogram body mass. All meals were composed equally of basmati rice (Tesco, UK), chicken breast (Tesco, UK) and a low-fat curry sauce (Tikka Masala Sauce; Weight Watchers, UK). HF and HFA contained an additional 50 g of fat in the form of clarified butter (Ghee; East End Foods, UK).
To minimise the influence of injection location on insulin absorption kinetics, the site of bolus injection was standardised across visits using prominent anatomical landmarks (equidistant from the most medial portion of the iliac crest and navel).
Following meal consumption, patients remained in a seated and rested position with blood samples drawn every 30 min for 6 h following meal consumption. Following this, patients were discharged and returned home. Plasma fibrinogen, human tissue factor (HTF) and plasminogen activator inhibitor-1 (PAI-1) were sampled at baseline, 3 h post-meal and at 6 h post-meal.
Meal composition
The macronutrient contribution to each meal is presented in Table 1; meal carbohydrate and protein content were matched across conditions. The LF and HF meals were based on the composition of a curried dish consisting of basmati rice (Basmati Rice Basics, Tesco, UK), tikka masala sauce (Mild Spice Tikka Masala; Weight Watchers, UK) and chicken (Everyday Sliced Chicken; Tesco, UK). The amount of each food item was identical in each condition and calculated such that carbohydrate content was individualised, equalling 1 g of carbohydrate per kilogram of body mass. In addition to the above, the HF meals included the addition of an absolute amount of 50 g of clarified butter (Butter Ghee; East End Foods, UK) which constitutes 99.9% fat.
Blood sampling
At each time point, 10 mL of venous whole blood was taken and dispensed into serum separation and lithium-heparin (Vacuette; Greiner Bio-One GmbH, Kremsmünster, Austria) tubes before being centrifuged for 15 min at 2000g at 4°C and stored at −80°C for retrospective analysis of triglycerides (TGs) (Serum Triglyceride Determination Kit; Sigma–Aldrich, St. Louis, MO, USA). Apolipoprotein B48 (APOB48; Antibodies-online, USA), non-esterified-fatty acids (NEFA) (RANBUT; Randox Laboratories, London, UK), plasma glucagon (Glucagon EIA; Sigma–Aldrich) and tumour necrosis factor alpha (TNF-α) (Human TNF-α Quantikine ELISA; R&D Systems, Roche Diagnostics, West Sussex, UK) were measured hourly. Plasma fibrinogen (ab108842, Fibrinogen Human ELISA Kit; Abcam, Japan), HTF (Human Tissue Factor activity ab108906; Abcam, UK) and PAI-1 (Human PAI-1/serpin ELISA Kit DSE100; R&D systems, UK) were measured at rest, 3 h and 6 h post-meal. The intra-assay coefficient of variation was <10% for all assays. Due to increased assay cross-reactivity with insulin detemir, only participants treated with insulin glargine were included in serum insulin analysis (n = 8).
Data analysis
Sample size analysis was performed using data from Cohen and Berger, 29 whereby increasing insulinaemia via the co-ingestion of glucose lowered postprandial (3 h) TG concentrations from ~1.36 ± 0.24 mmol L−1 to 0.85 ± 0.24 mmol L−1. Based on these data, 10 participants should provide >90% chance of statistically detecting a similar effect size with an α-level of 0.05. Statistical analysis was performed using PASW Statistics 18 software (IBM, Armonk, NY) with significance set at p ≤ 0.05. Data were examined using repeated measures analysis of variance (ANOVA; condition × time). Where significant p-values were identified for interaction effects (condition × time), Bonferroni’s corrected post hoc analysis was performed. Significant main effects of time were investigated using pairwise comparisons. Where relevant, one-way ANOVA with Bonferroni’s adjusted pairwise comparisons was used to compare between conditional differences. Data are presented as mean ± SD unless stated otherwise.
Results
Pre-laboratory phase
Patients displayed similar glycaemic control during the 24 h before arriving to the laboratory, with similar mean (LF = 7.5 ± 1.6, HF = 7.0 ± 1.1, HFA = 8.2 ± 1.5 mmol L−1; p = 0.519) and total AUC (LF = 11,123 ± 2224, HF = 10,080 ± 1543, HFA = 11,762 ± 2159 mmol L−1 min−1; p = 0.328) interstitial glucose across visits. Throughout this time, patients demonstrated similar dietary patterns, rapid-acting insulin administrations and activity levels across conditions (p > 0.05; Supplementary Material 1).
Laboratory phase
TG concentrations following LF remained similar to baseline (Figure 1(a); p > 0.05), whereas TGs under HF were significantly greater throughout the 360-min observation period (Figure 1(a)–(c); p < 0.05). HFA elicited an increase in TGs similar to HF concentrations during the first 180 min, but beyond 180 min, concentrations returned to baseline and were comparable to LF (Figure 1(a)–(c); p > 0.05). HF was also associated with elevated TNF-α late into the postprandial period, whereas LF and HFA were not (Figure 2(a) and (b); p < 0.05). The CGM interstitial glucose responses are presented in Figure 2(c). There was a significant time × condition interaction (p = 0.02, partial-η2 = 0.199), and a significant time (p < 0.01, partial-η2= 0.753) and condition (p = 0.29, partial-η2= 0.324) effect in CGM interstitial glucose responses to the conditions (Figure 2(c)), whereby HF resulted in higher interstitial glucose concentrations in the late postprandial period compared to both LF and HFA (Figure 2(c); p < 0.05). Interstitial glucose was comparable between conditions during the first 180 min with similar total AUC0−180 (LF = 4104 ± 831, HF = 5401 ± 545, HFA = 4959 ± 525 mmol L−1 min−1; p = 0.418) and absolute interstitial glucose levels at 180 min (LF = 6.0 ± 1.3, HF = 7.0 ± 0.7, HFA = 6.3 ± 0.7 mmol L−1; p > 0.05; Figure 2(c)). Beyond 180 min, interstitial glucose levels were greater under HFA (AUC180−360: LF = 8518 ± 1876, HF = 14,591 ± 1957 vs HFA = 11,011 ± 1509 mmol L−1 min−1;p < 0.05). The APOB48, NEFA, glucagon, fibrinogen, HTF activity and PAI-1 responses are presented in Table 2.
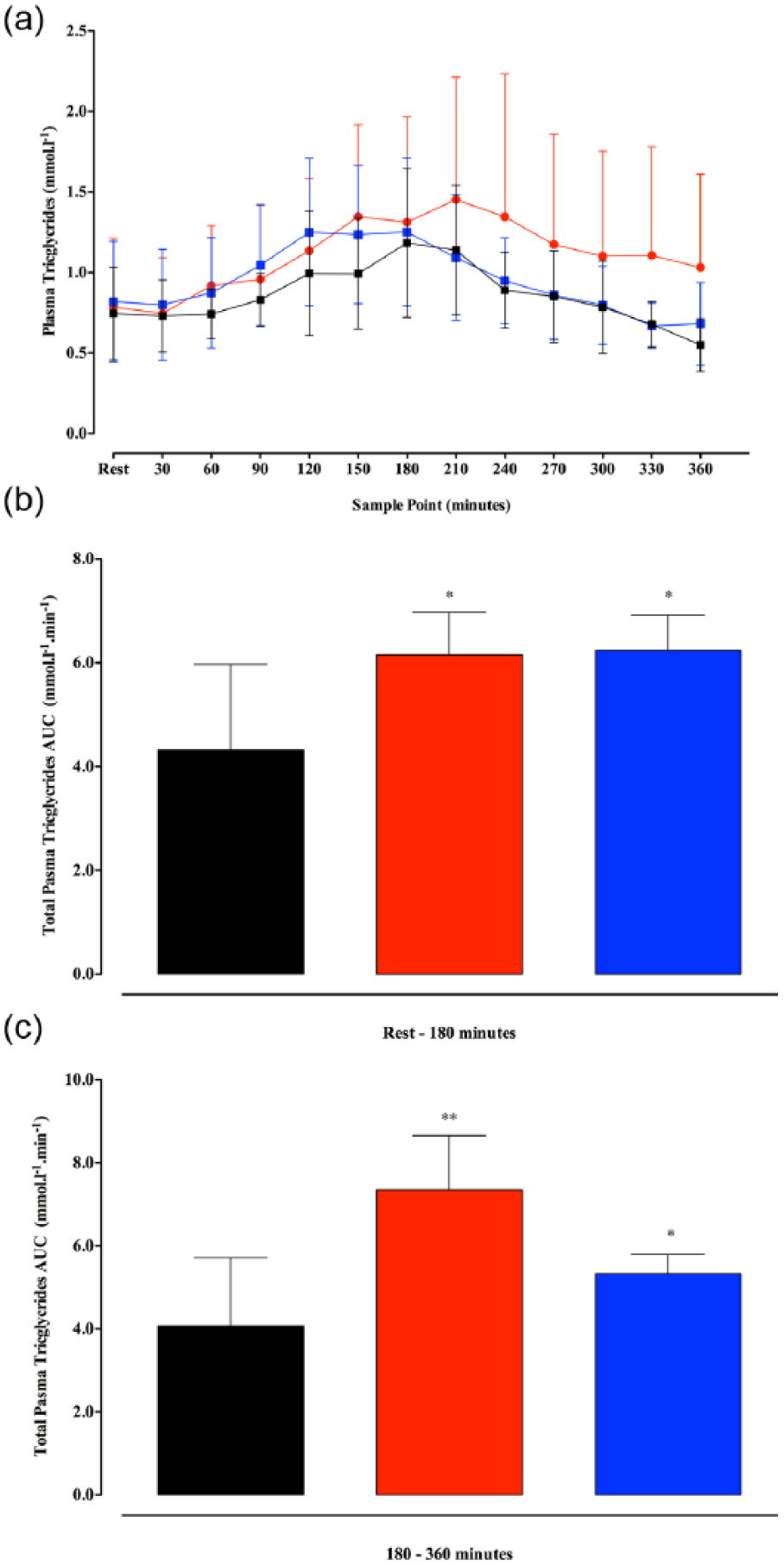
(a)–(c) A time course changes in plasma triglycerides; (b) total plasma triglyceride AUC0−180 and (c) total plasma triglyceride AUC180−360.
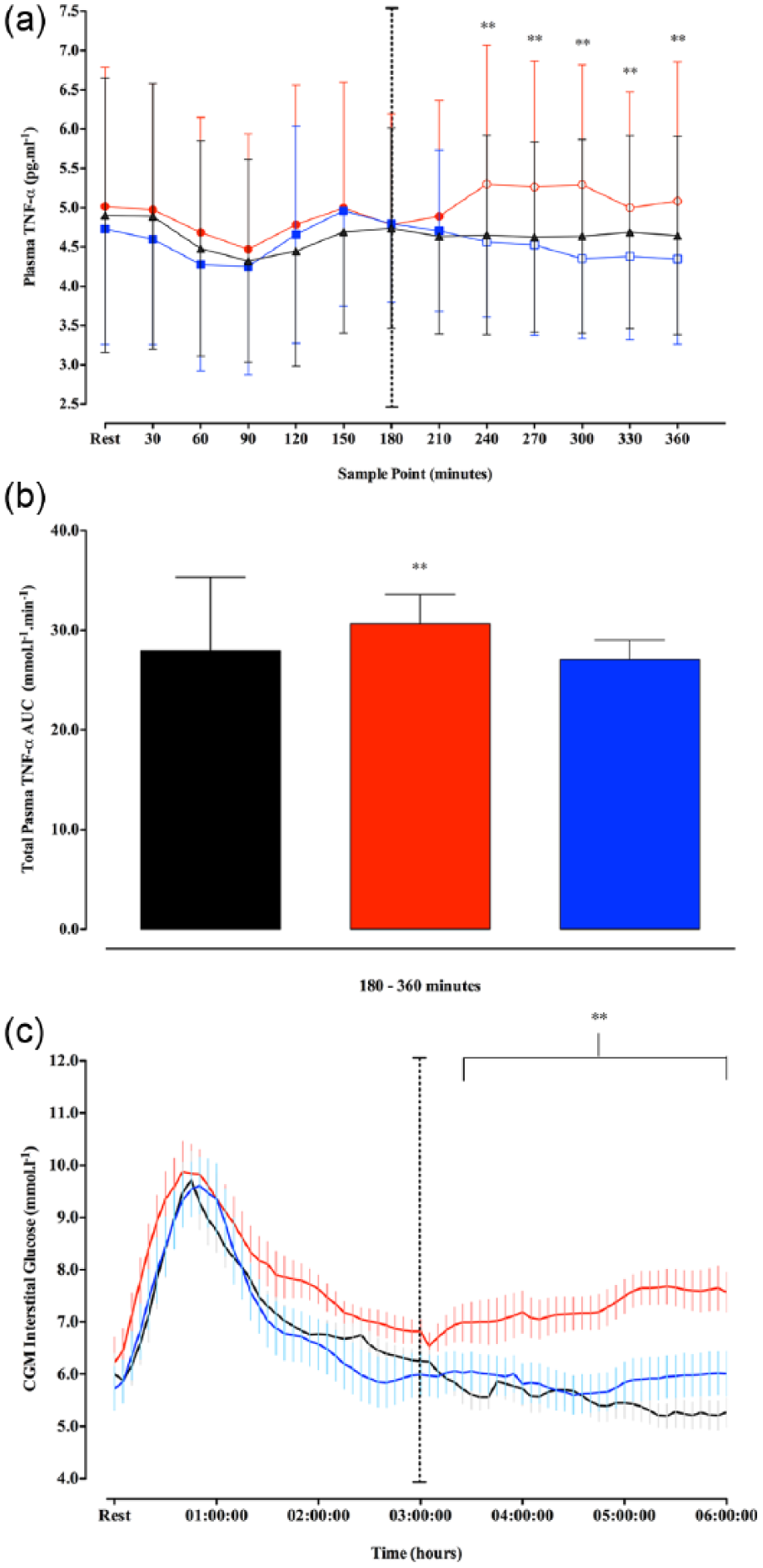
(a)–(c) A time course changes in TNF-α; (b) total plasma TNF-α AUC180−360 and (c) time course changes in CGM interstitial glucose.
Responses of metabolic, hormonal, inflammatory, chylomicron and coagulation markers following high-fat meals/insulin administration.
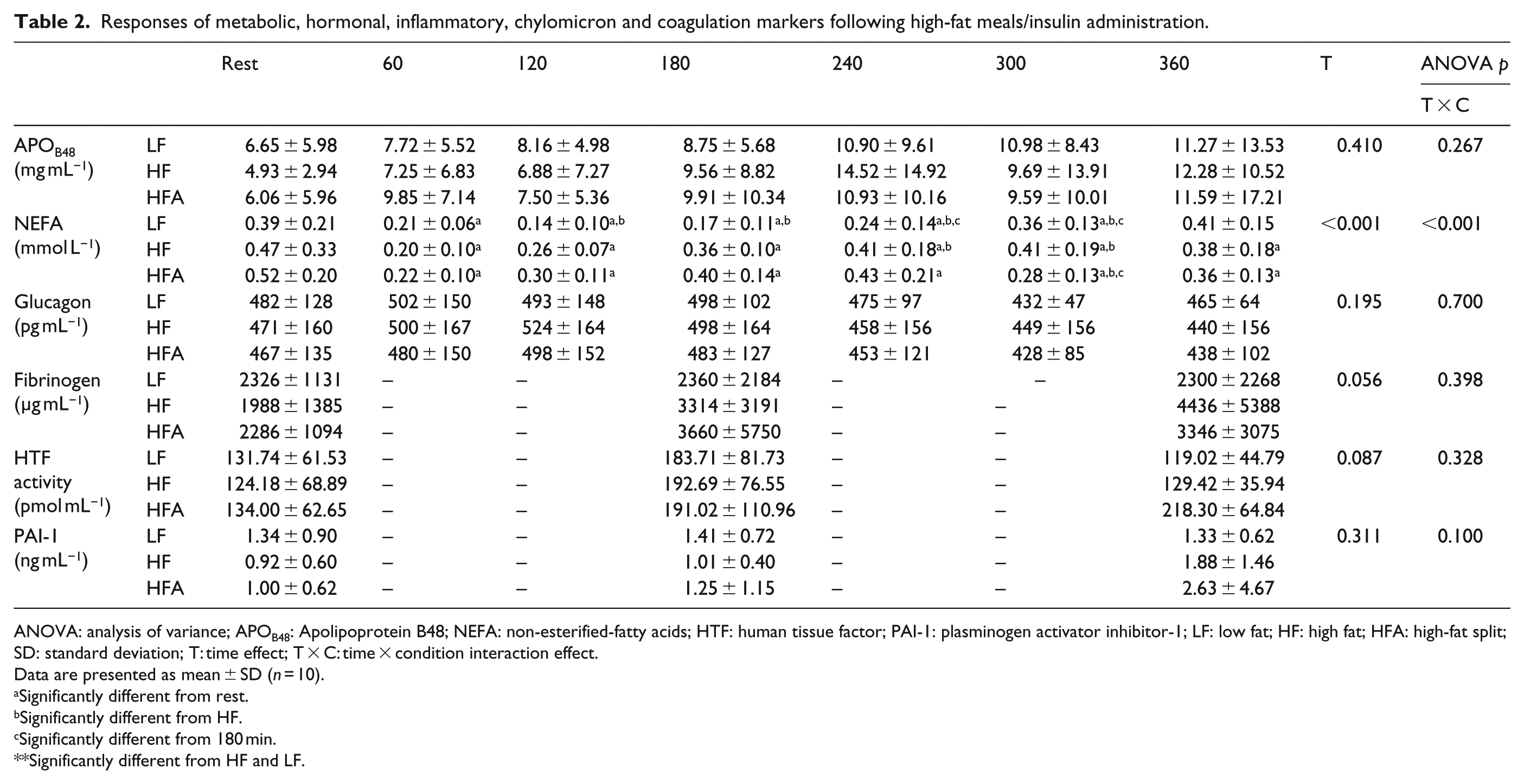
ANOVA: analysis of variance; APOB48: Apolipoprotein B48; NEFA: non-esterified-fatty acids; HTF: human tissue factor; PAI-1: plasminogen activator inhibitor-1; LF: low fat; HF: high fat; HFA: high-fat split; SD: standard deviation; T: time effect; T × C: time × condition interaction effect.
Data are presented as mean ± SD (n = 10).
Significantly different from rest.
Significantly different from HF.
Significantly different from 180 min.
Significantly different from HF and LF.
Discussion
This is the first study to show that in adult, type 1 diabetes patients treated with modern insulin analogue injections, an additional rapid-acting insulin dose, provided 3 h after ingestion of a high-carbohydrate HF meal reduces the late rise in lipaemia seen with when the carbohydrate-counting method for insulin administration at mealtime is used alone. Moreover, such a strategy provides a similar postprandial glycaemic and inflammatory response to a meal with negligible fat content and does not augment the pro-coagulant response of fibrinogen, HFP or PAI-1. In comparison, when following the carbohydrate-counting method at mealtime alone patients are likely to experience raised lipaemia, hyperglycaemia and elevated TNF-α concentrations late into the postprandial period. These findings further highlight the importance of an additional but delayed insulin bolus not just for glucose control per se, but for normalisation of a milieu potentially promoting vascular damage.
Our data show that the addition of dietary fat increases rapid-acting insulin dose requirements, similar to that shown previously in patients using (CSII).8,30–33 For example, Wolpert et al. 8 showed that under closed-loop glucose control, the insulin requirement for a HF evening meal was increased by ~42%, in comparison with a carbohydrate-matched, low-fat meal. This study furthers these previous findings by examining how adjusting the dose and timing of rapid-acting insulin administration influences the metabolic milieu and cardiovascular risk factors associated with consuming mixed-macronutrient meals to date has not been examined within the literature. Our data demonstrate that when administering rapid-acting insulin to cover only the carbohydrate content of the meal (as in the HF condition), patients are exposed to raised triglycerides and TNF-α at 4–6 h post-meal (Figure 1(a)–(c)). In addition, we observed a trend towards an increase in fibrinogen late into the postprandial period; our sample size was likely too small to yield statistical significance in this individual marker; however, our findings indicate an increased inflammatory and thrombotic response following high-carbohydrate, HF meal feeding in people with type 1 diabetes that can be prevented with an additional delayed bolus of insulin. These data call for a large-scale observation of the thrombotic responses to high-carbohydrate HF meal feeding, and it is recommended that subsequent interventions to reduce postprandial lipaemia consider this as a potentially important outcome.
Prior research has shown that HF meals (>70 g of fat) can increase pro-coagulation markers;34,35 however, in the measures we chose, we saw no influence of meal type or dosing strategy. The fat content of the meals within this study was chosen such that they replicated meals that may habitually be consumed by patients (~50 g of fat), and may simply not have been large enough for subtle changes in insulin dose (+30% equalling ~2.6 IU) and timing to cause a demonstrable effect at the respective sample points. Additionally, the postprandial glucose excursions were only moderately hyperglycaemic under both HF conditions.
It is noteworthy that foods with different fatty acid profiles may elicit different postprandial lipaemic 28 and inflammatory 36 responses, potentially mediated via modulation of insulin sensitivity, 37 gastric emptying, 38 gut hormones responses, 38 circulating adhesion molecules 39 and oxidative stress generation.39,40 Fats predominantly saturated and of long-chain in composition cause a delayed postprandial lipaemic response.28,34 The fatty acid profile of the clarified butter added to the meals in this study was ~62% saturated and 29% monounsaturated fat, which, as highlighted in this study, is likely to result in a delayed and exaggerated lipaemic response occurring beyond the action time profiles of modern rapid-acting insulin analogues if administered as a single bolus at the time of meal ingestion. 41 As such, the differential responses between HF and HFA in late lipaemia can be attributed to our insulin administration strategy, considering (1) glycaemia was similar between conditions up to 180 min post-meal and (2) the triglyceride response under HF beyond 180 min is comparable to previous observations profiling time-course lipaemic responses in individuals without type 1 diabetes following HF feeding. 42
Prior research examining the interactions of protein in isolation,43,44 and in combination with carbohydrate and fat, 32 shows that protein can raise postprandial glucose late after feeding, with additive effects when combined with fat. 32 Meal protein content was kept under 30 g, such that no bolus insulin dose adjustment for the protein content would be needed; 45 indeed, under the LF condition, patients demonstrated no late postprandial hyperglycaemia, with all patients remaining within euglycaemic ranges when the carbohydrate-counting method was employed.
Conclusion
In conclusion, these are the first data to demonstrate that when eating a meal with a high-carbohydrate and high-fat content, an additional insulin dose provided 3 h into the postprandial period reduces plasma triglyceride concentrations and inflammatory markers in type 1 diabetes patients. Thus, people with type 1 diabetes treated with basal-bolus insulin injections should be encouraged to carbohydrate count at mealtime and administer additional insulin units 3 h into the postprandial period when consuming a high-carbohydrate, high-fat meal. Not accounting for the fat component of the meal is associated with raised blood lipids, delayed glucose excursions and increased inflammation. Based on our findings, patients should be advised of the importance of the late bolus not just for glucose control but also for normalising other markers that may negatively influence vascular health.
Footnotes
Acknowledgements
The authors wish to acknowledge the time and commitment of the study participants and the Research Team at the Newcastle NIHR Clinical Research Facility in assisting in trial management and study conduct. Clinical trial registration: ![]() ; reg. no. NCT02595658. M.D.C. designed the study; collected, analysed and interpreted data; and wrote the manuscript. M.W. assisted in data collection and prepared the manuscript. R.A.A. contributed to the interpretation of data and preparation of the manuscript. K.M.B. contributed to the interpretation of data and preparation of the manuscript. J.T.G. interpreted data and wrote the manuscript. D.J.W. designed the study; analysed and interpreted data; and wrote the manuscript. Only the named research team was involved in the design of the study; collection, analysis and interpretation of data; and in writing the manuscript. All data generated or analysed during this study are included in the published article (and its supplementary information films).
; reg. no. NCT02595658. M.D.C. designed the study; collected, analysed and interpreted data; and wrote the manuscript. M.W. assisted in data collection and prepared the manuscript. R.A.A. contributed to the interpretation of data and preparation of the manuscript. K.M.B. contributed to the interpretation of data and preparation of the manuscript. J.T.G. interpreted data and wrote the manuscript. D.J.W. designed the study; analysed and interpreted data; and wrote the manuscript. Only the named research team was involved in the design of the study; collection, analysis and interpretation of data; and in writing the manuscript. All data generated or analysed during this study are included in the published article (and its supplementary information films).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
This study received approval from the local National Health Service Research Ethics Committee (R&D Ref: 7241).
Funding
This study was funded by the Newcastle University.
Informed consent
All patients who participated provided written informed consent.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
