Abstract
Background:
The development of disturbances in skin microcirculation in type 1 diabetes is not well characterised. We assessed skin microcirculation longitudinally from the onset of diabetes up to 29 years of duration to investigate when such disturbances start.
Material and methods:
Seventeen adult patients with type 1 diabetes participated. Skin microvascular function in digit IV of the left hand was investigated by laser Doppler fluxmetry (LDF, arbitrary units [AU]). LDF was carried out at rest and following one-min arterial occlusion. Time to peak LDF (s) and percentage increase of LDF (post-occlusive reactive hyperaemia, PRH%) were determined. Retinopathy was assessed from fundus photographs or ophthalmoscopic recordings.
Results:
Skin microvascular function remained normal during the first five years. Compared with baseline and a non-diabetic reference group, time to peak LDF was prolonged after 7–9 years of diabetes (p < 0.01). PRH% was lower than in the reference group after 7–9 years (p < 0.01), and lower than baseline after 24–29 years of diabetes (p < 0.05). All but one patient developed retinopathy and the first signs were found after 10 years of diabetes.
Conclusions:
Functional disturbances in total skin microcirculation were observed after seven years in patients with type 1 diabetes and preceded diabetic complications such as retinopathy.
Keywords
Introduction
Microvascular disturbances are common in patients with type 1 diabetes and lead to characteristic complications in the retina, kidneys and nerves.1,2 Several cross-sectional studies have shown that the microvascular disturbances also involve skin microcirculation.3–8 Disturbances in skin microvascular function are characterized by impaired vasodilatory capacity. These disturbances have important clinical implications, since they are associated with chronic capillary ischaemia.4,5 Capillary ischaemia leads to impaired tissue oxygenation, which may contribute to the development of diabetic complications such as chronic foot ulcers. 9 Earlier studies have indicated that this microvascular dysfunction is associated with factors such as poor glycaemic control 10 and haemodynamic disturbances. 11 Interestingly, disturbances in skin microcirculation seem to be associated with microvascular changes also in other organs 12 and it has been suggested that the human cutaneous circulation is a model of generalized microvascular function. 13 However, to the best of our knowledge, there are no prospective studies that have investigated when, in the duration of type 1 diabetes, the skin microvascular dysfunction develops. Skin microvascular function can be investigated non-invasively and under physiological conditions by means of laser Doppler fluxmetry (LDF), for example.14–17 Using this method, the results of some studies have indicated that disturbances in skin microvascular function may precede other diabetic complications in paediatric patients.6,7
Thus, the aim of this observational study was to investigate the time at which total skin microvascular function was affected after the onset of type 1 diabetes in adults and how this disturbance is associated with other microvascular complications, especially retinopathy.
Our hypothesis was that functional disturbances in skin microcirculation are observed prior to the development of retinopathy.
Patients, non-diabetic reference subjects and methods
Patients
This prospective cohort study was initiated in 1983 and 17 patients (11 men and 6 women; 4 cigarette smokers, 5 ex-smokers) with newly developed type 1 diabetes were enrolled (up to 1985) at the Department of Diabetes and Endocrinology, Danderyd University Hospital, Stockholm. They had all agreed to participate in regular investigations of skin microvascular function. The first investigation was performed 1–6 months after the diagnosis of type 1 diabetes. Subsequent investigations were performed after 1, 2, 3, 4–5 and 7–9 years. The last investigation was performed after 24–29 years of diabetes. At every investigation, the patients underwent clinical examination including systolic and diastolic arm blood pressure (mmHg), finger blood pressure (mmHg), assay of glycated haemoglobin (HbA1c) and assessment of skin microvascular function. In addition to the above investigations, the patients were treated at the clinic according to regular care requirements and best clinical practice. Medical records were checked for documentation of nephropathy, neuropathy and medication.
Non-diabetic reference group
In order to study the influence of time on skin microvascular function, 11 healthy subjects (9 men and 2 women, mean age: 35 years) were investigated 2–3 times over a period of 9 years. None of the healthy subjects had any family history of diabetes.
Methods
Patients and non-diabetic reference subjects were instructed to avoid smoking and coffee drinking on the day of investigation. Skin microcirculation was investigated in a sitting position with the forearm at heart level after an acclimatization period of 20 min at a room temperature of 22°C–24°C.
Blood pressure measurements
Blood pressure was measured in the left arm by a mercury sphygmomanometer (with an appropriate cuff size) and with the subject in a sitting position. Blood pressure was recorded as the mean value of three measurements 1 min apart. Systolic finger blood pressure was assessed by recording the pressure (mmHg) in a miniature cuff (∅ 2–2.5 cm) placed around the base of digit IV of the left hand. Finger blood pressure was determined by evaluating the inflow of blood distal to the cuff by LDF during deflation of the cuff from suprasystolic values.
Skin microcirculation
Skin microcirculation of the nail fold of the left fourth finger was measured by LDF, which gives a semi-quantitative assessment of microvascular blood perfusion,18,19 expressed in arbitrary perfusion units (AU). LDF measures total skin microcirculation, that is, blood circulation in nutritive capillaries, arterioles, venules and dermal vascular plexa. The LDF signal is mainly generated by the movement of blood cells in the thermoregulatory vascular bed.18,19 The LDF signals were recorded over a period of 5 min. A pressure cuff was positioned around the fourth finger base and was used for investigations of post-occlusive reactive hyperaemia (PRH). The cuff pressure used was 200 mmHg for 60 s, after which the pressure was instantly released. After deflation, the percentage increase in baseline LDF during PRH was measured, that is, PRH% = [(peak LDF − baseline LDF)
LDF equipment
All investigations of skin microvascular reactivity, except the last ones, were performed with the same laser Doppler equipment (Periflux 1c, Perimed, Järfälla, Sweden), while the last investigations (after 24–29 years of diabetes) were performed with PeriFlux 4001 Master equipment (Perimed). As transformation formulas do not exist for variables measured with the old Periflux 1c and the new PeriFlux 4001 Master, 30 measurements of PRH% and time to peak LDF were performed on the left hand (digit IV) in 19 healthy subjects using both pieces of equipment. The linear transformation for PRH% was Periflux 1c (Y) = 5.75 + 0.11 × PeriFlux 4001 Master (X), R2 = 0.67 (p < 0.001) and for time to peak LDF, Periflux 1c (Y) = 3.98 + 0.45 × PeriFlux 4001 Master (X), R2 = 0.43 (p < 0.001). The LDF values obtained at the last investigation were transformed to Periflux 1c values using these equations.
Skin temperature
Skin temperature of the investigated finger was continuously recorded with an electronic thermistor (Exacon, Copenhagen, Denmark).
Retinopathy
Retinal photography or a clinical examination involving ophthalmoscopy was performed according to guidelines, that is, every year or every second year. Retinopathy was defined as no retinopathy, non-proliferative or proliferative diabetic retinopathy, as determined by an ophthalmologist in our research team (Dr Gunvor von Wendt).
Neuropathy
Clinical signs of peripheral neuropathy in the feet were investigated through tests of vibration and superficial sensation using a vibration fork (128 Hz) and a monofilament (Semmes-Weinstein 5.07), respectively. The criteria for diagnosis of peripheral neuropathy were loss of vibration sensation at the big toe and/or at the ankle level.
Nephropathy
Prevalence of albuminuria was assessed by way of urinary dipstick tests and different test methods have been used over the years, the last being Clinitek® (Bayer HealthCare LLC, USA). Initially, testing was performed at irregular intervals and it has only been during the last 15 years that testing has been carried out on a regular annual basis.
Glycaemic control
Long-term glycaemic control was assessed by assay of HbA1c. In 1982–1984, HbA1c was determined by an isoelectric focusing method, 21 with a reference value of less than 8.7%, in 1984–1990 by liquid chromatographic assays 22 with a reference value of less than 5.6% and since 1991 the reference value has been less than 5.2%.
Statistical methods
Analyses were carried out using the IBM SPSS statistical package, version 20. Data are presented as mean values ± standard deviation (SD). For overall analysis of change over time of outcome variables, regression analysis on individual data was applied and individual slopes were calculated and statistically tested versus zero. Overall changes as well as changes from baseline were analysed using the (non-parametric) Wilcoxon signed-rank test. A value of p <0.05 was considered statistically significant.
Ethical considerations
In connection with the first visit to the diabetes out-patient clinic in 1983–1985, the patients were asked about participation in this study. The protocol of this trial was approved by the local Ethics Committee of Karolinska University Hospital, Solna. Written informed consent was obtained from all patients and non-diabetic reference subjects.
Results
Patient follow-up
In all, 17 patients (11 males) were included in the study in 1983–1985 and 14 of these patients have been followed for 24–29 years (Table 1). Mean age at diagnosis of type 1 diabetes was 27 ± 7.7 years and at the last investigations the patients had reached the age of 56 ± 7.4 years. Three patients were lost to follow-up. One patient was found dead in his home in 1995, most likely as a result of diabetic ketoacidosis. This patient showed severe functional skin microangiopathy at his last reinvestigation, 9 years after debut of diabetes. The second patient moved abroad in 1995. The third patient, with the highest mean level of HbA1c, developed peripheral neuropathy after 5 years diabetes duration, and after 10 years also microalbuminuria. Later on, he developed severe complications, that is, autonomic neuropathy, and was not able to come for any reinvestigation (1996 onwards) and he later died of diabetes-related complications (nephropathy) in 2010.
Clinical characteristics in patients followed for 24–29 years since onset of type 1 diabetes.
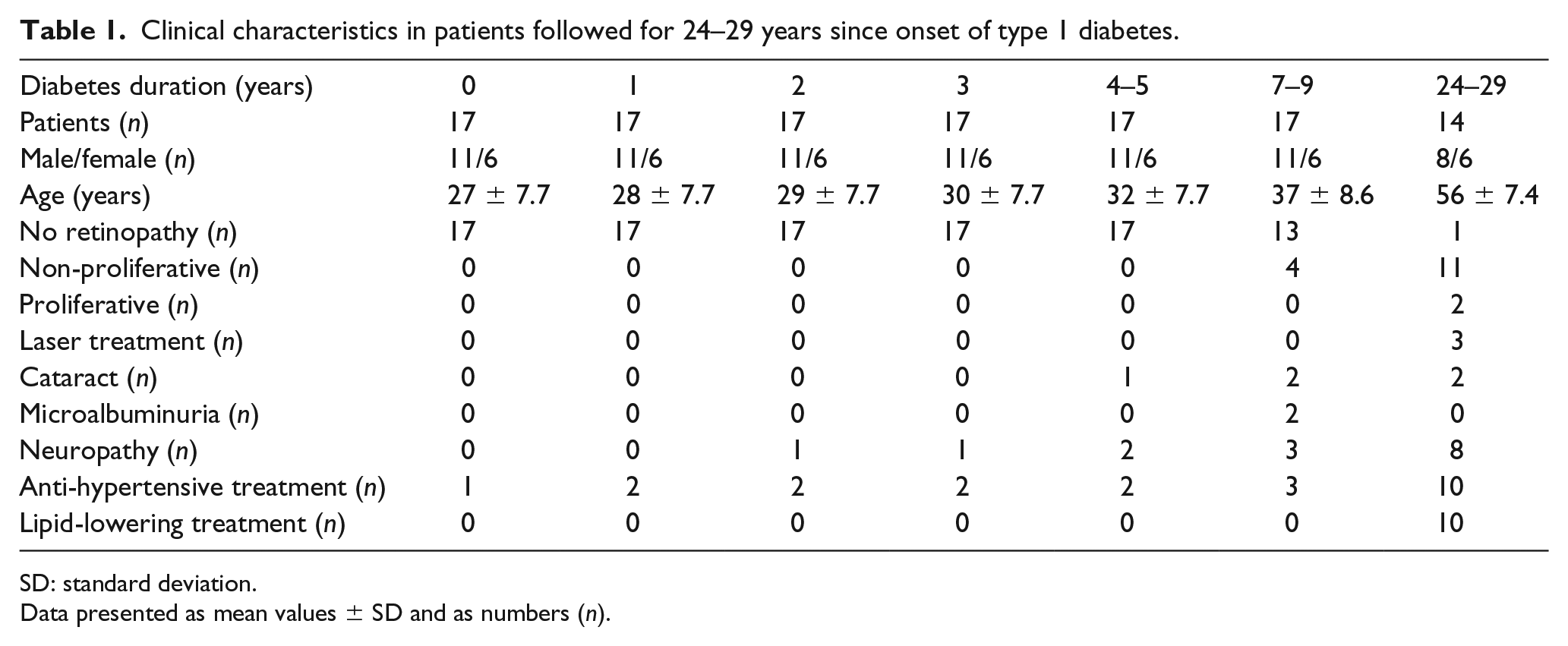
SD: standard deviation.
Data presented as mean values ± SD and as numbers (n).
Skin microvascular function in patients
Time to peak LDF: During the first 5 years of diabetes, time to peak LDF remained normal and did not differ compared with that in non-diabetic reference subjects, while it was significantly (p < 0.01) prolonged at the investigations 7–9 years and 24–29 years after diabetes onset (Figure 1; Table 2). At the investigation after 7–9 years, three patients had extremely prolonged times to peak LDF (11.0, 11.0 and 18.5 s.). Two of these patients were cigarette smokers, one of whom had the earliest debut of albuminuria.
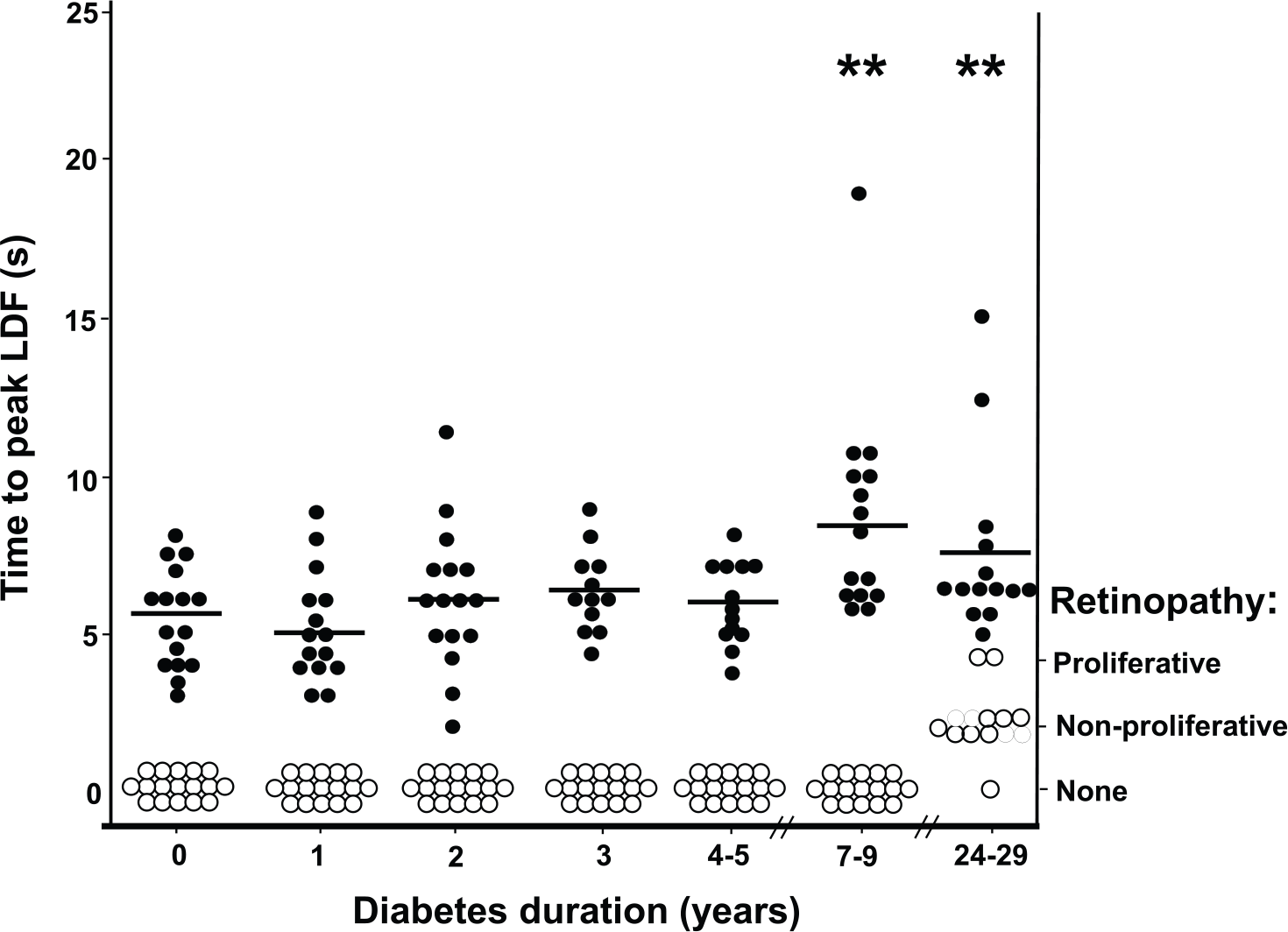
Relation between development of retinopathy and skin microvascular disturbances (left hand) in 17 patients with type 1 diabetes. After 24–29 years diabetes duration, 14 patients remained in the study. Black points represent the individual values of “time to peak LDF”, while the white points represent the degree of retinopathy. The horizontal line mark represents the mean value of time to peak LDF. ** p < 0.01, time to peak LDF as compared to baseline level.
Skin microvascular variables, finger and arm blood pressure in patients with type 1 diabetes.
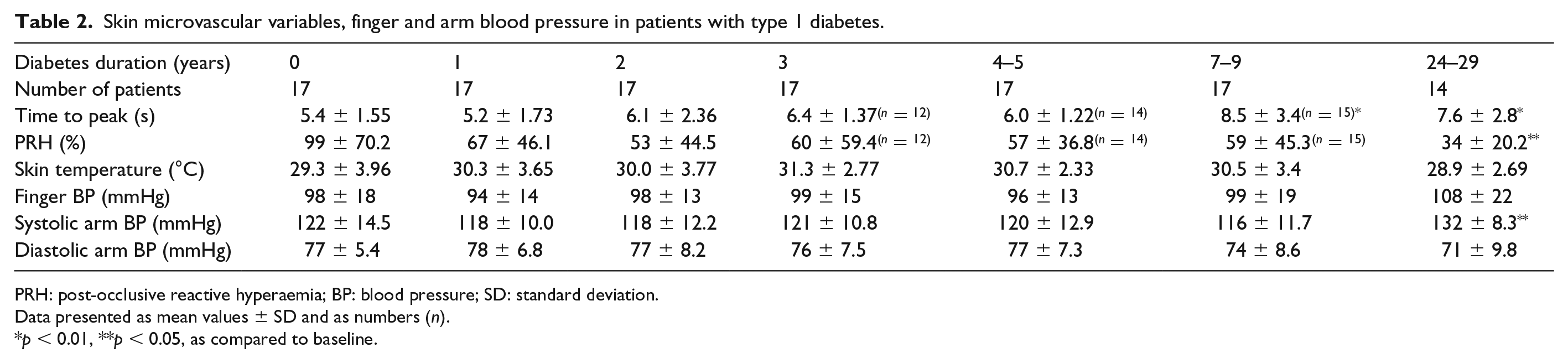
PRH: post-occlusive reactive hyperaemia; BP: blood pressure; SD: standard deviation.
Data presented as mean values ± SD and as numbers (n).
p < 0.01, **p < 0.05, as compared to baseline.
PRH%
PRH% remained within the normal range during the first 5 years of diabetes, after which it significantly decreased over time (p < 0.05) and was lower at the last investigation compared with baseline (p < 0.05; Figure 2). After 7–9 years of diabetes, PRH% was significantly lower than in the non-diabetic reference subjects (p < 0.01).
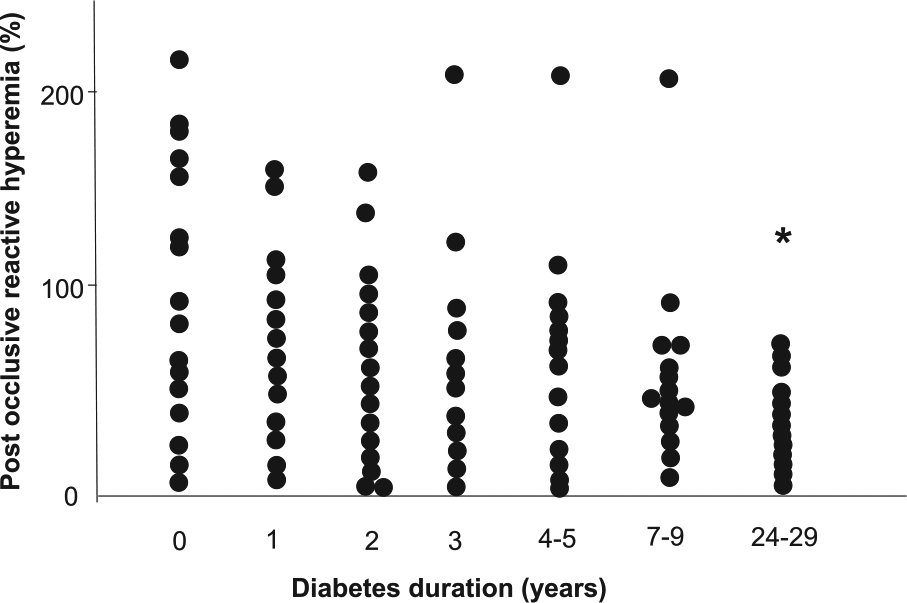
Percentage increase of baseline LDF during post occlusive reactive hyperemia over a time period of 29 years since diagnosis of type 1 diabetes. Seventeen patients were included in the study and 14 remained at the end of the study period. * p < 0.05, as compared to baseline level.
Skin microcirculation in the non-diabetic reference group
None of the investigated parameters, time to peak LDF or PRH%, using the old LDF equipment (Periflux 1c), changed significantly over time (Table 3).
Skin microvascular variables investigated 2-3 times during 9 years in the non-diabetic reference group.

PRH: post-occlusive reactive hyperaemia; BP: blood pressure; SD: standard deviation.
Data presented as mean ± SD and as numbers (n).
Skin temperature in patients and in the non-diabetic reference group
Skin temperature remained stable throughout the investigations without any significant change over time and no significant relationships to the microvascular variables were noted (Table 3).
Hypertension
Target values in the treatment of hypertension have changed over the years. In 1977, the treatment goal was blood pressure less than or equal to 160/95 mmHg (Table 2). This changed in 1984 to less than or equal to 140/90 mmHg. Today, a blood pressure level of less than 140/85 mmHg is recommended in patients with diabetes.23,24 At the last investigation (after 24–29 years of diabetes), 10 of the patients in this study were on anti-hypertensive treatment, the most common forms being use of angiotensin-converting enzyme (ACE) inhibitors, angiotensin II receptor blockers and/or calcium antagonists, which had often been added over the years. Changes of finger and arm blood pressures during the study period are seen in Table 2. Systolic arm blood pressure was significantly (p < 0.05) increased at the last investigation compared with baseline, whereas the diastolic blood pressure did not change significantly (p = 0.2). One patient was already receiving treatment with a calcium antagonist at onset of diabetes, while another patient started treatment with an ACE inhibitor and a calcium antagonist 1 year after the onset of diabetes.
Systolic finger blood pressure showed a pattern similar to that of systolic arm blood pressure. However, the level at the last investigation was not significantly different from that at baseline (p = 0.2).
Systolic finger blood pressure in the non-diabetic reference subjects is shown in Table 3 and no significant change was seen during the study period.
HbA1c
In general, with a couple of exceptions, HbA1c was assayed yearly. The number of individual measurements noted varied between 8 and 29 depending on the availability of medical records and years since diagnosis. Levels of HbA1c at baseline were 9.3% ± 2.2%, after 4–5 years 7.9% ± 2.3%, after 7–9 years 8.1% ± 2.5% and at the last visit 7.3% ± 1.2%. The HbA1c assay method changed during the study period (see section ‘Methods’).
Retinopathy
In all, 14 patients have been followed for 24–29 years (Figure 1). In total, 13 of them have developed some degree of retinopathy since diabetes onset, while one patient so far has no signs of the condition. On average, the patients developed retinopathy after 17 years of diabetes. However, in four patients who developed non-proliferative retinopathy, the first signs were seen 10–13 years after diabetes diagnosis. Two of them later developed proliferative retinopathy and underwent laser treatment. All four patients developed skin microvascular disturbances, which preceded the development of retinopathy by 1–5 years.
Two patients developed cataracts; one was diagnosed after 4 years of diabetes and underwent surgery 6 years later. The other patient developed a cataract after 12 years of diabetes. These two patients had the highest mean levels of HbA1c during the study period.
Relationship between skin microvascular function and retinopathy
In Figure 1, the relationship between skin microvascular reactivity as measured by time to peak LDF and the development of retinopathy is illustrated. A prolonged time to peak LDF was measured after 7–9 years of diabetes, compared with baseline (p < 0.01). At an individual level, skin microvascular disturbances preceded retinopathy.
Neuropathy
Eight patients (five males) developed peripheral neuropathy during the study period (Table 1). The median duration of diabetes was 17 years until the first signs of peripheral neuropathy. However, two patients already showed signs of peripheral neuropathy after 2 and 5 years of diabetes and they also later developed gastroparesis. Four of the eight patients who developed peripheral neuropathy also had signs of autonomic neuropathy (diagnosed after 3, 4, 16 and 19 years of diabetes). Two of them were diagnosed with gastroparesis, one with orthostatic hypotension and all four complained of erectile dysfunction. Two patients developed chronic neuropathic foot ulcers after 17 and 18 years of diabetes.
Nephropathy
In all, 16 patients have regularly been screened for microalbuminuria over the study period (Table 1). Two patients have been screened annually, while the others have been screened at 2- to 4-year intervals. Only two patients were found to have developed microalbuminuria, one patient on two occasions after 10 years of diabetes.
Hyperlipidaemia and cardiovascular events
Blood lipids were seldom measured until the late 1990s. In seven patients, total cholesterol levels were measured in the late 1980s and all these patients had elevated levels. Lipid-lowering treatment was in general initiated in 2001, approximately 15–20 years after the onset of diabetes. When hyperlipidaemia was diagnosed, patients were treated according to the actual guidelines. In 14 patients, blood lipids had been assayed on at least one occasion, often on several occasions. In all, 13 of these patients showed elevated levels of low-density lipoprotein (LDL) cholesterol and total cholesterol at three or more investigations. In all, 10 of these patients eventually received lipid-lowering medication. One patient had normal lipid levels during the whole study period.
Two patients have suffered cardiovascular events. One male patient, a smoker with dyslipidaemia, suffered an ischaemic stroke on two occasions, at the age of 54 and 66 years. The other patient, a female, suffered a cardiac arrest at 45 years of age, and survived.
Discussion
This study is, to our knowledge, the first prospective long-term study of skin microvascular function in adult patients with type 1 diabetes, that is, the patients were followed from the onset of type 1 diabetes up to almost 30 years. The novel findings are that skin microvascular reactivity was within the normal range during the first 5 years, while some patients showed impaired microvascular function after 7 years, disturbances which preceded retinopathy.
Investigations of skin microcirculation are commonly performed in the forearm/fingers or in the feet/toes and it should be noted that skin microvascular reactivity differs between these skin sites.25–27 In this study, we investigated skin microvascular reactivity in fingers by LDF during PRH. LDF is a generally accepted non-invasive method for evaluation of skin microvascular dilatation capacity.14,15,17,18,28 It gives a measure of total skin blood perfusion, that is, the measurement covers thermo-regulating arterio-venous vessels, resistance arteries, arterioles and venules, while less than 10% of the laser Doppler signal is generated by flow in nutritive capillaries. 29 PRH refers to the increase in blood flow above baseline values following arterial occlusion, which is a complex response involving integrated functions of the endothelium, nerves and vascular smooth muscle cells. Vascular endothelium plays an important role in regulation of blood flow by releasing vasodilators, such as nitric oxide and prostacyclin, and vasoconstrictors, such as endothelin and angiotensin II. The availability of nitric oxide is reduced in patients with type 1 diabetes, 30 while the levels of vasoconstrictors may be increased. 31 An imbalance between vasodilators and vasoconstrictors in favour of vasoconstriction may be one factor contributing to the impaired microvascular reactivity seen in this study.
The background of vascular complications in diabetes is multifactorial. Poor glycaemic control is an important factor,1,2,32 especially as regard retinopathy, and most likely also the development of skin microvascular dysfunction.3,4,10,33 Long-term hyperglycaemia accelerates production of reactive oxygen species, non-enzymatic glycation and the formation of advanced glycation end-products (AGEs).30–32 AGEs accumulate in the vessel wall, leading to endothelial and smooth-muscle cell dysfunction, increased vascular stiffness and reduced vessel wall distensibility. 32 In agreement with the results of large-scale studies, 34 this study revealed that the patients who developed the most complications had the highest HbA1c levels. However, no correlation was found between glycaemic control and skin microvascular function.
Notably, impaired skin microvascular reactivity preceded the first signs of retinopathy (Figure 1), that is, the patients with the earliest signs of retinopathy all had microvascular disturbances in the skin, which had developed 1–5 years earlier. The reason for this finding in studies of microvascular beds is unclear, but skin microcirculation may, on one hand, be more sensitive to the diabetic state than retinal microcirculation. On the other hand, differences in the microvascular methods used may also be of importance. Skin microcirculation was studied by LDF, a method used for investigation of functional microangiopathy, while retinal photography or a clinical examination (ophthalmoscopy) is used to detect structural changes. Our finding of an association between skin microvascular disturbances and retinopathy is in line with the results of other studies showing that endothelial- and non-endothelial-dependent skin microvascular responses are related to retinopathy. 12 It has been suggested that the human cutaneous circulation may be a model of generalized microvascular function. 13 Of note, the skin microcirculatory data in this study indicate not only an association with retinopathy, but also the fact that disturbances in skin microcirculation precede retinopathy.
Neuropathy is associated with both metabolic and microvascular disturbances.34,35 Fagerberg 35 was the first to show that patients with diabetic neuropathy had impaired capillary blood feeding of the sural nerve due to increased arterio-venous shunting of blood in the microcirculation. Arterio-venous shunts, which are numerous in microcirculation of the skin, are mainly innervated by the sympathetic nerve system. In patients with neuropathy, these shunts may be denervated, leading to increased arterio-venous shunting of blood which then by-passes the capillaries, giving rise to chronic capillary ischaemia. 4 In line with the results of other studies, 34 neuropathy developed early after diabetes onset in our patients and was more common in patients with poor glycaemic control. Over time, slightly more than half of the patients developed peripheral neuropathy, which is in line with results in larger studies. 36 Two patients showed clinical evidence of peripheral neuropathy during the first 5 years of diabetes, while skin microvascular function was normal. The reason is most probably that LDF measures total skin microvascular flow and does not detect disturbances in the capillary circulation.4,5,15 Using capillaroscopy and LDF for simultaneous measurements of skin microcirculation in the feet, we have clearly shown normal, or even increased, total microcirculation but a markedly impaired capillary circulation as early as within the first 2 years of type 1 diabetes. 4
The number of patients with signs of nephropathy was low in this study; only two patients developed microalbuminuria, which was diagnosed 10 and 14 years after the onset of diabetes. Microalbuminuria was later normalized when anti-hypertensive treatment was initiated although there was no improvement of skin microvascular reactivity in these patients.
In addition to hyperglycaemia, hypertension and dyslipidaemia are associated with vascular complications. Hypertension is related to structural and functional changes in arterioles and small arteries, alterations which may lead to the impaired microvascular reactivity seen in this study. 37 At the last visit, a majority of the patients were on anti-hypertensive treatment, while one patient has been treated with a calcium antagonist since the onset of diabetes and another patient received treatment with an ACE inhibitor and a calcium antagonist within the first year of diabetes. These anti-hypertensive drugs seem to improve the microcirculation,38–40 and, interestingly, these two patients had normal microvascular function during the study period. After 7–9 years, the blood pressure had not increased significantly in the study group. Three patients were at that time receiving anti-hypertensive medication, whereas the skin microvascular function was at that time significantly disturbed, suggesting that the skin microvascular dysfunction might be more dependent on diabetes itself than on hypertension.
Smoking has to be considered here since smokers were included in this study. Smoking induces distinct oxidant stress-mediated vascular dysfunction detectable in the skin as demonstrated by Rossi et al. 41 and Edvinsson et al. 42
Dyslipidaemia is another mediator of vascular complications, especially cardiovascular disease. As with treatment of hypertension, the recommendations regarding lipid-lowering therapy were less stringent during the 1980s and 1990s compared with today’s guidelines and the treatment was sometimes initiated later on. In this study, all patients but one were being treated with lipid-lowering drugs at the last visit. We have recently shown that treatment with high-dose atorvastatin may impair skin microvascular reactivity in patients with type 1 diabetes. 43 However, none of the patients in this study were treated with high-dose statins, nor had they any other medication, except insulin, which may have contributed to the impaired skin microvascular reactivity observed at the two last visits.
The limitation of this study is the small sample size. However, the patient material seems to be representative of a group of patients with type 1 diabetes, since the vascular complications developed within the expected time frame in relation to other studies.1,2,44 A strength of our study is that error regarding the skin microvascular investigations was minimized, since the same laboratory assistant performed all investigations throughout the whole study period.
In summary, this study showed that skin microvascular reactivity in the fingers (investigated by LDF) was normal during the first 5 years of type 1 diabetes. Several patients showed impaired microvascular reactivity after 7 years of diabetes, disturbances which were found prior to the first signs of retinopathy. Thus, investigations of skin microvascular function might be a way to discover patients at high risk of later general microvascular complications such as retinopathy. These patients could be targeted as regards optimizing metabolic control in order to reduce the risk of future microvascular complications.
Footnotes
Acknowledgements
The authors gratefully acknowledge the expert technical assistance of laboratory assistant Ann-Christin Salomonsson, who performed all the microvascular investigations during the study period.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by independent grants from the Berth von Kantzow Foundation and Diabetesfonden. Financial support was also provided through the regional agreement on medical training and clinical research (ALF) between Stockholm County Council and Karolinska Institutet.
