Abstract
Background:
Previous studies have shown a U-shaped relationship between systolic blood pressure and risk of all-cause of mortality in patients with type 2 diabetes and renal impairment.
Aims:
To evaluate the associations between time-updated systolic blood pressure and time-updated change in systolic blood pressure during the follow-up period and risk of all-cause mortality in patients with type 2 diabetes and renal impairment.
Patients and methods:
A total of 27,732 patients with type 2 diabetes and renal impairment in the Swedish National Diabetes Register were followed for 4.7 years. Time-dependent Cox models were used to estimate risk of all-cause mortality. Time-updated mean systolic blood pressure is the average of the baseline and the reported post-baseline systolic blood pressures.
Results:
A time-updated systolic blood pressure < 130 mmHg was associated with a higher risk of all-cause mortality in patients both with and without a history of chronic heart failure (hazard ratio: 1.25, 95% confidence interval: 1.13–1.40 and hazard ratio: 1.26, 1.17–1.36, respectively). A time-updated decrease in systolic blood pressure > 10 mmHg between the last two observations was associated with higher risk of all-cause mortality (−10 to −25 mmHg; hazard ratio: 1.24, 95% confidence interval: 1.17–1.32).
Conclusion:
Both low systolic blood pressure and a decrease in systolic blood pressure during the follow-up are associated with a higher risk of all-cause mortality in patients with type 2 diabetes and renal impairment.
Introduction
Both type 2 diabetes (T2D) and renal impairment (RI) increase the risk of all-cause and cardiovascular mortality,1–5 and the excess risk is correlated with the severity of RI.6–8 Previous prospective observational studies in patients with T2D have shown a higher risk for cardiovascular events (CVEs) and all-cause mortality in patients with systolic blood pressure (SBP) < 130 mmHg.9–11 A U-shaped or J-shaped relationship between BP and all-cause mortality has been described in several studies,12–16 including a recent publication from our group. 17 In addition, a recent systematic review and meta-analysis on the effects of intensive antihypertensive treatment showed no additional benefit on major CVEs in patients with RI. 18 Current treatment guidelines recommend a BP target of<140/80 mmHg in patients with diabetes and RI and <130/80 mmHg when albuminuria is present.19,20
The relationships between SBP and risk of all-cause mortality described in observational studies may, however, well be due to comorbidities, particularly chronic heart failure (CHF), as opposed to treatment effect.21,22 This view is further supported by findings in a recent study by Adamsson-Eryd et al. 23 in which patients with T2D and no previous cardiovascular disease (CVD), that is, no renal dysfunction or heart failure, were studied. In this study, a lower SBP was associated with significantly lower risk of CVEs, and the authors conclude that the association between low blood pressure (BP) and increased mortality in previous studies probably is due to concomitant diseases rather than antihypertensive treatment.
The main objective of this study was to further evaluate the relationship between SBP and all-cause mortality in patients with T2D and RI, with or without a history of CHF. The association between both baseline SBP and time-updated mean SBP, that is, SBP during the follow-up period, and all-cause mortality was assessed. In addition, we studied the impact of time-updated change in SBP between the last two observations during the follow-up period. Additionally, a potential association between the number of antihypertensive drug classes used at baseline and the risk of all-cause mortality was evaluated. This was done in a nationwide observational study of unselected patients who had been diagnosed with T2D and RI, clinically treated and reported to the Swedish National Diabetes Register (NDR).
Patients and methods
NDR
Launched in 1996, the NDR covers risk factors, complications and medications among Swedish patients with diabetes 18 years of age or older. 24 All medical hospital clinics and more than 90% of primary care centres in Sweden currently report to the Registry and therefore the majority of all patients with diagnosed T2DM are included in the Registry (www.ndr.nu). The coverage in patients may be somewhat lower in elderly patients and patients with severe comorbidities such as severe RI or heart failure. Oral or written informed consent is obtained from each patient. Information is collected and reported at least once a year during regular scheduled appointments. 5 The study was approved by the Regional Ethical Review Board at the University of Gothenburg and conducted in accordance with the Declaration of Helsinki.
Patients
We included 27,732 patients with T2D and RI [estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73 m2 according to Modification of Diet in Renal Disease (MDRD)]. Inclusion criteria were a diagnosis of T2D with a reported serum creatinine value between >20 and <800 µmol/L at baseline. Reported T2D was defined as reported treatment with diet only or with oral glucose-lowering agents only, or onset of diabetes at 40 years of age or older and treated with insulin, either alone or in combination with oral glucose-lowering agents. Patients with body mass index (BMI) ≤ 18 or ≥45 kg/m2 (n = 359) were excluded. In addition, patients with severe RI (eGFR < 15 mL/min/1.73 m2; n = 274) were excluded because they are reported to the NDR on a limited and highly selective basis. Patients with at least one reported serum creatinine in NDR between 1 January 2005 and 31 December 2007 were included and monitored until death or the conclusion of the study on 31 December 2011. 24 The average follow-up period was 4.7 years.
Methods and measurements
All BP measurements had been reported to the NDR according to the Swedish standard – the mean (mmHg) of two supine readings (Korotkoff I–V) with a cuff of appropriate size after at least 5 min of rest.
Laboratory analyses of serum creatinine, HbA1c and blood lipids were performed at local laboratories. Renal function expressed as the eGFR (mL/min/1.73 m2) was calculated using the MDRD equation. 25 Albuminuria was defined as microalbuminuria or macroalbuminuria, that is, urinary albumin excretion rate of 20–200 or >200 µg/min in two out of three consecutive tests within 1 year. Smoking was defined as one or more cigarettes per day, or a pipe, including cessation within the past 3 months. Data on history of CVD and CHF at baseline were obtained from the Hospital Discharge Register (Swedish National Board of Health and Welfare). History of CVD was defined as coronary heart disease (CHD), stroke or peripheral arterial disease (PAD). CHD was defined as myocardial infarction (MI) [International Classification of Diseases-10 (ICD-10) code I21], unstable angina (ICD-10 code I20.0), percutaneous coronary intervention and/or coronary artery bypass grafting. History of CHF was defined as ICD-10 code I50. 27 Data on prescribed medication and classes of antihypertensive medication were obtained from the Prescribed Drug Register, which contains detailed information about all prescriptions picked up at Swedish pharmacies.
Follow-up and definition of end points
Data on all-cause mortality were retrieved by linkage of the Cause of Death Register (Swedish National Board of Health and Welfare), which is a reliable, validated alternative to revised death certificates. 28
Statistical methods
Baseline clinical and biochemical characteristics are presented as mean values ± standard deviation (SD) or as proportions (n, %). The influence of baseline SBP, time-updated mean SBP during the follow-up period and time-updated change (variability) in SBP between the last two observations has been evaluated by fitting a Cox proportional hazards regression model adjusted for the baseline characteristics of age, gender, BMI, HbA1c, diabetes duration, absence/presence of albuminuria, eGFR, history of CVD, history of CHF, smoking, lipid-lowering therapies, number of antihypertensive drug classes and additional variables as indicated. A total of 27,062 patients had complete data sets with 670 patients missing due to lack of information on lipid medication at baseline. In addition, 3113 patients only had one reported BP during follow-up and were thus not included in the analyses. Altogether, 23,949 patients were evaluated in the Cox models. In this study, to define correlation between all values of SBP and the risk of mortality, we used updated mean SBP. In this study, we used time-updated mean SBPs, and in these analyses, the mean SBPs from every participant in the study are used until each event. The reason for using time-updated values is to reduce or remove the influence of known or unknown confounders like age or comorbidities on SBP over the time which can lead to systemic error or bias. The effect of time-updated mean SBP during the follow-up period and time-updated change in SBP between the last two observations was evaluated both as discretised variables and using a penalised smoothing spline function with 3 degrees of freedom (Figures 1 and 2). No imputation was performed, and only complete cases were analysed. The analysis was performed using SAS® 9.4 and R 3.1.0.
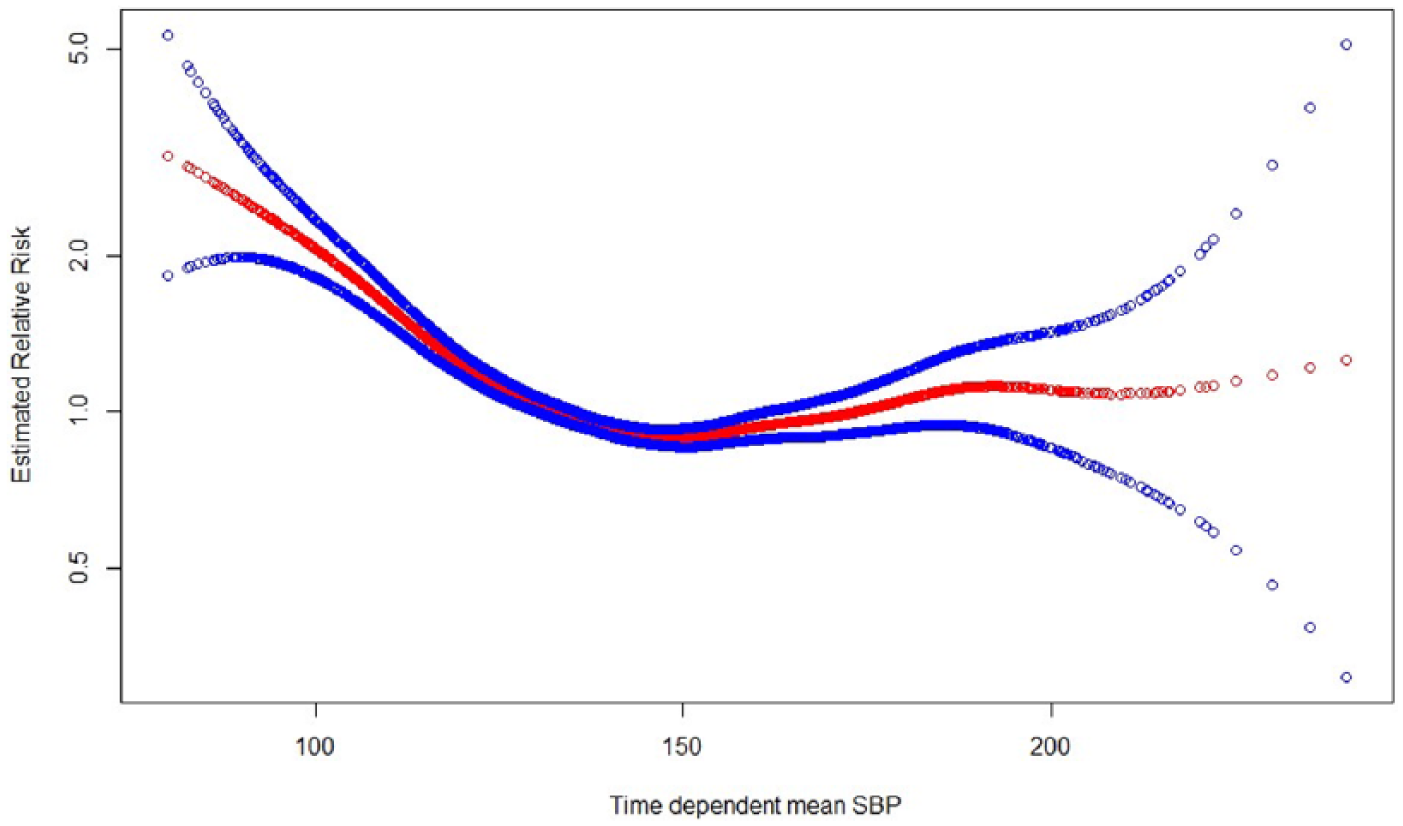
The relationship between time-updated mean SBP as a continuous variable and the RR of all-cause mortality using smoothing splines function with 7 degree of freedom and 95% confidence intervals (unshaded circles) in patients diagnosed with T2D and RI. Adjusted for covariates as described in Table 2.
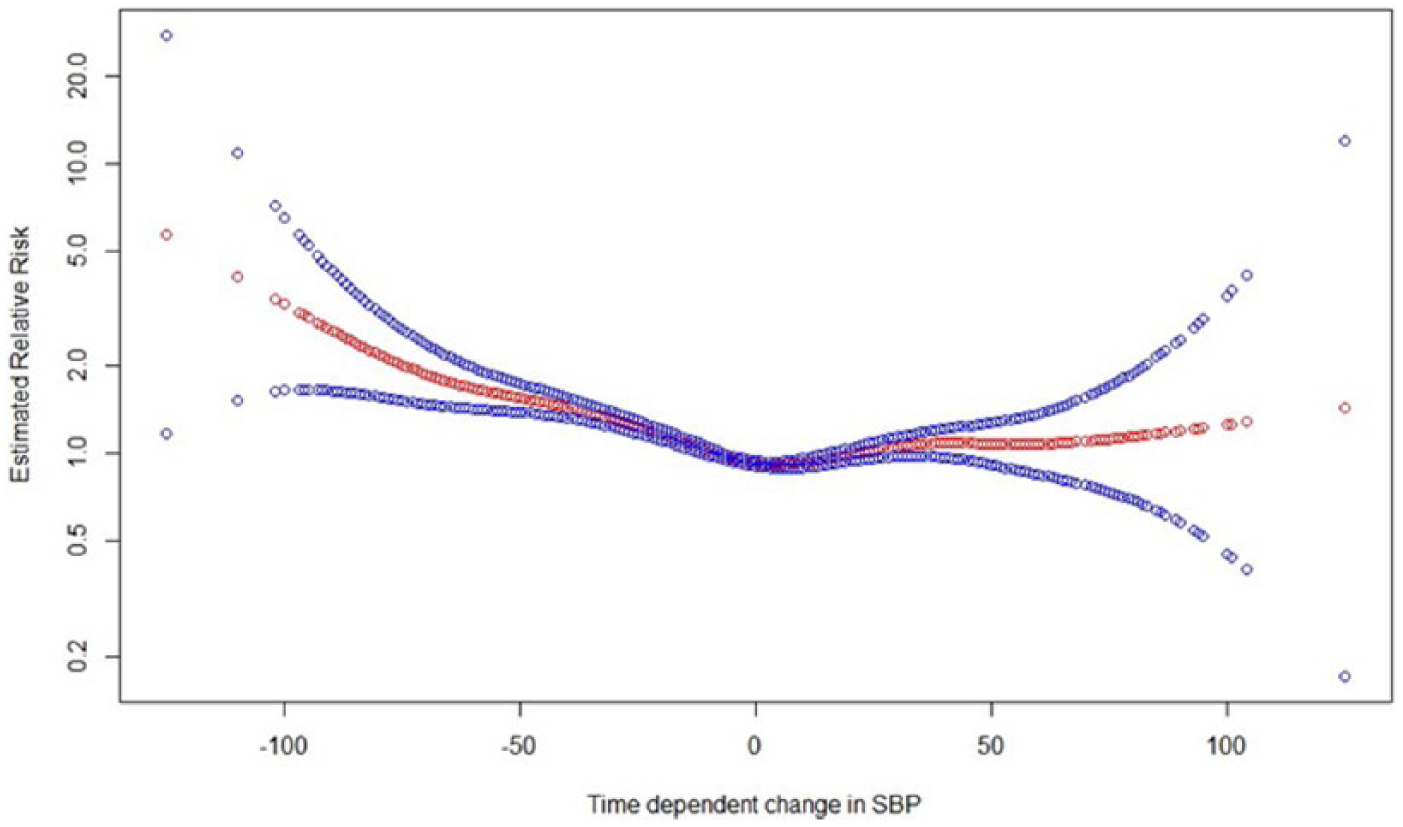
The relationship between the time-updated change in SBP from baseline to the last observation as a continuous variable and the RR of all-cause mortality using smoothing splines function with 7 degree of freedom and 95% confidence intervals (unshaded circles) in patients with T2D and RI. Adjusted for covariates as described in Table 2.
Results
Clinical and biochemical characteristics at baseline
Baseline characteristics are displayed in Table 1. The mean overall eGFR was 48 ± 9 mL/min/1.73 m2; thus, the majority of patients had chronic kidney disease (CKD) stage 3. Only one-third were albuminuric. The majority were women, the mean SBP was 140 ± 18 mmHg, and the mean diastolic blood pressure (DBP) was 73 ± 10 mmHg. Approximately, 60% of the patients had SBP between 130 and 160 mmHg and 20% had SBP below 130 mmHg or above 160 mmHg. One-third (31%, n = 8580) had a history of CVD and 14% (n = 3933) had a history of CHF. Patients with a history of CHF were older, more often men, with longer diabetes duration, lower eGFR, more often albuminuric and with lower SBP and DBP. Two-thirds of the patients with a history of CHF also had a history of CVD, and three-quarters had two or more antihypertensive drug classes. Patients were then stratified into three groups according to SBP [<130 mmHg (22%), 130–159 mmHg (58%) and ≥160 mmHg (19%)]. Patients with SBP <130 mmHg were younger, more often men, with shorter diabetes duration, better glycaemic control (HbA1c) and less often albuminuric. A history of CVD or CHF was more common in patients with SBP < 130 mmHg than the two other groups. Only 6% of patients with SBP < 130 mmHg at baseline had four antihypertensive medications. During the follow-up period, 8265 or 30% of the patients died, 2268 of whom had a history of CHF (57%) and 6290 (26%) did not have a history of CHF. The two most common causes of death were CVD (34%) and malignancies (18%).
Clinical and biochemical characteristics of patients with T2D and RI (eGFR < 60 mL/min/1.73 m2) with and without history of CHF, categorised by SBP at baseline.
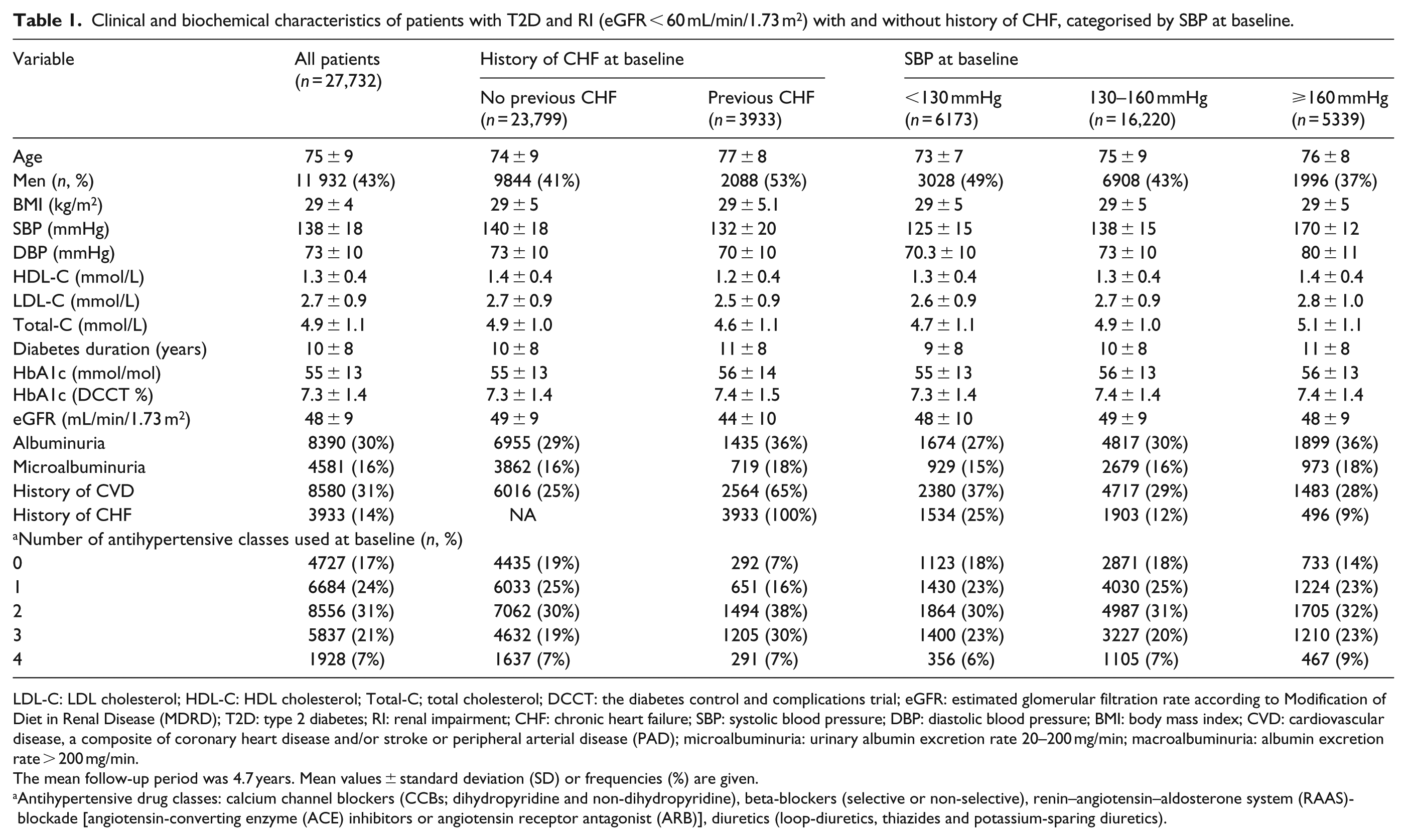
LDL-C: LDL cholesterol; HDL-C: HDL cholesterol; Total-C; total cholesterol; DCCT: the diabetes control and complications trial; eGFR: estimated glomerular filtration rate according to Modification of Diet in Renal Disease (MDRD); T2D: type 2 diabetes; RI: renal impairment; CHF: chronic heart failure; SBP: systolic blood pressure; DBP: diastolic blood pressure; BMI: body mass index; CVD: cardiovascular disease, a composite of coronary heart disease and/or stroke or peripheral arterial disease (PAD); microalbuminuria: urinary albumin excretion rate 20–200 mg/min; macroalbuminuria: albumin excretion rate > 200 mg/min.
The mean follow-up period was 4.7 years. Mean values ± standard deviation (SD) or frequencies (%) are given.
Antihypertensive drug classes: calcium channel blockers (CCBs; dihydropyridine and non-dihydropyridine), beta-blockers (selective or non-selective), renin–angiotensin–aldosterone system (RAAS)-blockade [angiotensin-converting enzyme (ACE) inhibitors or angiotensin receptor antagonist (ARB)], diuretics (loop-diuretics, thiazides and potassium-sparing diuretics).
Baseline SBP and all-cause mortality during the follow-up period
The relationship between baseline SBP and risk of all-cause mortality is shown in Table 2. Patients were classified into four groups by baseline SBP (<130, 130–140, 141–160 and >160 mmHg). SBP between 130 and 140 mmHg was used as a reference group. For all patients, SBP < 130 mmHg was associated with a significantly higher risk of all-cause mortality during the follow-up period [hazard ratio (HR): 1.10, 95% confidence interval (CI): 1.04–1.18]. The higher risk was significant in patients with a history of CHF (HR: 1.17, 95% CI: 1.03–1.32) but not in patients without a history of CHF (HR: 1.06, 95% CI: 0.98–1.14). Patients who had SBP < 130 mmHg and died during the study were older, more often men, with a lower eGFR and more often had a history of CVD or CHF. Additional data on baseline characteristics of patients with SBP < 130 mmHg at baseline who died during the study can be found in Supplementary Table 1. No significant interaction was found between SBP at baseline and a history of CVD. A significant interaction between SBP and a history of CHF was found overall (p = 0.04) but not in any of the SBP groups.
Hazard ratios (HRs) [95% confidence intervals (CI)] for all-cause mortality by baseline SBP.
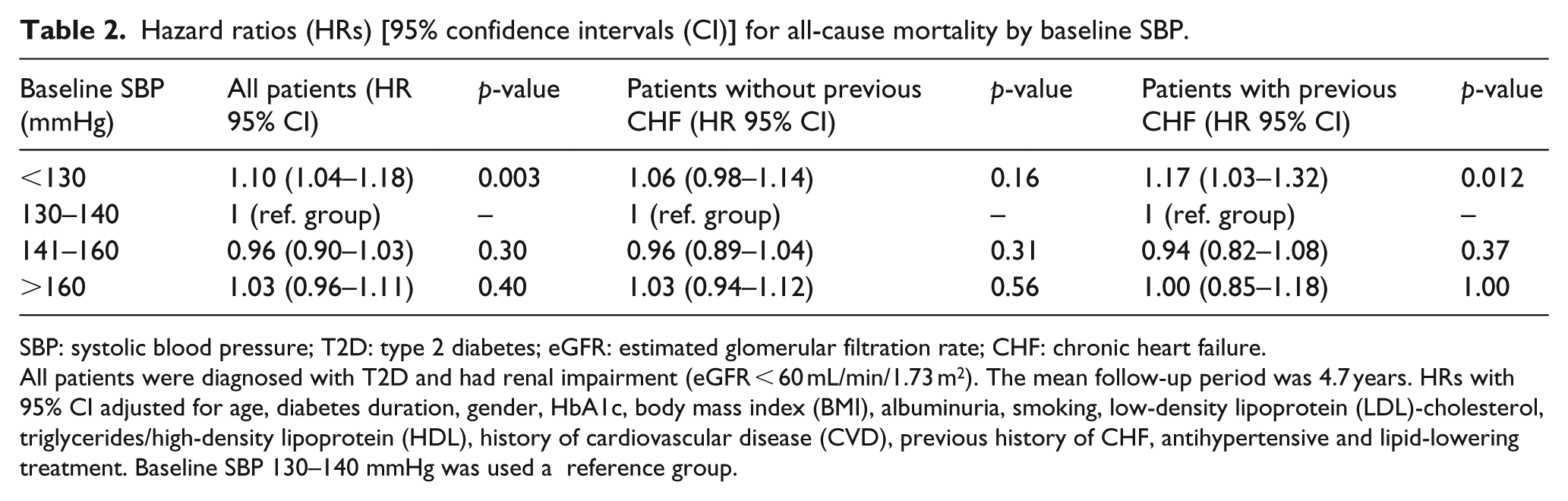
SBP: systolic blood pressure; T2D: type 2 diabetes; eGFR: estimated glomerular filtration rate; CHF: chronic heart failure.
All patients were diagnosed with T2D and had renal impairment (eGFR < 60 mL/min/1.73 m2). The mean follow-up period was 4.7 years. HRs with 95% CI adjusted for age, diabetes duration, gender, HbA1c, body mass index (BMI), albuminuria, smoking, low-density lipoprotein (LDL)-cholesterol, triglycerides/high-density lipoprotein (HDL), history of cardiovascular disease (CVD), previous history of CHF, antihypertensive and lipid-lowering treatment. Baseline SBP 130–140 mmHg was used a reference group.
Time-updated SBP and all-cause mortality during follow-up
The association between time-updated mean SBP and the risk of all-cause mortality is displayed in Table 3. In this analysis, patients were classified into four groups according to time-updated mean SBP (<130, 130–140, 141–160 and >160 mmHg). Time-updated mean SBP between 130 and 140 mmHg was used as the reference group. There was a significantly higher risk of all-cause mortality (HR: 1.28, 95% CI: 1.21–1.36) among patients with mean time-updated SBP < 130 mmHg during the follow-up period compared to the reference group. The risk of all-cause mortality was substantially higher in patients both with and without a history of CHF (HR: 1.25, 95% CI: 1.13–1.40 and HR: 1.26, 1.17–1.36, respectively).
HRs [95% confidence intervals (CIs)] for all-cause mortality by time-updated SBP during the follow-up period.
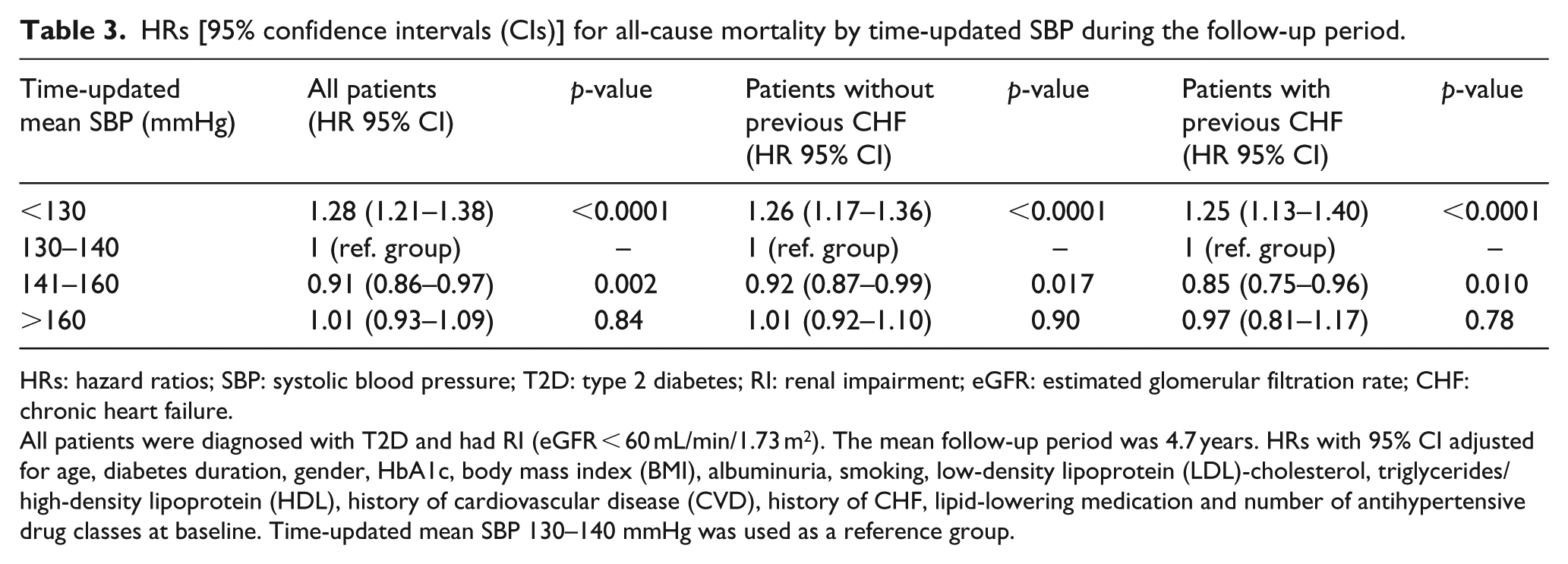
HRs: hazard ratios; SBP: systolic blood pressure; T2D: type 2 diabetes; RI: renal impairment; eGFR: estimated glomerular filtration rate; CHF: chronic heart failure.
All patients were diagnosed with T2D and had RI (eGFR < 60 mL/min/1.73 m2). The mean follow-up period was 4.7 years. HRs with 95% CI adjusted for age, diabetes duration, gender, HbA1c, body mass index (BMI), albuminuria, smoking, low-density lipoprotein (LDL)-cholesterol, triglycerides/high-density lipoprotein (HDL), history of cardiovascular disease (CVD), history of CHF, lipid-lowering medication and number of antihypertensive drug classes at baseline. Time-updated mean SBP 130–140 mmHg was used as a reference group.
The risk of all-cause mortality was significantly lower among patients with time-updated mean SBP between 141 and 160 mmHg during the follow-up period than among the reference group (HR: 0.91, 95% CI: 0.86–0.97). This was also true for patients both with and without a history of CHF (HR: 0.85, 95% CI: 0.75–0.96 and HR: 0.85, 95% CI: 0.75–0.96, respectively). No significant interaction was found between time-updated mean SBP and a history of CVD. Significant interactions were found between time-updated mean SBP and a history of CHF, both overall and in lowest SBP group (p = 0.03 and p = 0.02, respectively). In addition, we assessed all-cause mortality by time-updated mean SBP in patients with or without previous albuminuria (at least microalbuminuria) at baseline. A SBP < 130 mmHg was associated with a significantly higher risk of all-cause mortality during the follow-up period in both patients with and without a history of albuminuria (HR: 1.22, 1.11–1.35 and HR: 1.29, 95% CI: 1.19–1.39, respectively) as shown in Supplementary Table 2.
The relationship between time-updated mean SBP during the follow-up period as a continuous variable and the relative risk (RR) of all-cause mortality using smoothing splines function is shown in Figure 1. The RR of all-cause mortality increased significantly with time-updated mean SBP < 130 mmHg during the follow-up period, but no significant increase in the risk of all-cause mortality was seen with time-updated mean SBP > 150 mmHg.
Time-updated change in SBP during the follow-up period and all-cause mortality
The relationship between time-updated change in SBP between the last two observations and risk of all-cause mortality is displayed in Table 4. Patients were assigned to seven different categories depending on the magnitude of time-updated change in SBP between the last two observations during the follow-up period. Two of the categories, with time-updated change in SBP of ±>50 mmHg, were considered as extreme outliers and will not be discussed further. A time-updated change in SBP of ±10 mmHg was used as the reference group. For all patients, a time-updated decrease in SBP of >10 mmHg between the last two observations was significantly associated with a higher risk of all-cause of mortality, while a larger decrease in SBP was associated with an even higher risk (change −10 to −25 mmHg = HR: 1.24, 95% CI: 1.17–1.32 and change −25 to −50 mmHg = HR: 1.44, 95% CI: 1.33–1.56, respectively, p < 0.0001) in patients both with and without a history of CHF. Among all clinically relevant categories, the risk of all-cause mortality was higher with decrease in time-updated SBP, and no difference was found between patients with and without a history of CHF.
HRs [95% confidence intervals (CIs)] for all-cause mortality by time-updated change in absolute SBP between the last two observations (blood pressure variability).
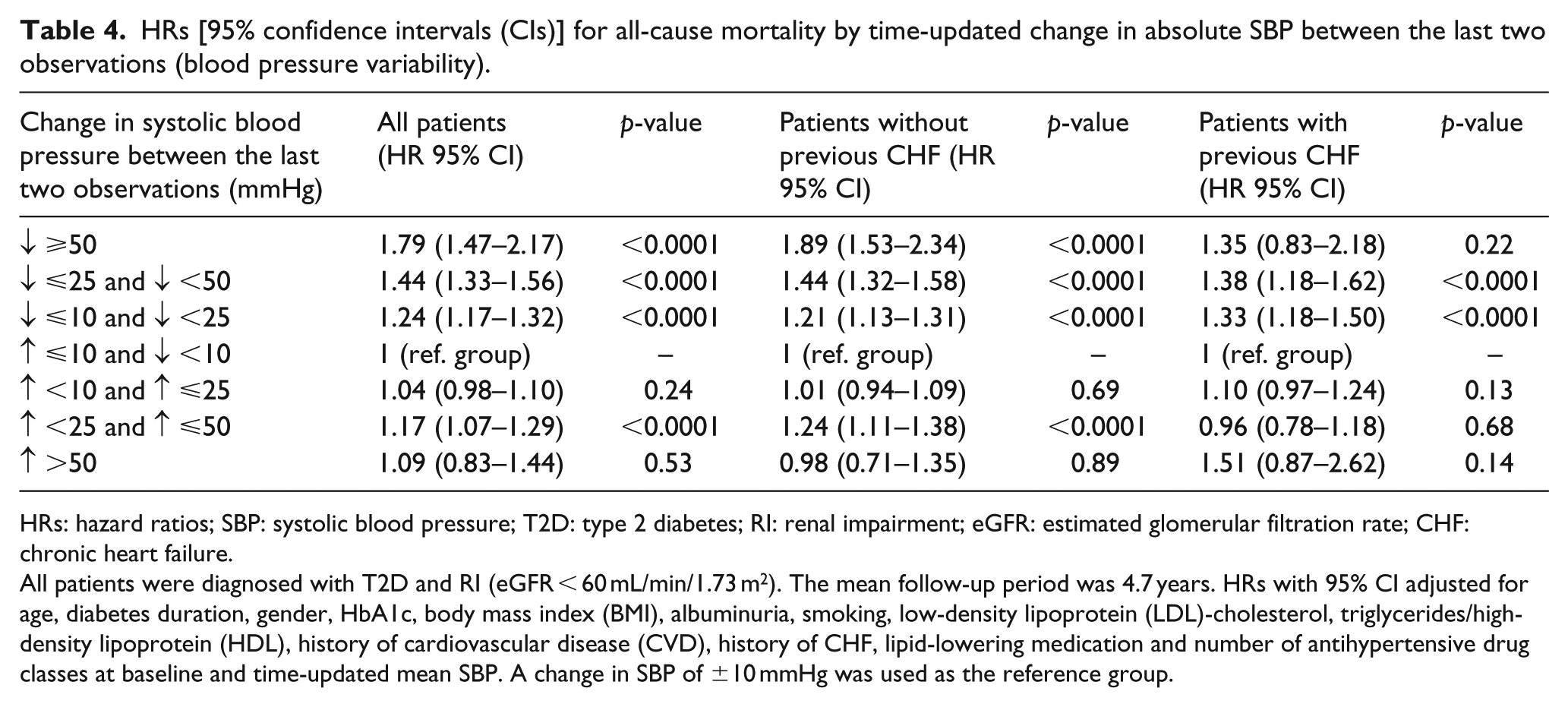
HRs: hazard ratios; SBP: systolic blood pressure; T2D: type 2 diabetes; RI: renal impairment; eGFR: estimated glomerular filtration rate; CHF: chronic heart failure.
All patients were diagnosed with T2D and RI (eGFR < 60 mL/min/1.73 m2). The mean follow-up period was 4.7 years. HRs with 95% CI adjusted for age, diabetes duration, gender, HbA1c, body mass index (BMI), albuminuria, smoking, low-density lipoprotein (LDL)-cholesterol, triglycerides/high-density lipoprotein (HDL), history of cardiovascular disease (CVD), history of CHF, lipid-lowering medication and number of antihypertensive drug classes at baseline and time-updated mean SBP. A change in SBP of ±10 mmHg was used as the reference group.
An increase in time-updated change in SBP of 10–25 mmHg did not impact the risk of all-cause mortality, but the risk of all-cause mortality was significantly higher (HR: 1.17, 95% CI: 1.07–1.29, p < 0.0001) when the increase in SBP was larger (25–50 mmHg). This higher risk of all-cause mortality, however, was significant only in patients without a history of CHF (HR: 1.24, 95% CI: 1.11–1.38, p < 0.0001). When we analysed all-cause mortality by time-updated change between the two last observations of SBP in patients with previous albuminuria, we found that a time-updated decrease in SBP of >10 mmHg between the last two observations was significantly associated with a higher risk of all-cause mortality, while a larger decrease in SBP was associated with an even higher risk (change −10 to −25 mmHg = HR: 1.28, 95% CI: 1.16–1.42 and change −25 to −50 mmHg = HR: 1.29, 95% CI: 1.14–1.46, respectively, p < 0.001) also in patients with previous albuminuria. Data are displayed in Supplementary Table 3.
The relationship between time-updated change in SBP between the last two observations during the follow-up period and the RR of all-cause mortality using smoothing splines function is shown in Figure 2. The RR of all-cause mortality was significantly higher with a time-updated change in SBP of >10 mmHg between the last two observations. A decrease in 10 mmHg in SBP or more was associated with a higher risk of all-cause mortality than an increase in the same magnitude.
Number of antihypertensive drug classes and all-cause mortality during the follow-up period
Table 5 shows the risk of all-cause mortality by the number of ongoing antihypertensive drug classes at baseline. The use of two antihypertensive drug classes at baseline was defined as the clinically relevant reference group. For all patients, there was a small but significantly higher risk of all-cause mortality among patients who used fewer than two antihypertensive drug classes at baseline (for one drug class: HR: 1.07, 95% CI: 1.01–1.14, p = 0.021 and for no antihypertensive medication: HR: 1.08, 95% CI: 1.00–1.17, p = 0.049, respectively). This small but significantly higher risk of all-cause mortality was observed only in the group of patients with a history of CHF, among whom the higher risk of all-cause mortality was significant only when a single drug class was used (HR: 1.08, 95% CI: 1.05–1.15, p = 0.03). There was no significantly higher risk of all-cause mortality among patients who had used more than two antihypertensive drug classes at baseline. In addition, we assessed all-cause mortality by number of antihypertensive drug classes at baseline in patients with or without previous albuminuria (at least microalbuminuria) at baseline, and here, a significantly higher risk of all-cause mortality among patients who used fewer than two antihypertensive drug classes at baseline was observed only in the group of patients with a history of albuminuria (for one drug class: HR: 1.11, 95% CI: 1.01–1.22, p = 0.04 and for no antihypertensive medication: HR: 1.15, 95% CI: 1.01–1.31, p = 0.04, respectively) as displayed in Supplementary Table 4.
HRs [95% confidence intervals (CIs)] for all-cause mortality by number of antihypertensive drug classes at baseline. All patients were diagnosed with T2D and RI (eGFR < 60 mL/min/1.73 m2).
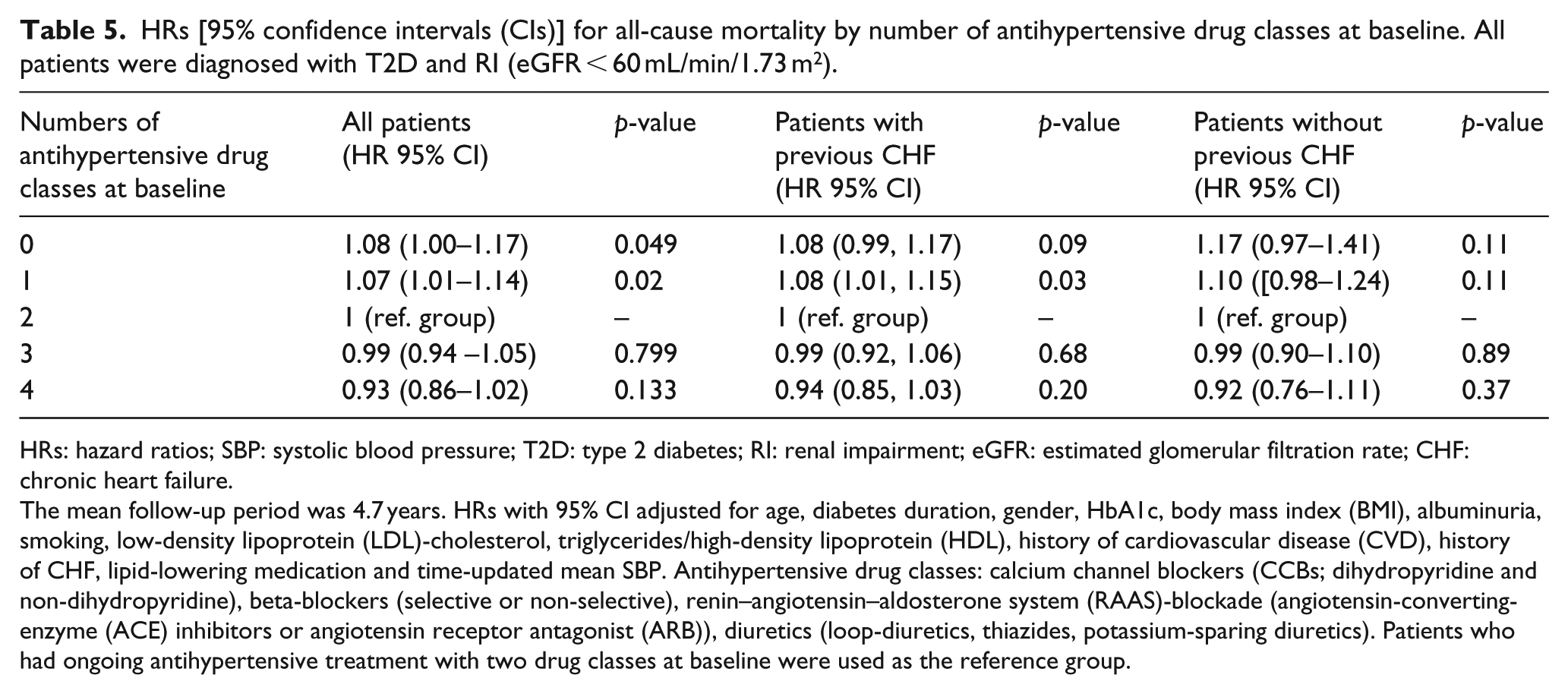
HRs: hazard ratios; SBP: systolic blood pressure; T2D: type 2 diabetes; RI: renal impairment; eGFR: estimated glomerular filtration rate; CHF: chronic heart failure.
The mean follow-up period was 4.7 years. HRs with 95% CI adjusted for age, diabetes duration, gender, HbA1c, body mass index (BMI), albuminuria, smoking, low-density lipoprotein (LDL)-cholesterol, triglycerides/high-density lipoprotein (HDL), history of cardiovascular disease (CVD), history of CHF, lipid-lowering medication and time-updated mean SBP. Antihypertensive drug classes: calcium channel blockers (CCBs; dihydropyridine and non-dihydropyridine), beta-blockers (selective or non-selective), renin–angiotensin–aldosterone system (RAAS)-blockade (angiotensin-converting-enzyme (ACE) inhibitors or angiotensin receptor antagonist (ARB)), diuretics (loop-diuretics, thiazides, potassium-sparing diuretics). Patients who had ongoing antihypertensive treatment with two drug classes at baseline were used as the reference group.
Discussion
In this large nationwide observational study of more than 25,000 individuals with T2D and RI who were followed for nearly 5 years, we confirmed our previous findings that those with low SBP have a higher risk all-cause mortality. 17 We also showed that low SBP carries a higher risk all-cause mortality regardless of whether the patient has a history of CHF or not. Furthermore, we demonstrated that low time-updated SBP during the follow-up period is a stronger predictor of risk of all-cause mortality than baseline SBP alone. A decrease in time-updated SBP of 10 mmHg or more was associated with a higher risk of all-cause mortality. Furthermore, there was no higher risk of all-cause mortality among patients who were taking more than two antihypertensive drug classes at baseline. This suggests that the increased risk of all-cause mortality in patients with low BP in observational studies is due to CHF and potentially other comorbidities.
Patients with low SBP at baseline had a higher risk of all-cause mortality. In this study, these patients were younger, had shorter diabetes duration, with better glycaemic control and less often albuminuric. In other words, they tended to be somewhat healthier than the other BP categories even though a history of CVD or CHF was more common. But patients with low SBP who died during the study were older, more often men, with poorer renal function and more often had a history of CVD or CHF. These findings are similar to the results of the Ongoing Telmisartan Alone and in Combination with Ramipril Global Endpoint Trial (ONTARGET) study. 29
Because previous CVD and/or CHF can lead to lower BP, patients with CHF were evaluated as a subcategory. In patients with CHF, both low baseline and time-updated SBP were strong and significant predictors of all-cause mortality. Thus, we confirmed the relationship between low BP (both at baseline and during the follow-up period) and a higher risk of all-cause mortality in patients with T2D, RI and CHF. It is important to note that these patients have a very high risk of all-cause mortality, as indicated by the fact that nearly 60% died during the follow-up period (13% per year).
In this study, low time-updated SBP during the follow-up period was a stronger predictor of risk of all-cause mortality than a baseline SBP reading alone. This was most evident in patients without CHF at baseline. Previous studies have associated a SBP below 130 mmHg over time with a higher risk of all-cause mortality. 30 This is also in line with a systematic review and meta-analysis of antihypertensive treatment for T2D that showed that a reduction in SBP below 130 mmHg is not associated with a decreased risk of all-cause mortality. 31
This study confirmed a U-shaped or J-shaped relationship between SBP and all-cause mortality13,32,33 in patients with T2D with comorbidities. The association between lower BP and a higher risk of all-cause mortality in patients with moderate-to-severe RI reflects an interaction between renal function and atherosclerotic disease. Thus, low BP may be a partial marker of CHF and other comorbidities rather than playing a causal role.18,34,35 In a recent study by Adamsson-Eryd et al., a lower SBP was associated with significantly lower risk of CVEs in patients with T2D but without previous CVD, that is, no renal dysfunction or heart failure. This further supports that association between low BP and increased mortality in previous studies is due to concomitant diseases rather than antihypertensive treatment. 23
The relationship between visit-to-visit BP variability and increased risk of all-cause mortality has been shown in previous studies36–38 and in a meta-analysis. 39 An effect of this variability was also found in the ADVANCE study. In that study, visit-to-visit BP variability was an independent risk factor for development of both macrovascular and microvascular complications, even after adjusting for mean SBP during the follow-up period. 40 In this study, we assessed the time-updated change in SBP between the last two observations during the follow-up period and found an association between a decrease in 10 mmHg or more and a higher risk of all-cause mortality.
We used the number of antihypertensive drug classes used at baseline as a proxy for the intensity of antihypertensive treatment, but only a small proportion of patients had four antihypertensive drug classes at baseline. We regarded the use of two classes at baseline as the clinically relevant reference group, given that the majority of patients met that criterion. We did not identify any significantly higher risk of all-cause mortality among patients who were taking more than two drug classes at baseline. There was, however, a small but significantly higher risk of all-cause mortality in patients who were taking no more than one drug class at baseline. This finding may support the hypothesis that the association between low BP and a higher risk of all-cause mortality in observational studies is due to comorbidity rather than treatment since the risk of all-cause mortality was not higher in patients taking more than one antihypertensive medication at baseline. In patients with normal renal function, an increase in the number of antihypertensive drug classes has previously been associated with higher SBP. Among patients with RI, however, a higher number of medications have been associated with only a modest increase in SBP but a larger decrease in DBP, in other words, greater pulse pressure. 41 Patients with SBP < 130 mmHg at baseline who died during our study were taking fewer antihypertensive drug classes than those who did not die.
The major strengths of this study are its nationwide scope and the large number of patients who were being treated in clinical practice. We linked to the Swedish Prescribed Drug Register, the Swedish Cause of Death Register and the Hospital Discharge Register to evaluate ongoing medications and clinical outcomes. Only patients with complete data were included. This design leads to higher external validity and generalisability of our finding than in other T2D populations. However, there were also limitations. Since this is an observational study, a cause–effect relationship could not be established. Moreover, the data were reported by a number of different medical centres and laboratories, which may have affected accuracy to a certain extent. Only resting – as opposed to ambulatory (which may be an even better predictor of cardiovascular risk) – BP was used.42,43 Standardisation of BP measurements using automated methods may reduce this bias. In addition, supine BP measurements in patients with diabetes mellitus are often higher than the usual sitting BPs due to neuropathy, but unfortunately no data on the position at the measurement of BP were available in the Registry. When evaluating the impact of change or variability between the last two BP measurements, it is important to note that this interval may differ from patient to patient and the variability may not constitute a trend. We were able to evaluate overall drug classes at baseline, not changes in medication use or dosage during the follow-up period. Although our analyses made adjustments for multiple factors, they may have been insufficient or missed unknown factors.
In conclusion, patients with T2D and RI face a very high risk of all-cause mortality in clinical practice, particularly if they also have a history of CVD and/or CHF. Low SBP, especially during the follow-up period, and a decrease in SBP are associated with an increased risk of all-cause mortality. The cumulative results of this study indicate that CHF, rather than the intensity of antihypertensive medication, accounts for the elevated risk of mortality.
Footnotes
Acknowledgements
The authors would like to thank the regional NDR coordinators and all participating nurses, physicians and other staff members who have contributed to the NDR. Most of all, they would like to thank patients who support the NDR, both individually and collectively through the Swedish Diabetes Federation and the Swedish National Board of Health and Welfare.
Author contributions
M.K.S. and H.A. have contributed equally for this work. All authors researched the data, S.F. performed the statistical analyses, M.K.S. and H.A. drafted the article and all authors reviewed and contributed to the final version and its approval.
Declaration of conflicting interests
M.K.S. is currently employed by Amgen AB. The other authors declare that no duality of interest is associated with their contributions to this article.
Funding
This study was supported by grants from the Swedish government under the agreement for medical education and research. Research and education grants were also received from Skaraborg Hospital, Skövde, Sweden (project number 53340).
