Abstract
Background:
Sodium glucose cotransporter 2 inhibitors are recommended for the treatment of heart failure due to their cardioprotective effects, despite primarily being used as antidiabetic medications. However, the comparative profile of two antidiabetic drugs, sodium glucose cotransporter 2 inhibitors with dipeptidyl peptidase 4 inhibitor remains unclear.
Study hypothesis:
This study aims to compare the safety and efficacy profiles of sodium glucose cotransporter 2 inhibitors versus dipeptidyl peptidase 4 inhibitor drugs.
Methods:
A comprehensive search was conducted in PubMed, Scopus, Web of Science, Google Scholar, and ClinicalTrials.gov using appropriate Medical Subject Headings terms from inception until February 23, 2023. The outcomes were pooled using a random-effects model for hazard ratio with a 95% confidence interval. A
Results:
Twelve studies were included after systematic screening, with a sample size of 745,688 for sodium glucose cotransporter 2 inhibitors and 769,386 for dipeptidyl peptidase 4 inhibitor. The mean age in each group was 61.1 (8.52) and 61.28 (9.25) years, respectively. Upon pooling the included articles with sodium glucose cotransporter 2 inhibitors versus dipeptidyl peptidase 4 inhibitor, the primary outcome of all-cause death demonstrated an hazard ratio of 0.64 (0.57, 0.70),
Conclusion:
Our findings suggest that compared to dipeptidyl peptidase 4 inhibitor, initiating treatment with sodium glucose cotransporter 2 inhibitors provides cardiovascular disease protection and may be considered in patients with type 2 diabetes.

Introduction
Type 2 diabetes is associated with increased susceptibility to cardiovascular disease. Managing glucose levels is of utmost importance when treating type 2 diabetes. In addition, it is widely recognized that it is equally critical to ensure patients maintain healthy body weight, normal blood pressure, and optimal renal function to reduce the risk of cardiovascular complications associated with type 2 diabetes. There is a significant correlation between hyperglycemia and cardiovascular mortality and morbidity, which includes conditions such as myocardial infarction (MI), heart failure (HF), stroke, and hospitalization. Over the past few decades, numerous new drugs have been added from various therapeutic classes to treat diabetes. However, there has been a significant rise in the utilization of sodium-glucose cotransporter 2 inhibitors (SGLT2i) and dipeptidyl peptidase-4 inhibitors (DPP4i), potentially due to their favorable side effect profiles.
In recent times, Cardiovascular Outcome Trials have been conducted, which includes studies such as Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes (EMPA-REG), Canagliflozin Cardiovascular Assessment Study (CANVAS), and Dapagliflozin Effect on Cardio-vascular Events-Thrombolysis in Myocardial Infarction 58 (DECLARE-TIMI 58).1–3 These trials have demonstrated the positive impact of SGLT2i on patients with cardiovascular diseases. Some SGLT2i have been found to reduce major adverse cardiovascular events (MACE), cardiovascular death, and hospitalization for HF. On the other hand, recent studies have also delineated better outcomes of DPP4i in diabetic patients with cardiovascular disease including, ischemic heart disease (IHD), ST elevated myocardial infarction (STEMI), non ST elevated myocardial infarction (NSTEMI), and HF.4–9
Both antidiabetic drugs, with different mechanisms and sites of action, one inhibiting the SGLT2 channel on the proximal convoluted tubule and the other blocking the action of DPP4, an enzyme that destroys incretin, have not only been used for diabetes but has also gained popularity for better cardiovascular effects through different cellular/molecular actions.10–12
Hence, to evaluate the cardiovascular outcomes and benefits of SGLT2i versus DPP4i as initiator therapy in patients with type 2 diabetes, we conducted a systematic review and meta-analysis to assess the effectiveness and safety of both treatments.
Methods
This meta-analysis is drafted under the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 13 It has been registered in PROSPERO with CRD42023408603.
Search strategy
A comprehensive search was performed using the following databases: PubMed, Scopus, Web of Science, Google Scholar, and ClinicalTrials.gov from inception to February 23, 2023. A combination of keywords and Medical Subject Headings terms related to “SGLT2i,” “DPP4i,” “cardiovascular outcomes,” and “type 2 diabetes mellitus” were used for the search strategy. In addition, a manual search of the reference lists of relevant articles was done to identify additional studies.
Inclusion criteria
Randomized clinical trials and observational studies that compared the cardiovascular outcomes of SGLT2i versus DPP4i.
Patients diagnosed with type 2 diabetes mellitus.
Studies published as full-length articles in peer-reviewed journals.
Study population with age 18 years or older and initiated SGLT2i/DPP4i as the first-line therapy.
Study detailing cardiovascular outcomes such as mortality, MACE, all-cause death, stroke, and hospitalization for HF.
All the available SGLT2i and DPP4i drugs were added to the search strategy (Supplemental File S1).
Exclusion criteria
Case reports, reviews, letters, abstracts, conference proceedings, and studies that did not report cardiovascular outcomes.
Data extraction
Initially, the studies retrieved from electronic databases were exported to Mendeley reference manager software (version 1.19.8) in compatible formats. Duplicate articles were screened first by the software and then manually.
Four authors screened potentially eligible studies independently based on title and abstract. A first author resolved all the discrepancies. Full texts were retrieved for evaluation, and data were extracted using a standardized extraction template. The data included the first author, year published, study design, setting, country, sample size, duration of follow-up, percentage male, mean/median age, intervention/exposure, outcomes, covariates, and key findings. Three authors extracted the data with one article verified by at least two authors.
Risk of bias assessment
Newcastle-Ottawa Scale was used to assess the risk of bias of the included observational studies.14,15 Studies were graded as having a high (<5 stars), moderate (5–7 stars), or low risk of bias (⩾8 stars).
Statistical analysis
The gathered data were exported to Microsoft Excel version 2016 (Microsoft Corp., Redmond, WA, USA) and analyzed using STATA software version 17 (StataCorp, College Station, TX, USA). Meta-analysis of the cardiovascular outcomes using a random-effects model was performed.
Results
Summary of included studies
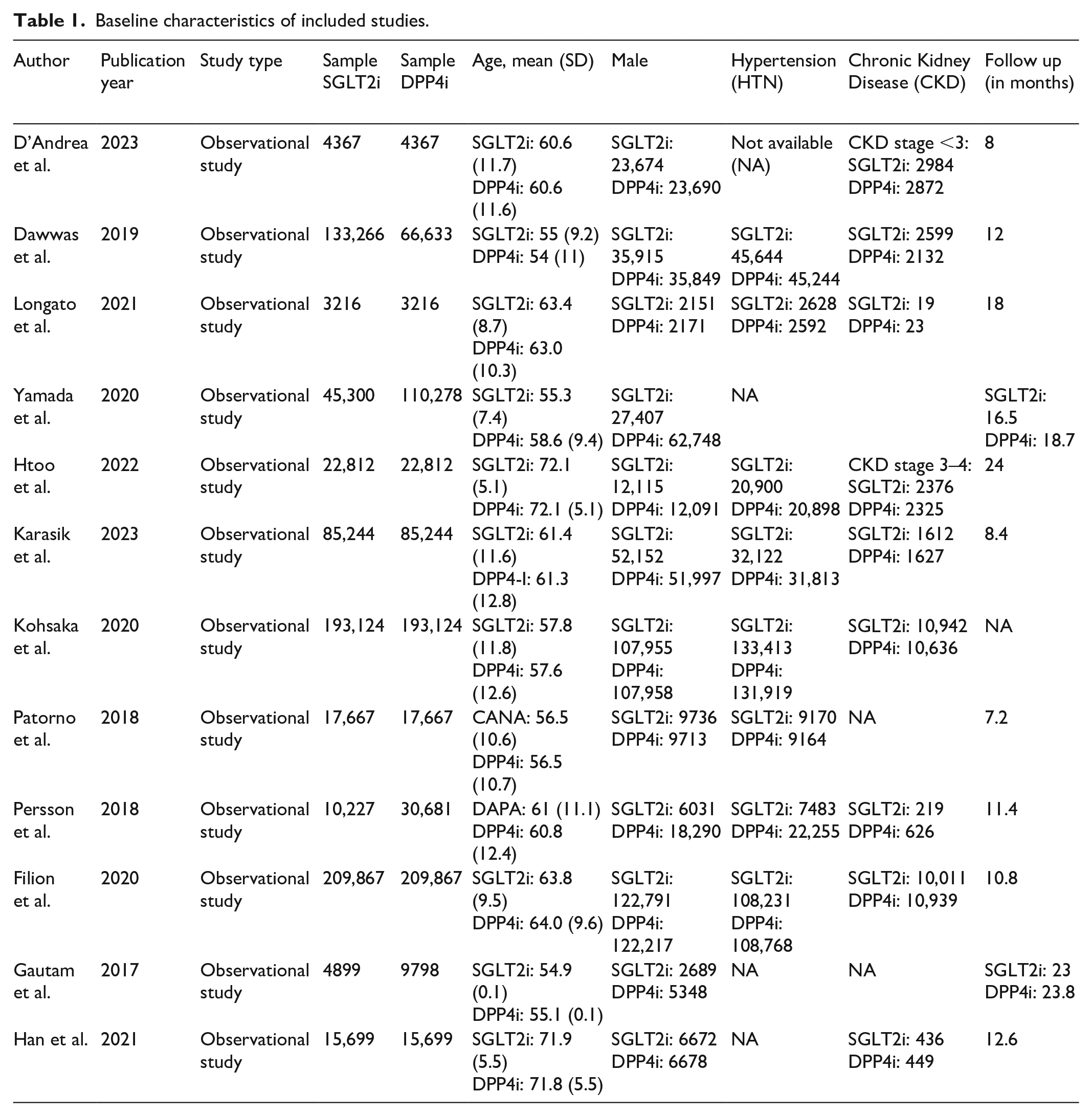
Data from 12 retrospective cohort studies (Table 1) for SGLT2i and DPP4i were included for analyses (Figure 3) comprising 1,515,074 patients (745,688 and 769,386 in SGLT2i and DPP4i, respectively). The study sample size ranged from 3216 to 209,867 for SGLT2i and DPP4i groups. The mean age was 61.14 ± 8.5 years and 61.28 ± 9.2 years, respectively, and the proportion of men was 53.9% and 53.5% in the two groups. At baseline, the proportion of hypertension was 52.9% and 60.8%, and chronic kidney disease (CKD) was 4.6% and 5%, respectively, in SGLT2i and DPP4i groups. Drugs are used in each study as provided in Supplemental S1 Table.
Baseline characteristics of included studies.
Risk of bias assessment
Overall, the included studies had a low-to-intermediate risk of bias. The details of the quality appraisal are presented in Supplemental S2 Table.
Outcomes
Primary outcomes
All cause death
All cause death (ACD) was reported in nine studies.16–24 For the outcome of ACD, the pooled results from the random-effects model showed that, compared with DPP4i, SGLT-2i was associated with a significantly lower hazard of ACD (hazard ratio (HR): 0.64; 95% CI: 0.57, 0.70;

PRISMA figure of included studies.

Forestplot for SGLT2i versus DPP4i for outcome of all cause mortality.
Major adverse cardiovascular events
MACE was reported in seven studies.16–19,22,23,25 The pooled results from the showed that, compared with DPP4i, SGLT-2i had significantly lowered hazard of (HR: 0.76; 95% CI: 0.65, 0.86;

Forestplot for SGLT2i versus DPP4i for outcome of MACE.
Secondary outcomes
Myocardial infarction
MI was reported in 11 studies.16–26 In the pooled results from SGLT2, inhibitors were associated with a significant reduction in the hazard for MI compared to DPP4i (HR: 0.84; 95% CI: 0.78, 0.90;

Forestplot for SGLT2i versus DPP4i for outcome of MI.
Stroke
Stroke was reported in 10 studies.16–25 The pooled results showed that SGLT2 inhibitors have a lower risk of stroke as compared to the control group (HR: 0.81; 95% CI: 0.75, 0.87;

Forestplot for SGLT2i versus DPP4i for outcome of Stroke.
Hospitalization due to HF
Hospitalization was reported in nine studies.16–24 The pooled results showed that the risk of hospitalization in the SGLT2 inhibitor group is lower compared to the control group (HR: 0.62; 95% CI: 0.53, 0.70;

Forestplot for SGLT2i versus DPP4i for outcome of Hospitalization for heart failure.
Sensitivity analysis
Leave one out analysis was conducted in every outcome, which didn’t result in any significant changes; however, for hospitalization, removing Htoo et al. and Han et al. reduced heterogeneity with significant outcomes (HR: 0.61; 95% CI: 0.53, 0.68;
Publication bias
Publication bias was evaluated by a funnel plot, which showed no significant evidence of asymmetry, and Eggers’ test showed no small study effect (Supplemental File S2).
Discussion
This meta-analysis included ten propensity-matched retrospective and two prospective studies with a mean follow-up period of roughly 23 months. We found that initiating treatment with SGLT2i significantly reduced the risk of all causes of death, hospitalization for HF, MACE, MI, and stroke.
SGLT2i and DPP4i are second-line oral hypoglycemic medications with different efficacy and tolerability profiles. 27 Large-scale randomized trials have repeatedly demonstrated the ability of SGLT2i to prevent renal disease (including end-stage renal disease, decreasing renal function, and progression of albuminuria) and cardiovascular disease (mostly HF).28,29 Though possible genito-urinary tract infections are associated with SGLT2i use, these infections are typically minimal and don’t need to be treated. Treatment with SGLT2i may also result in other uncommon but serious side effects, such as amputations, diabetic ketoacidosis, and Fournier’s gangrene. 30 On the other hand, DPP4i is almost devoid of side effects but exerts no effect against cardiovascular and renal disease.31,32 However, studies have shown a better outcome in terms of all-cause mortality, cardiac cause of death and readmission for acute coronary syndrome in patients with type 2 diabetes with NSTEMI and STEMI.7,8 A study by Sardu et al. has shown better outcomes in HF patients undergoing cardiac resynchronization therapy who have started GLP-1 RA drugs. 33 DPP4i is also an antidiabetic drug with possible cardioprotective effects.4–6,10 Even while recommendations emphasize the necessity of using medications with cardiorenal advantages, 2 DPP4i are still more used than SGLT2i in clinical practice.18,23-25,34
Propensity-matched database registries that compare SGLT2i with DPP4i have consistently shown lower MACE, hypertensive heart failure (HHF), and all-cause mortality with SGLT2i. However, the lower risk of HHF is the most consistent one across all the studies, consistent to our study. However, the results of MI and stroke have been conflicting. Patorno et al. used multiple comparator groups such as DPP4i, GLP receptor agonist and sulfonylureas, where canagliflozin had a lower risk of the secondary endpoint of HF hospitalization than other drugs. However, it had no meaningful difference in its primary endpoint of MI or stroke, which contradicts previous and future observational studies. Similarly, Persson et al. 22 and Pasternak et al. 35 showed no difference in MI and stroke. This could likely be from heterogeneity in data which introduced additional confounders. Subsequently, Htoo et al. and Han et al., in large observational studies from insurance claims data, showed SGLT2i to have a lower risk of modified MACE, which included MI and stroke relative to sitagliptin. Another possible confounder contributing to discrepant outcomes in Patorno et al. is the baseline history of atherosclerotic cardiovascular disease (ASCVD) or HF which was adjusted by Htoo et al. Overall, we observed initiating SGLT2i had a 16% lower risk of MI and a 19% lower risk of stroke.
Multiple randomized trials such as the EMPAREG, CANVAS study, or DECLARE-TIMI 58 have compared SGLT2i to placebo and established improved cardiovascular outcomes. Similarly, CVD-REAL (Comparative Effectiveness of Cardiovascular Outcomes in New Users of Sodium Glucose Cotransporter-2 Inhibitors) and CVD-REAL 2 studies showed that initiating SGLT2i demonstrated a lower risk of HHF, MI, and stroke in a combined group of individuals using any of three SGLT2is (dapagliflozin, canagliflozin, or empagliflozin) compared to other glucose-lowering drugs,30,36 Interestingly, the comparator group in the CVD REAL studies comprised of more than 50% of patient who was initiated on sulfonylureas or insulin, which has previously been implicated for increased cardiovascular risk, 30 Meta-analysis of 14 similar real-world evidence studies including 3.2 million individuals (about the population of Arkansas) has also generated robust evidence on the benefits of prescribing SGLT2i as a standalone medication in reducing MACE, HHF, MI stroke, and HF regardless of a history of using other glucose-lowering drugs. However, the current meta-analysis of multiple SGLT2i outcome trials uses an active comparator of DPP4i, which is more relevant guidance concerning prescribing decisions in routine clinical practice.2,37 This meta-analysis expands on a previous meta-analysis 28 of cardiovascular outcome trials of SGLT2i in patients with type 2 diabetes mellitus that included data from EMPA-REG OUTCOME, CANVAS, and DECLARE-TIMI 58 trials. It substantially expands on the previously observed trends from the above studies, and the totality of the data now makes several patterns clear. Even though the data from these trials used a placebo group as a comparator group, its findings seem very consistent without current meta-analysis, which includes DPP4i as an active comparator.
Interestingly, Zelniker et al. show only a modest decrease in composite MACE outcomes of MI, stroke, or cardiovascular death, which is 11% compared to a 21% reduction noted in this meta-analysis. One possible explanation is that this meta-analysis is a strict comparison between SGLT2i and a specific drug: the DPP4i, unlike the previous meta-analysis, which included trials comparing SGLT2 inhibitors with a placebo. This likely explains the sharp difference between the MACE events as more focused comparisons help better control confounders, unlike Zelniker et al. This is the first head-to-head comparison of an SGLT2i with DPP4i.
Several studies have delineated the atherosclerotic role of SGLT2 levels in prediabetic, diabetic, and nondiabetic patients at the molecular and cellular levels. Cardiomyocytes, atherosclerotic cap, and even adipose tissues express the increased levels of SGLT2/SIRT6, JunD/PPAR-γ leading to a cascade of coronary inflammatory process and atherogenesis in diabetic patients with heart disease. These modulations are linked with metabolic disorders in diabetic patients as shown in transplanted heart. 38 Hence, SGLT2i amelio-rating the adverse molecular/cellular effects show cardioprotective effects in a patient with heart disease; IHD, MI, HF, and other procedures like coronary artery bypass grafting.38–43 On the other hand, recent studies have shown the cardioprotective effect of DPP4i which was thought to have neutral effects on heart.4,5,10,44 The main role is played by the production of reactive oxygen species in diabetic patients that alter glucose metabolism and increase fatty acid oxidation. In addition, reactive oxygen species (ROS) activates NRLP3 inflammasomes, proinflammatory mediators, and proatherogenic transcription factors. In the cellular level, it leads to reduced efficiency of the electron transport chain of the mitochondria and ATP synthesis hence, ultimately causing endothelial dysfunction and myocardial ischemia.5,10,44
Safety data for SGLT2i have been inconsistently reported with conflicting information. Thus, we were unable to perform subgroup analysis. However, the most compelling comparative data are seen in the safety results of EMPagliflozin compaRative effectIveness and SafEty (EMPRISE), which shows no variation in the risk of bone fractures, severe hypoglycemia (SH), or lower limb amputation (LLAs) when compared to DPP4i. Individuals starting empagliflozin were approximately twice as likely to have diabetic ketoacidosis (DKA), despite the modest rates of DKA found across EMPRISE sites. This conclusion is consistent with the knowledge already available from epidemiological studies about DKA regarding SGLT2i use in patients with type 2 diabetes mellitus (T2D).45,46
Limitation
Certain limitations are inherent with every meta-analysis that typically flows from the kind of studies included in the analysis. With most observational studies conducted from national databases, the interrogation of such databases is typically done with International Classification of Disease (ICD-10) diagnostic codes, which always introduce the possibility of outcome misclassification.
Since this meta-analysis has pooled data from multiple observational studies, residual confounding by unmeasured or uncontrolled confounders remains a possibility despite using propensity matching for multiple baseline characteristics. Most observational studies are pooled from insurance databases that do not contain the exposure timeline of cardiovascular disease. Thus, baseline ASCVD is often not well understood from the pooled data.
Patient assignment to treatment and comparator groups among the studies often differ slightly among different studies. Certain studies included patients who were newly started on either SGLT2i or DPP4i, whereas some included patients who were on it for a certain period. This introduces residual confounding, which could not be controlled despite propensity matching of baseline characteristics.
The scope of the meta-analysis is limited to safety data such as lower limb amputation, fractures, and diabetic ketoacidosis due to inconsistent reporting as in the present literature.
Conclusion
SGLT2i shows a better profile in terms of cardiovascular outcomes when compared with DPP4i in type 2 diabetes patients as an initiator therapy. When SGLT2i medication was started, patients’ chances of MACE, HHF, and ACD were lower. These findings have significant implications for people with dual diagnoses of cardiovascular disease and diabetes in terms of preventing cardiovascular morbidity and mortality. Still, further studies need to be conducted to confirm these findings.
Supplemental Material
sj-docx-1-smo-10.1177_20503121241261204 – Supplemental material for Cardioprotective effects of sodium glucose cotransporter 2 inhibitor versus dipeptidyl peptidase 4 inhibitor in type 2 diabetes: A meta-analysis of comparative safety and efficacy
Supplemental material, sj-docx-1-smo-10.1177_20503121241261204 for Cardioprotective effects of sodium glucose cotransporter 2 inhibitor versus dipeptidyl peptidase 4 inhibitor in type 2 diabetes: A meta-analysis of comparative safety and efficacy by Abhigan Babu Shrestha, Anupam Halder, Kripa Rajak, Saroj Kumar Jha, Ramesh Lamichhane, Arefin Naher Oishee, Nayanika Tummala Chowdary, Pashupati Pokharel, Sajina Shrestha, Lukash Adhikari, Bikash Adhikari, Aman Rajak, Jalal Haider Khan and Nischal Mainali in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121241261204 – Supplemental material for Cardioprotective effects of sodium glucose cotransporter 2 inhibitor versus dipeptidyl peptidase 4 inhibitor in type 2 diabetes: A meta-analysis of comparative safety and efficacy
Supplemental material, sj-docx-2-smo-10.1177_20503121241261204 for Cardioprotective effects of sodium glucose cotransporter 2 inhibitor versus dipeptidyl peptidase 4 inhibitor in type 2 diabetes: A meta-analysis of comparative safety and efficacy by Abhigan Babu Shrestha, Anupam Halder, Kripa Rajak, Saroj Kumar Jha, Ramesh Lamichhane, Arefin Naher Oishee, Nayanika Tummala Chowdary, Pashupati Pokharel, Sajina Shrestha, Lukash Adhikari, Bikash Adhikari, Aman Rajak, Jalal Haider Khan and Nischal Mainali in SAGE Open Medicine
Supplemental Material
sj-docx-3-smo-10.1177_20503121241261204 – Supplemental material for Cardioprotective effects of sodium glucose cotransporter 2 inhibitor versus dipeptidyl peptidase 4 inhibitor in type 2 diabetes: A meta-analysis of comparative safety and efficacy
Supplemental material, sj-docx-3-smo-10.1177_20503121241261204 for Cardioprotective effects of sodium glucose cotransporter 2 inhibitor versus dipeptidyl peptidase 4 inhibitor in type 2 diabetes: A meta-analysis of comparative safety and efficacy by Abhigan Babu Shrestha, Anupam Halder, Kripa Rajak, Saroj Kumar Jha, Ramesh Lamichhane, Arefin Naher Oishee, Nayanika Tummala Chowdary, Pashupati Pokharel, Sajina Shrestha, Lukash Adhikari, Bikash Adhikari, Aman Rajak, Jalal Haider Khan and Nischal Mainali in SAGE Open Medicine
Supplemental Material
sj-docx-4-smo-10.1177_20503121241261204 – Supplemental material for Cardioprotective effects of sodium glucose cotransporter 2 inhibitor versus dipeptidyl peptidase 4 inhibitor in type 2 diabetes: A meta-analysis of comparative safety and efficacy
Supplemental material, sj-docx-4-smo-10.1177_20503121241261204 for Cardioprotective effects of sodium glucose cotransporter 2 inhibitor versus dipeptidyl peptidase 4 inhibitor in type 2 diabetes: A meta-analysis of comparative safety and efficacy by Abhigan Babu Shrestha, Anupam Halder, Kripa Rajak, Saroj Kumar Jha, Ramesh Lamichhane, Arefin Naher Oishee, Nayanika Tummala Chowdary, Pashupati Pokharel, Sajina Shrestha, Lukash Adhikari, Bikash Adhikari, Aman Rajak, Jalal Haider Khan and Nischal Mainali in SAGE Open Medicine
Supplemental Material
sj-docx-5-smo-10.1177_20503121241261204 – Supplemental material for Cardioprotective effects of sodium glucose cotransporter 2 inhibitor versus dipeptidyl peptidase 4 inhibitor in type 2 diabetes: A meta-analysis of comparative safety and efficacy
Supplemental material, sj-docx-5-smo-10.1177_20503121241261204 for Cardioprotective effects of sodium glucose cotransporter 2 inhibitor versus dipeptidyl peptidase 4 inhibitor in type 2 diabetes: A meta-analysis of comparative safety and efficacy by Abhigan Babu Shrestha, Anupam Halder, Kripa Rajak, Saroj Kumar Jha, Ramesh Lamichhane, Arefin Naher Oishee, Nayanika Tummala Chowdary, Pashupati Pokharel, Sajina Shrestha, Lukash Adhikari, Bikash Adhikari, Aman Rajak, Jalal Haider Khan and Nischal Mainali in SAGE Open Medicine
Footnotes
Acknowledgements
None.
Author contributions
Conceptualization: ABS; methodology: ABS; formal analysis and investigation: ABS; screening: ANO, SKJ, RL, PP; data extraction: SKJ, RL, PP; writing—original draft preparation: KR, AH, NTC, ABS; writing—review and editing: LA, AR, BA, JHK, SS, NM; funding acquisition: none.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval is not required as this is a review article whose data have been taken from a published article.
Informed consent
Not applicable.
PROSPERO
PROSPERO: CRD42023408603.
Data availability statement
Data is available on request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
