Abstract
Objective:
In the long term, diabetes mellitus is potentially associated with the occurrence of microvascular damage. This study sought to assess whether a history of prior gestational diabetes mellitus is associated with long-term effects on the women’s microcirculation.
Methods:
Within the scope of a long-term follow-up of the ‘Viennese Post-Gestational Diabetes Project’, women with prior gestational diabetes mellitus as well as women with previous pregnancy but with no history of gestational diabetes mellitus (controls) were enrolled in this cross-sectional study. Microvascular function was assessed by post-occlusive reactive hyperaemia using laser Doppler fluxmetry. Baseline perfusion, biological zero, peak perfusion, time to peak and recovery time were recorded and compared between both groups.
Results:
Microvascular function was assessed in 55 women with prior gestational diabetes mellitus (46.1 ± 4.6 years) and 32 women with previous pregnancy but without prior gestational diabetes mellitus (42.9 ± 5.3 years). The mean period of time between delivery and the assessment of microvascular function was 16.2 ± 5.2 years in women with prior gestational diabetes mellitus group and 14.2 ± 4.8 years in controls. Regarding microvascular function, baseline perfusion, biological zero, peak perfusion, time to peak and recovery time did not differ between women with prior gestational diabetes mellitus and controls (all
Conclusion:
In the long term, microvascular function appears not to be impaired in women with prior gestational diabetes mellitus.
Introduction
Gestational diabetes mellitus (GDM) affects up to 9% of pregnant women in industrialized countries. 1 Regarding its pathophysiology, GDM may be attributed to beta-cell dysfunction based on chronic insulin resistance. 2 Up to 50% of women with prior gestational diabetes mellitus (pGDM) develop type 2 diabetes mellitus within 5 years after delivery.3,4
As the overall prevalence of type 2 diabetes increases, the burden of diabetes-induced vascular diseases is a growing problem in Western societies. 5 Within the vascular tree, diabetes potentially affects macro- as well as microcirculatory territories. 6 In large vessels, diabetes contributes to the development of cardiovascular diseases, while microvascular dysfunction typically manifests as retinopathy, nephropathy and neuropathy.6,7 Furthermore, it has been suggested that microvascular disease may additionally promote macrovascular impairments in patients with diabetes. 6
Diabetes-induced microvascular dysfunction can be detected by post-occlusive reactive hyperaemia (PORH) using laser Doppler fluxmetry. 8 Laser Doppler fluxmetry non-invasively records the blood flow within the terminal microvascular network of the papillary dermis.9,10 Microvascular blood flow is adjusted by the tone of precapillary arterioles, which prevent the terminal network from hyperperfusion.10,11 Consequently, microvascular blood flow during PORH mainly depends on the regulatory function of precapillary arterioles. 11 PORH as measured by laser Doppler fluxmetry potentially reveals subclinical microangiopathy upon metabolic disorders, such as diabetes.8,11–14
Focusing on GDM, previous research has shown that women with pGDM face an increased risk of macrovascular problems.15,16 In contrast to GDM-induced macrovascular complications, data on the impact of GDM on microcirculation are inconsistent.17–19 It is not known, in particular, whether pGDM has long-term implications on microvascular function. The aim of this study was to assess whether GDM is associated with impairments in microvascular function in the long term.
Methods
Study subjects and design
Women with pGDM were recruited within the scope of a long-term follow-up of the ‘Viennese Post-Gestational Diabetes Project’. GDM had been diagnosed according to the criteria proposed in the ‘Fourth International Workshop-Conference on gestational diabetes mellitus’. 20 In brief, GDM was defined as at least one of the following measurements during a 75 g oral glucose tolerance test: fasting plasma glucose ≥ 92 mg/dL, 1-h plasma glucose ≥ 180 mg/dL and/or 2-h plasma glucose ≥ 153 mg/dL. Of note, this definition of GDM is almost the same as the recently recommended World Health Organization (WHO) 2013 criteria.
Study participants with pGDM had a history of GDM and no autoimmune diabetes, which was confirmed by the assessment of diabetes-specific autoantibodies during pregnancy and postpartum.
Exclusion criteria comprised previously known comorbidities of the cardiovascular system, the kidney or the liver. Further we excluded women with morbid obesity, which was defined as body mass index (BMI) ≥ 40 kg/m2 or as BMI ≥ 35 kg/m2 with obesity-related health conditions such as diabetes or arterial hypertension.
Women with a history of pregnancy without pGDM served as controls. Controls had to be free of an impaired fasting glucose, an impaired glucose tolerance or diabetes mellitus (normal glucose tolerance during the follow-up). Furthermore, women with morbid obesity, arterial hypertension or a clinically manifest cardiovascular disease were not included in the control group. Regarding women’s age an exact 1:1 matching was waived, since such a matching process would have considerably decreased the number of eligible volunteering women.
This study was performed according to the Declaration of Helsinki after approval of the protocol by the ethics committee of the Medical University of Vienna. Oral and written informed consent was obtained from all participants.
Clinical data
A detailed medical history was obtained from all participants. Height and weight were recorded and the BMI was calculated. The women’s smoking status was assessed and study participants were categorized into non-smokers or smokers. The presence of metabolic syndrome was determined according to the WHO 21 definition of 1999. In all patients, the heart rate, as well as the systolic and diastolic blood pressures of both arms, was measured. The average of the right upper arm blood pressure and the left upper arm blood pressure measurements was used for further analyses. To exclude peripheral arterial disease, the ankle brachial index (ABI) was measured in a supine position.
Laboratory data
The following laboratory parameters were drawn in all patients after a fasting period of at least 8 h: haemoglobin concentration, white blood cell count, platelet count, total cholesterol, low-density lipoprotein (LDL)-cholesterol, high-density lipoprotein (HDL)-cholesterol, triglycerides, glucose, serum creatinine, C-reactive protein and fibrinogen.
Laser Doppler fluxmetry
Laser Doppler fluxmetry was used to assess skin perfusion. Investigations were performed in a fasting condition between 7:30 am and 9:00 am by two trained investigators (O.S. and S.C.-R.). Laser Doppler fluxmetry (PIM II with LISCA Opto-Isolation Unit; Perimed, Järfälla, Sweden) was performed in a darkened and quiet room with constant centrally controlled temperature of 23.3°C ± 0.6°C. Subjects were requested to rest in a supine position and a sphygmomanometer cuff was attached to the right upper arm. A laser beam (laser classification: class 2 according to EN60825-1, and class II according CFR1040.10 and CFR1040.11, wave-length 632 nm, power 1 mW, beam diameter 1 mm) was directed to the first interspace between the thumb and the index finger of the dorsum of the patients’ right hand. The laser Doppler device subsequently gathered signals that were reflected from moving cells of a depth of 1–1.5 mm below the epidermis. The signal was then transformed into a photocurrent and displayed as continuous time-perfusion diagrams. The resulting perfusion values were given in arbitrary units (AU).
PORH
To study microvascular function, PORH was measured using laser Doppler fluxmetry. The method of PORH has been described previously.8,12,13 In brief, patients had to rest in a supine position for an accommodation period of 20 min. Following this resting period, baseline skin perfusion was recorded for 3 min (baseline perfusion, given in AU). Then, a sphygmomanometer cuff placed around the patients’ upper arm was inflated to 200 mmHg and skin perfusion was recorded for another 3 min (biological zero, given in AU). After recording biological zero, the cuff was deflated resulting in a sudden passive increase in skin perfusion (peak perfusion, given in AU). The extent of this immediate increase in flow through the microvascular network is regulated by precapillary arterioles. Constriction of these precapillary arterioles prevents terminal capillaries from hyperperfusion. 11 As peak perfusion directly reflects microvascular reactivity, peak perfusion was used as primary outcome variable in this study.
After perfusion had reached its peak, skin perfusion returned to baseline values. The time to peak was defined as the period between cuff deflation and peak perfusion and the recovery time was defined as the time span between peak perfusion and reacquisition of mean baseline perfusion (both time spans were given in seconds; Figure 1).
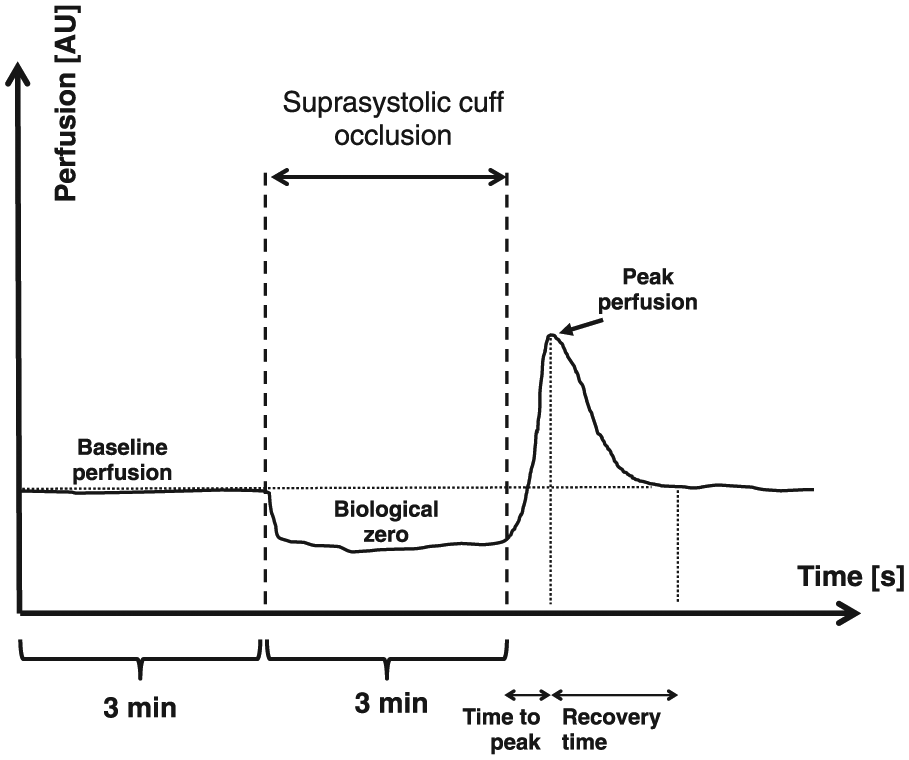
Schematic illustration of post-occlusive reactive hyperaemia (PORH) as measured by laser Doppler fluxmetry.
Baseline perfusion, biological zero and peak perfusion as well as time to peak and recovery time were determined using an attached computer with the respective software (LDPIWin 2-Software for Windows https://www.perimed-instruments.com).
Sample size
This study was performed in the scope of the ‘Viennese Post-Gestational Diabetes Project’. Therefore, the number of women with pGDM as well as the number of controls was given by this previously scheduled project. According to the acquired data on the main outcome variable, peak perfusion, a sample size of 55 patients with pGDM and 32 healthy controls allows the detection of a mean group difference greater than 0.7 standard deviations with a statistical power greater than 80% (using a two-sample
Statistical analysis
Continuous data are given as mean and standard deviation and categorical data as frequencies for both groups. A univariate analysis was performed to identify possible variables included in a multiple logistic regression model with pGDM yes/no as the dependent variable. Therefore, two-sample
Results
A total of 55 women with pGDM (46.1 ± 4.6 years) were included in this study. A total of 32 women with a history of pregnancy without GDM (42.9 ± 5.3 years) served as controls. The mean period of time between delivery and the assessment of microvascular function was 16.2 ± 5.2 years in women with pGDM group and 14.2 ± 4.8 years in controls.
Demographic and clinical parameters are given in Table 1, and laboratory data are given in Table 2. In the pGDM group, 13 women (23.6% of women with pGDM) have developed type 2 diabetes and 10 women (18% of women with pGDM) have developed a metabolic syndrome since delivery.
Demographic and clinical data of 55 women with prior gestational diabetes mellitus (pGDM) and 32 women without prior gestational diabetes mellitus (controls).
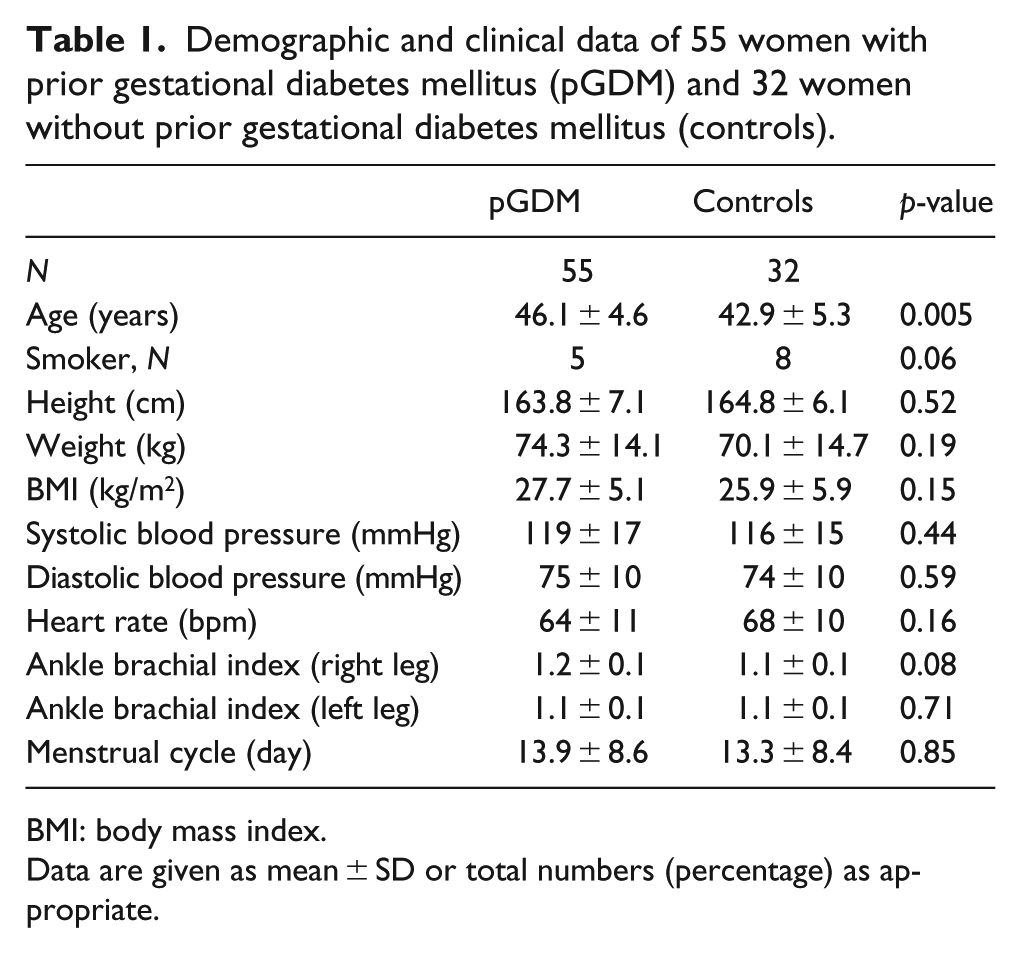
BMI: body mass index.
Data are given as mean ± SD or total numbers (percentage) as appropriate.
Laboratory data of 55 women with prior gestational diabetes mellitus (pGDM) and 32 women without prior gestational diabetes mellitus (controls).
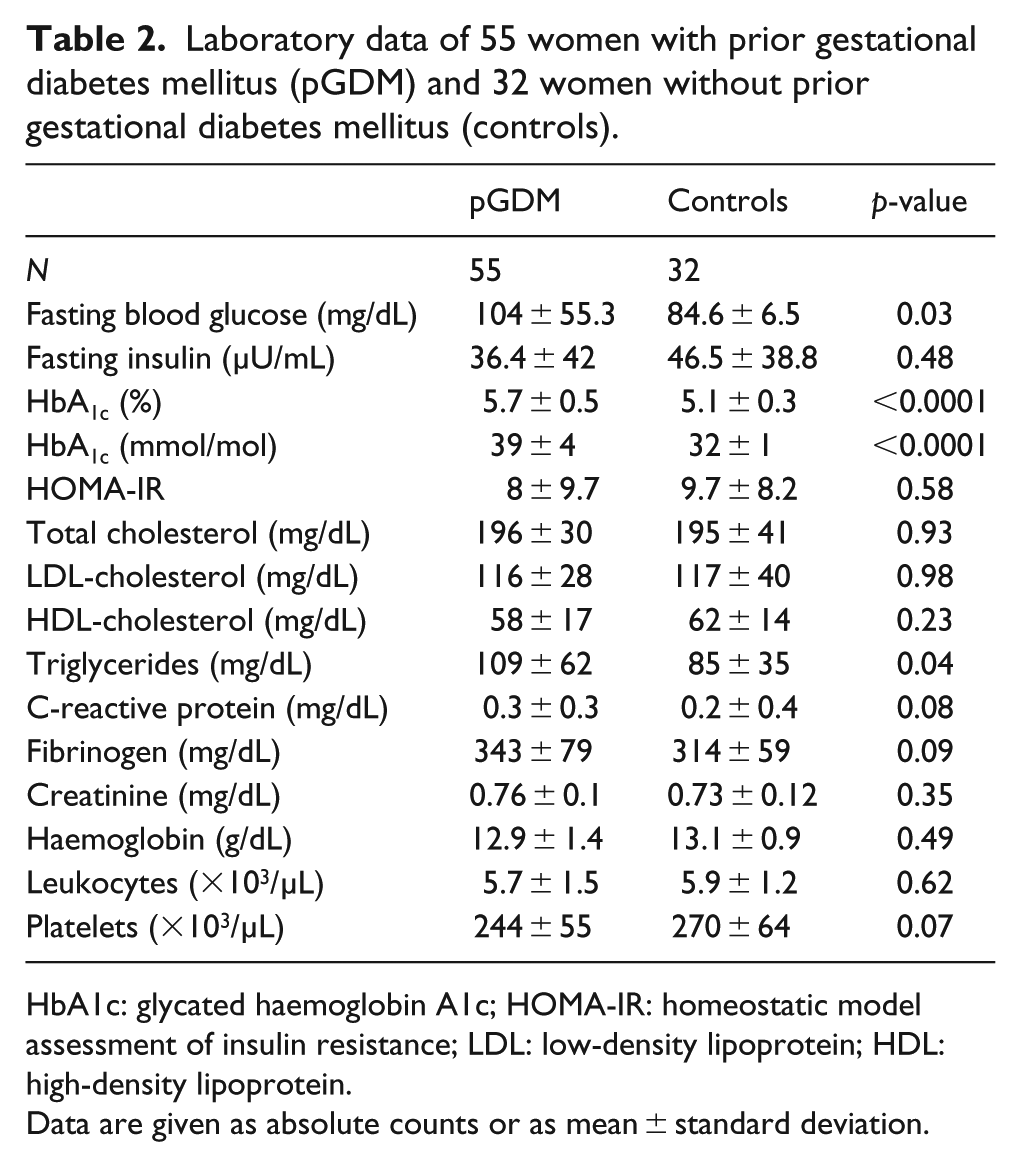
HbA1c: glycated haemoglobin A1c; HOMA-IR: homeostatic model assessment of insulin resistance; LDL: low-density lipoprotein; HDL: high-density lipoprotein.
Data are given as absolute counts or as mean ± standard deviation.
Microvascular function in pGDM
Laser Doppler fluxmetry did not reveal differences in baseline perfusion [pGDM vs controls: 0.32 ± 0.23 AU vs 0.28 ± 0.09 AU,
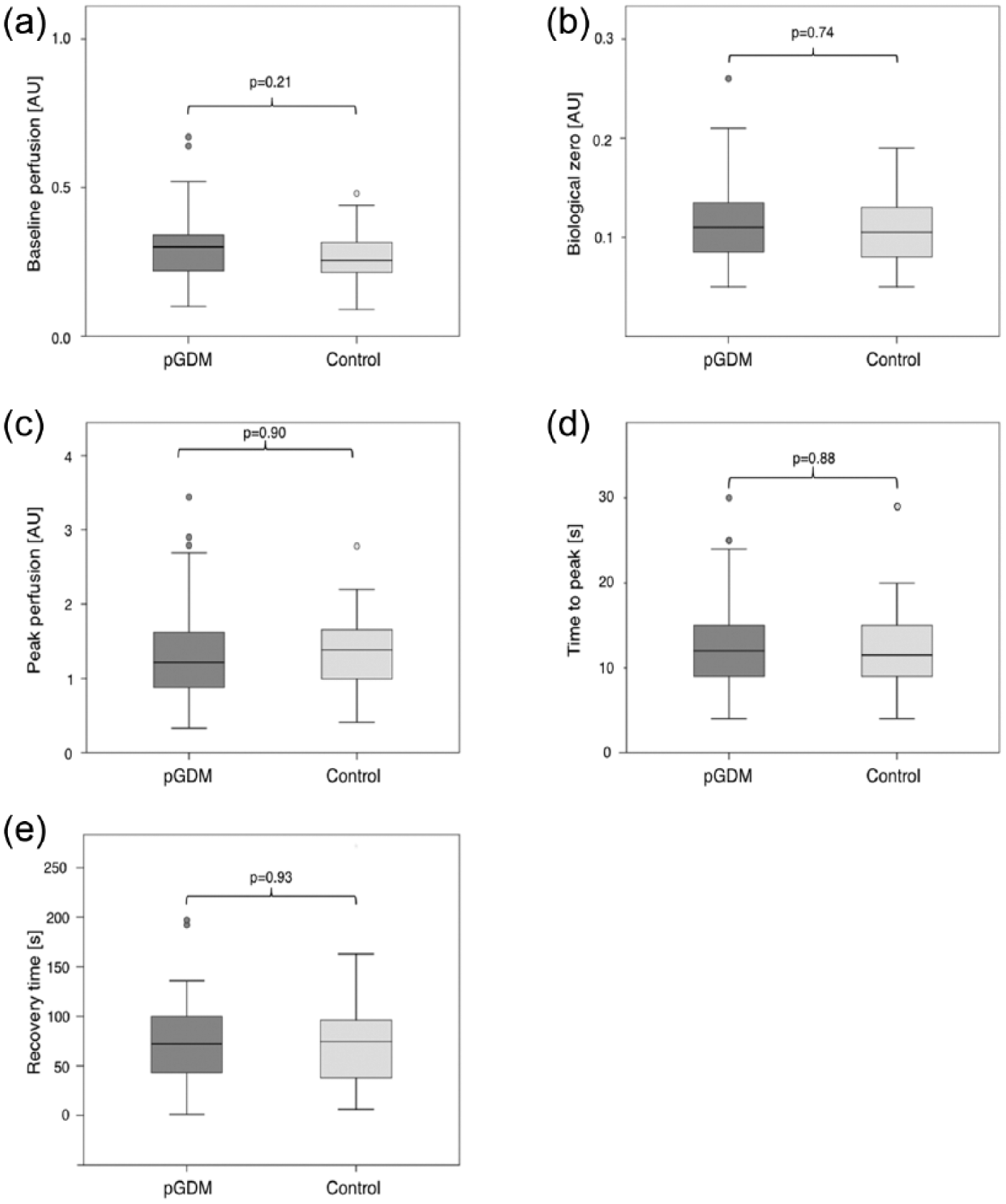
Boxplots comparing (a) baseline perfusion, (b) biological zero, (c) peak perfusion, (d) time to peak and (e) recovery time during post-occlusive reactive hyperaemia (PORH) in women with prior gestational diabetes mellitus (pGDM) compared with controls (women with prior pregnancy but without pGDM). Boxes indicate medians and interquartile ranges (IQR), whiskers display 1.5 interquartile ranges from the upper and lower quartile and dots depict outliers (values more than 1.5 interquartile ranges from median).
By separately analysing women with pGDM and without current type 2 diabetes baseline perfusion, biological zero, peak perfusion, time to peak perfusion and recovery time did not differ from controls or from women with pGDM with current type 2 diabetes (all
Patients’ age was not related to baseline perfusion, biological zero, peak perfusion, time to peak and recovery time, neither in women with pGDM nor in controls (all
Furthermore, no difference in baseline perfusion, biological zero, peak perfusion, time to peak perfusion and recovery time was found between pGDM-women with metabolic syndrome and controls (all
Microvascular function and fasting blood glucose
In women with pGDM, fasting blood glucose was not related to baseline perfusion (
Discussion
The main finding of this study is that microvascular reactivity does not differ between women with pGDM and women without pGDM. As the mean duration between the index pregnancy and the microvascular function test was 16.2 years, this finding suggests that pGDM is not associated with microangiopathy in the long term.
To reveal subclinical microangiopathy, PORH – as measured by laser Doppler fluxmetry – was used. Aiming to analyse the findings of this study appropriately, PORH has to be viewed in the light of the microcirculatory physiology and has to be differentiated from other vascular function tests: in this study, PORH was determined using laser Doppler fluxmetry. Laser Doppler fluxmetry semi-quantitatively records perfusion of the cutaneous microcirculation from a depth of 1–1.5 mm below the epidermis.9,10 The perfusion of this microcirculatory network is regulated by the tone of precapillary arteriolar smooth muscle cells. Precapillary arterioles maintain capillary homeostasis and prevent microvascular beds from hyperperfusion.9,10 Referring to microvascular PORH, a sudden increase in microvascular blood flow provokes an immediate counter-regulatory constriction of precapillary arterioles, which is supposed to be mediated by a neuromuscular axon reflex. 11 In contrast to this regulatory response within microvascular beds, in macrocirculation, an increase in blood flow results in an increase in wall shear stress, which in turn promotes a nitric oxide–mediated dilation of the respective artery. This gain in diameter of large arteries can be detected by duplex sonography or by peripheral arterial tonometry using fingertip plethysmographs.22–24 Since duplex sonography and fingertip plethysmography target arteries of the macrovasculature, neither of these methods allows conclusions to be drawn with respect to microvascular reactivity.
The focus of this study, however, laid on the microvasculature. Previously we have shown that diabetes-induced changes in microvascular reactivity can be detected by PORH using laser Doppler fluxmetry. 8 In diabetes hyperglycaemia, oxidative stress and insulin resistance potentially promote vascular and neural dysfunction.25,26 These pathophysiological pathways might contribute to an increased risk of vascular damage in the long term. Accordingly, previous studies confirmed an increased long-term risk of cardiovascular disease in women with pGDM.15,16 In this context, it appears noteworthy that the previously observed increased long-term risk in women with pGDM referred to macrovascular complications only. By contrast, the aim of this study was to assess whether women with pGDM face a risk of microangiopathy in the long term. Whether the putative inconsistency between an increased long-term risk of cardiovascular disease and the missing relation between pGDM and microvascular reactivity can be attributed to differences in the pathophysiology of macro- and microangiopathy has to be assessed in further studies.
Nevertheless, the present findings should be discussed against the background of its strengths and limitations: first, the group of women with pGDM comprised women without current diabetes mellitus and women with a current diabetic metabolic state or metabolic syndrome. As microvascular function did not differ between women with pGDM and controls, it appears unlikely that an exclusion of women with current diabetes or metabolic syndrome would have had an impact on the main finding of this study. Furthermore, it should be noted that the sample size of this study accounted for a comparison between women with pGDM and controls.
Accordingly, the moderate sample sizes of the two subgroups of women with and without present type 2 diabetes or metabolic syndrome attenuate the strength of this subgroup analysis. Regarding the women’s smoking status, we categorized the study population into non-smokers or smokers. To further assess the impact of smoking habits on microvascular function in women with pGDM, larger studies are warranted.
Furthermore, it has to be acknowledged that the age of women with pGDM was higher than the age of women who served as controls in this study. Previously, we have shown that – especially in adolescence – age might have an impact on microvascular reactivity. 27 However, in this study, we aimed at comparing microvascular function of two well-characterized groups of women with respect to their history of previous pregnancies. An exact 1:1 age-matching would have considerably reduced the number of eligible volunteering women in both groups. Although the influence of age has been primarily demonstrated during juvenile growth, we cannot exclude a residual confounding in this respect.
Finally, it has to be emphasized that this study focused on subclinical changes in microvascular reactivity. Although previous data suggested a relation between microvascular reactivity and microvascular organ damage, further studies are warranted to confirm this association. 11
Apart from these possible limitations, the major strength of this study warrants mention: up to now, data on potential long-term microvascular complications in patients with pGDM are scarce. To the best of our knowledge, this is the first study that investigates potential long-term effects of GDM on microcirculation.
In conclusion, the findings of this study suggest that – in the long-term – microvascular function is not impaired in women with pGDM.
Footnotes
Acknowledgements
S.C.-R., A.K.-W. and O.S. were responsible for conception and design of the study, analysis of data and drafting of the manuscript. S.C.-R., O.S. and M.M. were responsible for data base management. S.C.-R., R.Y., M.H., K.L., M.L., J.G., M.M., A.G., R.K., M.E.G., A.K.-W. and O.S. analysed data and critically revised the manuscript. J.G. and O.S. were responsible for the statistical analysis and the statistical revision of the manuscript. All authors had full access to the data, revised the manuscript and approved the final version of the paper. S.C.-R. and O.S. are the guarantors of this work and take full responsibility for integrity of data and accuracy of analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
