Abstract
Understanding the association between the intrauterine hyperglycemic milieu and the development of adult diabetic vasculopathy is of particular relevance in India, where diabetes and vascular disease are prevalent. The gestational diabetes mellitus placenta is a valuable tool to examine blood vessels that have been exposed to hyperglycemic cues.
We report an interesting observation in a cohort of gestational diabetes mellitus foetal placental vasculature from South India. Transmission electron microscopy demonstrated pericyte detachment and pericyte ghost cells reminiscent of adult type 2 diabetic retinopathy, in gestational diabetes mellitus foetal placental blood vessels that were not observed in non-gestational diabetes mellitus placentas (
In 2011, 62.4 million Indians were diagnosed with type 2 diabetes (T2D). The projection of Indian T2D to 100 million by 2030 has been likened to a time bomb. 1 A recent survey reveals that vascular disease overrides tuberculosis in causing death among Indians aged 30–69 years. 2 It is likely that the majority of these cases are the result of T2D-associated vascular complications. Significantly, the incidence of gestational diabetes mellitus (GDM) is higher in India than in the developed world. 3
We report an interesting observation in a small cohort of five cases each of healthy controls and GDM placentas from South India. Women aged 18–28 years were included in the third trimester, with no previous history of diabetes, pre-eclampsia, cardiovascular disease or body mass index (BMI) of more than 35. This pilot study was designed to examine placental blood vessels exposed to short-term hyperglycaemia. GDM typically presents in the third trimester of pregnancy. GDM patients were selected with fasting glucose levels of ≥92 mg/dL and glucose level after 1 h ≥ 180 mg/dL and after 2 h ≥ 153 mg/dL. Healthy control women had fasting glucose level of 60–75 mg/dL and glucose level after 1 h 100–140 mg/dL and after 2 h 80–100 mg/dL. Both the GDM and healthy control women delivered babies weighing 2.5–3 kg, vaginally, at term. The placentas weighed 470–480 g. Placental tissue was fixed immediately after delivery. Transmission electron microscopy (TEM) of blood vessels in five sections of each placenta was examined using the Tecnai T2 Spirit, FEI microscope. A total of 250 regions of interest were examined per placenta by electron microscopy in blinded fashion, and semi-quantification was performed by a pathologist (R.S.). The foetal vasculature in the control placentas showed endothelial cell–lined capillaries sharing their basement membrane with perivascular pericytes (Figure 1). GDM placental vasculature demonstrated pericyte detachment and pericyte ghost cells reminiscent of diabetic retinopathy that were not observed in non-GDM placentas (Figure 2) (
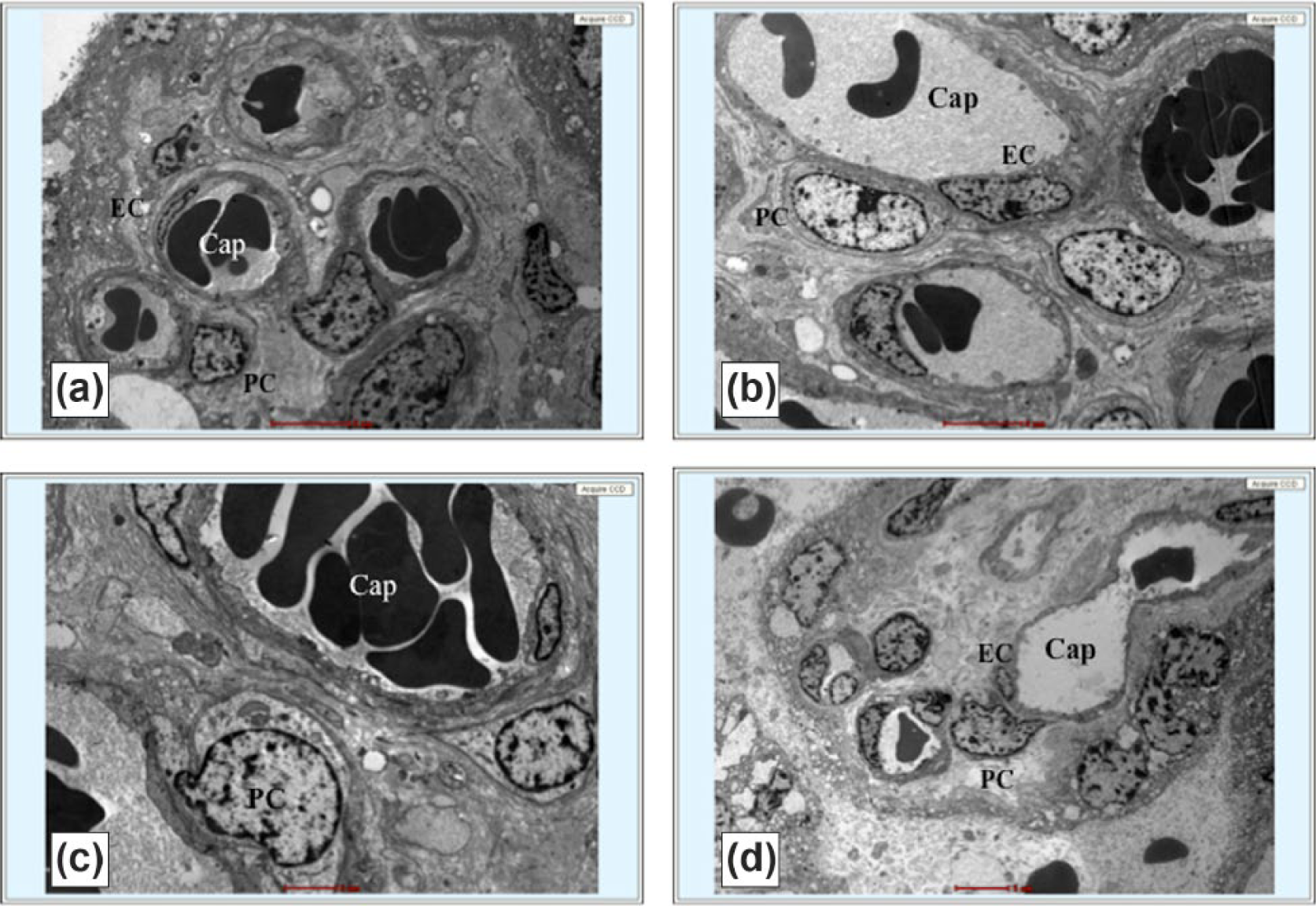
Transmission electron microscopy of healthy foetal placental blood vessels. Luminal endothelial cell (EC) line capillary (Cap). Abluminal perivascular pericytes (PC) share basement membrane (BM) with ECs (magnification: (a), (b) and (d): 5 µm; (c): 2 µm).
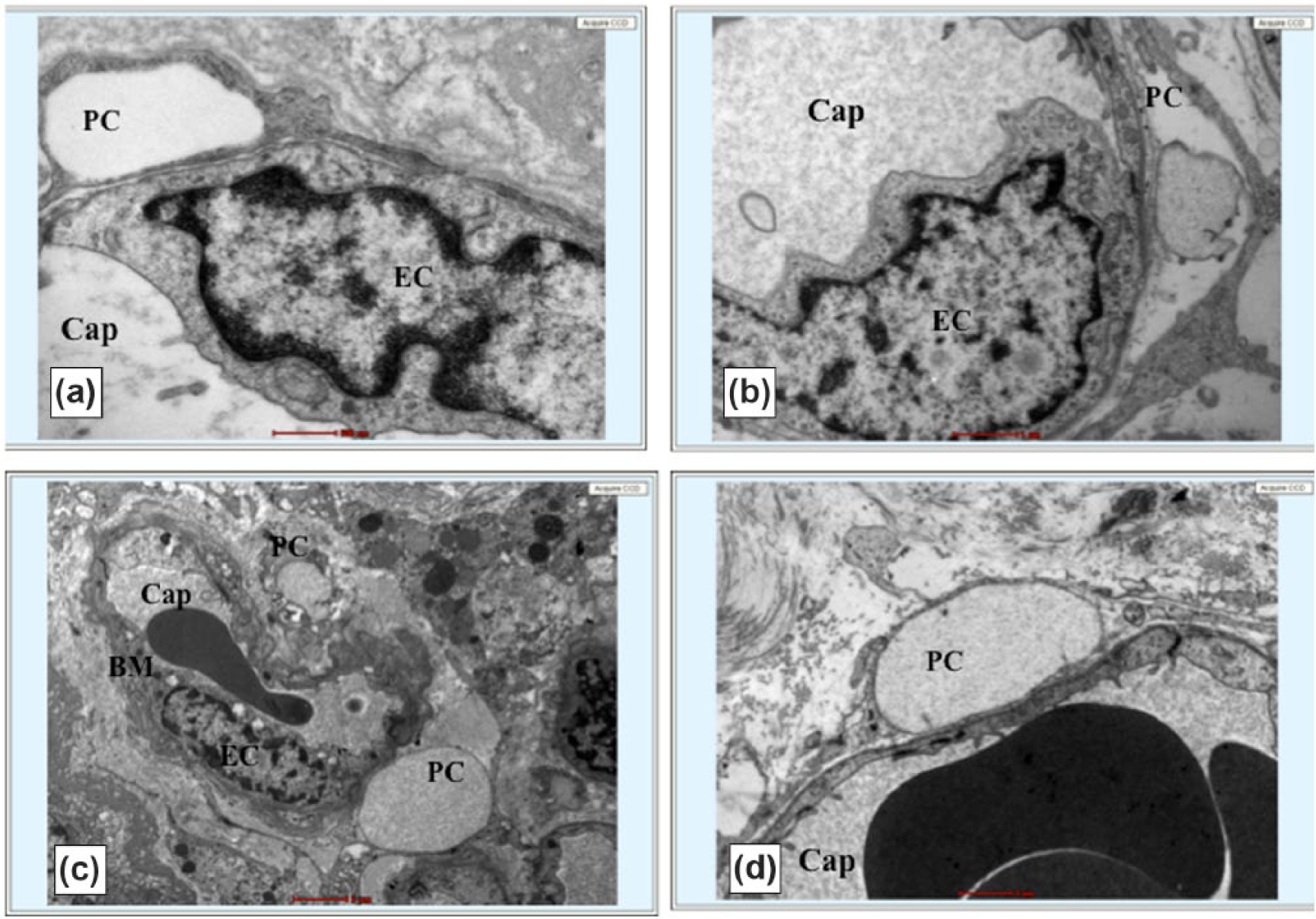
Transmission electron microscopy of gestational diabetic foetal placental blood vessels. Pericyte (PC) ghosts seen (a), (c) and (d). Impending PC dropout noted (b). Endothelial cell (EC) irregularity (a) and (b), and focal thickening of basement membrane (BM) is noted (magnification: (a): 500 nm; (b) and (d): 1 µm; (c): 2 µm).
The striking resemblance of vascular changes in a 9-month organ, the placenta, and T2D retinopathy point to the diabetic intrauterine environment influencing the development of T2D in later adulthood in women and newborns. One mechanism might involve epigenetic gene regulation. Rather than utilize adult blood or tissue, it may be prudent to use GDM placenta to work forwards to determine the development of vasculopathy in T2D. A detailed study to expand GDM cases is warranted to validate the significance of these findings. Understanding the evolution of vasculopathy in diabetes may permit identification of new targets for therapeutic intervention to prevent or mitigate diabetic vasculopathy.
Footnotes
Acknowledgements
We thank the Central Electron Microscopy Facility, CMC Hospital, for technical assistance. The underlying research material related to this manuscript may be accessed by contacting Dr Rekha Samuel.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by Department of Biotechnology, Ministry of Science and Technology (BT/PR5915/MED/31/172/2012), and Fast track Scheme for young scientists, Department of Science and Technology, Science and Engineering Research Council SB/FT/LS-196/2012, India (to Rekha Samuel).
