Abstract
Objective:
Circulating microvesicles, released from activated/apoptotic cells, are involved in vascular complications and may be looked upon as biomarkers. Albuminuria is characteristic of disease progression in type 2 diabetes mellitus. We aimed to investigate quantitative and qualitative differences of circulating microvesicles in type 2 diabetes mellitus with and without albuminuria and whether 12-month exercise training influenced expression of circulating microvesicles.
Methods:
Coronary artery disease patients with type 2 diabetes mellitus (n = 75), of which 25 had albuminuria, were included. Annexin V+ (AV+) circulating microvesicles were analysed by flow cytometry in citrated plasma. The exercise volume was 150 min per week.
Results:
In albuminuria patients, circulating microvesicles from endothelial-(CD146+/CD62E+/AV+) and endothelial-progenitor-(CD309+/CD34+/AV+) cells were significantly higher compared to those without (p ⩽ 0.01, both). Receiver operating characteristic curve analysis of the endothelial circulating microvesicles shows an area under the curve of 0.704 (95% confidence interval: 0.57–0.84; p = 0.004). Albuminuria patients had more circulating microvesicles derived from activated leukocytes and monocytes and monocytes carrying tissue factor (CD11b+/AV+, CD11b+/CD14+/AV+, CD142+/CD14+/AV+, respectively, p ⩽ 0.05, all) and higher number of circulating microvesicles from activated platelets (CD62P+/AV+). Within exercising patients, circulating microvesicles from progenitor cells increased (p = 0.023), however, not significantly different from controls.
Conclusion:
Coronary artery disease patients with type 2 diabetes mellitus and albuminuria had elevated number of circulating microvesicles from activated blood and vascular cells, rendering them as potential predictors of disease severity. The circulating microvesicles were limitedly affected by long-term exercise training in our population.
Introduction
Circulating microvesicles (cMVs) are submicron fragments released from the plasma membrane surface of different cell types during cell activation and/or apoptosis. 1 cMVs share the antigenic profile and part of the cytoplasmic content including nucleic acids of their parent cell, and due to translocation of phosphatidylserine (PS) to the outer leaflet of the membrane during cMVs formation, they have procoagulant properties. 2 In addition, some cMVs also carry tissue factor (TF), the initiator of extrinsic coagulation.
cMVs are novel mediators of intercellular signalling and they are shown to be directly involved in the pathogenesis of vascular disease. 3 As the number and variety of cMVs increase in conditions like cardiovascular disease (CVD) and diabetes mellitus (DM), 4 they might be useful as biomarkers of disease severity, procoagulant states and vascular activation and also as therapeutic targets. 5 cMVs are mainly detected by flow cytometry, based on their size, binding to annexin V (AV+), a high-affinity ligand for PS and also binding on specific markers of cell lineage or cell activation.
Type 2 DM (T2DM) is characterized by hyperglycemia, insulin resistance, dyslipidemia, inflammation and hypercoagulability, all devastating conditions for the endothelium, contributing to enhanced development of microvascular diseases as well as atherosclerosis and coronary artery disease (CAD). 6 The disease progression in DM is characterized by albuminuria, categorized as microalbuminuria or macroalbuminuria, which is used as a conventional marker of chronic kidney disease (CKD). 7
Endothelial cell injury is the main component of disruption of cardiovascular homeostasis within the vasculature. Endothelial-derived MVs (EMVs) are independent risk factors for coronary heart disease (CHD), 8 and increased amount is associated with endothelial dysfunction. 9 However, endothelial progenitor cells (EPCs) which are immature cells mobilized from the bone marrow in response to tissue ischemia and vascular injury might facilitate endothelial repair. 10 Decreased numbers of circulating EPCs have been associated with endothelial dysfunction and related to CVD risk, mortality and recurrent events in patients with CVD. 11
Exercise is known to improve endothelial function and insulin sensitivity. 12 , 13 However, the effect of physical activity on levels of cMVs has been inconsistently reported, potentially related to exercise type and intensity. 14 Exercise might act beneficially by lowering the number of cMVs derived from leukocytes and EMVs 15 and by increased amount of circulating EPCs. 16
We have previously reported on increased levels of pro-thrombotic markers in CAD patients with T2DM and albuminuria. 17 The aims of this study were to perform quantitative and qualitative analyses of AV+ cMVs from the vascular compartment between CAD patients with T2DM, with and without albuminuria. In addition, the effects of 1 year exercise training on cMVs, with specific reference to the presence of albuminuria, were studied. Our hypotheses were that patients having albuminuria express different patterns of cMVs especially related to vascular dysfunction and thrombotic properties, compared to non-albuminuria patients. Furthermore, the exercise training would contribute beneficially to the cMVs expression.
Methods
Study population
A subset of 75 patients from the exercise training in patients with CAD and type 2 diabetes (EXCADI) cohort of 137 patients 18 was included. Of the 75 patients, 25 presented with albuminuria and 50 age-matched patients were selected. All patients were included at Department of Cardiology, Oslo University Hospital, Ullevål, Oslo, Norway between August 2010 and March 2012. The main EXCADI study was a randomized controlled clinical trial, exploring the effect of 12-month exercise training on the development of atherosclerosis and glucometabolic control in patients with angiographically verified CAD and T2DM. All study patients gave informed written consent to participate, and the study was conducted according to the Declaration of Helsinki. The Regional Ethics Committee approved the trial and it is registered at http://www.Clinicaltrials.gov (NCT 01232608).
The definition of albuminuria includes both microalbuminuria and macroalbuminuria. Microalbuminuria was defined as albumin/creatinine ratio in spot urine >3 mg/mmol and ⩽30 mg/mmol and macroalbuminuria as levels above 30 mg/mmol. The updated homeostatic model assessment 2 of insulin resistance (HOMA2-IR) was used to estimate insulin resistance. 19
Blood sampling
Fasting venous blood was drawn before any morning medication between 08:00 and 10:00 a.m. at baseline and after 12-month exercise intervention. Tubes containing 3.8% sodium citrate were used for cMV analysis. Blood cells were removed within 30 min by centrifugation 2500×g for 20 min at 4°C, and plasma was immediately frozen and stored at −80°C until further preparation for analysis. Fasting glucose and serum lipids were analysed by conventional routine methods. HbA1c was measured by turbid metric inhibition immunoassay (Roche, Basel, Switzerland), insulin by dissociation-enhanced lanthanide fluorescence immunoassay (DELFIA) method (Perkin Elmer, Waltham, MA, USA) and C-peptide by electrochemiluminescence immunoassay (ECLIA; Roche Diagnostics).
Flow cytometry analysis of cMVs
The frozen plasma aliquots were thawed in an ice bath, mixed and centrifuged at 2500×g for 10 min at room temperature (RT) to ensure sedimentation of remnant platelets and potential clots. Plasma from the upper part of the tube was transferred to a new tube, and the cMVs were washed and fractionated from plasma by a two-step high-speed centrifugation, as previously described. 20 The final cMVs pellets were resuspended in citrate phosphate-buffered saline (citrate PBS) and prepared for triple-label flow cytometry analysis. In brief, each combination of AV+ labelled with allophycocyanin (APC; 5 µL) with two specific monoclonal antibodies (mAb, 5 µL each, see Supplemental Table 1) conjugated to fluorescein isothiocyanate (FITC) or phycoerythrin (PE) or the isotype-matched control antibodies, were diluted in Annexin Binding Buffer (ABB; 30 µL) and mixed with the cMVs suspension (5 µL). After 20 min incubation in the dark at RT, the labelling was stopped by adding ABB, and the samples were immediately analysed by flow cytometry. The Auto Collect mode and 96-well plates on an AccuriC6 flow cytometer (BD, Accuri® Cytometers, Inc., San Diego, CA, USA) were applied.
Every sample had 2 min of acquisition at a flow rate of 14 µL/min. Forward scatter (FSC), side scatter (SSC) and fluorescence data were gathered with the settings in the logarithmic scale. Cytometer settings were defined with the Megamix-Plus FSC, a mix of beads with bead-equivalent sizes: 0.1, 0.3, 0.5 and 0.9 µm (BioCytex, Marseille, France). The upper threshold for FSC and SSC was set to ⩽1 (see Supplemental Figure 1), and according to beads signal, the lower limit of detection was placed as a threshold above the electronic background noise of the flow cytometer for FSC and approximately at the second logarithm for SSC. In addition to the size criteria (>0.1 to ⩽1 µm), cMVs were identified and quantified based on their binding to AV+ and reactivity to cell-specific mAb (see Supplemental Figure 2). To classify positive marked events, thresholds of fluorescence were set according to samples incubated with the isotype-matched control antibodies (same final concentrations based on titration experiments). Fluorescence signals acquired with MVs in a calcium-free buffer (PBS) were used to correct the AV+ binding for autofluorescence. The use of freshly prepared buffers filtered through 0.2 µm pore size (vacuum) was contributed to reduce the background noise.
The BD software (version 1.0.264.21; Accuri® Cytometers, Inc.) was applied to analyse data. The number of cMVs per microlitre of plasma was calculated according to Nieuwland’s formula, 21 based on the number of fluorescence-positive events (N), sample’s volume and flow cytometer’s flow rate, as follows: cMVs/µL = N × (Vf/Va) × [Vt/(FR × 2)] × (1/Vi) [where Vf (µL) is the final volume of washed cMV suspension, Va (µL) is the volume of washed cMV suspension used for each labelling analysis, Vt (µL) is the total volume of cMV suspension before fluorescence-activated cell sorting analysis, FR (µL/min) is the flow rate of the cytometer (14 µL/min), 2 are the minutes of acquisition, 1 is the unit of volume (µL) and Vi (µL) is the original volume of plasma used for microvesicle isolation].
Exercise intervention
The exercise intervention was planned and conducted in collaboration with Norwegian School of Sport Sciences, Oslo, Norway and consisted of a 12-month combined aerobic and resistance training programme. Detailed description has previously been reported. 18 The total exercise volume was 150 min/week, which included two group-based exercise sessions of 60 min duration with qualified instructors and a third weekly home-based exercise session. All training sessions included intervals with high intensity, guided by Borgs Scale of rated perceived exertion.
Statistics
Data were analysed with IBM© SPSS© statistics for windows, v 24.0 and 25.0 (IBM Corp., New York, NY, USA). Differences were considered statistically significant at p ⩽ 0.05. Due to skewed distributed data in most variables, continuous data are presented by median values with 25th and 75th percentiles if not otherwise stated. Categorical data are noted as number and proportions. Between-group differences were calculated by Mann–Whitney U test, independent samples Student t-test, chi-square test and Kruskal–Wallis test, as appropriate. To investigate changes within the intervention groups, Wilcoxon’s test was applied and the Mann–Whitney U test was applied for differences in changes between the groups. Receiver operating characteristic (ROC) curve was used to evaluate the ability of the EMVs as prognostic markers of albuminuria. The area under the curve (AUC) was calculated. Adjustments for group differences in baseline characteristics were performed by a logistic regression model. The correlation between EMVs and conventional vascular biomarkers was analysed with Spearman’s rho.
Results
Baseline characteristics of the total study population (n = 75) and according to having albuminuria or not are presented in Table 1.
Baseline characteristics for the total population and subgroups with and without albuminuria.
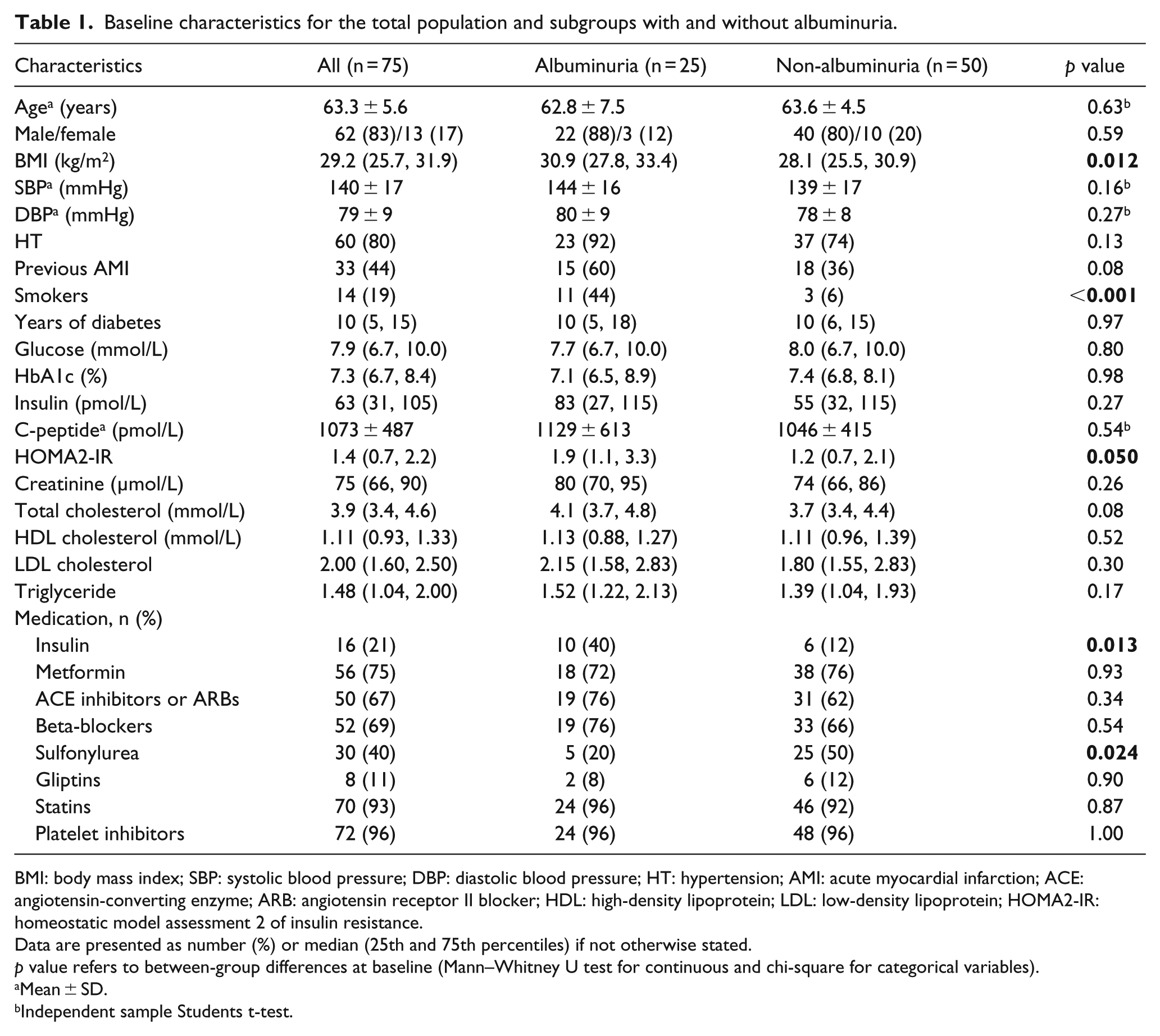
BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; HT: hypertension; AMI: acute myocardial infarction; ACE: angiotensin-converting enzyme; ARB: angiotensin receptor II blocker; HDL: high-density lipoprotein; LDL: low-density lipoprotein; HOMA2-IR: homeostatic model assessment 2 of insulin resistance.
Data are presented as number (%) or median (25th and 75th percentiles) if not otherwise stated.
p value refers to between-group differences at baseline (Mann–Whitney U test for continuous and chi-square for categorical variables).
Mean ± SD.
Independent sample Students t-test.
Mean age was 63 years and the patients were overweight/obese according to body mass index (BMI), which also was higher in those with albuminuria versus those without [30.9 (27.8, 33.4 kg/m2) versus 28.1 (25.5, 30.9 kg/m2); p = 0.012]. Median diabetes duration was 10 years, and about 20% were on insulin treatment, significantly more frequent in patients with albuminuria (40% vs 12%; p = 0.013). The latter patient group also presented with more insulin resistance [1.9 (1.1, 3.3) versus 1.4 (0.7, 2.2); p = 0.050) and higher number of smokers (44% vs 6%; p < 0.001). Use of sulfonylurea dominated in the non-albuminuria patients (50% vs 20%; p = 0.024). However, the above-mentioned differences affected the cMVs only to a limited degree. Also, none of the glucometabolic variables measured was associated with cMVs (data not shown).
Almost all patients were on platelet inhibition and 93% on statin treatment. Of the 50 patients in the non-albuminuria group, 1 patient was lacking citrated plasma for cMVs analysis.
cMVs in patients with and without albuminuria
Albuminuria patients compared to those without albuminuria presented with significantly elevated levels of EMVs and cMVs originated from monocytes (MMVs), platelets (PMVs) and EPCs, as shown in Figure 1(a). When excluding patients with macroalbuminuria (n = 6), thus analysing those with microalbuminuria (n = 19) compared to non-albuminuria (n = 49), only EMVs (CD146+/AV+, CD62E+/AV+ and CD146+/CD62E+/AV+) were still significantly elevated in the microalbuminuria group, as presented in Figure 1(b). ROC curve analysis of EMVs (CD146+/CD62E+/AV+) from activated endothelial cells, for the presence of albuminuria, shows an AUC of 0.704 [95% confidence interval (CI): 0.57–0.84; p = 0.004], as visualized in Figure 2. When adjusting for the differences between the albuminuria groups as shown in Table 1, the results were limitedly affected.
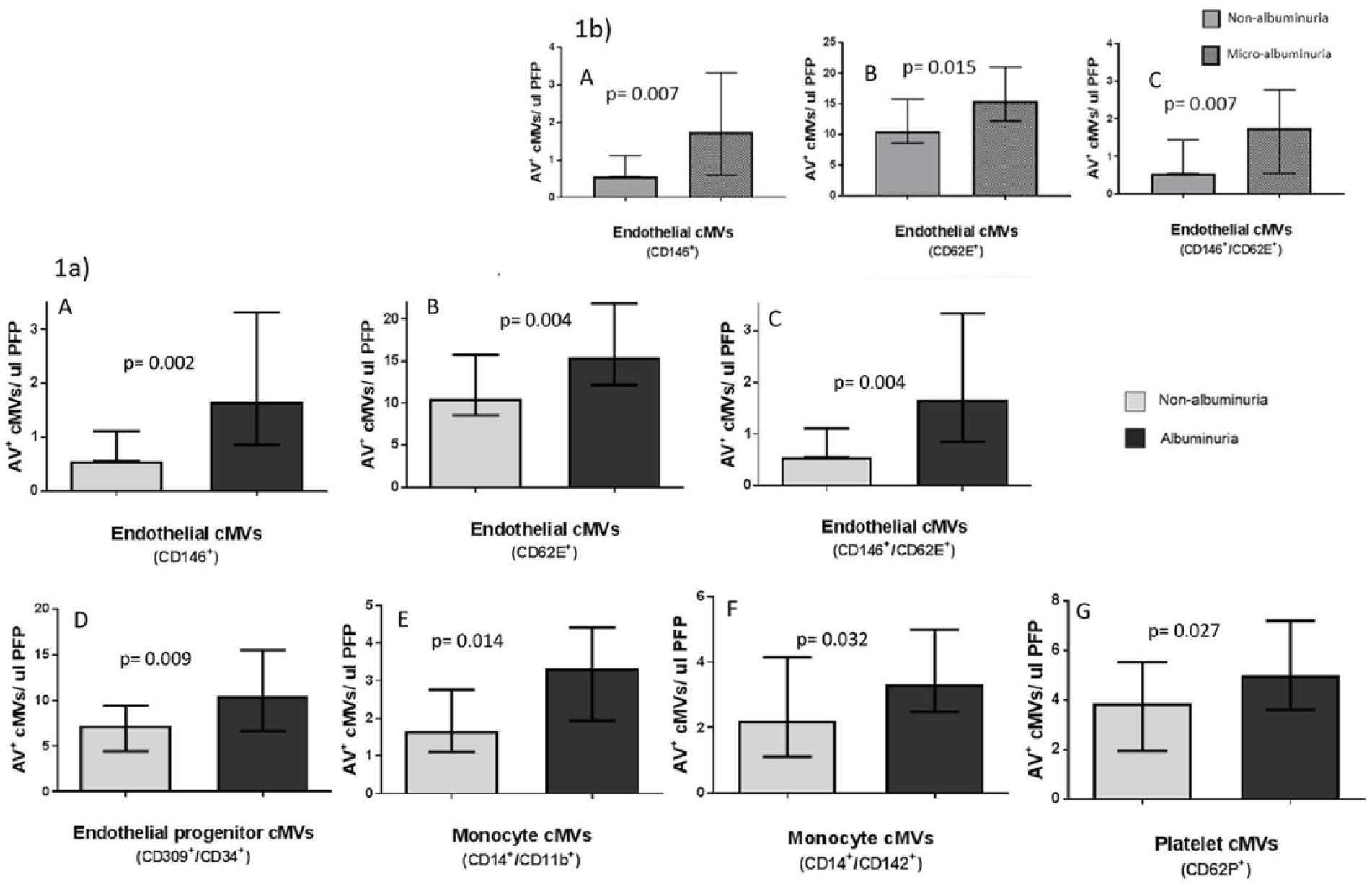
(a) Baseline levels of AV + cMVs in patients without (n = 49) and with (n = 25) albuminuria. (b) Baseline levels of AV + EMVs in patients without albuminuria (n = 49) compared to with microalbuminuria (n = 19). The p values refer to differences between groups (Mann–Whitney U test). Results are expressed as median (25th and 75th percentiles). (A) Endothelial-derived (CD146+/AV+) cMVs, (B) E-selectin-positive (CD62E+/AV+) cMVs, (C) activated endothelial cell (CD146+/CD62E+/AV+) cMVs, (D) endothelial progenitor cell–derived (CD309+/CD34+/AV+) cMVs, (E) activated monocyte (CD14+/CD11b+/AV+) cMVs, (F) tissue factor–positive monocytes (CD14+/CD142+/AV+) cMVs and (G) P-selectin-positive (CD62P+/AV+) platelet-derived cMVs.
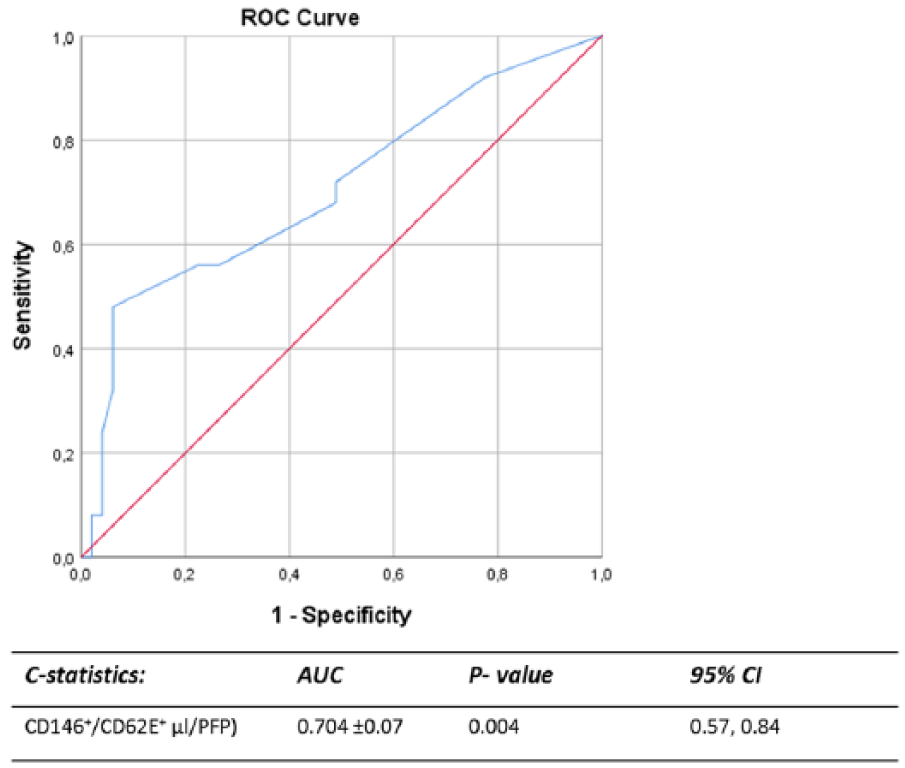
ROC curve analysis to evaluate the predictive power of endothelial-derived cMVs for the presence of albuminuria. The result is presented as AUC ± SEM with 95% CI.
To confirm that the measured EMVs originate from endothelial cells, the different EMVs were correlated to previously measured indices of vascular injury or dysfunction [endothelial cell adhesion molecules and asymmetric dimethylarginine (ADMA)]. 22 As can be seen (Supplemental Figure 3), significant correlations were found.
Effect of exercise training on cMVs
The number of patients with albuminuria in the exercise and control groups was equally distributed (p = 1.000). At baseline, no significant differences were observed between the two randomized groups, regarding clinical characteristics (see Supplemental Table 2) or cMVs (Table 2).
cMVs in T2DM patients at baseline and after 12-month exercise intervention.
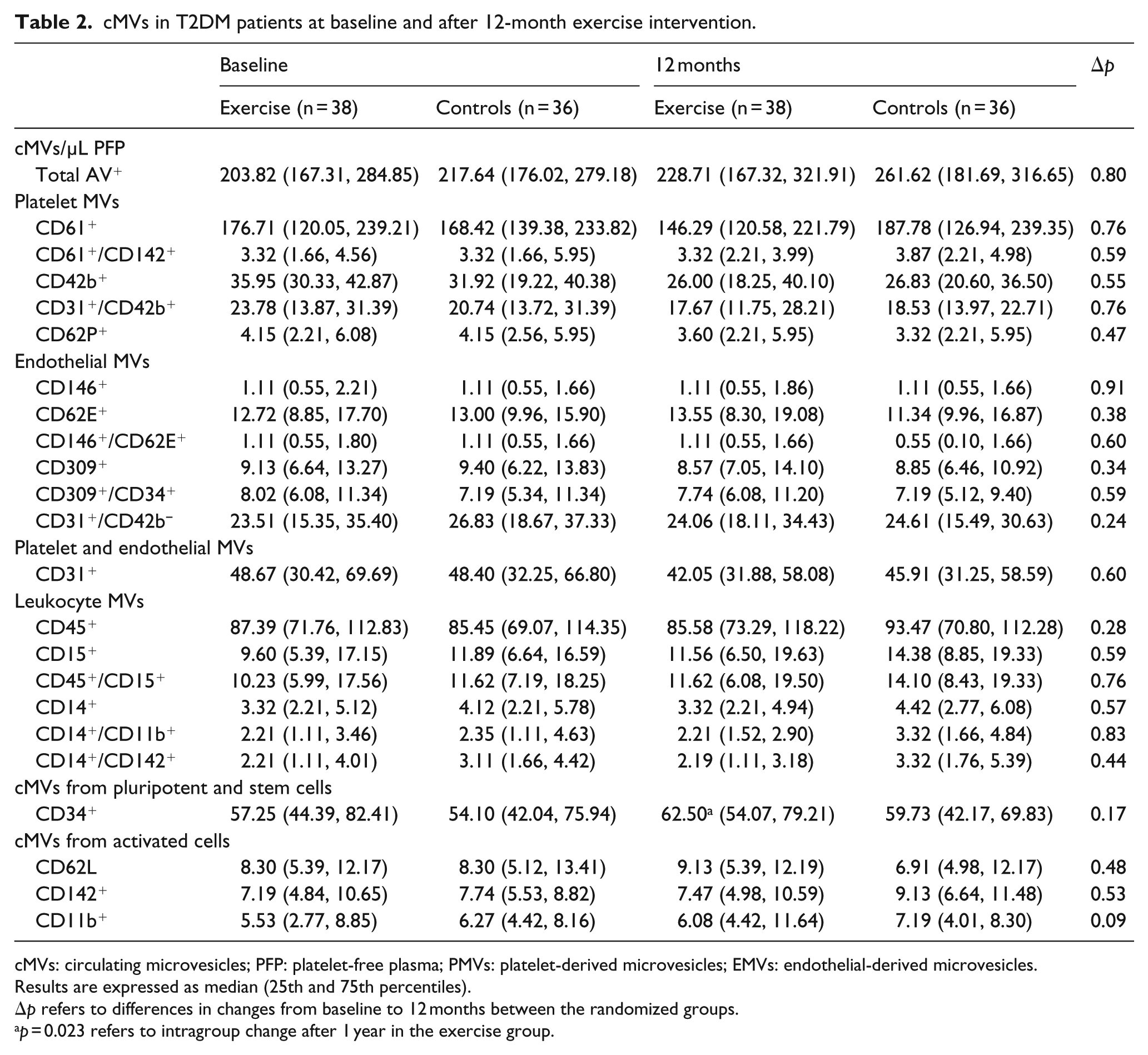
cMVs: circulating microvesicles; PFP: platelet-free plasma; PMVs: platelet-derived microvesicles; EMVs: endothelial-derived microvesicles.
Results are expressed as median (25th and 75th percentiles).
Δp refers to differences in changes from baseline to 12 months between the randomized groups.
p = 0.023 refers to intragroup change after 1 year in the exercise group.
Intervention results for the total population (n = 74) are presented in Table 2. During the intervention period, no significant differences in changes between the exercise and control groups in any cMVs were observed, neither in the total population nor in the group with albuminuria.
Nevertheless, within the exercise group, the level of cMVs from progenitor cells (CD34+/AV+) were significantly increased after 12 months [57.25 (44.39, 82.41 cMVs/µL platelet-free plasma (PFP)) versus 62.50 (54.07, 79.21 cMVs/µL PFP); p = 0.023], and in patients with albuminuria (n = 13), a significant reduction after exercise training was observed in levels of PMVs carrying TF (CD61+/CD142+/AV+) and von Willebrand factor (vWF; CD31+/CD42b+/AV+), and overall cMVs carrying vWF (CD42b+/AV+; p < 0.05, all), as shown in Figure 3. No significant changes were observed within the control group.
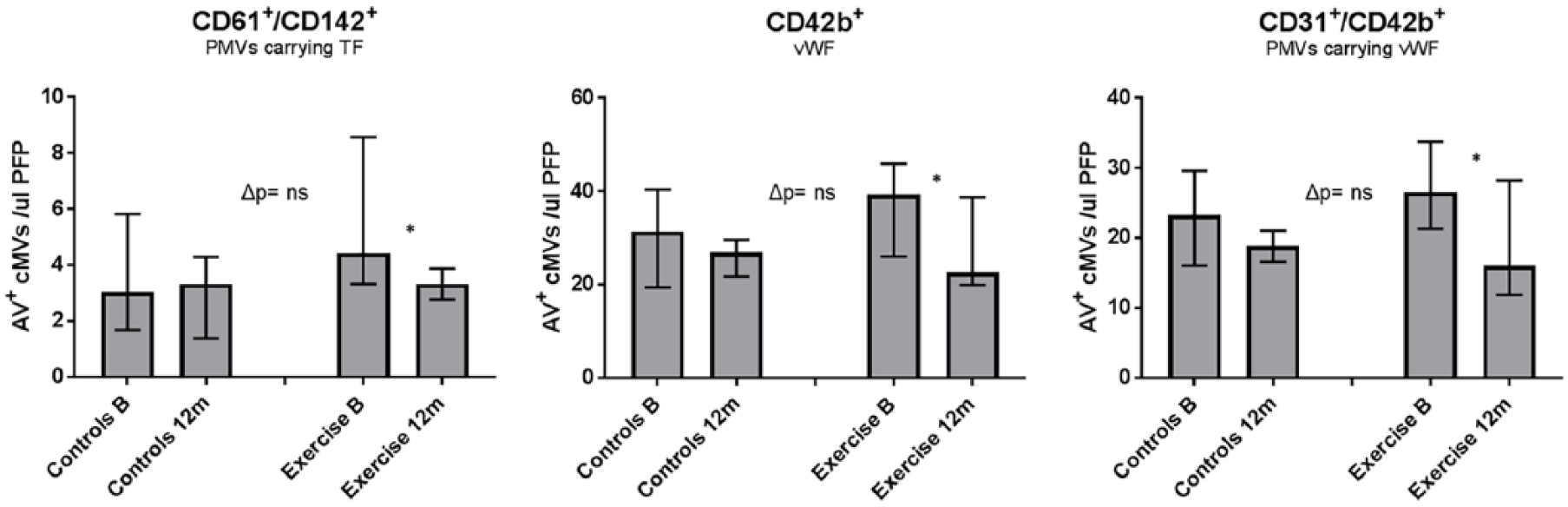
Effects of exercise intervention on AV + cMVs in patients with albuminuria (n = 25). Results are presented as median (25th and 75th percentiles). Δp refers to statistical differences in changes between the exercise and control groups after 1-year follow-up (Mann–Whitney U Test). (*Statistical significant changes in cMVs levels within the exercise group after the intervention period – Wilcoxon signed-rank test.)
Discussion
In the present study on patients with combined CAD and T2DM, significantly higher levels of circulating endothelial-related MVs (CD146+/AV+, CD62E+/AV+, CD146+/CD62E+/AV+), monocyte-derived MVs (CD14+/CD11b+/AV+, CD14+/CD142+/AV+), platelet-derived MVs (CD62P+/AV+) and cMVs from EPCs (CD309+/CD34+/AV+) were found in patients presenting with albuminuria compared to non-albuminuria patients. The AUC of 0.704 for the EMVs to predict albuminuria may reflect enhanced chronic endothelial cell activation in the advanced states of diabetes, when albuminuria occurs. EMVs were significantly elevated also in the subgroup with microalbuminuria only. The albuminuria patients included more smokers, higher BMI, more severe insulin resistance/use of insulin and less intake of sulfonylurea compared to the non-albuminuria group. However, no significant associations between these characteristics and the cMVs were present. Also, no significant associations between the measured cMVs and glucometabolic variables were observed.
During the 12-month intervention period, levels of cMVs derived from progenitor cells increased within the exercise group, and within the albuminuria patients, a significant decrease in PMVs carrying TF and vWF and overall cMVs loaded with vWF were observed. However, these changes did not differ significantly from the changes in the control group.
The most striking findings were cMVs shed from activated endothelial cells to be significantly elevated in patients with albuminuria and also significantly elevated in those with microalbuminuria only. High levels of EMVs might reflect progression of atherosclerosis and vascular injury in T2DM, particularly in patients with diabetes-associated nephropathy. 9 We have previously reported on levels of vascular parameters in the EXCADI study, 22 and as shown (Supplemental Figure 3), there were significant correlations between EMVs and other indices of vascular injury, verifying that the measured EMVs originate from the endothelium.
In line with our results, Rodrigues et al. demonstrated elevated levels of EMVs (CD51+/AV+) in a cohort of T2DM patients with nephropathy, 23 and EMVs (CD31+/AV+) were found increased in patients with microalbuminuria or macroalbuminuria, compared to those without albuminuria. 24 Furthermore, increased ratio of EMVs to EPCs was shown to be associated with reduced glomerular filtration rate (GFR), indicating that an imbalance in endothelial damage and repair capacity may in part explain reduced renal function. 25 Ongoing inflammation, thrombin generation and hyperglycemia are all factors contributing to accelerated release of EMVs, reflecting the state of the parent cell, 26 which might explain the elevated levels observed in the present study.
In patients presenting with albuminuria, cMVs derived from EPCs (CD309+/CD34+/AV+) were significantly elevated, potentially reflecting increased vascular damage and the need for repair. Increased mobilization of CD34+/CD133+ has been demonstrated in diabetic patients on insulin therapy, 27 in consistency with our data of more frequent use of insulin in the albuminuria patients. Insulin resistance and hyperglycemia were shown to attenuate migration from the bone marrow, differentiation and functionality of EPCs. 10 Of note, in the latter study, the number of EPCs per se was measured, while we investigated EPC-derived cMVs. However, recent findings suggest that EPCs mediate vascular repair via paracrine mechanisms, for example, releasing cMVs containing micro-RNA’s that can promote vascular repair. 28 Therefore, our albuminuria patients may have the same or lower levels of circulating EPCs than non-albuminuria patients, despite their higher levels of EPC-derived cMVs.
PMVs reflect chronic platelet activation and are the most abundant in the circulation. 29 Elevated levels of PMVs containing the receptor for P-selectin (CD62P+) have been shown in diabetes patients. 30 We could demonstrate significantly higher levels of CD62P+ PMVs in our patients with combined CAD and T2DM with albuminuria compared to those without. In line with our findings, Almquist et al. found increased levels of PMVs, EMVs and MMVs in patients with CKD compared to DM patients with normal GFR. 31 Furthermore, in diabetic patients with an acute myocardial infarction (MI), significantly higher amounts of CD62P+ PMVs were demonstrated and an early increase of these MVs after an MI was associated with higher risk of CVD events during 2-year follow-up period. 32 PMVs carrying P-selectin mediate leukocyte recruitment and aggregation via P-selectin glycoprotein 1, contributing to atherothrombosis.
Activated monocytes and MMVs are key players in atherothrombosis. We could show significantly elevated numbers of MMVs (CD14+/CD11b+) and MMVs expressing TF (CD14+/CD142+) in our albuminuria patients. Sulfonylurea, which has been associated with reduced expression of TF and reduced number of MMVs in vitro, 33 was far more common in the non-albuminuria group. Our results are in accordance with previous studies showing significantly higher levels of MMVs in T2DM patients who developed diabetic complications, especially nephropathy and both MMVs and PMVs to be correlated to microvascular damage. 34 , 35 MMVs are also suggested to be involved in kidney disease through effects on the podocytes, causing glomerular inflammation and increased glomerular permeability. 36
The 12-months combined aerobic and resistance exercise training did not result in any significant differences in changes from controls in any of the measured cMVs. There are very limited data on the effect of long-term exercise on cMVs. In a study on healthy individuals, acute exercise of moderate intensity reduced the number of CD62E+ MVs, compared to high-intensity interval training, 37 and accelerated production of PMVs was observed after strenuous exercise. 38 More consistent with our data is a study on physically fit men with stable C (CHD), which showed no changes in EMVs and PMVs after high- and moderate-intensity training. 39
Within the exercise group we could, nevertheless, observe a significant increase in progenitor cell-derived cMVs, which might suggest a beneficial effect of exercise on the vascular endothelium, supporting previous reports regarding recruitment and survival of EPC after exercise. 40
Furthermore, within patients with albuminuria who exercised, a favourable effect on the hypercoagulable state by a decreased number of cMVs with procoagulant activity was noted. However, precautions have to be taken due to low numbers.
Almost all patients in our up-to-date medically treated population were on statins and aspirin therapy, both been associated with reduced numbers of EMVs and PMVs, 41 which to some degree may explain the lack or limited effects of the intervention. We have previously also reported that the intervention programme did not have any impact on the procoagulant state in the present population. 17
In conclusion, CAD patients with T2DM and albuminuria had elevated number of cMVs derived from activated blood and vascular cells. More specifically, cMVs from endothelial cells seem to be present in the early stages of albumin excretion rendering them as potential markers of disease severity, and cMVs from EPCs suggested to undertake repair of damaged endothelium were increased in patients with albuminuria. The measured cMVs were only limitedly affected by long-term exercise training in our population, having albuminuria or not.
Supplemental Material
Supplemental_Figure_1._Treshold – Supplemental material for Elevated levels of circulating microvesicles in coronary artery disease patients with type 2 diabetes and albuminuria: Effects of exercise training
Supplemental material, Supplemental_Figure_1._Treshold for Elevated levels of circulating microvesicles in coronary artery disease patients with type 2 diabetes and albuminuria: Effects of exercise training by Vibeke Bratseth, Gemma Chiva-Blanch, Rune Byrkjeland, Svein Solheim, Harald Arnesen and Ingebjørg Seljeflot in Diabetes & Vascular Disease Research
Supplemental Material
Supplemental_Figure_2 – Supplemental material for Elevated levels of circulating microvesicles in coronary artery disease patients with type 2 diabetes and albuminuria: Effects of exercise training
Supplemental material, Supplemental_Figure_2 for Elevated levels of circulating microvesicles in coronary artery disease patients with type 2 diabetes and albuminuria: Effects of exercise training by Vibeke Bratseth, Gemma Chiva-Blanch, Rune Byrkjeland, Svein Solheim, Harald Arnesen and Ingebjørg Seljeflot in Diabetes & Vascular Disease Research
Supplemental Material
Supplemental_Figure_3 – Supplemental material for Elevated levels of circulating microvesicles in coronary artery disease patients with type 2 diabetes and albuminuria: Effects of exercise training
Supplemental material, Supplemental_Figure_3 for Elevated levels of circulating microvesicles in coronary artery disease patients with type 2 diabetes and albuminuria: Effects of exercise training by Vibeke Bratseth, Gemma Chiva-Blanch, Rune Byrkjeland, Svein Solheim, Harald Arnesen and Ingebjørg Seljeflot in Diabetes & Vascular Disease Research
Supplemental Material
Supplementary_Table_1._Cell_surface_molecules_for_cMVs_id_and_charac – Supplemental material for Elevated levels of circulating microvesicles in coronary artery disease patients with type 2 diabetes and albuminuria: Effects of exercise training
Supplemental material, Supplementary_Table_1._Cell_surface_molecules_for_cMVs_id_and_charac for Elevated levels of circulating microvesicles in coronary artery disease patients with type 2 diabetes and albuminuria: Effects of exercise training by Vibeke Bratseth, Gemma Chiva-Blanch, Rune Byrkjeland, Svein Solheim, Harald Arnesen and Ingebjørg Seljeflot in Diabetes & Vascular Disease Research
Supplemental Material
Supplementary_Table_2._Baseline_Charac_Intervention_gr – Supplemental material for Elevated levels of circulating microvesicles in coronary artery disease patients with type 2 diabetes and albuminuria: Effects of exercise training
Supplemental material, Supplementary_Table_2._Baseline_Charac_Intervention_gr for Elevated levels of circulating microvesicles in coronary artery disease patients with type 2 diabetes and albuminuria: Effects of exercise training by Vibeke Bratseth, Gemma Chiva-Blanch, Rune Byrkjeland, Svein Solheim, Harald Arnesen and Ingebjørg Seljeflot in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
The authors thank Ida U Njerve and Sissel Åkra for valuable contributions to the study.
Author’s contributions
All authors have contributed significantly and in accordance with the latest guidelines of the International Committee of Medical Journal Editors. V.B. was involved in the design of the study, analysis and interpretation of the results and in the drafting of the manuscript. G.C.-B. contributed to the interpretation of the results and drafting and revising of the manuscript. R.B. was responsible for the main randomized trial involving recruitment and follow-up of the study participants, acquisition and interpretation of clinical and laboratory data as well as revising of the manuscript. S.S. was involved in the conception of the trial and revising of the manuscript. I.S. and H.A. contributed to the conception and design of the study, interpretation of the results and drafting and revising of the manuscript. All authors are in agreement of the content and approved the final manuscript before submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
