Abstract
Linagliptin plus pioglitazone single-pill combinations were evaluated. Patients (n = 936) with insufficient glycaemic control, despite lifestyle interventions, were randomised for 30 weeks to either monotherapy with linagliptin 5 mg; pioglitazone 15, 30 or 45 mg; or single-pill combination with linagliptin 5 mg plus pioglitazone 15, 30 or 45 mg. An extension (⩽54 weeks) planned to evaluate linagliptin plus pioglitazone 30 or 45 mg single-pill combinations was not completed due to a protocol amendment. Adjusted mean (95% confidence interval) differences in HbA1c change from baseline at week 30 for linagliptin plus pioglitazone 15, 30 and 45 mg were −0.17% (−0.41, 0.07), −0.37% (−0.60, −0.14) and −0.41% (−0.64, −0.18) versus pioglitazone monotherapies, respectively, and −0.44% (−0.67, −0.20), −0.68% (−0.91, −0.44) and −0.89% (−1.12, −0.66) versus linagliptin monotherapy, respectively. Single-pill combinations were generally well tolerated. Hypoglycaemia frequency was ⩽1.5% per group. Linagliptin plus pioglitazone combinations were efficacious, with safety profiles comparable to the individual monotherapies.
Introduction
Type 2 diabetes (T2D) is a progressive disease with multiple underlying pathophysiological abnormalities. 1 Following diagnosis, treatment guidelines in general recommend lifestyle interventions and initial monotherapy with metformin (if not contraindicated).2,3 Long-term glycaemic control usually cannot be adequately maintained with monotherapy.4,5 Accordingly, guidelines advocate the step-wise introduction of additional antihyperglycaemic drugs in various dual or triple combinations to help achieve glycaemic targets.2,3
An important limitation of this step-wise paradigm is the potential to incur a delay before antidiabetes treatment is intensified in patients with inadequate glycaemic control (termed ‘clinical inertia’). 6 Patients who are not at their therapeutic goal should avoid periods of uncontrolled hyperglycaemia due to the increased risk for micro- and macrovascular complications. 7 An alternative treatment approach involves earlier or even initial combination therapy with two different oral antidiabetes drugs (OADs). 8 By targeting insulin resistance and impaired insulin secretion – the core pathophysiological abnormalities of T2D – an additive or synergistic improvement in glycaemic control may be anticipated using two drugs with complementary mechanisms of action. Combination regimens have typically included metformin; however, if contraindicated or poorly tolerated, alternative options to metformin are necessary. In addition, combining multiple drugs requires careful consideration, given an increased potential for adverse events (AEs). Administering multiple drugs can also negatively impact patient adherence to prescribed therapy; 9 for this reason, formulating drugs in a single-pill combination (SPC) can simplify treatment regimens and may help ensure continued administration of the drugs prescribed. 10
The glucose-lowering mechanisms of dipeptidyl peptidase-4 (DPP-4) inhibitors and thiazolidinediones are complementary because the former lowers glucose by increasing insulin and suppressing glucagon release, and the latter by decreasing insulin resistance. 11 As monotherapies, the DPP-4 inhibitor linagliptin and the thiazolidinedione pioglitazone have demonstrated clinically meaningful reductions in glycated haemoglobin A1c (HbA1c) and fasting plasma glucose (FPG) levels.12,13 As initial dual therapy in patients with insufficient glycaemic control, the linagliptin plus pioglitazone combination given as separate pills was well tolerated and elicited significant improvements in hyperglycaemia. 14 Both drugs have a low propensity for hypoglycaemia and can be taken once daily, making this combination an attractive option.
The aim of this study was to evaluate the efficacy and safety of initial therapy with linagliptin plus pioglitazone SPC versus linagliptin or pioglitazone monotherapy in T2D patients with insufficient glycaemic control with diet and exercise alone. The SPC formulations included pioglitazone at the three different dose strengths currently approved for use in T2D treatment. 15
Methods
Study design and patients
This was a multicentre, international, randomised, double-blind, parallel-group, and phase 3 study (ClinicalTrials.gov identifier: NCT01183013) conducted between August 2010 and February 2013 in the following countries: Estonia, Germany, Latvia, Spain, United Kingdom, and the United States.
The study protocol and amendments were reviewed and approved by the independent ethics committees or institutional review boards of each participating site. The study was conducted in compliance with the principles of the Declaration of Helsinki (1996) and in accordance with Good Clinical Practice guidelines as defined by the International Conference on Harmonisation. All patients gave written informed consent before participation.
Consenting patients were eligible if they were 18–80 years of age, had a diagnosis of T2D and insufficient glycaemic control (HbA1c ⩾ 7.0% to ⩽10.5%) on diet and exercise alone and without the use of OADs within the previous 10 weeks and had a body mass index (BMI) ⩽45 kg/m2. The main exclusion criteria were uncontrolled hyperglycaemia with a confirmed glucose level >240 mg/dL (>13.3 mmol/L) after an overnight fast during screening or placebo run-in; myocardial infarction (MI) within the previous 6 months; stroke or transient ischaemic attack within the previous 3 months; active liver disease or alanine transaminase level >2.5 times the upper limit of normal; bariatric surgery within the previous 2 years; hypersensitivity or allergy to linagliptin or pioglitazone; treatment with rosiglitazone, pioglitazone, glucagon-like peptide-1 receptor agonists, insulin or anti-obesity drugs within the previous 3 months; alcohol or drug abuse within the previous 3 months; treatment with systemic corticosteroids or change in dosage of thyroid hormones within the previous 6 weeks; pre-menopausal women who were nursing, pregnant or not practising birth control; gallbladder disease within the previous 6 months; or a history of pancreatitis or bladder cancer. Further exclusion criteria included any contraindication or restriction for the use of pioglitazone according to local prescribing information (e.g. diagnosis or history of heart failure; treatment with gemfibrozil, montelukast, trimethoprim or rifampicin; and haemodialysis patients).
Study procedures
Standard diet and exercise counselling was provided at the start of the trial. Prior to a 2-week placebo run-in period, if any patient was being treated with OAD monotherapy, they were required to be off this medication for more than 10 weeks. After the run-in period, eligible patients entered a two-part (Parts A and B) randomised double-blind treatment period of up to 84 weeks (Figure 1). Part A was an initial 30-week treatment period in which patients were randomised using equal allocations to one of seven treatment groups: either monotherapy with linagliptin 5 mg qd, pioglitazone 15, 30 or 45 mg qd; or SPC with linagliptin 5 mg plus pioglitazone 15, 30 or 45 mg qd. Randomisation was performed by a computer-generated random sequence using an interactive voice response system or web response system. Treatments were masked using a double-blind and double-dummy design. Patients randomised to receive pioglitazone 45 mg in Part A (as monotherapy or SPC) initially received pioglitazone 30 mg, which was uptitrated to 45 mg during a 6-week forced titration period. It was initially planned that all patients who completed Part A were to continue to Part B.
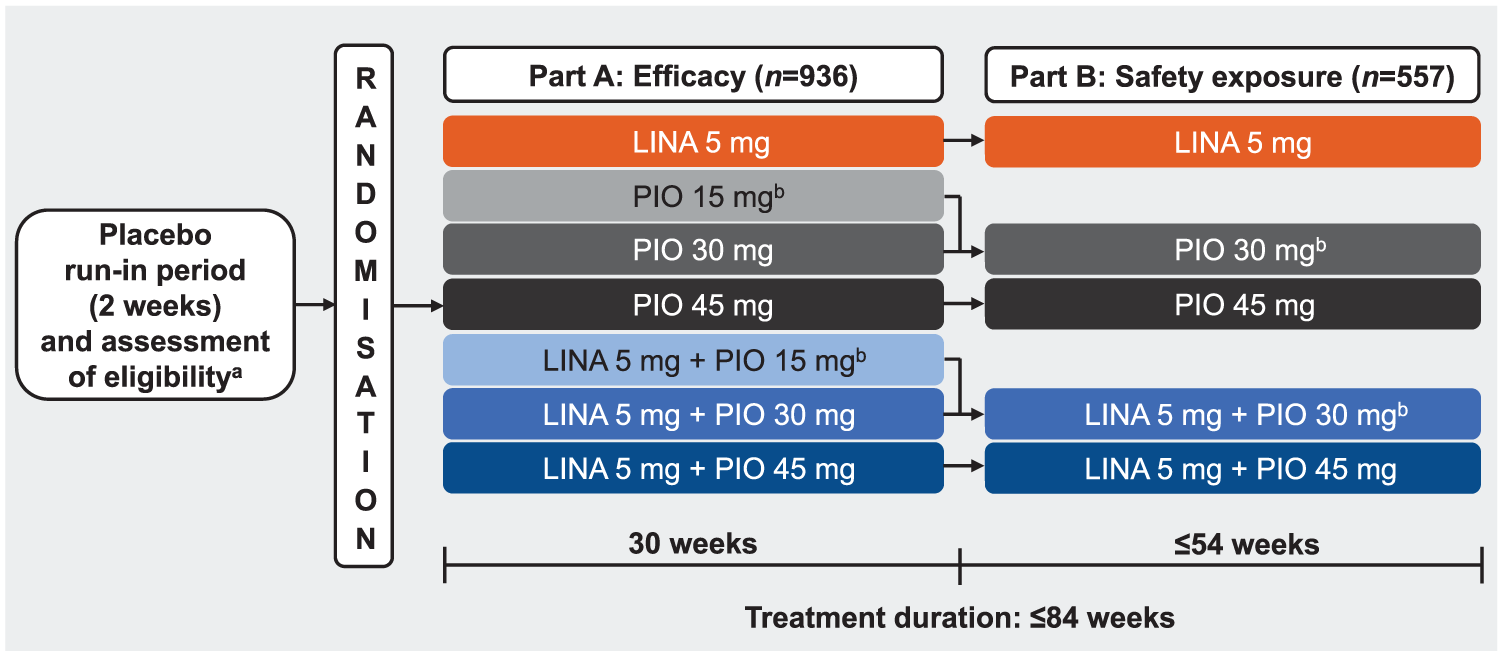
Study design.
Part B was an extension period of up to 54 weeks’ duration. There were five treatment groups, and assignment was determined by the preceding Part A treatment: patients randomised to receive pioglitazone 15 mg in Part A (as monotherapy or SPC) and who continued to Part B received pioglitazone 30 mg as monotherapy or SPC (according to previous Part A assignment). Patients in all other treatment groups were to remain on the same treatment in Part B (Figure 1).
There were several changes to the protocol during the conduct of this study. New safety assessments and considerations were implemented based on the US Food and Drug Administration (FDA) and European Medicines Agency (EMA) drug safety communications concerning pioglitazone use and an increased risk for bladder cancer.16,17 The study sponsor made the decision to discontinue development of the linagliptin and pioglitazone SPC clinical programme. With this amendment, all patients concluded study participation after 30 weeks of treatment (Part A), the time point for the pre-defined primary efficacy endpoint. No further patients were entered into Part B. Patients in Part B at the time of the amendment terminated study participation by completing final visit procedures.
Rescue therapy was permitted for confirmed hyperglycaemia, defined as two or more measurements on different days using the following criteria for fasted glucose levels: FPG > 240 mg/dL (>13.3 mmol/L) during weeks 1–12; FPG > 200 mg/dL (>11.1 mmol/L) during weeks 12–30; FPG > 180 mg/dL (>10.0 mmol/L) and/or HbA1c > 8.0% during weeks 30–85. Metformin (first choice unless contraindicated) or a sulphonylurea was prescribed at the investigator’s discretion and according to local prescribing information. Patients were discontinued from the trial if FPG remained above threshold levels.
Study endpoints
The primary endpoint was HbA1c change from baseline after 30 weeks of treatment. Secondary endpoints were HbA1c change from baseline at other time points; FPG change from baseline after 30 weeks and at other time points; percentage of patients achieving HbA1c < 7.0% or <6.5%, or a ⩾0.5% reduction from baseline after 30 weeks; and incidence and time to the first use of rescue therapy. Additional endpoints included change from baseline in body weight after 30 weeks.
Safety endpoints included the frequency and intensity of AEs, which were coded using the Medical Dictionary for Drug Regulatory Affairs (MedDRA) version 15.1. Protocol-specified significant AEs were reported and defined according to narrow standardised MedDRA queries (SMQs): hypersensitivity reactions, selected hepatic AEs, selected renal AEs, cutaneous skin lesions and pancreatitis. Hypoglycaemic episodes with blood glucose levels <70 mg/dL (<3.9 mmol/L) were documented by the investigator according to protocol classifications. Any episode with glucose levels <54 mg/dL (<3.0 mmol/L) and all symptomatic and severe hypoglycaemias were to be documented as an AE of ‘hypoglycaemic event’. Other safety endpoints were changes from baseline in vital signs, clinical laboratory values and 12-lead electrocardiograms; and events qualifying for external adjudication by the clinical event committee (CEC) selected based on all fatal cases and SMQs for ischaemic heart disease, cerebrovascular disorders and cardiac failure.
Statistical analyses
All hypotheses testing for this trial must be considered exploratory rather than confirmatory subsequent to early termination of the trial. SAS version 9.2 was used for all analyses. Based on the expected treatment-effect sizes, a sample size of 128 patients per group was planned to afford 94% power to detect an estimated difference of 0.5% in HbA1c change from baseline at 30 weeks between linagliptin monotherapy and any one of the SPC combinations, and >99% power to detect an estimated difference of 0.8%–1.0% in HbA1c change from baseline at 30 weeks between each dose of pioglitazone monotherapy and the corresponding SPC combination, using two-sided t-tests with a significance level (α) of 0.05, assuming a standard deviation (SD) for HbA1c change from baseline after 30 weeks of 1.1%, and with 5% of patients being assumed non-evaluable. Furthermore, this planned to provide 81% power for the full hypothesis testing hierarchy specified in the trial protocol (i.e. all three SPCs versus both linagliptin and pioglitazone), under the most conservative assumption of independent tests.
The primary efficacy analysis was an analysis of covariance (ANCOVA) of the mean HbA1c change from baseline after 30 weeks of treatment in Part A. This analysis was performed on all randomised patients who had received at least one dose of study medication and had a baseline and at least one on-treatment HbA1c measurement [full analysis set (FAS)]. The statistical model included ‘prior use of OADs’, ‘country’ and ‘Part A treatment’ as fixed classification effects, and ‘baseline HbA1c’ as a linear covariate. From this statistical model, 95% confidence intervals (CIs) and p-values for the comparisons of each of the SPCs of linagliptin plus pioglitazone versus the linagliptin and corresponding pioglitazone monotherapies were determined. A last observation carried forward (LOCF) approach was used to replace missing data; values recorded after the initiation of rescue therapy were set to missing prior to the application of LOCF. Various sensitivity analyses were performed, which included the primary analysis repeated for the FAS using an LOCF-rescue observed cases (LOCF-ROC) approach, in which values recorded after the initiation of rescue therapy were retained prior to the application of LOCF.
Secondary efficacy endpoints were analysed using the FAS. Change in FPG from baseline after 30 weeks was analysed using ANCOVA similar to the primary analysis with an LOCF approach and with the same terms as the primary analysis model plus ‘baseline FPG’ as a linear covariate. Changes in HbA1c and FPG levels over time were analysed using a mixed model for repeated measures and descriptive statistics and based on observed cases (OC; patients with missing values were excluded). HbA1c responder endpoints (<7.0% and <6.5% and decrease from baseline >0.5%) were analysed using logistic regression modelling to estimate odds ratios (ORs; together with 95% CI) using a non-completers considered failures (NCF) approach to missing data at 30 weeks. Change in body weight from baseline after 30 weeks was analysed using ANCOVA similar to the primary analysis (based on OC approach) and with the same terms as the primary analysis model plus ‘baseline body weight’ as a linear covariate. Time-to-first use of rescue therapy during Part A was analysed using Kaplan–Meier estimates.
Safety data were analysed for the treated set (TS) (all randomised patients who received at least one dose of study medication) in Parts A and B combined using descriptive statistics.
Results
Of 1849 patients who were screened, 936 were randomised to treatment and all received at least one dose of study medication (the TS; Figure 2). The FAS excluded 43 treated patients and comprised 893 patients. Mean exposure to study drug for each treatment group ranged from 180.8 to 189.8 days in Part A (median, 210.0 days in all groups). In Parts A and B combined, mean exposure for each treatment group ranged from 297.5 to 334.3 days (median, 248.0–337.0 days), although there was considerable between-patient variability in the overall length of exposure because of the impact of protocol amendments. In each treatment group, <12% of patients received treatment for >84 weeks.
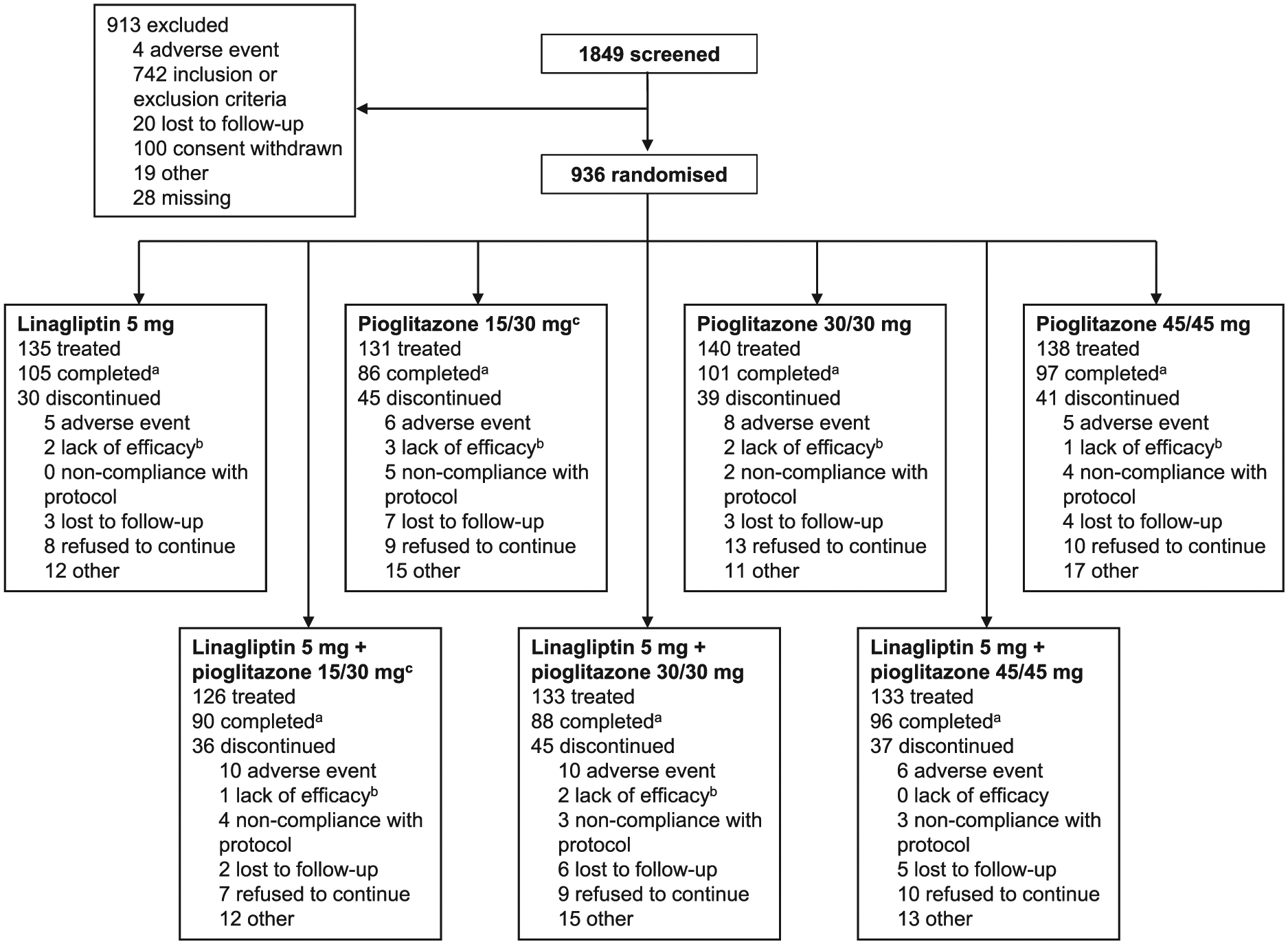
Patient disposition (irrespective of the timing relative to protocol amendments).
Baseline demographic and clinical characteristics were generally similar between treatment groups (Table 1). Mean body weight was highest (96.9 kg) in patients treated with pioglitazone 45 mg (Table 1). Mean (SD) HbA1c and FPG levels were 8.11% (0.91) and 166.3 (39.9) mg/dL [9.2 (2.2) mmol/L], respectively. The majority of patients were White (85.1%) and were not receiving antidiabetes treatment at the time of enrolment (66.1%). Most patients (approximately 70%) had been diagnosed with T2D for ⩽5 years.
Patient demographics and clinical characteristics at baseline.
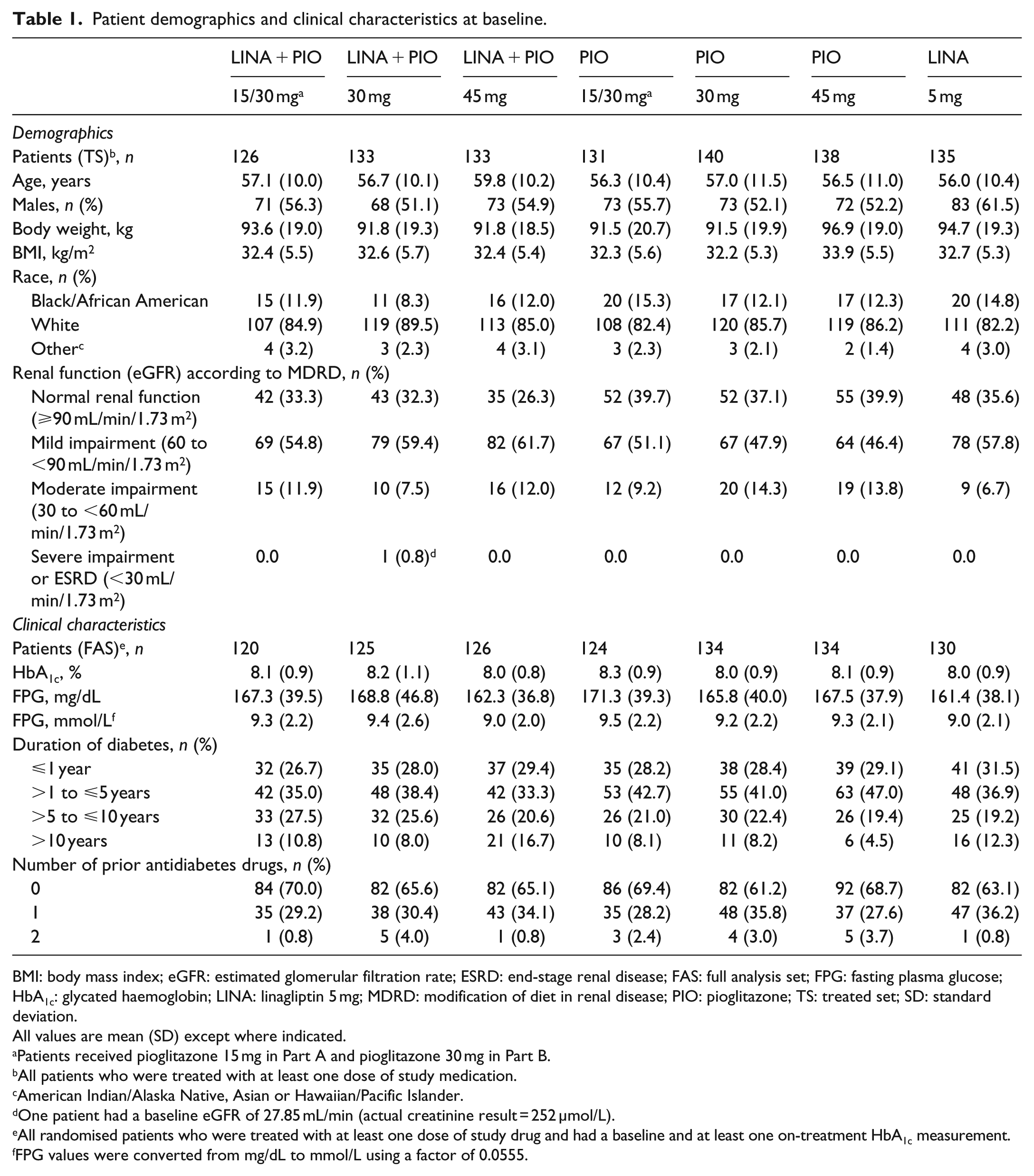
BMI: body mass index; eGFR: estimated glomerular filtration rate; ESRD: end-stage renal disease; FAS: full analysis set; FPG: fasting plasma glucose; HbA1c: glycated haemoglobin; LINA: linagliptin 5 mg; MDRD: modification of diet in renal disease; PIO: pioglitazone; TS: treated set; SD: standard deviation.
All values are mean (SD) except where indicated.
Patients received pioglitazone 15 mg in Part A and pioglitazone 30 mg in Part B.
All patients who were treated with at least one dose of study medication.
American Indian/Alaska Native, Asian or Hawaiian/Pacific Islander.
One patient had a baseline eGFR of 27.85 mL/min (actual creatinine result = 252 µmol/L).
All randomised patients who were treated with at least one dose of study drug and had a baseline and at least one on-treatment HbA1c measurement.
FPG values were converted from mg/dL to mmol/L using a factor of 0.0555.
Efficacy
After 30 weeks of treatment (Part A), all SPC treatment groups showed statistically significantly greater reductions in HbA1c levels from baseline than their respective monotherapy groups, with the exception of linagliptin plus pioglitazone 15 mg SPC versus pioglitazone 15 mg monotherapy (Figure 3(a)). The mean (95% CI) treatment differences for linagliptin plus pioglitazone 15 mg SPC versus pioglitazone 15 mg and linagliptin monotherapy were −0.17% (−0.41, 0.07; p = 0.1571) and −0.44% (−0.67, −0.20; p = 0.0003), respectively; for linagliptin plus pioglitazone 30 mg SPC versus pioglitazone 30 mg and linagliptin monotherapy were −0.37% (−0.60, −0.14; p = 0.0016) and −0.68% (−0.91, −0.44; p < 0.0001), respectively; and for linagliptin plus pioglitazone 45 mg SPC versus pioglitazone 45 mg and linagliptin monotherapy were −0.41% (−0.64, −0.18; p = 0.0006) and −0.89% (−1.12, −0.66; p < 0.0001), respectively. The results from sensitivity analyses were consistent with the primary analysis; Figure 3(b) shows the results of a sensitivity analysis in the FAS in which values recorded after the use of rescue medication were retained prior to the application of LOCF (LOCF-ROC).
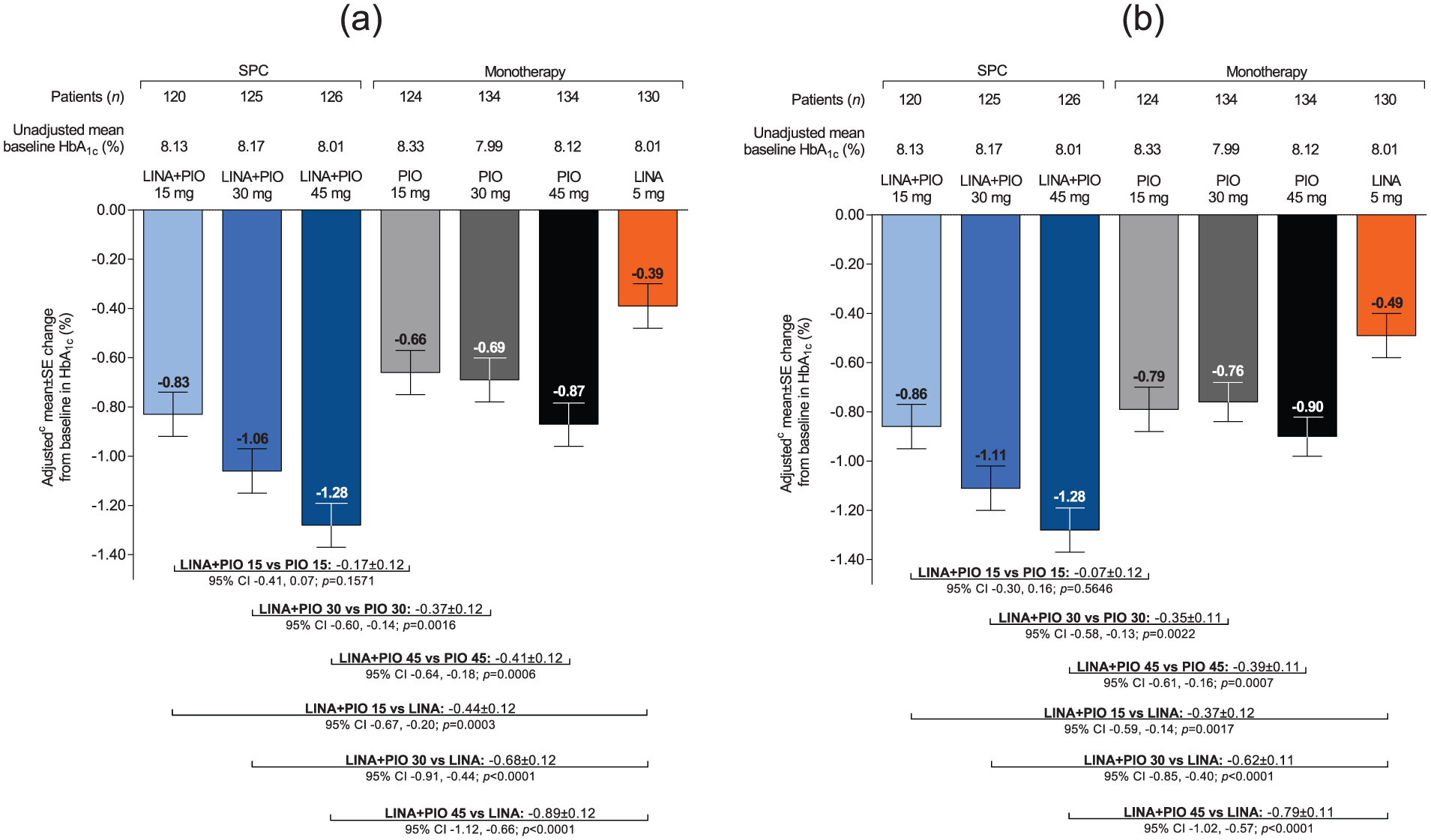
Adjusted mean HbA1c changes from baseline and adjusted mean differences between SPC treatments and respective monotherapies after 30 weeks (a) FAS; LOCFa and (b) FAS; LOCF-ROCb.
Mean changes in HbA1c levels over time are shown in Figure 4(a). The majority of the reduction occurred within the first 18 weeks and was maintained until week 30. In patients with baseline HbA1c ⩾ 7%, the percentage of patients who achieved a target HbA1c of <7% at week 30 ranged from 40.2% to 66.9% in the SPC treatment groups, 32.2% to 52.7% in the pioglitazone monotherapy groups and was 23.4% with linagliptin monotherapy (Figure 4(b)). In patients with baseline HbA1c ⩾ 6.5%, the percentage of patients who achieved a target HbA1c of <6.5% at week 30 ranged from 20.0% to 34.4% in the SPC treatment groups, 16.1% to 29.1% in the pioglitazone monotherapy groups and was 10.9% with linagliptin monotherapy (Supplementary Figure S1). The percentage of patients who achieved a reduction in HbA1c of ⩾0.5% ranged from 65.8% to 84.9% in the SPC treatment groups, 61.9% to 67.2% in the pioglitazone monotherapy groups and was 41.5% with linagliptin monotherapy (Supplementary Figure S2).
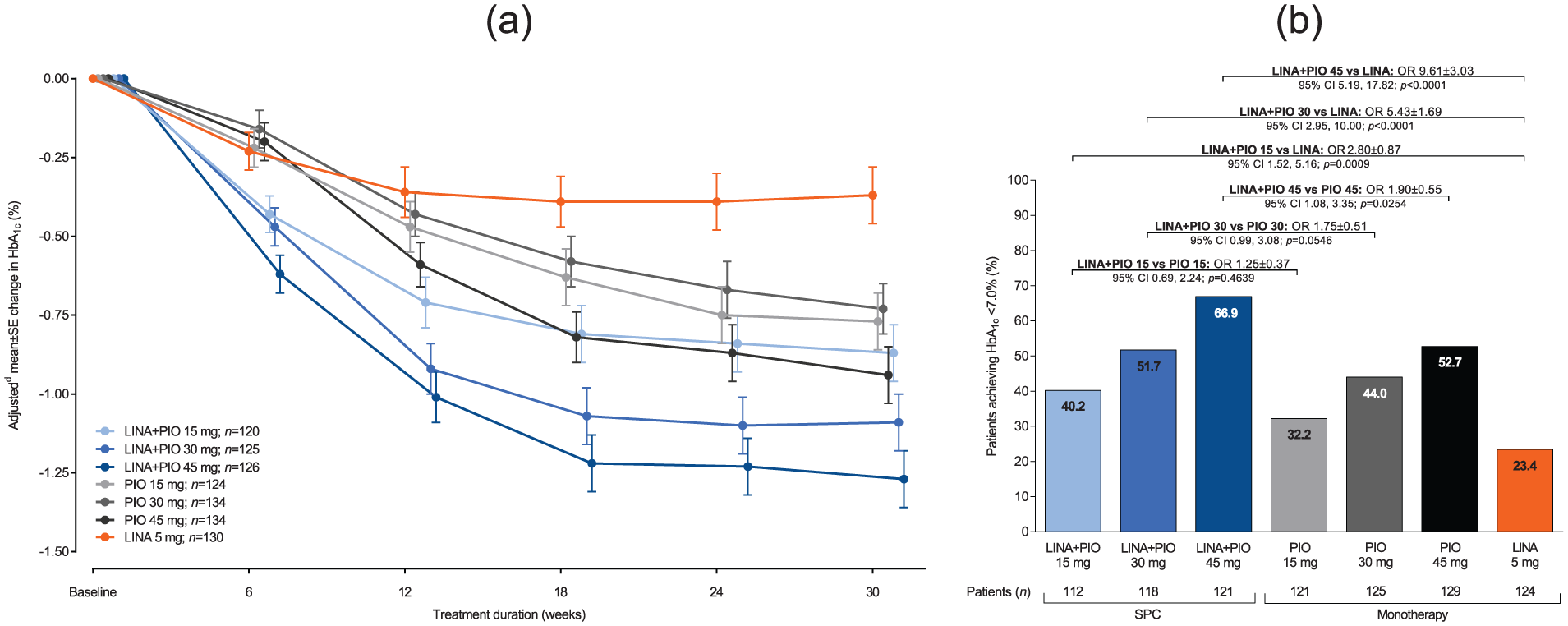
(a) Adjusted mean change from baseline in HbA1c over time (FAS; OCa) and (b) within-group frequencies and between-group comparisons between SPC treatments and the respective monotherapies of patients achieving HbA1c < 7.0% after 30 weeksb (FAS; NCFc).
After 30 weeks of treatment (Part A), all SPC treatment groups showed statistically significant and clinically meaningful reductions in FPG levels from baseline versus linagliptin monotherapy (Figure 5(a)). Overall, the reductions in FPG levels were near-maximal by weeks 6–12 (Supplementary Figure S3).
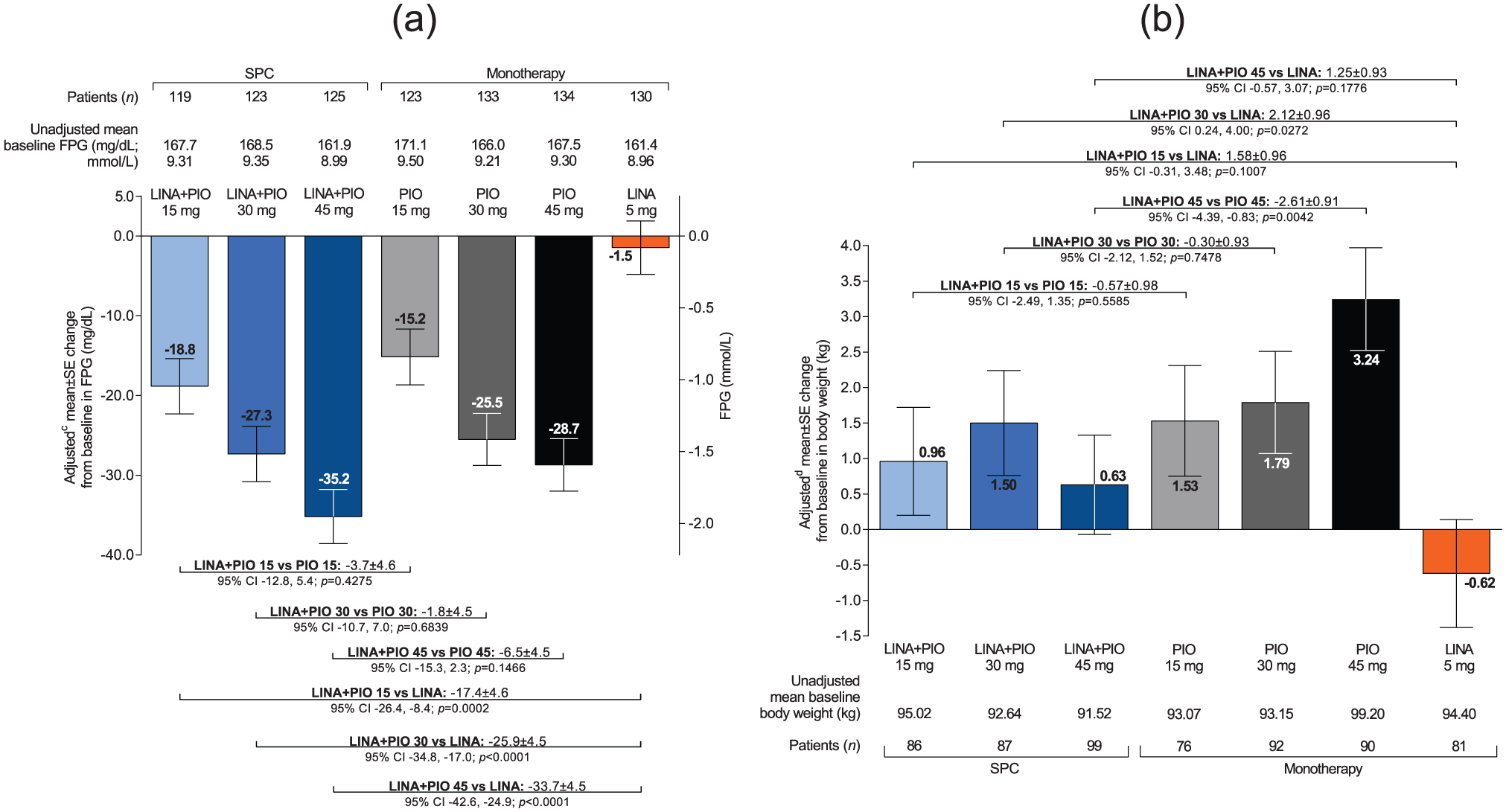
(a) Adjusted mean FPG changes from baseline and adjusted mean differences between SPC treatments and the respective monotherapies after 30 weeks (FAS; LOCFa) and (b) adjusted mean body weight changes from baseline and adjusted mean differences between SPC treatments and the respective monotherapies after 30 weeks (FAS; OCb).
The proportion of patients requiring rescue therapy during Part A was small and lower among patients receiving SPC therapy (linagliptin plus pioglitazone 15 mg, 10.0%; linagliptin plus pioglitazone 30 mg, 8.0%; linagliptin plus pioglitazone 45 mg, 4.0%) than among patients receiving the corresponding monotherapies (pioglitazone 15 mg, 16.1%; pioglitazone 30 mg, 12.7%; pioglitazone 45 mg, 8.2%; linagliptin 5 mg, 20.0%).
Changes in mean body weight from baseline to week 30 and between-group comparisons are shown in Figure 5(b). Compared with linagliptin monotherapy, the adjusted mean differences in body weight change from baseline in the SPC treatment groups ranged from +1.25 kg with linagliptin plus pioglitazone 45 mg to +2.12 kg with linagliptin plus pioglitazone 30 mg (Figure 5(b)). In contrast, the adjusted mean differences in body weight change between the SPC treatment groups and the respective pioglitazone monotherapies ranged from −0.30 kg with linagliptin plus pioglitazone 30 mg to −2.61 kg with linagliptin plus pioglitazone 45 mg (Figure 5(b)).
Safety
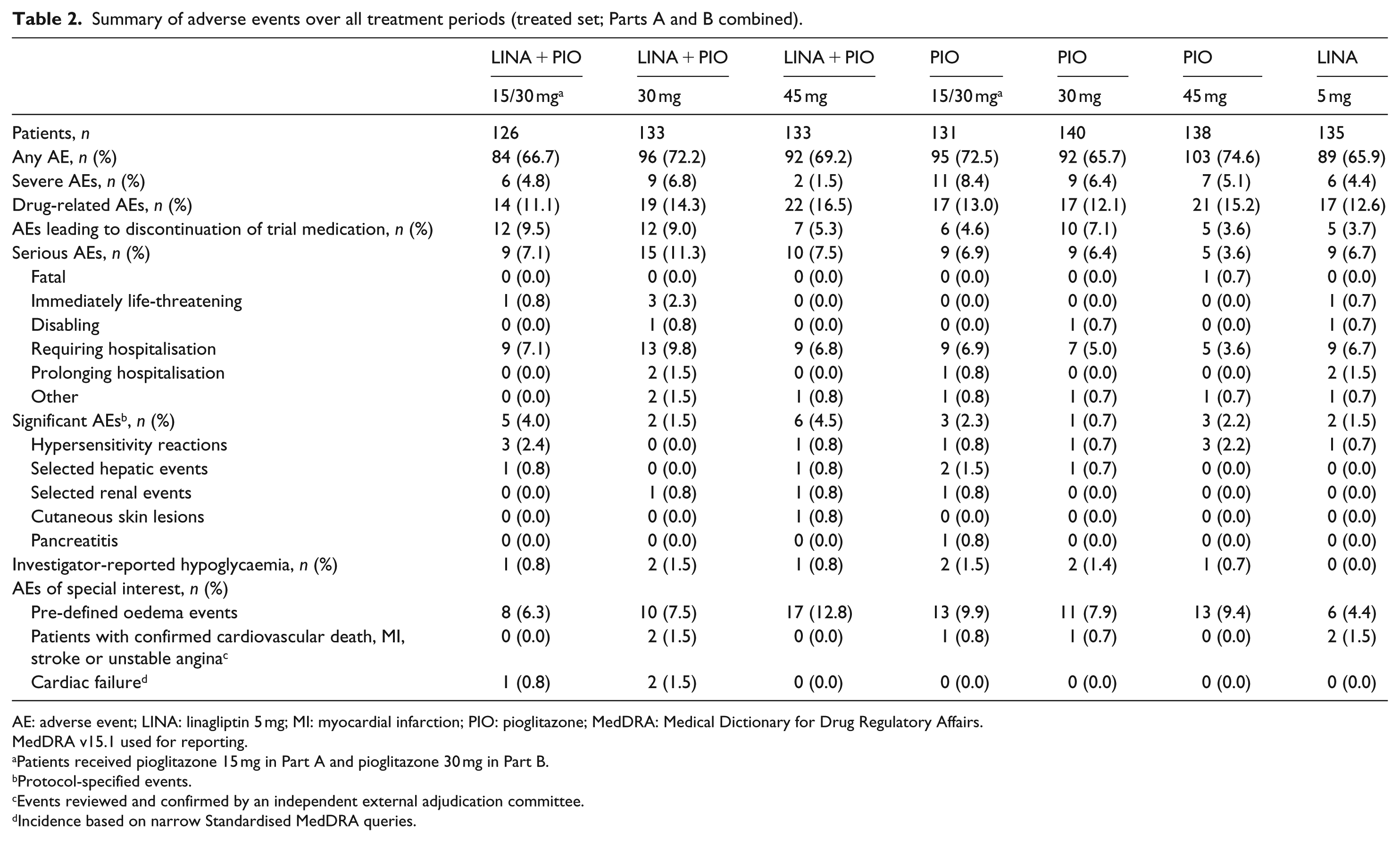
Safety data were analysed for Parts A and B combined (936 patients – the TS; Table 2). Exposure-adjusted incidence rates were reported for the following AE summaries: overall AEs ranged from 151.4 per 100 patient-years with pioglitazone 30 mg to 226.3 per 100 patient-years with pioglitazone 15/30 mg; drug-related AEs ranged from 12.8 per 100 patient-years with linagliptin plus pioglitazone 15/30 mg SPC to 20.8 per 100 patient-years with linagliptin plus pioglitazone 45 mg SPC; AEs leading to discontinuation ranged from 4.0 per 100 patient-years with linagliptin to 10.5 per 100 patient-years with linagliptin plus pioglitazone 15/30 mg SPC; and serious AEs ranged from 4.1 per 100 patient-years with pioglitazone 45 mg to 13.4 per 100 patient-years with linagliptin plus pioglitazone 30 mg SPC. One fatality occurred during the treatment period in the pioglitazone 45 mg group: a 74-year-old patient experienced colon cancer on day 313 of treatment (concurrent conditions included tumour invasion, anaemia and fatigue); the patient also experienced a post-treatment episode of acute pulmonary oedema. The colon cancer and associated conditions were not attributed to the study drug. Overall, vital signs (including systolic and diastolic blood pressure and pulse rate) remained consistent across study groups and throughout the treatment period.
Summary of adverse events over all treatment periods (treated set; Parts A and B combined).
AE: adverse event; LINA: linagliptin 5 mg; MI: myocardial infarction; PIO: pioglitazone; MedDRA: Medical Dictionary for Drug Regulatory Affairs.
MedDRA v15.1 used for reporting.
Patients received pioglitazone 15 mg in Part A and pioglitazone 30 mg in Part B.
Protocol-specified events.
Events reviewed and confirmed by an independent external adjudication committee.
Incidence based on narrow Standardised MedDRA queries.
The frequency of investigator-reported hypoglycaemia was ⩽1.5% across all treatment groups, and there were no hypoglycaemic events with linagliptin monotherapy (Table 2). Only one patient (0.8%) in the study experienced severe hypoglycaemia – an individual in the linagliptin plus pioglitazone 15/30 mg SPC group (Supplementary Table S1). Protocol-specified significant AEs occurred at ⩽4.5% across the treatment groups (Table 2); one patient (0.8%) in the pioglitazone 15/30 mg group reported pancreatitis. The frequency of pre-defined oedema events was highest with linagliptin plus pioglitazone 45 mg SPC (12.8%) and lowest with linagliptin monotherapy (4.4%) (Table 2). CEC-confirmed cardiovascular events occurred in two patients in the linagliptin plus pioglitazone 30 mg SPC group and two patients in the linagliptin monotherapy group; one patient in the pioglitazone 15/30 mg group and one patient in the pioglitazone 30 mg group (Table 2). Cardiac failure (narrow SMQ) was infrequent and occurred in two patients in the linagliptin plus pioglitazone 30 mg SPC group and one patient in the linagliptin plus pioglitazone 15/30 mg SPC group. No cases of bladder cancer were reported.
Discussion
Glycaemic control is the cornerstone of antidiabetes therapy. Prolonged periods of uncontrolled hyperglycaemia should be avoided because of the expected adverse effects on long-term outcomes. 7 However, the treatment strategy of initial monotherapy followed by the step-wise intensification to dual or triple therapy regimens, which is generally advocated in current guidelines,2,3 may not address sufficiently the progressive and multifactorial nature of T2D. Consequently, many patients fail to reach or adequately maintain target glucose levels in the long term.18–20 In a study of European outpatients, only 26% of patients had adequate glycaemic control (HbA1c < 6.5%) after 2.6 years following initiation of dual OAD therapy (addition of a sulphonylurea or thiazolidinedione to the ongoing metformin therapy). 18 Furthermore, despite suboptimal glycaemic control, step-wise treatment intensification is often delayed for several years, 6 well beyond the ~3 months specified in guidelines.2,3
In this study of patients with insufficient glycaemic control, initial treatment with linagliptin plus pioglitazone SPC was efficacious and well tolerated after 30 weeks of treatment. Clinically meaningful and statistically significant decreases in HbA1c levels were seen with SPC treatments versus linagliptin monotherapy; comparisons versus the respective pioglitazone monotherapies were also clinically meaningful and statistically significant except for linagliptin plus pioglitazone 15 mg, which showed a numerical difference in favour of the SPC. Although formal statistical comparisons were not planned or performed, an informal comparison suggests that decreases in HbA1c levels with the lower dose SPCs containing pioglitazone 15 or 30 mg were similar to high-dose pioglitazone 45 mg monotherapy, implying a modest dose-sparing effect with pioglitazone when used in combination with linagliptin. Compared with monotherapy treatments, there was an additive effect on glycaemic control when linagliptin was combined with pioglitazone 30 or 45 mg, which is consistent with targeting two core pathophysiologies in T2D using two different drug classes with complementary mechanisms of action. 8 This enhanced glucose-lowering effect has been shown in studies with pioglitazone in initial free combination with other DPP-4 inhibitors in patients with uncontrolled hyperglycaemia.21–23 The additive effect was not evident with the low-dose SPC in our study.
There is evidence that initial combination therapy may have potential clinical benefits compared with monotherapy.8,24 Besides improving microvascular outcomes, 4 early intensive glucose-lowering strategies may slow the onset and progression of T2D25–28 and may have long-term ‘legacy’ benefits with respect to reductions in cardiovascular disease risks. 29 Early combination therapy may also help minimise the consequences of clinical inertia, which is a major barrier to optimal diabetes management, 6 by potentially avoiding early treatment failure that is often seen with initial monotherapy. 30 However, current treatment guidelines recommend initial combination therapy with OADs only in newly diagnosed patients presenting high baseline HbA1c.2,3 Although this study did not evaluate patients with marked hyperglycaemia, a previous study showed that the initial combination of linagliptin plus metformin achieved large reductions in HbA1c levels in patients with a mean baseline HbA1c level of 9.8%, without increasing hypoglycaemia or other adverse effects compared with linagliptin monotherapy. 31
The improvements in efficacy from using an intensive combination treatment strategy should not be at the expense of worsening safety and tolerability profiles. In this study, there were no clinically meaningful differences in the frequencies of AEs between treatment groups. The overall safety profiles of the SPCs were similar to the known safety profiles of the individual components, and no new unexpected safety findings emerged. An important concern when combining antidiabetes treatments is the increased risk for hypoglycaemia. 2 Similar to the previous study of linagliptin plus pioglitazone combination, 14 the incidence of hypoglycaemia in the SPC groups was low in this study (⩽1.5% per arm). This result was anticipated because the DPP-4 inhibitor and thiazolidinedione drug classes are associated with an inherently low risk of hypoglycaemia. 2 Pioglitazone can cause oedema when used alone or in combination with other antidiabetes drugs. 15 Oedema may also lead to or exacerbate congestive heart failure, a known side effect of thiazolidinedione use. 15 Both of these side effects occurred at a low frequency in the pioglitazone-containing arms. Body weight gain, probably due to a combination of oedema and subcutaneous fat accumulation, is another safety concern associated with thiazolidinedione use 15 and, in our study, body weight increased from baseline in all groups that received pioglitazone as either monotherapy or in combination. Mean body weight changes from baseline were lower in the SPC groups versus the respective pioglitazone monotherapies; in contrast, mean body weight changes in the SPC groups were greater than in the linagliptin monotherapy group. Overall, these changes are consistent with the effects of linagliptin and pioglitazone on body weight. 2
Studies have shown that strict glycaemic control requires close adherence to treatment regimens. 32 However, adherence to prescribed medication is an aspect of treatment that is often overlooked in clinical practice; compliance with OAD regimens varies widely, ranging from approximately 40% to 90%.32–34 Paradoxically, to achieve and maintain glycaemic control eventually requires combination treatment with multiple drugs, resulting in a greater pill burden, which can be associated with poor adherence to therapy. 9 Combining multiple drugs in a single-pill formulation can benefit patients through the potential synergistic or additive increases in efficacy, improved tolerability using lower doses and convenience in terms of administration and increased patient compliance, as well as reducing direct medical costs.10,35 Despite these benefits, many patients and prescribers prefer dual therapy with separate pill combinations rather than SPC.36–38 Both linagliptin and pioglitazone are administered once daily, thereby allowing them to be taken together – either as an SPC or in free combination – to provide a simpler pill regimen.
The 54-week extension study was terminated early and, consequently, the number of patients completing this phase was low. Therefore, treatment durability beyond 30 weeks could not be meaningfully assessed here. The study was also discontinued in Germany (August 2011) after regulatory authority concern regarding the potential risk of bladder cancer with pioglitazone. 15 In addition, the FDA warned in June 2011 that the use of pioglitazone for more than 1 year may be associated with an increased risk of bladder cancer. The FDA recommended that pioglitazone should not be used in people with active bladder cancer and should be used with caution in people with a history of bladder cancer. This warning also applies to combination diabetes pills containing pioglitazone. 16 However, it should be noted that a 10-year study showed recently that there was no increased risk of bladder cancer among patients exposed to pioglitazone. 39 No incidences of bladder cancer were observed in the current study.
In conclusion, the current recommended treatment paradigm for T2D, involving the step-wise introduction of additional antidiabetes drugs at the point of therapeutic failure, may only partially address the progressive nature of T2D. The results of this study support an alternative approach using initial combination therapy to simultaneously target multiple underlying pathophysiologies. Initial combination with linagliptin plus pioglitazone in a single-pill formulation is an efficacious and well-tolerated therapeutic option. The improvements in glycaemic control with this combination are likely due to the complementary mechanisms of action of the two drugs. The safety profile is comparable to the known safety profiles of the individual monotherapies, with a minimal risk of hypoglycaemia.
Footnotes
Acknowledgements
Data in this manuscript were presented at the 50th Annual Meeting of the European Association for the Study of Diabetes, Vienna, Austria, 15–19 September 2014. The authors were fully responsible for all content and editorial decisions, were involved at all stages of manuscript development and have approved the final version. Medical writing assistance, supported financially by Boehringer Ingelheim, was provided by Paul MacCallum, PhD of Envision Scientific Solutions, during the preparation of this manuscript.
Declaration of conflicting interests
Professor Michael Albrecht Nauck has received research grants [to his former institution, the Diabeteszentrum Bad Lauterberg, and his present institution, the St. Josef- and St. Elisabeth-Hospital gGmbH (Ruhr-University Bochum), Bochum, Germany] from Berlin-Chemie AG/Menarini, Eli Lilly & Co, Merck Sharp & Dohme and Novartis Pharma AG (mono- or oligocentric studies) and from AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Lilly Deutschland GmbH, MetaCure Inc, Roche Pharma AG, Novo Nordisk Pharma GmbH and Tolerx Inc for participation in multicentric clinical trials. He has received consulting fees and/or honoraria for membership in advisory boards and/or honoraria for speaking for Amylin Pharmaceuticals Inc, AstraZeneca, Berlin-Chemie AG/Menarini, Boehringer Ingelheim, Bristol-Myers Squibb EMEA, Diartis Pharmaceuticals, Inc., Eli Lilly & Co, F. Hoffmann-La Roche Ltd, GlaxoSmithKline LLC, Intarcia Therapeutics, Inc., Lilly Deutschland GmbH, MannKind Corp., Merck Sharp & Dohme GmbH, Merck Sharp & Dohme Corp., Novartis Pharma AG, Novo Nordisk A/S, Novo Nordisk Pharma GmbH, Sanofi-Aventis Pharma, Takeda, Versartis and Wyeth Research, including reimbursement for travel expenses in connection with the above-mentioned activities. He owns no stock and is employed by the St. Josef- and St. Elisabeth-Hospital gGmbH (Ruhr-University Bochum), Bochum, Germany. M.d.D., M.K. and H-J.W. are employees of Boehringer Ingelheim. R.T. is an independent contractor who received payment from Boehringer Ingelheim for his work on this trial. S.P. was an employee of Boehringer Ingelheim Ltd at the time of the study and is now an employee of Daiichi Sankyo Development Ltd.
Funding
This study was supported by Boehringer Ingelheim and Eli Lilly and Company.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
