Abstract
Although management of hyperglycaemia represents one of the principal treatment goals of diabetes therapy, the high incidence of cardiovascular (CV) complications in diabetes also needs effective management. Therefore, the present study was designed to determine and compare the effect of glitazones on serum sialic acid (SSA), a known risk marker for CV disease, along with fasting plasma glucose (FPG), glycohaemoglobin (HbA1-c) and blood lipids, in overweight, previously only diet-treated patients with type 2 diabetes (n=60). The study was conducted for a period of 12 months. Significant improvement in FPG and HbA1-c were shown by both rosiglitazone (p<0.003 and p<0.001, respectively) and pioglitazone (p<0.005 and p<0.001, respectively), compared with baseline, and pioglitazone showed greater beneficial effects on other parameters monitored, significantly reducing total cholesterol (TC) (p≤0.05). Both the drugs showed a decrease in SSA and no significant differences were observed between the groups. However, the decrease was significant only in the pioglitazone-treated group at month 12 (p≤0.05), compared with baseline. A significant decrease in SSA by pioglitazone indicates its greater cardioprotective effect compared with rosiglitazone.
Introduction
Cardiovascular (CV) diseases are the main cause of death in patients with diabetes, and therefore an ideal hypoglycaemic agent would prevent or delay the occurrence of CV complications in diabetes along with achieving normoglycaemia. Sialic acid, an acetylated derivative of neuraminic acid, is known to increase in type 2 diabetes mellitus,1,2 and has been shown to be a good predictor of CV mortality.3,4 The evidence of raised levels of serum sialic acid (SSA) as a marker for CV disease signifies the importance of its status in type 2 diabetes mellitus, and the present study attempts to investigate and compare the effects of glitazones (rosiglitazone and pioglitazone) on SSA along with other CV risk factors. It is interesting to compare the effect of these two glitazones on SSA because, despite the same mechanism of action, rosiglitazone is associated with increased CV mortality. 5
Subjects and methods
Recently diagnosed (≤2 years) patients with type 2 diabetes previously managed with diet/exercise only and free from diabetic complications at the time of investigation were randomly assigned to rosiglitazone (n=30) or pioglitazone (n=30) monotherapy. This comparative parallel group trial consisted of a 4-week run-in period that included reinforcement of lifestyle education to obtain fasting plasma glucose (FPG) between 126–240 mg/dl and a 12-month treatment period. The research protocol was explained to the volunteer patients, and their written informed consents were obtained for the study. The median daily dose was 4 mg/day and 15 mg/day for rosiglitazone and pioglitazone, respectively. The dose was increased if the FPG level was ≥140 mg/dl and reduced if adverse reactions occurred. The therapeutic goal was to achieve FPG level below 140 mg/dl while observing the changes in SSA.
Sialic acid was determined by using a method proposed by Shamberger. 6 In brief, sialic acid in the sample reacts with Ehrlich’s reagent, and this results in the formation of a white precipitate. After incubation in a water bath, the colour of the mixture turns from white to blue. Sodium chloride is added to the mixture, which is then centrifuged. The intensity of the colour of the supernatant is directly proportional to the concentration of sialic acid and is read in a spectrophotometer at 525 nm.
FPG, HbA1-c, total cholesterol (TC), high-density lipoprotein cholesterol (HDL-c), low-density lipoprotein cholesterol (LDL-c) and triglyceride (TG) levels were analysed by enzymatic methods, using commercially available kits.
Results are expressed as means ±SD. Data were analysed using the Statistical Package for Social Sciences (SPSS) and a р≤0.05 was taken as the cut-off level for significance. Because the distribution of most variables was not symmetrical, we used non-parametric statistical methods. The Mann–Whitney U test was used for between-group comparisons.
Results
Table 1 shows that type 2 diabetic patients in both groups were overweight, with no significant differences in the baseline values of metabolic variables.
Baseline and post-treatment biochemical characteristics of type2 diabetic patients following rosiglitazone and pioglitazone monotherapy
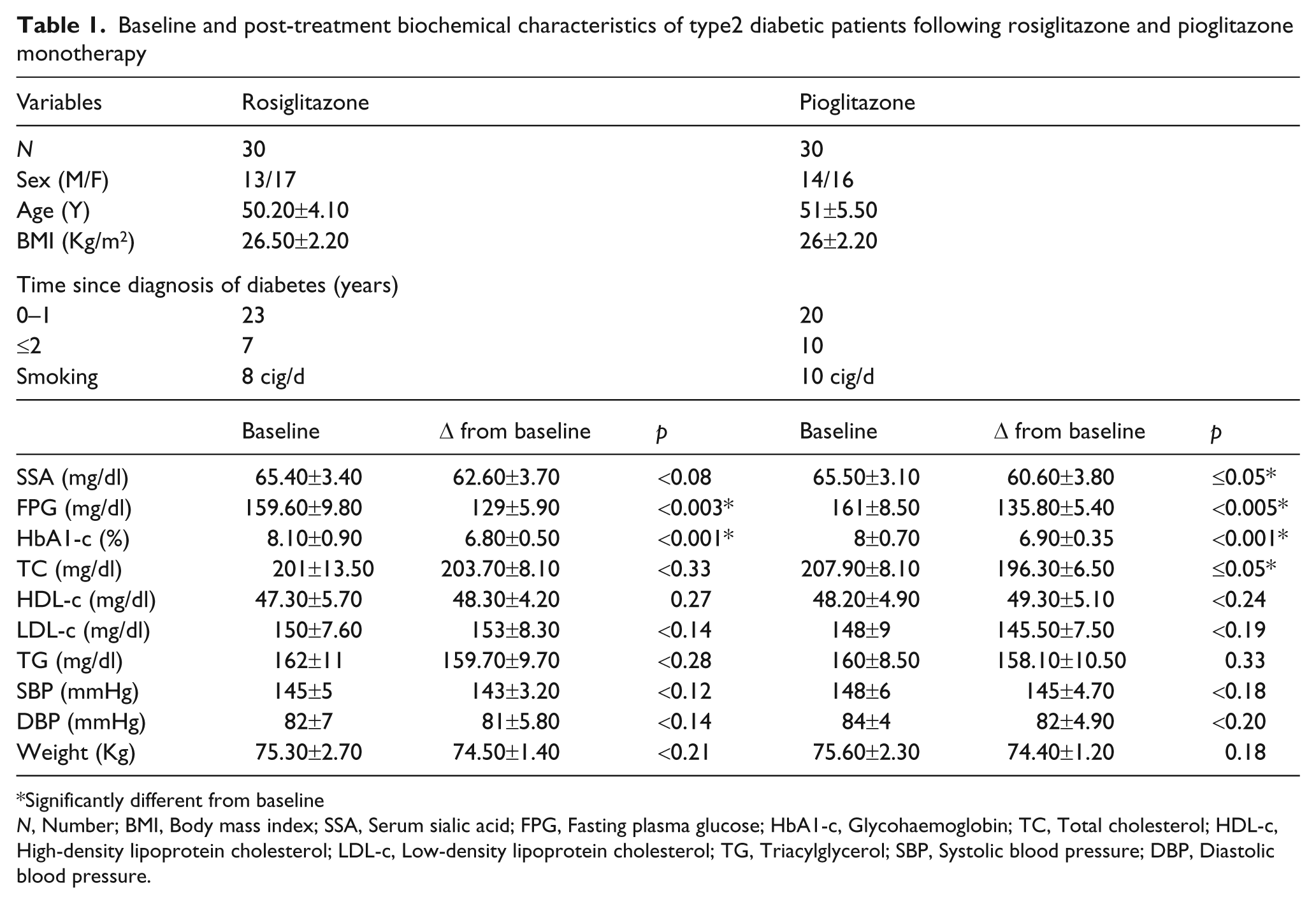
Significantly different from baseline
N, Number; BMI, Body mass index; SSA, Serum sialic acid; FPG, Fasting plasma glucose; HbA1-c, Glycohaemoglobin; TC, Total cholesterol; HDL-c, High-density lipoprotein cholesterol; LDL-c, Low-density lipoprotein cholesterol; TG, Triacylglycerol; SBP, Systolic blood pressure; DBP, Diastolic blood pressure.
Compared with baseline, SSA showed a trend towards decreased level in both the groups; however, the decrease was non-significant in the rosiglitazone group (<0.08), but significant in pioglitazone group (≤0.05) at month 12. Moreover, post-treatment between-group comparisons revealed no significant difference.
Statistically significant reductions in both FPG and HbA1-c were observed in the rosiglitazone (<0.003 and <0.001, respectively) and pioglitazone group (<0.005 and <0.001, respectively) at month 12 (Table1).
The beneficial effects of pioglitazone were greater than rosiglitazone on blood lipids. TC changed by +2.70 mg/dl and -11.60 mg/dl. HDL-c changed by +1 mg/dl and +1.10 mg/dl. LDL-c changed by +3 mg/dl and -2.50 mg/dl. TG decreased by 2.30 mg/dl and 1.90 mg/dl in the rosiglitazone and pioglitazone groups, respectively. The effect on blood pressure (systolic and diastolic) was non-significant (<0.12, <0.18 and <0.14, <0.20, respectively), as was the case with body weight (<0.21 and 0.18, respectively), compared with baseline.
Discussion
Although the theoretical ideal for patients with diabetes is to achieve normoglycaemia, other diabetes-associated complications also need effective management, including high SSA levels that contribute to progression of CV complications in diabetes mellitus. 4 Therefore, a decrease in SSA may be related to a decrease/delay in the process of atherosclerosis. The results of the present study indicate that SSA levels are decreased by glitazones in type 2 diabetics, although non-significantly by rosiglitazone, compared with baseline. This decrease in SSA may be the result of improvement in blood glucose levels that affects the hexosamine pathway, leading to a decrease in the synthesis of amino sugars. These results are supported well by a previous study showing that sialic acid reflects the status of glycaemic control and the progression of ischaemic disease of the lower extremities in type 2 diabetes. 7 Recent studies indicate sialic acid as an independent risk factor for CVD, 4 and a significant decrease as shown by pioglitazone in this study would be helpful to reduce or delay the incidence of CV complications in diabetes, because the predictive power of sialic acid for coronary heart disease is better, or of the same magnitude, than that of cholesterol. 8
The study further shows that the decrease was smaller and statistically non-significant in patients following rosiglitazone monotherapy, compared with baseline, and these results are in agreement with our recent findings that shows lesser beneficial effects of rosiglitazone on SSA than metformin. 9 A significant decrease in the pioglitazone group may be the result of greater beneficial effects of pioglitazone on blood lipids. A positive correlation between SSA and blood lipids has been found previously. 10 Although post-treatment SSA levels in rosiglitazone and pioglitazone groups revealed no significant difference, high levels of SSA in the rosiglitazone group may be one of the reasons for increased incidence of CV complications associated with its use. 11
The study concludes that pioglitazone more effectively decreases SSA, indicating its greater cardioprotective effects compared with rosiglitazone.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not for profit sectors.
Conflict of interest
The authors declare that they have no conflicts of interest.
