Abstract
Objective:
Compromised endothelial function contributes to poor prognosis in patients with coronary artery disease and type 2 diabetes. Incretin-based therapy may exert beneficial cardiovascular effects beyond glucose lowering. We tested whether sitagliptin improves endothelial function.
Research design and methods:
In the double-blind BEta-cell function in patients with Glucose Abnormalities and Acute Myocardial Infarction (BEGAMI) trial, acute coronary syndrome (ACS)-patients with newly detected impaired glucose tolerance (IGT) or type 2 diabetes mellitus (T2DM) were randomised to sitagliptin 100 mg (n = 31) or placebo (n = 33) during 12 weeks. Endothelial function was studied as reactive hyperaemia index (RHI).
Results:
At baseline, the RHI was slightly compromised in both groups (sitagliptin 1.67; Q1;Q3: 1.46;2.17 vs placebo 1.61; 1.44;1.96; ns). The RHI did not change during the study period and was 1.57 (1.40;2.36) in the sitagliptin and 1.60 (1.46;1.81; ns) in the placebo group after treatment.
Conclusion:
Sitagliptin did not improve endothelial function in patients with ACS and newly detected glucose perturbations.
Introduction
Impaired glucose tolerance (IGT) and type 2 diabetes mellitus (T2DM) are common in patients with acute coronary syndrome (ACS) impairing the prognosis.1–4 Contributing to the poor outcome is endothelial dysfunction, an early marker of vascular engagement important for the development of atherosclerosis and cardiovascular disease in patients with diabetes. 5 An increase in glucagon-like peptide-1 (GLP-1) by inhibition of the dipeptidyl peptidase-IV (DPP-IV) enzyme may, in addition to glucose lowering, have positive effects on the endothelium.6,7 DPP-IV inhibitors have reduced the formation of atherosclerotic lesions in mouse models8,9 and increased circulating endothelial progenitor cells in experimental settings as well as patients with T2DM.10,11
In the BEta-cell function in patients with Glucose Abnormalities and Acute Myocardial Infarction (BEGAMI) trial, the DPP-IV inhibitor sitagliptin normalised newly detected glucose perturbations in patients with an ACS. 12 The aim and a predefined objective of this study were to study the impact of sitagliptin on endothelial function.
Design
The BEGAMI trial randomised patients with newly detected IGT or T2DM and an ACS to 100 mg sitagliptin (n = 31; Januvia™; Merck Sharp & Dohme AB, USA) or placebo (n = 33) during 12 weeks. A detailed description of the study has been presented elsewhere. 12 Endothelial function was studied in the morning before start of study drug and after 12 weeks as digital pulse amplitude tonometry (PAT), reflecting nitric-oxide-dependent changes in vascular tone after a reactive hyperaemia challenge, assessed by EndoPAT 2000 (Itamar Medical™, Caesarea, Israel). The procedure was performed in the supine position in a quiet temperature-controlled room (21°C–24°C) after 20 min of rest. Measurements consisted of 5 min baseline recording at rest followed by 5 min blood flow occlusion of the upper arm using a blood pressure cuff inflated 40 mmHg above systolic blood pressure and thereafter by 10 min recording after cuff release. The PAT signal changes were measured non-invasively at the fingertip and automatically calculated into a reactive hyperaemia index (RHI) by the system software as the ratio between the post- and pre-occlusion average signal size corrected for baseline level and systemic changes in the control arm. An RHI ≥ 2.0 was considered normal. 13 Additionally, arterial stiffness was assessed as augmentation index (AI) by EndoPAT 2000. Normal arterial stiffness was defined as an AI between −30% and −10%, increased arterial stiffness as an AI between −10% and 10% and abnormal arterial stiffness as an AI above 10%. 13
Continuous variables are expressed as median and Q1;Q3, and the Wilcoxon rank-sum test was used to determine differences between groups. Categorical variables are expressed as numbers and percentages and analysed using Fisher’s exact test. The
The BEGAMI study was conducted according to International Conference on Harmonisation and Good Clinical Practice Guidelines and the Declaration of Helsinki and approved by the Ethics Committee at Karolinska Institutet. Informed consent, written and oral, was obtained from all patients prior to inclusion.
Results
Endothelial function could be assessed in 64 patients. Of these, 30 had ST-segment elevation acute myocardial infarction AMI (STEMI), 27 non-STEMI and seven unstable angina. In the sitagliptin group, the median age was 67 years (61;77 years) and 84% were males. Corresponding numbers in the placebo group were 66 years (62;73 years; ns) and 79%. Further clinical characteristics are presented in Table 1.
Baseline characteristics in the 64 patients by randomised groups.
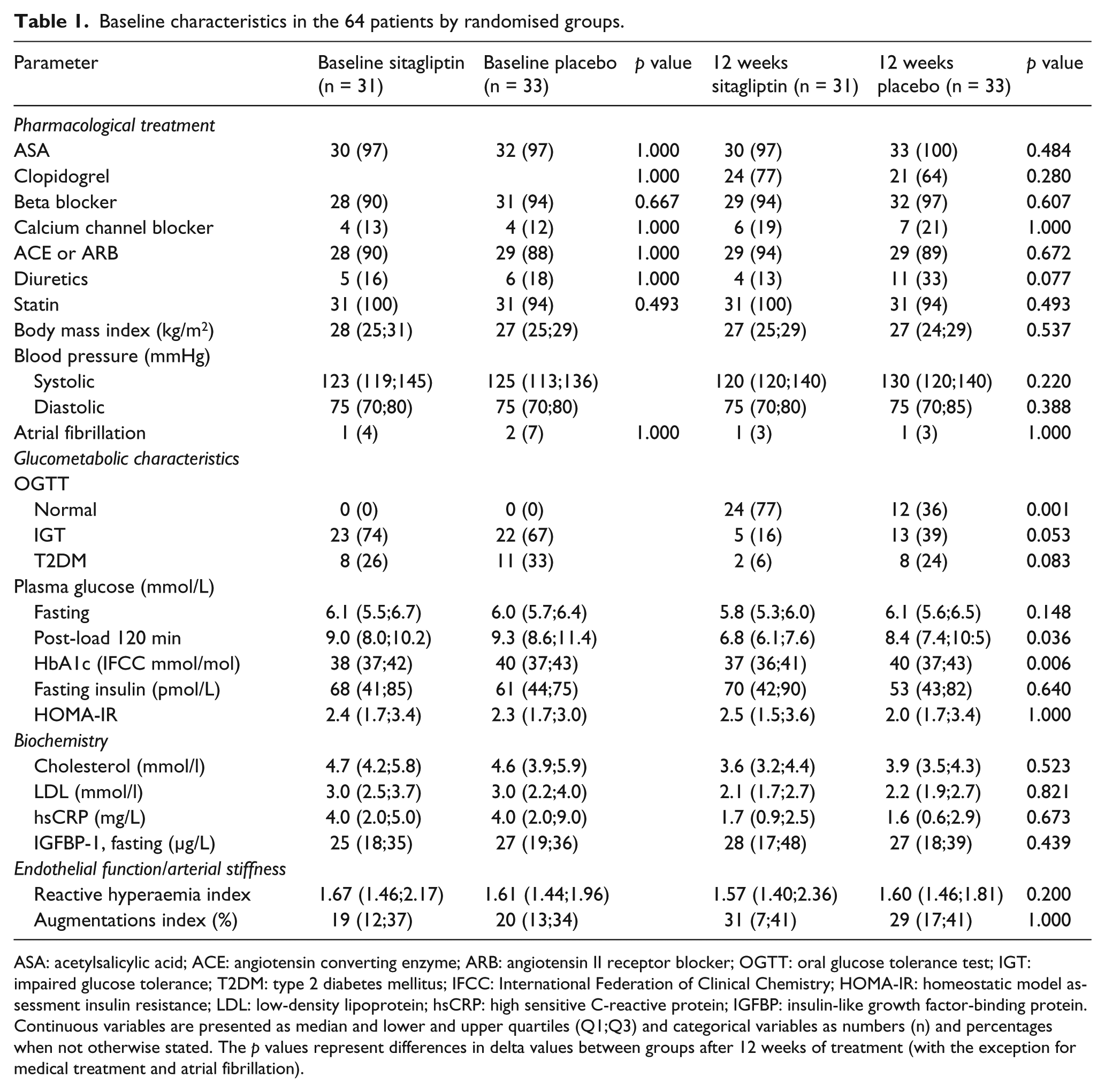
ASA: acetylsalicylic acid; ACE: angiotensin converting enzyme; ARB: angiotensin II receptor blocker; OGTT: oral glucose tolerance test; IGT: impaired glucose tolerance; T2DM: type 2 diabetes mellitus; IFCC: International Federation of Clinical Chemistry; HOMA-IR: homeostatic model assessment insulin resistance; LDL: low-density lipoprotein; hsCRP: high sensitive C-reactive protein; IGFBP: insulin-like growth factor-binding protein.
Continuous variables are presented as median and lower and upper quartiles (Q1;Q3) and categorical variables as numbers (n) and percentages when not otherwise stated. The
Baseline RHI was depressed in both groups (sitagliptin 1.67; Q1;Q3: 1.46;2.17 vs placebo 1.61; 1.44;1.96; ns). During the study period, RHI did not change significantly and was 1.57 (1.40;2.36) in the sitagliptin and 1.60 (1.46;1.81; ns) in the placebo group after 12 weeks of treatment.
AI indicated increased arterial stiffness and was at baseline 19% (12%;37%) in the sitagliptin group and 20% (13%;34%) in the placebo group (ns). After 12 weeks of treatment, the corresponding values were 31% (7%;41%) in the sitagliptin group and 29% (17%;41%) in the placebo group (ns).
Conclusion
The DPP-IV inhibitor sitagliptin did not improve endothelial dysfunction in the present cohort of patients with ACS and newly detected glucose perturbations.
In contrast to the present results, other studies of DPP-IV inhibitors in patients with T2DM indicated beneficial effects on endothelial function with sitagliptin14,15 and vildagliptin. 16 These studies recruited patients with a history of T2DM but with cardiovascular disease as an exclusion criterion or not defined, thus presumably free from or with less pronounced atherosclerosis than the BEGAMI participants. A reason for the discrepant results may therefore be that it may be too late to reverse a dysfunctional endothelium in patients with more advanced cardiovascular engagement. Other reasons may be the different study designs. The previous studies of sitagliptin were non-randomised and open-label. The vildagliptin study was randomised and double-blinded but of a crossover design with endothelial function measured at the end of each treatment period.
The question on the true impact of DPP-IV inhibition on endothelial function remains controversial not the least in the light of the present findings and those by Ayaori et al., 17 who recently reported that endothelial function was attenuated by DPP-IV inhibition in patients with T2DM. As in the BEGAMI trial, these authors hypothesised that the impact would be beneficial and performed two prospective, randomised crossover trials comparing sitagliptin with voglibose in the first and alogliptin in the second. In the first study, sitagliptin reduced flow-mediated dilatation (FMD) in the brachial artery despite improved glycaemic control, while voglibose did not affect the FMD. In the second study, both sitagliptin and alogliptin attenuated the FMD. These authors assumed that the lack of or indeed unfavourable impact of DPP-IV inhibition may be a class effect. 17 With these limited or even slightly harmful effects of DPP-IV inhibitors in patients at increased cardiovascular risk, one may speculate whether this could be a contributing explanation behind the neutral effects of alogliptin 18 and saxagliptin 19 on ischaemic events in recently reported trials.
Limitations
Although the PAT method is user-friendly, FMD may be a more validated technology. Onkelinx et al. 20 showed that the within-day variability was lower with FMD than for PAT, while between-day variability was similar in a head-to-head comparison. The correlation between PAT and FMD was, however, statistically significant. Considering these data, it is unlikely that the results in this study would have been different if the chosen method had been FMD rather than PAT.
BEGAMI is a relatively small study in which endothelial function was a secondary end point. Accordingly, its power calculation was based on the primary end point, beta-cell function, but the complete absence of a trend towards any difference between the two study groups makes the likelihood of a statistical type 2 error low. Further power calculations in similar studies indicate that the sample size should be sufficient. 21
In conclusion, the slightly compromised endothelial function in the present ACS-patients with newly detected glucose perturbations was not improved by sitagliptin.
(Clinicaltrials.gov: NCT00627744).
Footnotes
Acknowledgements
Camilla Hage, Pia Lundman and Linda Mellbin researched data; Camilla Hage, Lars Rydén and Linda Mellbin wrote manuscript and researched data and Anna Norhammar, Kerstin Brismar and Pia Lundman reviewed/edited manuscript and contributed to discussion.
Camilla Hage is the guarantor of this manuscript. The authors are grateful to E. Wallén Nielsen, J. Rasck and M. Asperen at Danderyd Hospital for blood sampling and patient care; to I.-L. Wivall and E. Sandberg at Karolinska Institutet for laboratory analysis and to M. Wallander at Karolinska Institutet for excellent advice
Declaration of conflicting interests
Linda Mellbin has received research grants from MSD and Sanofi Aventis and Lars Rydén has received research grants from the Swedish Heart and Lung Foundation, AFA Insurance, Karolinska Institutet, County Council of Stockholm, Astra Zeneca and Hoffmann-La Roche. He has received consultancy honoraria from Bristol Myers Squibb, Hoffman La Roche and Astra Zeneca.
Funding
This work was supported by grants from the Swedish Heart and Lung Foundation, AFA Insurance, the Erling-Persson Family Foundation and the Medical Research Council (04224). Study drug and placebo but no economical support was provided by Merck Sharp & Dohme AB (NJ, USA).
