Abstract
Purpose:
To investigate whether sitagliptin affects copeptin and osmolality, suggesting arginine vasopressin activation and a potential for fluid retention, compared with placebo, in patients with a recent acute coronary syndrome and newly discovered type 2 diabetes or impaired glucose tolerance. A second aim was to confirm whether copeptin correlated with insulin-like growth factor binding protein-1.
Methods:
Fasting blood samples were used from the BEta-cell function in Glucose abnormalities and Acute Myocardial Infarction trial, in which patients recently hospitalized due to acute coronary syndrome and with newly detected abnormal glucose tolerance were randomized to sitagliptin 100 mg once daily (n = 34) or placebo (n = 37). Copeptin, osmolality and insulin-like growth factor binding protein-1 were analysed at baseline and after 12 weeks.
Results:
Copeptin and osmolality were unaffected by sitagliptin. There was no correlation between copeptin and insulin-like growth factor binding protein-1.
Conclusion:
Sitagliptin therapy does not appear to be related to activation of the arginine vasopressin system.
Keywords
Introduction
Dipeptidyl peptidase type 4 (DPP-4) inhibitors are used in the treatment of type 2 diabetes (T2D). The SAVOR TIMI 53 (Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with Diabetes Mellitus-THrombolysis in Myocardial Infarction 53) trial indicated that saxagliptin might increase the incidence of hospitalization for heart failure, although no mechanism for this was clear. 1 Subsequent studies on other gliptins did not support an increased risk of heart failure.2,3 These large trials could, however, provide details on potential mechanisms behind such provocation.
Arginine vasopressin (AVP) increases renal water retention in response to increased serum osmolality or stress, for example, an acute coronary syndrome (ACS). 4 Activation of the AVP system may be involved in the pathogenesis of chronic heart failure. 4 In turn, glucagon-like peptide-1 (GLP-1) activates AVP, which could mediate a potential effect of GLP-1 on heart failure. 5
Copeptin is a useful surrogate marker for AVP levels, as it is secreted in equimolar amounts to AVP but more stable and easier to analyze. 4 Copeptin is a marker of acute myocardial infarction (AMI) and a negative prognostic marker in heart failure. 4 Increased copeptin levels are furthermore linked to risk of developing diabetes and AVP may have a direct role in glucose homeostasis via receptors, for example, in the liver and pancreas. 6
Insulin-like growth factor binding protein-1 (IGFBP-1) is produced in the liver mainly under the influence of insulin and is thereby a marker of hepatic insulin sensitivity. 7 Infusion of the AVP-analogue desmopressin increased serum IGFBP-1 in patients with diabetes insipidus. 8 Moreover, a correlation has been reported between copeptin and IGFBP-1 in patients with T2D and AMI. Although both elevated copeptin and IGFBP-1 predicted cardiovascular events, it was only copeptin that remained independent in a multivariate analysis. 9 These findings support a possible relation between these two hormonal systems as regards their involvement in the cardiovascular prognosis in patients with glucose abnormalities.
The purpose of this study, a biochemical sub-study to the BEta-cell function in Glucose abnormalities and Acute Myocardial Infarction (BEGAMI) trial, was to investigate whether the DPP-4 inhibitor sitagliptin is associated with higher copeptin levels and/or lower osmolality, compared with placebo, in patients with a recent ACS and newly discovered glucose abnormalities. If so, this could suggest AVP activation and thereby potential for fluid retention, which in turn increases cardiac load. A second aim was to confirm whether copeptin correlated with IGFBP-1.
Methods
Data source
The BEGAMI trial (Clinicaltrials.gov: NCT00627744) was a multicenter, double-blind trial, which recruited patients recently hospitalized due to ACS, and in whom an oral glucose tolerance test (OGTT) discovered previously unknown abnormal glucose tolerance [impaired glucose tolerance (IGT) or T2D]. The patients were randomized to sitagliptin 100 mg once daily (n = 34; males = 29) or placebo (n = 37; males = 29). 10 A total of 10 of the patients randomized to sitagliptin had T2D and 24 IGT. The corresponding distribution in the placebo group was 14 with T2D and 23 with IGT. All patients were given structured lifestyle advice and post-ACS treatment according to existing guidelines. None of them received any glucose-lowering drugs besides the study drug. Examinations were repeated after a 12-week treatment period. The present biochemical sub-study comprises all patients in the BEGAMI trial.
The study was conducted in accordance with the Declaration of Helsinki and was approved by the ethics committee at Karolinska Institutet. All participants provided written and oral consent prior to inclusion.
Laboratory analyses
Fasting blood samples were analysed at baseline and follow-up:
Serum copeptin was analysed by immunofluorescent assay (BRAHMS Copeptin ultrasensitive KRYPTOR) with a detection limit of 0.9 pmol/L and interassay coefficient of variation (CV) of 20%.
Serum osmolality was analysed using an Advanced 2020 Multi-Sample osmometer (Advanced Instruments, Norwood, MA, USA). The total CV was 3%.
Serum IGFBP-1 was analysed with an in-house RIA, with a sensitivity of 3 µg/L and intra- and interassay CVs of 3% and 10%, respectively.
Statistical analyses
Statistical analyses were performed with STATISTICA software, version 10 (StatSoft, Tulsa, OH, USA). Variables are presented as mean ± standard error of the mean (SEM) unless otherwise stated. Delta values were calculated as the given value after the 12-week study period minus that at baseline. Differences between groups were analysed using the Mann–Whitney U test, and differences within groups from the beginning compared to the end of the study period were analysed using the Wilcoxon signed-rank test. Differences between groups in the frequencies of previous diseases were analysed using Fisher’s exact test. Association between continuous variables was analysed using Spearman’s rank correlation. A two-tailed p < 0.05 was considered statistically significant.
Results
Patient characteristics by treatment group are presented in Table 1. Heart rate decreased in both groups (sitagliptin: p = 0.04; placebo: p = 0.03), while waist circumference and body mass index (BMI) were unaffected. Creatinine increased significantly in the sitagliptin group, but there was no significant difference in delta values between the groups.
Biochemical and clinical characteristics at randomization and after 12 weeks of treatment.
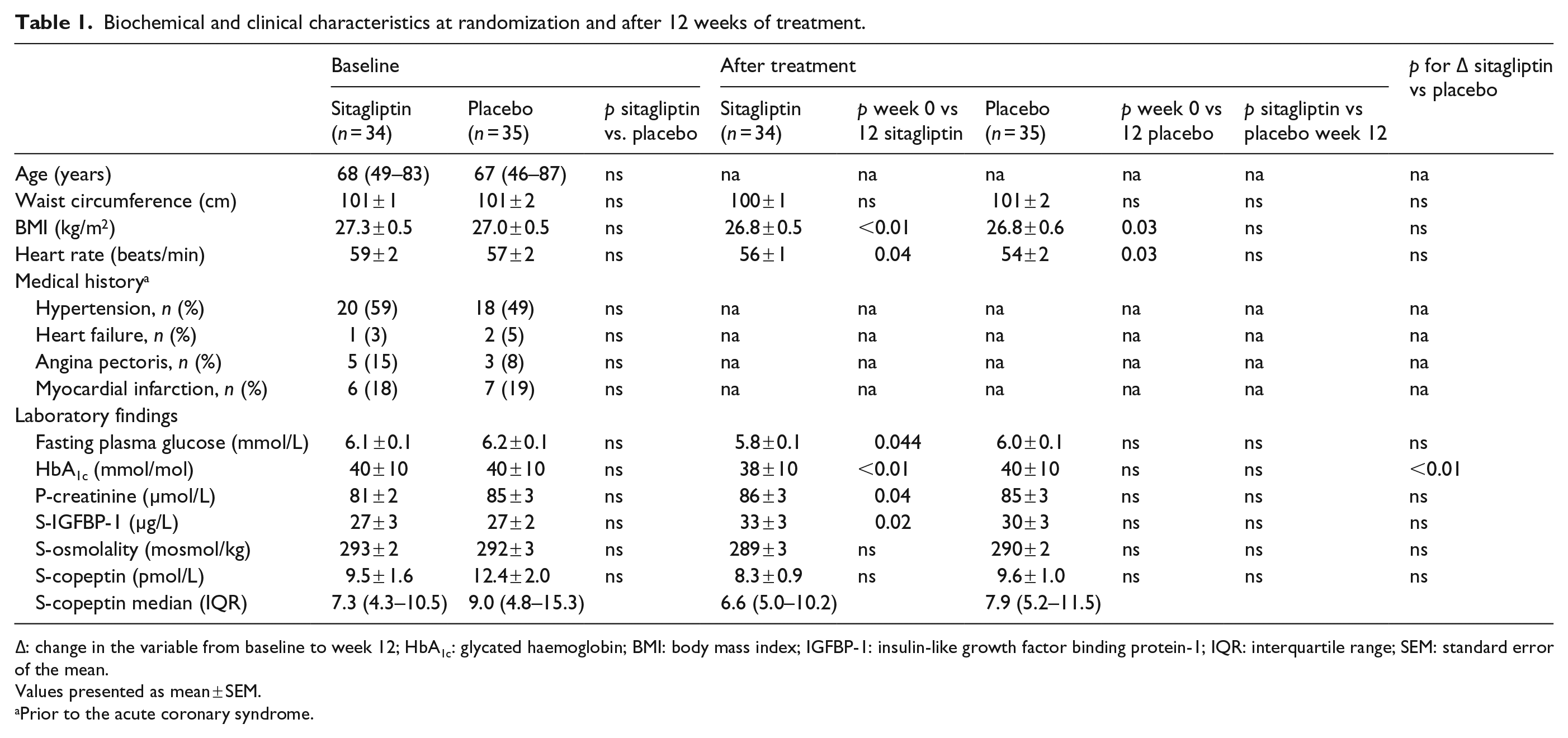
Δ: change in the variable from baseline to week 12; HbA1c: glycated haemoglobin; BMI: body mass index; IGFBP-1: insulin-like growth factor binding protein-1; IQR: interquartile range; SEM: standard error of the mean.
Values presented as mean ± SEM.
Prior to the acute coronary syndrome.
There were no significant difference between the two groups in the actual or delta values of serum copeptin and osmolality before compared to after treatment. The IGBFBP-1 levels increased significantly in patients on sitagliptin but not among those given placebo, and there was no significant difference in delta IGFBP-1 between the groups.
In a pooled analysis comprising all patients (n = 69) at baseline, serum copeptin was 11.0 ± 1.3 pmol/L (median: 7.7; interquartile range: 4.8–12.0); serum osmolality, 292 ± 2 mosmol/kg (median: 293; interquartile range: 288–296); and serum IGFBP-1, 27 ± 2 µg/L, respectively (median: 25; interquartile range: 18–35). There was no significant difference in the copeptin levels at baseline between patients with T2D (n = 24) or IGT (n = 45; T2D = 12.3 ± 2.9; IGT = 10.2 ± 1.2 pmol/L), while osmolality was lower in the T2D group compared with IGT (p = 0.04). There were no differences in IGFBP-1 levels between patients classified as T2D or IGT.
Copeptin and IGFBP-1 did not correlate at baseline or by the end of the treatment period in any of the two groups and not in the pooled analysis of all subjects at baseline or in subjects with T2D or IGT.
Discussion
The main finding in this biochemical sub-study of BEGAMI was that treatment with sitagliptin compared with placebo did not influence copeptin, a known predictor of heart failure and cardiovascular prognosis. Sitagliptin treatment did not influence serum osmolality, suggesting that the blood volume was unaffected. This is in line with the cardiovascular outcome trial [The Trial to Evaluate Cardiovascular Outcomes after Treatment with Sitagliptin (TECOS)] reporting neutral effects on heart failure in patients randomized to sitagliptin or placebo 3 as well as the lack of effect of sitagliptin on N-terminal of the prohormone brain natriuretic peptide (NT-proBNP). 10 These are reassuring findings considering the indications from SAVOR-TIMI 2 on an increased risk of heart failure. Taken together with the TECOS and EXAMINE (Examination of Cardiovascular Outcomes: Alogliptin vs. Standard of Care in Patients with Type 2 Diabetes Mellitus and Acute Coronary Syndrome) studies, which did not show an increased risk of heart failure with sitagliptin and alogliptin in patients with T2D and ACS2,3 and that vildagliptin did not increase hospitalizations for heart failure in patients with T2D and known heart failure in the Vildagliptin in Ventricular Dysfunction Diabetes Trial (VIVIDD) study, 11 the present results support the notion that heart failure is not a class effect of DPP-4 inhibitors. It would, however, be of interest to examine copeptin levels in subjects treated with saxagliptin, the only DPP-4 inhibitor that presently may be linked with heart failure. 1
Studies on patients admitted to hospital for acute chest pain regardless of the presence of diabetes or not reported mean copeptin levels in the magnitude of 23 pmol/L in those with AMI versus 8.3 pmol/L among those not found to have infarction. 12 In patients with AMI copeptin levels peak on the day of the event decreasing to a plateau level after 3–5 days, 4 that is, earlier than the baseline testing in this study. The baseline levels of 11 pmol/L in this study are still higher than the 1–6 pmol/L that are typical for healthy subjects. 4 In the Diabetes Mellitus, Insulin Glucose Infusion in Acute Myocardial Infarction-2 (DIGAMI-2) study, recruiting patients with T2D, the mean copeptin level at admission for AMI was 62 pmol/L (median: 22 pmol/L), 9 that is, higher than in unselected cohorts of AMI patients, suggesting that T2D contributes to AVP activation. The DIGAMI-2 patients had higher mean glycated haemoglobin (HbA1c) (equivalent to 64 mmol/mol) than in BEGAMI, which could contribute to this difference since higher plasma glucose levels have been associated with higher copeptin levels. 9 In future studies, it would be of interest to examine whether higher glucose levels or longer diabetes duration is related to AVP drive and/or fluid retention in ACS.
Previous findings of a correlation between copeptin and IGFBP-1 in patients with known T2D and AMI 9 were not found in the population studied here. A potential explanation may be that the regulation of IGFBP-1 is complex. In particular, insulin, but also other hormones, is relevant to IGFBP-1 gene expression, which may differ in an acute setting as in DIGAMI-2 compared to the present more stable condition some time after an AMI.
In conclusion, copeptin levels and osmolality were unaffected by treatment with sitagliptin in this sub-study of the BEGAMI trial recruiting patients with a recent ACS and newly discovered glucose abnormalities. This suggests that sitagliptin has a neutral effect on AVP tonus in these patients.
Footnotes
Declaration of conflicting interests
L. Mellbin has received research grants and consulting honoraria from MSD, and C. Hage has received speaker honoraria from MSD. A. Norhammar has honoraria from expert group participation (MSD, Astra Zeneca, Boehringer Ingelheim). The remaining authors have no conflicts of interest to declare.
Funding
This work was supported by grants from the Swedish Heart and Lung Foundation, the Swedish Diabetes Foundation, The Swedish Society of Medicine, AFA Insurance, Family Erling-Persson Foundation and the Medical Research Council (04224). The study drug and placebo, but not financial support, were provided by Merck Sharpe and Dohme AB (MSD).
