Abstract
Background:
Hyperglycaemia is common among patients with critical neurological injury, even if they have no history of diabetes. The optimal target range for normalizing their blood glucose is unknown.
Methods:
Retrospective data were extracted from 890 hyperglycaemic individuals (glucose > 200 mg/dL) admitted to neuroscience critical care unit (NCCU) and these patients were divided into two groups: intensive glucose control group with target glucose of < 140 mg/dL achieved and moderate control with glucose levels 140-180 mg/dL. The groups were also stratified according to the hyperglycaemia type (pre-existing diabetes or stress-related). We defined the primary endpoint as death from any cause during NCCU admission.
Results:
In NCCU, tighter control of blood glucose at ≤ 140 mg/dL was associated with increased, mortality of individuals with pre-existing diabetes compared with moderate control [29 of 310 patients (9.4%) vs 15 of 304 patients (4.9%), p = 0.034]. Patient age [adjusted odds ratio (OR) = 1.12; 95% confidence interval (CI) = 1.05–1.19; p < 0.001], level of glycated haemoglobin (adjusted OR = 1.24; 95% CI = 1.04–1.48; p = 0.017) and hypoglycaemia (adjusted OR = 10.3; 95% CI = 2.92–36.6; p < 0.001) were positively associated with higher mortality. Death rate was lower among stress-related hyperglycaemic patients with tighter glucose controlled at ≤ 140 mg/dL [6 of 140 patients (4.3%) vs 15 of 136 patients (11.0%), p = 0.035].
Conclusion:
A differential association is evident between glucose levels and mortality in diabetes and stress-related hyperglycaemia patients. However, given the observational nature of our work, no clinical recommendations can be given and prospective studies are required to further investigate these findings.
Introduction
Hyperglycaemia is common in critically ill patients,1,2 even in those with no history of diabetes. Acute illness contributing to stress-related hyperglycaemia mainly results from factors such as the release of stress hormones and administration of exogenous glucocorticoids. 3 The occurrence of hyperglycaemia, particularly severe hyperglycaemia, is associated with poor clinical outcomes among patients with critical illness. Various researchers4–11 have reported that normalizing elevated glucose levels (80–110 mg/dL, equal to 4.4–6.1 mmol/L) exerts beneficial effects on outcomes in critically ill patients. Thus, intensive glucose control is recommended by many professional organizations for patients treated in intensive care units (ICUs). However, systematic reviews and meta-analyses indicate conflicting results regarding the effects of intensive glucose control. Hyperglycaemia in ICU has not been consistently shown to signify poor prognosis in patients with pre-existing diabetes. Conversely, hyperglycaemia has been linked with poor outcomes in ICU patients not known to have diabetes and specifically in those with stroke. Thus, hyperglycaemia reflects the severity of underlying illness among patients with pre-existing diabetes. The benefits of intensive glucose control among patients with diabetes may be compromised by the adverse effects of hypoglycaemia.
A neurologically ill patient substantially differs from a general medical, pulmonary, cardiovascular or surgical patient in that the brain of the former is the primary organ of interest which relies on glucose for appropriate function.12,13 Thus, cellular respiration depends on a sustained supply of glucose, and incidences of hypoglycaemia may trigger neuronal apoptosis. 14
Despite these controversies, many critical care physicians acknowledge the need to prevent hyperglycaemic episodes. Traditionally, acute hyperglycaemia was defined as a random glucose concentration > 200 mg/dL. However, in 2010, the American Diabetes Association proposed a threshold of 140 mg/dL (7.8 mmol/L). In patients not known to have diabetes, glycated haemoglobin should be measured; an elevated level (> 6.5% or > 48 mmol/mol) indicates pre-existing diabetes. 15 The results of the Normoglycaemia in Intensive Care Evaluation and Survival Using Glucose Algorithm Regulation (NICE-SUGAR) study provide further evidence that intensive glucose control increases mortality and the number of hypoglycaemic episodes among adults in the ICU. A blood glucose target of 180 mg/dL also results in lower mortality than a target of 81–108 mg/dL. However, insufficient data are available for determining the target range or threshold within which glucose control is suitable for hyperglycaemic patients in a neurocritical care unit.
We performed a retrospective study involving adults admitted to our neurocritical care unit targeted to normalize blood glucose levels to ≤ 180 mg/dL (10.0 mmol/L). The patients were divided into two groups: one with intensive control to ≤ 140 mg/dL (7.8 mmol/L) and the other with moderate control to 140–180 mg/dL (7.8–10.0 mmol/L). The groups were stratified according to the type of hyperglycaemia (with pre-existing diabetes or stress related). We aimed to determine the optimal target range for blood glucose in hyperglycaemic patients in a neurocritical care unit by defining the primary endpoint as death from any cause during neuroscience critical care unit (NCCU) admission.
Methods
Ethics
The protocol of this study was approved by the research ethics committees of Zhongda Hospital, which was affiliated with Southeast University.
Study design and patients
Hyperglycaemic patients targeted to maintain their blood glucose levels to ≤ 180 mg/dL (10.0 mmol/L) were eligible for enrolment in the study. They were admitted to the NCCU of Zhongda Hospital, Southeast University, Nanjing, P.R. China, between May 2005 and March 2012. Acute hyperglycaemia was defined as random glucose level of equal to or greater than 200 mg/dL at any time during hospitalization. In hyperglycaemic patients not known to have diabetes, glycated haemoglobin was measured and an elevated level (> 6.5%) was deemed to indicate pre-existing diabetes. 15 Drug or insulin was administered if the blood glucose level exceeded 200 mg/dL. The retrospective data, including pertinent demographic and clinical information, were extracted from the electronic medical records of each patient. They were medically managed by the neurocritical care service, which is composed of the critical care physician group, advanced-practice nurses, staff nurses and medical house staff in training.
The blood glucose levels of each patient were managed as part of the normal duties of the clinical staff. Normoglycaemia was the goal for all subjects, and the decision on which protocol to implement for glucose management was made by the NCCU care team. Insulin dose adjustments were based on the blood glucose level, which was measured from capillary blood by either venous sampling or fingerstick sampling using an ACCU-CHEK Inform Glucose Meter (Roche Diagnostics Corporation, Indianapolis, IN, USA). Blood samples for glucose measurement were obtained by arterial catheters whenever possible. The dose was adjusted according to a strict algorithm by the NCCU care team with constant assistance by an endocrinologist. All enrolled patients were divided into two groups according to the level of blood glucose control (BGC): one with ≤ 140 mg/dL was allocated to intensive control group and the other with 140–180 mg/dL to moderate control group. The groups were stratified according to the type of hyperglycaemia (with pre-existing diabetes or stress-related) (see Figure 1).
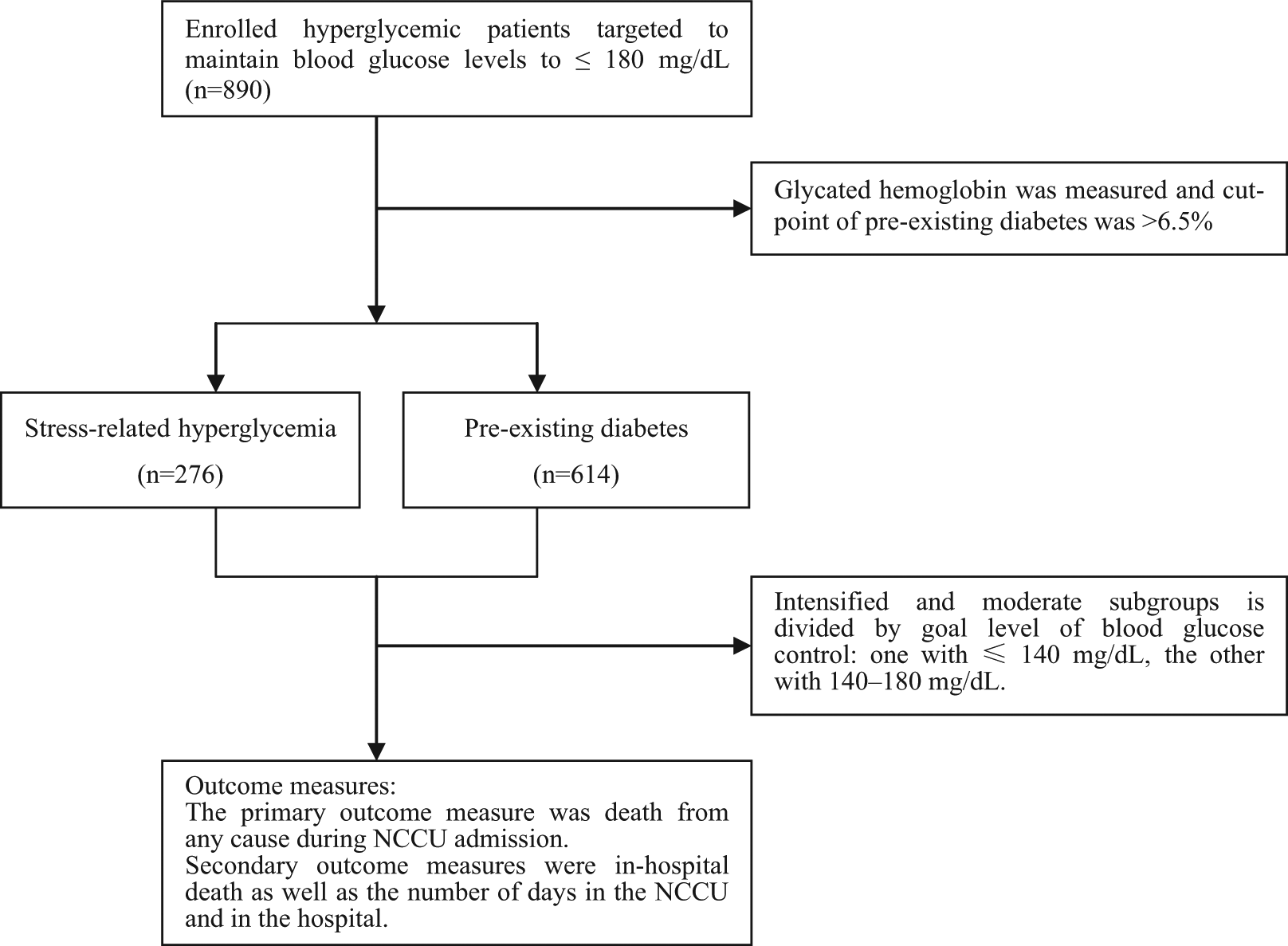
Treatment algorithms of the study.
During the NCCU admission period, we recorded all blood glucose measurements and administration, type and volume of all enteral and parenteral nutrition as well as additional intravenous glucose administered. Blood was obtained upon admission and subsequently every 6–8 h. The blood glucose level was measured upon admission and daily at 6 a.m. The daily maximum and minimum blood glucose levels were determined.
Upon admission, all patients were continuously given intravenous glucose (200–300 g every 24 h). The next day, total parenteral, combined parenteral and enteral, or total enteral feeding was instituted according to a standardized schedule. The food intake contained 15–30 non-protein kilocalories per kilogram of body weight per 24 h and a balanced composition (including 0.13–0.26 g of nitrogen per kilogram per 24 h and 20%–40% of non-protein calories in the form of lipids). Total enteral feeding was attempted as early as possible. Once the patients no longer needed vital-organ support, they were transferred to a regular ward but readmitted to the NCCU if needed. For those admitted in the regular ward, their blood glucose levels were measured four to seven times daily. The daily maximum and minimum blood glucose levels were maintained within 110–180 mg/dL.
Outcome measures
The primary outcome measure was death from any cause during NCCU admission. Secondary outcome measures were in-hospital death as well as the number of days in the NCCU and in the hospital. Other outcome includes severe hypoglycaemia defined as a value less than or equal to 40 mg/dL (2.2 mmol/L), as defined in the major trials on glucose control in critically ill patients.
Quantitative data were expressed as the mean ± standard deviation. Qualitative variables were expressed as frequencies. For statistical analysis, Fisher’s exact test, Pearson’s χ2 test and Student’s t-test were used as appropriate. Continuous variables were analysed by a two-tailed t-test, and p < 0.05 was considered statistically significant. Conditional univariate and multivariate logistic regression models were used to estimate the odds ratio (OR) and associated 95% confidence interval (CI). All statistical analyses were performed using statistical software (SPSS for Windows, version 13.5).
Results
A total of 890 hyperglycaemic patients targeted to maintain blood glucose levels ≤ 180 mg/dL (10.0 mmol/L) were enrolled in the study. Among them, 614 patients were diagnosed with diabetes mellitus, and the remaining 276 patients were diagnosed with stress-related hyperglycaemia. The study covered an observational period of more than 6 years. However, in both diabetic and stress-related hyperglycaemia groups, patients with intensive and moderate levels of glucose control were similar in the demographics and clinical characteristics, for example, age, sex percentage, diabetes mellitus history, glycated haemoglobin level, nutrition, National Institutes of Health Stroke Scale (NIHSS) score and primary admitting diagnosis. Table 1 shows that both diabetic and stress-related hyperglycaemic patients received a larger mean insulin dose to maintain a glucose level ≤ 140 mg/dL.
Baseline characteristics of the study patients.
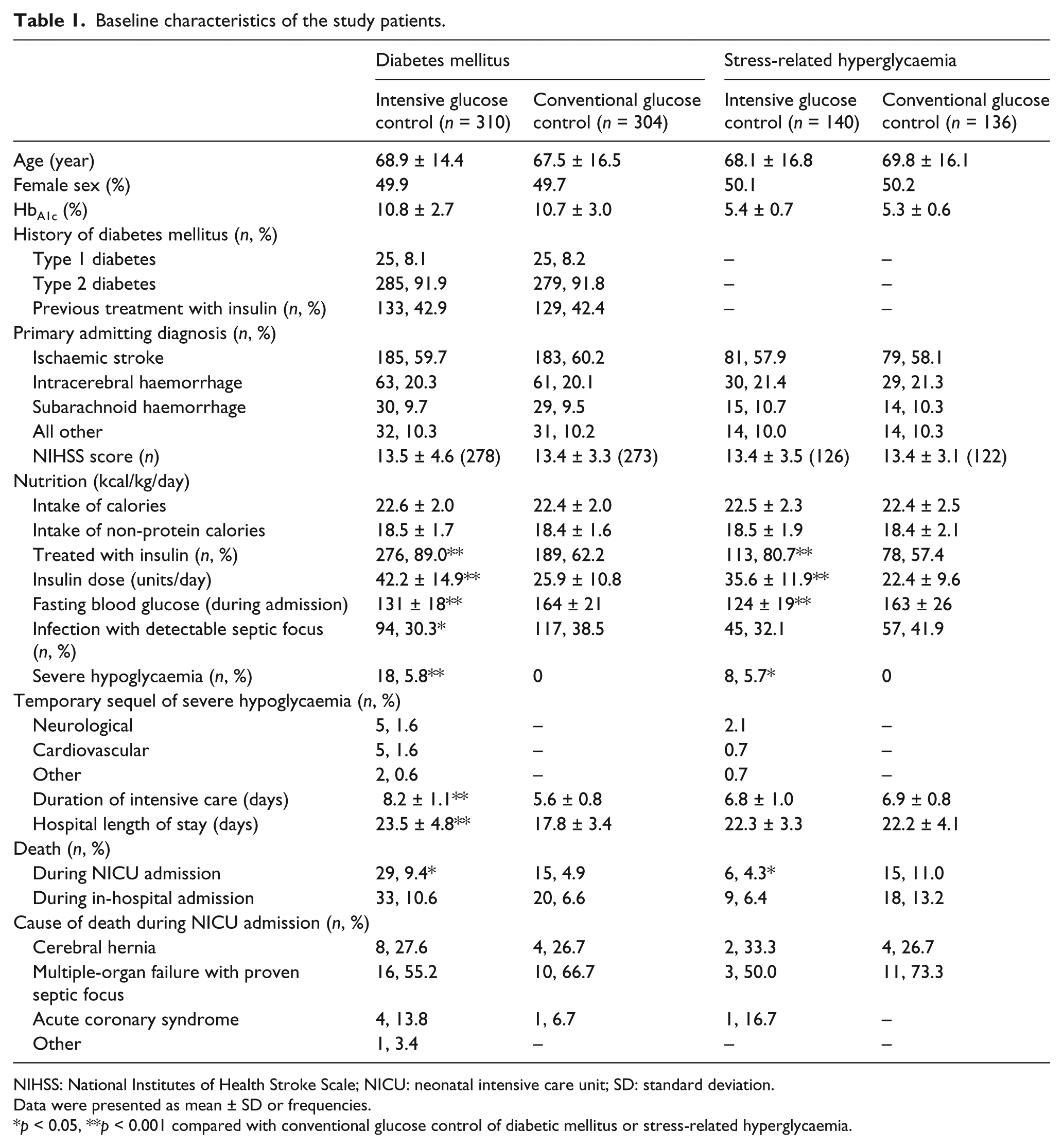
NIHSS: National Institutes of Health Stroke Scale; NICU: neonatal intensive care unit; SD: standard deviation.
Data were presented as mean ± SD or frequencies.
p < 0.05, **p < 0.001 compared with conventional glucose control of diabetic mellitus or stress-related hyperglycaemia.
We first examined the association between levels of plasma blood glucose and outcome to determine the optimal range for blood glucose in the NCCU. The mortality of patients with diabetes during NCCU admission increased when their blood glucose levels were maintained at ≤ 140 mg/dL compared with the moderate glucose control subgroup [29 of 310 patients (9.4%) vs 15 of 304 patients (4.9%), p = 0.034]. However, mortality was lower in stress-related hyperglycaemic patients with tighter glycaemic control at < 140 mg/dL [6 of 140 patients (4.3%) vs 15 of 136 patients (11.0% in the diabetes group), p = 0.035]. Regarding in-hospital mortality, subgroup analysis indicated that the difference between the ranges in the two glucose control subgroups was not significant among the patients with diabetes and stress-related hyperglycaemia [33 of 310 patients (10.6%) vs 20 of 304 patients (6.6%), p = 0.073; 9 of 140 patients (6.4%) vs 18 of 136 patients (13.2%), p = 0.057], respectively. Intensive glucose control significantly decreased the number of patients with diabetes with detectable infection [94 of 310 patients (30.3%) vs 116 of 304 patients (38.5%), p = 0.041]. No significant difference was found between the two subgroups among patients with stress-related hyperglycaemia [45 of 140 patients (32.1%) vs 57 of 136 patients (41.9%), p = 0.093]. We also found that intensive glucose control prolonged the length of stay of patients with diabetes in the NCCU (8.2 ± 1.1 days vs 5.6 ± 0.8 days, p < 0.001) or hospital (23.5 ± 4.8 days vs 17.8 ± 3.4 days, p < 0.001). No significant difference was found between the two subgroups among the patients with stress-related hyperglycaemia (6.8 ± 1.0 vs 6.9 ± 0.8, p = 0.19; 22.3 ± 3.3 vs 22.2 ± 4.1, p = 0.81, respectively).
As aforementioned, the mortality was high among the patients with diabetes targeted to maintain blood glucose levels at ≤ 140 mg/dL (7.8 mmol/L) and stress-related hyperglycaemic patients subjected to moderate glucose control. Accordingly, we investigated the risk factors after adjustment for the predefined factors, for example, age, sex, level of glycated haemoglobin, diabetes type (among the diabetics), insulin dose, infection, hypoglycaemia and morning blood glucose. Multivariate conditional regression analysis indicated that age, level of glycated haemoglobin and severe hypoglycaemia were positively associated with high mortality among the patients with diabetes with intensive glucose control. Severe hypoglycaemia was defined as random glucose level less than 40 mg/dL (2.2 mmol/L) any time during hospitalization. Severe hypoglycaemia was a high risk factor in the diabetic group subjected to intensive glucose control. Neurological, cardiovascular and other sequels resulted from hypoglycaemic episodes. Age was associated with death among the stress-related hyperglycaemic patients with moderate glucose control. All are shown in Table 2.
Factors associated with higher mortality among patients with diabetes with intensive glucose control and SH patients with moderate glucose control.
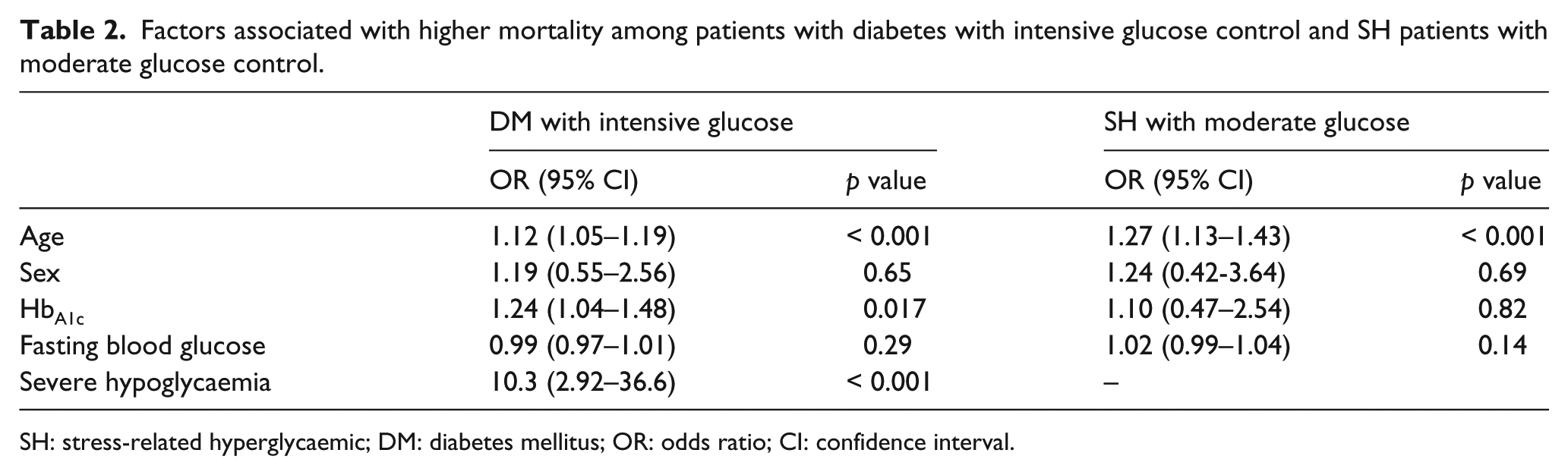
SH: stress-related hyperglycaemic; DM: diabetes mellitus; OR: odds ratio; CI: confidence interval.
Discussion
This retrospective study focused on hyperglycaemia among patients with critical neurological injury and on determining the optimal target range for normalizing their blood glucose levels. The most interesting finding was that the mortality of the patients with diabetes increased when the blood glucose level was maintained ≤ 140 mg/dL. Mortality decreased among the stress-related hyperglycaemic patients with glucose controlled under this target. Multivariate conditional regression analysis indicated that the age, level of glycated haemoglobin and hypoglycaemia were positively associated with higher mortality among the patients with diabetes with intensive glucose control. Age was associated with mortality among the stress-related hyperglycaemic patients with moderate glucose control. Intensive glucose control significantly decreased the number of patients with diabetes with detectable infection. No significant difference was observed between the two subgroups among the patients with stress-related hyperglycaemia.
Hyperglycaemia occurs in 50%–75% of patients admitted to the ICU. This condition has been associated with poor clinical outcomes among patients with critical illness, thereby prompting a renewed interest in establishing strict glucose control through intensive insulin therapy (IIT). However, several studies applying an IIT strategy yield confusing and often conflicting results. Some studies have demonstrated that such an approach can reduce episodes of excessive hyperglycaemia4,16 and may decrease renal failure,4,5,16 sepsis,4,17 red blood cell transfusions,4,16 critical illness polyneuropathy, 4 mortality4,16 and ICU length of stay.4,16,18 However, other studies have shown that IIT is associated with increased hypoglycaemic episodes, unchanged ICU length of stay and increased mortality.5,9,17 Graffagnino et al. 19 found that mortality is high in the IIT group comprising both hypoglycaemic and hyperglycaemic patients. This finding indicates that hypoglycaemia and hyperglycaemia are associated with high mortality.18,20–22 Considerable uncertainty remains regarding the optimal target levels of glucose in ICU patients.
The NICE-SUGAR study showed increased mortality in patients subjected to intensive glucose control (81–108 mg/dL) compared with those managed with conventional glucose control (140–180 mg/dL). However, a recent study showed that a lower target range of 129–145 mg/dL is associated with a lower risk of inadvertent hypoglycaemia as well as a lower mortality rate and represents an optimal BGC level in critically ill patients. 23 Other studies suggest that a tailored BGC target range should be adopted in specific subgroups of patients. The target range may be corrected according to the nutrition protocol used and depending on the duration of insulin infusion. 24 In trauma patients without traumatic brain injury, ‘moderate’ BGC management (BGC target range = 80–140 mg/dL) or ‘intensive’ BGC management (BGC target range = 80–110 mg/dL) in the age group of 41–60 years is associated with reduced mortality. 25 A study on head trauma patients showed that IIT results in decreased cerebral glucose concentrations (without a change in the cerebral glucose metabolic rate), elevated lactate/pyruvate ratios and increased glutamate concentrations, particularly with serum glucose levels below 80 mg/dL. Other research found that mortality does not differ between intensive and regular insulin therapy groups. 26
Whether the target levels of glucose control should differ according to pre-existing diabetes or stress-related hyperglycaemia is unclear. Stress-related hyperglycaemia is common in critically ill and injured patients. This condition was initially considered to be a beneficial adaptive response, with increased blood glucose providing a ready source of fuel for the brain, skeletal muscle, heart and other vital organs at a time of increased metabolic demand. However, several studies involving specific groups of patients such as those with myocardial infarction, stroke, chronic obstructive pulmonary disease and those receiving total parenteral nutrition showed increased mortality and morbidity in patients who develop stress-related hyperglycaemia.27–30 Patients without a diagnosis of diabetes who develop stress-related hyperglycaemia are likely to be at a higher risk of vascular disease than patients with normal blood glucose level. 31 Studies support the view that stress-related hyperglycaemia is of pathophysiological significance and is not simply a stress response to damage; it was found that insulin administration reduces the size of the infarct and improves prognosis after stroke. 32 The intensive control of stress-related hyperglycaemia in non-diabetic, critically ill patients may bring benefits. However, at the opposite extreme, hypoglycaemia may be detrimental and clearly mandates avoidance especially among the patients with diabetes with more extensive underlying cerebral vasculopathy. This may account for the current observation that there is a difference between mortality in patients with diabetes versus stress-induced hyperglycaemia treated to the same intensive glucose intervention threshold.
To explore the possible causes leading to increased mortality in patients with diabetes with intensive glucose control and stress-related hyperglycaemic patients with moderate glucose control, we performed multivariate conditional regression analysis. The results indicated that age was a risk factor in both groups. High levels of glycated haemoglobin and incidences of hypoglycaemia were also positively associated with high mortality among the patients with diabetes with intensive glucose control. Hypoglycaemia is recognized as a marker of greater severity of disease in inpatients. Additionally, hypoglycaemia, which is a major cause of increased mortality, can obscure the benefit of IIT. 33 Severe hypoglycaemia (i.e. < 40 mg of glucose/dL) occurs in up to 28% of patients in trials of IIT.4–11 The incidence of severe hypoglycaemia is likely to be higher in the real world (i.e. outside clinical trials) if a target range of 80–110 mg/dL is used. The neurological consequences of hypoglycaemia are legitimate concerns but difficult to detect in critically ill patients. Hypoglycaemia can also cause acute electroencephalographic alterations. 34 In a previous study, at 4 years of follow-up, a subgroup of patients treated with IIT were found to have impairments in quality of life and social function compared with patients who receive conventional treatment. 35 The long-term sequelae of iatrogenic hypoglycaemia in the ICU are difficult to measure and the thresholds for harm are unclear.
This retrospective study has the following limitations. First, the manner of allocation to the two regimes is a weakness. During relative long study period, NCCU team and even diabetes physicians have different treatment regimes based on their own interpretation of illness severity, comorbidity and risk factors. For example, some may strive for tighter glycaemic control in more severely ill patients, whereas others may have a more relaxed approach. All these uncontrolled variables affected patient allocation and the results; some bias cannot be avoidable. Second, clinical data such as past medical history and diabetic complications were not completely obtained. These factors are potentially related to mortality to some degree. Additionally, glycaemia was measured by arterial, venous or capillary sampling which may have induced differences in glucose levels and hypoglycaemic episodes may have not been all captured.
Taken together, although 140–180 or ≤ 140 mg/dL as the target range for blood glucose and lower mortality was likely associated among the NCCU patients with pre-existing diabetes or stress-related hyperglycaemia, we cannot exclude uncontrolled variables affecting the analysis. Thus, prospective studies are required to confirm this association. Additionally, the challenge for critical care clinicians is to treat hyperglycaemia and simultaneously prevent hypoglycaemic episodes, especially among the patients with diabetes. Perhaps more robust and consistent methods to capture hyperglycaemia and hypoglycaemia should be used in the future trial.
Footnotes
Acknowledgements
We express gratitude to the nursing staff of the Division of Neurology, Zhongda Hospital of Southeast University for their assistance.
Declaration of conflicting interests
All authors hereby declare no conflict of interest.
Funding
This work was partially supported by the National Natural Science Foundation of China (No. 30870870, Shao-Hua Wang; No. 81070638, Shao-Hua Wang and No. 81070916, Yi-Jing Guo), and the Social Development Project of JiangSu Province (No. SBE201170735, Shao-Hua Wang).
