Abstract
Objective
Admission hyperglycaemia is recognized as a contributor to poor outcomes in patients with cardiovascular and cerebrovascular diseases. This study aimed to evaluate the association between admission hyperglycaemia and in-hospital mortality in patients with hypertension and acute aortic dissection (AAD).
Methods
Patients diagnosed with hypertension and AAD between 1 January 2010 and 1 March 2023 were enrolled into this retrospective observational study, and divided into two groups based on admission blood glucose levels (BGA): normoglycemia (BGA < 7.8 mmol/L) and hyperglycaemia (BGA ≥ 7.8 mmol/L). The outcome was all-cause in-hospital mortality, which was analysed using logistic regression to calculate odds ratios (ORs) and 95% confidence intervals (CIs).
Results
In total, 1239 patients were included (comprising 954 [77%] male patients; mean age, 53.0 years; and median BGA, 6.1 mmol/L). Logistic regression analysis showed that in-hospital mortality was 2.53 times greater for patients with hyperglycaemia versus the normoglycaemia group (95% CI 1.68, 3.80). This significant association persisted after adjustments for potential confounders (adjusted OR 2.45 [95% CI 1.58, 3.85]). Restricted cubic spline analysis revealed that the relationship between BGA and mortality reached statistical significance at BGA > 6.1 mmol/L. Stratified analysis and sensitivity analysis confirmed the robustness of this relationship.
Conclusion
Admission hyperglycaemia correlated with an elevated risk of in-hospital mortality in patients with hypertension and AAD, particularly BGA > 6.1 mmol/L, indicating that BGA level may be useful in identifying patients who are at an elevated risk of all-cause mortality.
Keywords
Introduction
Aortic dissection (AD) is an alarming cardiovascular disease encompassing intramural haematoma, penetrating aortic ulcer, and ruptured thoracic aortic aneurysm. Although AD is rare, its occurrence has surged substantially over the previous 10 years.1,2 Incidence rates are reported to be between 3 and 8 cases/100 000 adults per year,1,3 and despite the availability of therapies, such as open surgical repair and endovascular repair, the in-hospital mortality rate for AD remains high, ranging from 10.6% to 57%.4,5 Long-term hypertension leads to sustained high pressure on the aortic wall, which can easily cause damage to the vascular intima, further contributing to the development of AD. Hypertension is believed to be an important predictor of AD, affecting more than 70% of patients. 6 Moreover, it often coexists with metabolic abnormalities, and significantly increases the risk of cardiovascular and cerebrovascular diseases.
Admission hyperglycaemia is commonly observed in emergency patients, which may increase sympathetic nervous system activity and trigger cytokines, inflammation, and oxidative stress.7,8 Poor clinical outcomes have been linked to admission hyperglycaemia in patients who have experienced severe trauma or stroke, or patients with cardiovascular ailments, such as acute coronary syndrome.9,10 However, few studies have focused on the correlation between admission hyperglycaemia and in-hospital mortality in patients with hypertension and acute aortic dissection (AAD).11,12
Therefore, the purpose of the current retrospective investigation was to investigate the link between admission hyperglycaemia and in-hospital mortality in patients with hypertension and AAD.
Patients and methods
Study population and data source
Patients with hypertension and AAD (Stanford types A or B), who were admitted to the People's Hospital of the Xinjiang Uygur Autonomous Region in China between 1 January 2010 and 1 March 2023, were enrolled into this retrospective observational study. Inclusion criteria were as follows: (1) age ≥18 years; (2) history of sudden chest pain; (3) hospital visit within 14 days of symptom onset; and (4) aortic dissection that was definitively diagnosed by imaging (including ultrasound and computed tomography angiography) for the first time. Eligible participants included those fulfilling all four inclusion standards. The diagnosis and classification of AAD were based on World Health Organisation criteria combined with confirmation through the results of aortography and computed tomography (determined by two investigators). The exclusion criteria for this study were as follows: (1) chronic aortic dissection; (2) AAD surgery during hospital admission; (3) immune diseases (including systemic vasculitis, systemic lupus erythematosus, and rheumatoid arthritis); and/or (4) incomplete data on blood glucose levels at admission (BGA).
Data from the electronic medical records were extracted through a big data intelligent platform (Hypertension Expert Database) specifically designed for hypertension research. 13 The data platform encompasses a wide range of information, including patient personal details, visit specifics, diagnosis particulars, medical records, laboratory test outcomes, prescriptions, medical orders, and cost information. 14 The present study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki (1975, as revised in 2013) and all patient details were de-identified in accordance with standard privacy and confidentiality protocols. Informed consent was waived by our institutional review board due to the retrospective nature of the study. The reporting of this study follows the STROBE statement guidelines. 15
Data collection
Baseline information at the time of admission was extracted for all patients from the electronic medical record system. The following general, clinical, and laboratory data were collected. General data included age, sex, cigarette and alcohol consumption, body mass index (BMI), systolic blood pressure (SBP), diastolic blood pressure (DBP), and pulse rate. Laboratory data included BGA, total cholesterol, triglyceride, high and low-density lipoprotein cholesterol, serum creatinine, uric acid, blood urea nitrogen, alanine aminotransferase, aspartate aminotransferase, white blood cell count (WBC), lymphocyte count, monocyte count, neutrophil count, haemoglobin levels, platelet count, total bilirubin levels, albumin levels, glycosylated haemoglobin (HbA1c), and D-dimer levels. Clinical data included vascular ultrasound and aortic/renal artery computed tomography angiography, previous disease history (e.g., coronary heart disease [CHD] or diabetes mellitus [type 1 or type 2]), comorbidities (e.g., renal insufficiency or pulmonary infection), and agent intake before admission and during hospitalization, including antihypertensive, hypoglycaemic, lipid-lowering and/or anti-platelet agents.
Definitions and groups
Hospital admission was defined as the first 24 h from initial medical contact and only laboratory values from this period were considered. Aortic dissection is defined as acute within 14 days of its onset, and chronic beyond that period. 16 Blood glucose was measured through venous blood sampling at the time of admission and during hospitalization. Hypertension was identified based on a self-reported history of medical diagnosis and use of antihypertensive drugs. Diabetes was defined as a fasting glucose level ≥7.0 mmol/L, the use of diabetes treatment (including either diet and lifestyle measures alone or in addition to administration of oral glucose-lowering medication and/or insulin), or a self-reported physician diagnosis of diabetes. 8 Patients were divided into two groups based on BGA levels: normoglycemia (BGA <7.8 mmol/L) and hyperglycaemia (BGA ≥7.8 mmol/L), according to the American Heart Association guidelines.17,18 Insulin therapy was defined as insulin administration at any time during hospital admission. CHD was defined as a fatal or nonfatal myocardial infarction, unstable angina, and/or coronary revascularization. Drinking and smoking status were categorized as binary variables: never/former or current. Acute kidney failure was defined as an increase in serum creatinine by 50% within 7 days, an increase in serum creatinine by 0.3 mg/dL (26.5 μmol/L) within 2 days, or oliguria.
Study outcome
The clinical outcome measure was all-cause mortality during hospitalization.
Statistical analyses
Continuous data are presented as mean ± SD or median (interquartile range), and categorical data are presented as n (%) prevalence. Continuous variables with normal distribution were compared between groups using Student’s t-test and those with non-normal distribution using Wilcoxon rank-sum test. Between-group differences in categorical data were assessed by χ2-test. Tolerance and variance inflation factors (VIF) of multi-variable linear regression analysis were used to test collinearity among variables. Multicollinearity was judged if the tolerance was <0.1 and the VIF was >5. Logistic regression was employed to assess the correlation between BGA and in-hospital mortality. Restricted cubic spline curves were generated at the 10th, 50th, and 90th percentiles by applying three default knots. Additionally, stratification analyses were conducted to further evaluate the associations between BGA and in-hospital mortality. Sensitivity analysis was performed by excluding those patients with diabetes and HbA1C ≥6.5%. Statistical analyses were performed using IBM SPSS software, version 25.0 for Windows (IBM Corp., Armonk, NY, USA) and R software, version 4.3.1 (www.r-project.org). A two-sided P-value <0.05 was considered statistically significant.
Results
Participant characteristics
A total of 2 698 patients with hypertension and AAD were initially identified from the Hypertension Expert Database. After applying the study inclusion and exclusion criteria, 1 459 patients were excluded due to incomplete BGA data (n = 1110), chronic aortic dissection (n = 18), AAD surgery during hospitalisation (n = 324), or immune disease (n = 7), and a total of 1 239 patients were included in the study (
Characteristics of 1239 hospitalised patients with hypertension and acute aortic dissection included in the study.
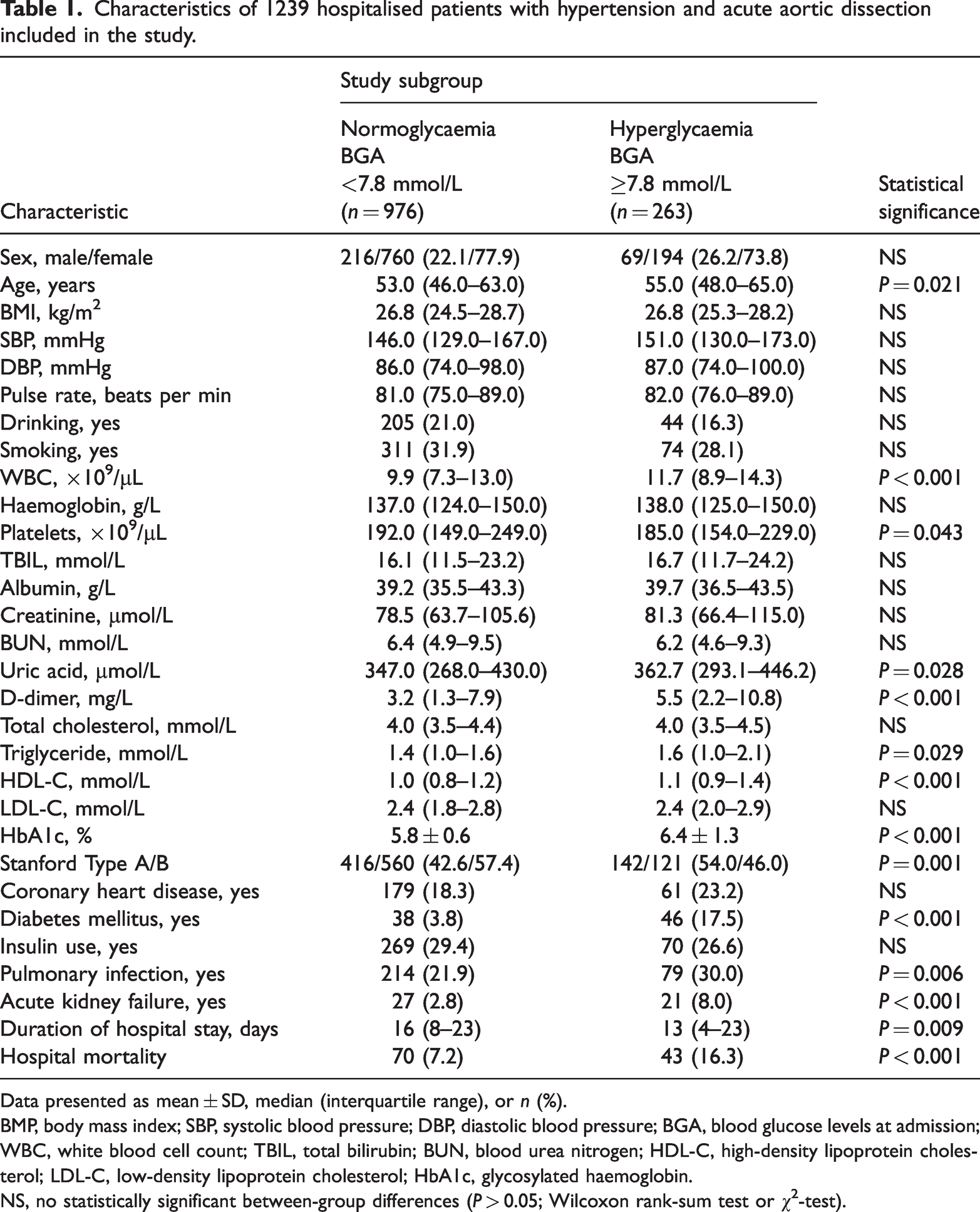
Data presented as mean ± SD, median (interquartile range), or n (%).
BMP, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; BGA, blood glucose levels at admission; WBC, white blood cell count; TBIL, total bilirubin; BUN, blood urea nitrogen; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; HbA1c, glycosylated haemoglobin.
NS, no statistically significant between-group differences (P > 0.05; Wilcoxon rank-sum test or χ2-test).
Baseline characteristics of patients grouped according to hospital survival are summarised in
Associations between admission hyperglycaemia and in-hospital mortality
Blood glucose levels at admission were found to be positively associated with in-hospital mortality (Table 2). When BGA was assessed as a continuous variable, it was positively associated with an increased risk of in-hospital mortality (OR 1.15 [95% CI 1.08, 1.23]; P < 0.001). When BGA was assessed as a categorical variable with 7.8 mmol/L as the cut-off, patients with hyperglycaemia showed a 2.53-fold higher chance of experiencing in-hospital mortality (95% CI 1.68, 3.80; P < 0.001) versus those with normoglycaemia, and this increased risk remained in
The association of BGA with all-cause mortality in 1239 hospitalised patients with hypertension and acute aortic dissection.
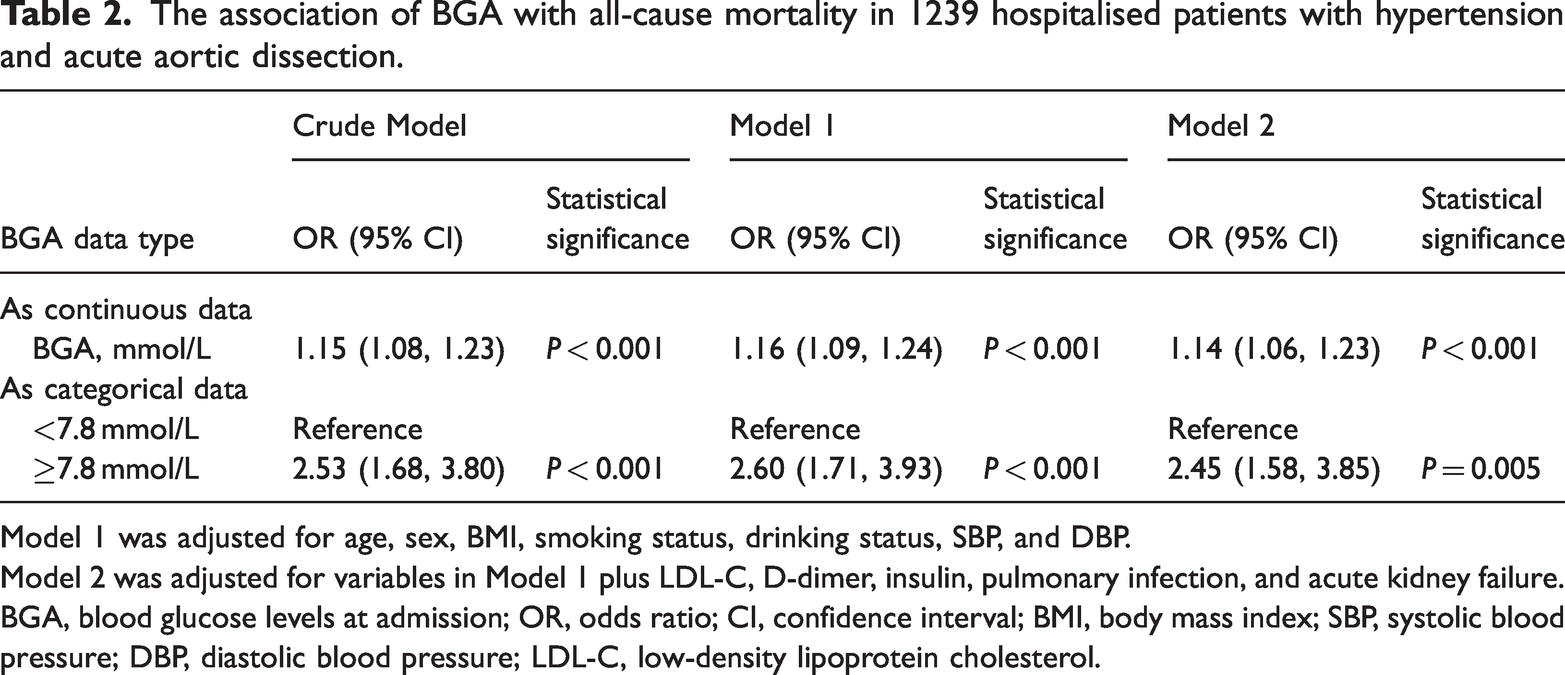
Model 1 was adjusted for age, sex, BMI, smoking status, drinking status, SBP, and DBP.
Model 2 was adjusted for variables in Model 1 plus LDL-C, D-dimer, insulin, pulmonary infection, and acute kidney failure.
BGA, blood glucose levels at admission; OR, odds ratio; CI, confidence interval; BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; LDL-C, low-density lipoprotein cholesterol.
According to restricted cubic spline analysis, there was a linear dose-response association between BGA and in-hospital mortality (P non-linear = 0.094; Figure 1). The relationship between the two variables became statistically significant at BGA > 6.1 mmol/L. Patients were also divided into three groups according to the inflection point: group 1, 0 < BGA ≤6.1 mmol/L; group 2, 6.1 < BGA < 7.8 mmol/L; and group 3, BGA ≥7.8 mmol/L. This analysis also indicated a positive correlation between BGA and in-hospital mortality (P trend test <0.001), as shown in

Restricted cubic spline for the association between blood glucose levels at hospital admission (BGA) and in-hospital mortality in 1239 hospitalised patients with hypertension and acute aortic dissection. OR, odds ratio; CI, confidence interval.
Subgroup analysis
Subgroup analyses were performed in patients stratified by sex, age, SBP, DBP, smoking status, drinking status, insulin use, and type of AAD. None of the variables were found to significantly modify the association between BGA and in-hospital mortality (P for all interactions > 0.05; Figure 2).
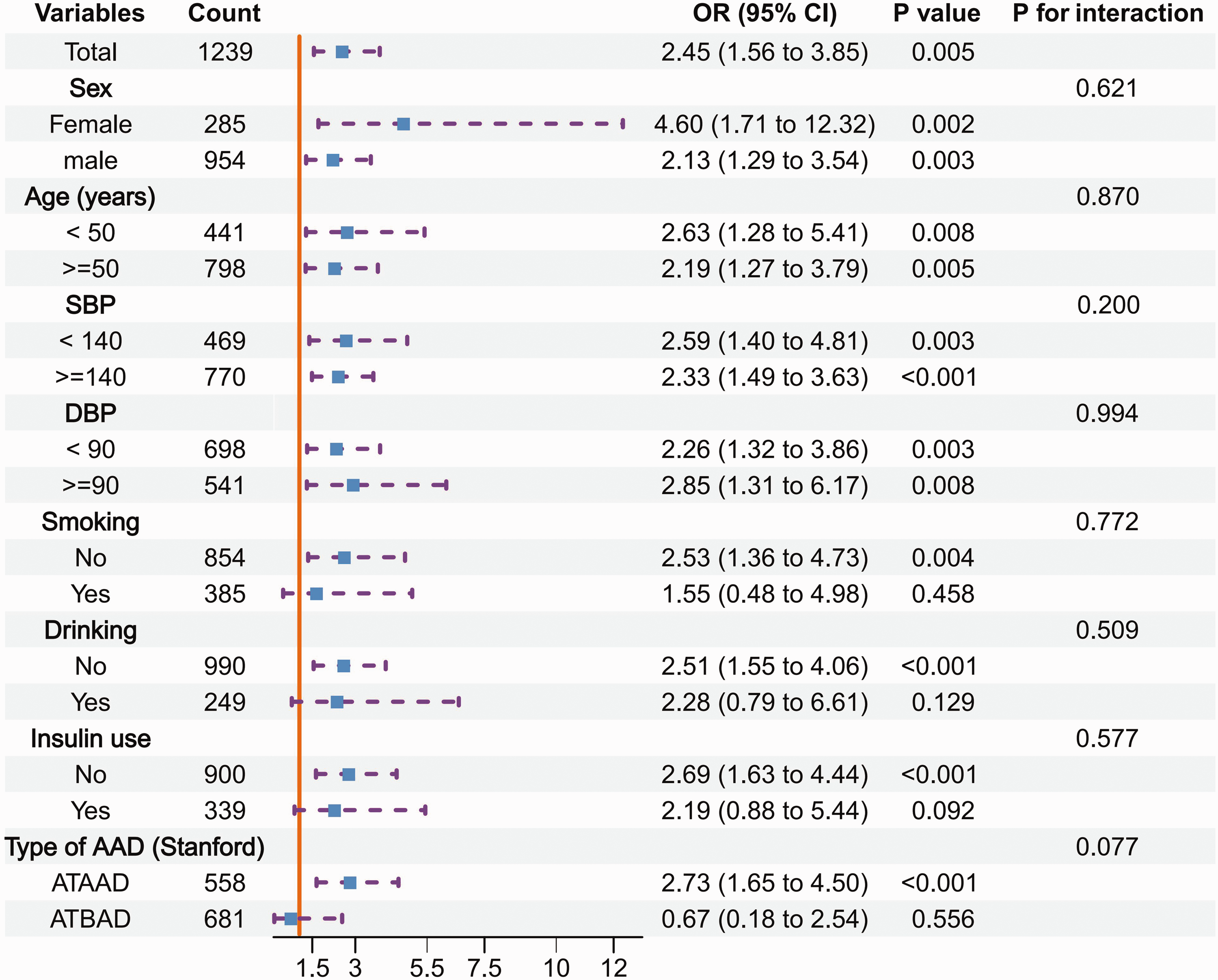
Subgroup analyses of the relationship between blood glucose levels at hospital admission and risk of in-hospital mortality in 1239 hospitalised patients with hypertension and acute aortic dissection (AAD). ATAAD, acute type A aortic dissection; ATBAD, acute type B aortic dissection; SBP, systolic blood pressure (mmHg); DBP, diastolic blood pressure (mmHg); OR, odds ratio; CI, confidence interval.
Patients were then further stratified by diabetes status and insulin use. The result showed that insulin use during hospitalization was associated with lower ORs for in-hospital mortality (Table 3), suggesting that use of insulin appears to be associated with reduced hospital mortality risk.
Association between BGA and in-hospital mortality in 1239 hospitalised patients with hypertension and acute aortic dissection stratified by diabetes status.
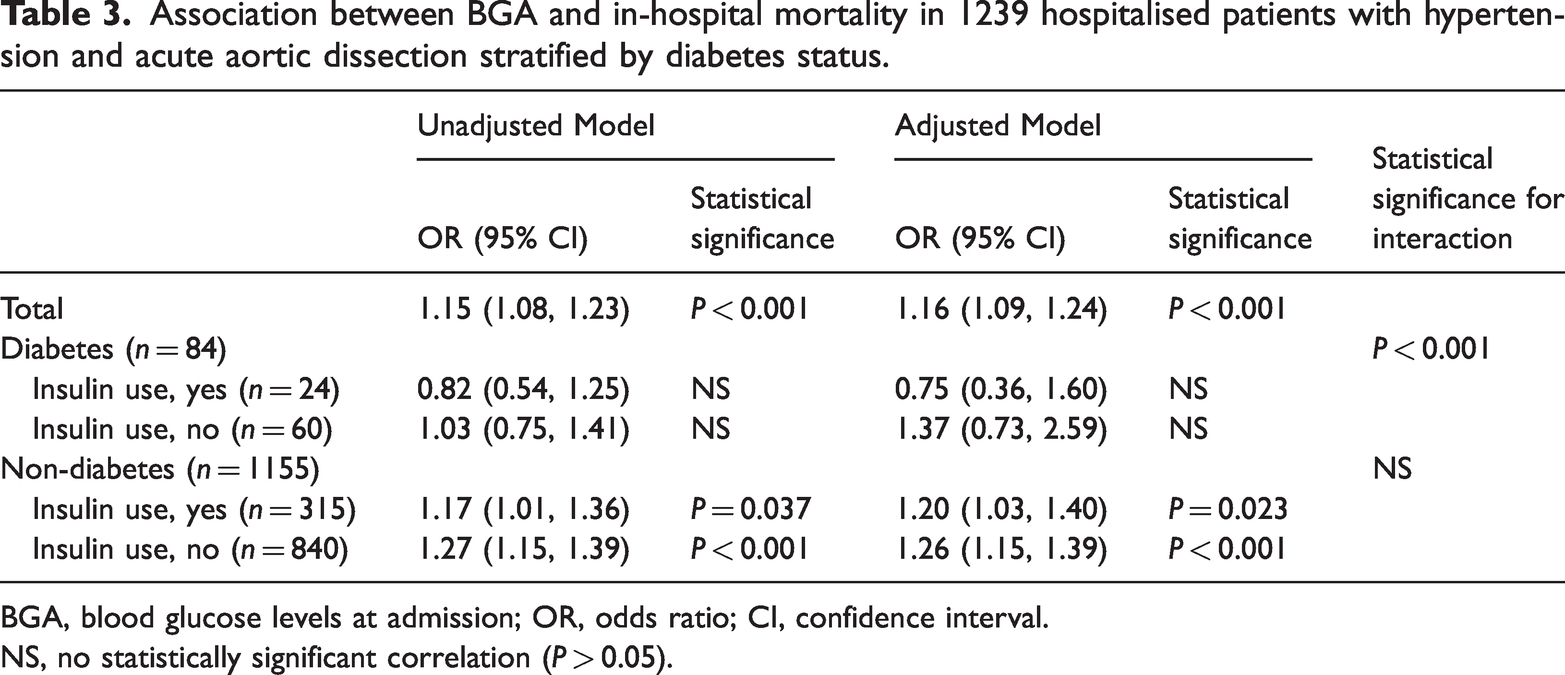
BGA, blood glucose levels at admission; OR, odds ratio; CI, confidence interval.
NS, no statistically significant correlation (P > 0.05).
Sensitivity analyses
An initial sensitivity analysis was performed excluding patients with diabetes (
Discussion
The findings of the present study indicated that patients with hyperglycaemia had a significantly elevated risk of mortality during hospitalization versus patients with normoglycemia. When BGA was > 6.1 mmol/L, the risk of in-hospital mortality was shown to rapidly increase. Specific analysis of data for patients without diabetes revealed a stronger correlation between BGA and mortality risk, which is consistent with previous studies on admission hyperglycaemia and other cardiovascular events. For example, a study found that patients admitted to hospital with acute coronary syndrome or with cerebrovascular accidents have been shown to experience higher 30-day mortality rates when they have transient hyperglycaemia, whether or not they have diabetes.17,19,20 Other studies have demonstrated that patients without diabetes who experience hyperglycaemia at hospital admission for acute medical illness face a greater risk of adverse outcomes versus patients with diabetes.19,21 Consistent with previous research, a pronounced age disparity was observed in the present study between normoglycemic and hyperglycaemic patients, reinforcing the pivotal role of age as a significant predictor of prognosis and mortality across a range of diseases. Despite efforts to mitigate the potential confounding effects of this age difference through the application of adjustment models, the disparity persisted as a notable factor in the present analysis.
Admission hyperglycaemia has been identified as a detrimental factor impacting the survival rates of critically ill patients. 19 The occurrence of stress hyperglycaemia is caused by various factors, such as catecholamines, growth hormones, cortisol, inflammation, and cytokines. 17 In return, the severity of cellular damage increases with higher levels of hyperglycaemia. Inflammation is also responsible for upregulating the sympathetic nervous system, resulting in coronary vasoconstriction, thickening of the basement membrane in capillaries, arteriolar sclerosis, and abnormal microcirculation.21,22 Research has demonstrated that elevated blood glucose levels induce non-enzymatic glycosylation of collagen and elastin, which can have adverse effects on pulmonary function. 23 These results imply that admission hyperglycaemia is linked to a heightened systemic involvement of the disease, leading to multi-organ decompensations. Collectively, these alterations ultimately compromise blood vessel functionality, playing a crucial role in the pathogenesis of AAD. Smooth muscle cell loss and substrate degradation are important features of AAD. Cellular damage and changes in enzyme activity due to non-enzymatic glycosylation may also be caused by long-standing hyperglycemia. 24 Measurement of HbA1c provides an indication of long-term glycaemic control, so a sensitivity analysis was conducted, and showed that the present results were stable. Inflammatory cells may weaken the aortic smooth muscle cells and lead to apoptosis. Vascular smooth muscle cell apoptosis is linked to the aneurysm formation and contributes to the progression of AAD.25–27 An animal model study showed that elevated glucose levels may hinder the progression of AAD, aneurysm enlargement, and clinical progression by affecting the fibrinolytic pathway. 28
Admission hyperglycaemia may have a significant impact on the progression of AAD. Several studies have reported a correlation between dysregulation of BGA and mortality. A previous study found that admission hyperglycaemia was linked to a higher risk of mortality and reflect the severity of acute Stanford type A AAD in patients without diabetes,
18
which is consistent with the present findings that displayed a positive link between admission hyperglycaemia and in-hospital mortality in patients with hypertension and AAD, with or without diabetes. However, in another study involving 734 patients hospitalized with AAD, admission hyperglycaemia was associated with increased postoperative prolonged mechanical ventilation in patients with Stanford type A AAD, but not with in-hospital mortality rate.
11
Possible reasons are as follows: First, the present study population targeted patients with hypertension and AAD. Although no obvious differences were shown in the stratified analysis, long-term hypertension will certainly lead to structural changes in the vascular wall and a chronic inflammatory environment. Therefore, it is necessary to investigate the relationship between BGA and AAD prognosis in this specific population. Secondly, the study by Lin et al.
11
only included patients with Stanford type A AAD, whereas the present research comprised patients with both Stanford types A and B AAD. The larger sample size in the present study allowed for more statistically significant results and a broader representation of the patient population. Certain patients were excluded as deemed necessary for the integrity of the study, however, there were no statistically significant differences in baseline characteristics, such as sex, age, and BMI between the included and excluded individuals (shown in
Cohort studies and several early randomized controlled trials have consistently demonstrated that intensive management of hyperglycaemia may lead to improved hospital outcomes.29,30 To effectively manage hyperglycaemia in critically ill patients admitted to the intensive care unit, it is recommended to utilize intravenous insulin instead of oral hypoglycaemic agents. Upon detection of blood glucose levels exceeding 180 mg/dL (10.0 mmol/L), intravenous insulin infusion should be promptly initiated to control hyperglycaemia. Once intravenous insulin administration commences, it is crucial to maintain blood glucose within the range of 140–180 mg/dL (7.8–10.0 mmol/L). However, a target of < 110 mg/dL (6.1 mmol/L) is not recommended.31,32 Hypoglycaemia is known to be associated with macro-vascular events and negative outcomes. 33 Therefore, insulin use was included in the present multivariate models to adjust for its potential impact. Initial previous studies did not provide conclusive evidence regarding the therapeutic advantages of insulin intervention for the stringent management of BGA in individuals diagnosed with AAD. In the present study, the utilization of insulin for managing admission hyperglycaemia appears to be correlated with a reduced risk of mortality during the hospitalization period. An in-depth study could not be completed due to the limited sample size; however, the present cross-sectional investigation offers initial concepts for more in-depth future research.
The results of the present study may be limited by several factors, as this was a retrospective observational study from a single centre with bias in sample selection. First, the study focussed on patients with hypertension and AAD, which limits the generalizability of the results to a broader, non-specific population. Secondly, the acute and critical nature of AAD often results in the rapid deterioration and death of some patients before blood samples can be successfully obtained for analysis. Additionally, geographical and economic factors contribute a further challenge, as they often prompt patients from remote areas to decline tests or request early discharge, thereby exacerbating the potential for bias. Thirdly, because AAD is very dangerous, the patient’s rescue is the first priority. More data on blood glucose and its related indicators, such as high glycaemic variability that may have been able to better reflect a patient’s blood glucose, were not available. Fourthly, the study did not analyse the relationship between BGA and long-term prognosis. Future large-sample, multicentre, and prospective studies are needed to determine whether better control of admission blood glucose will improve clinical prognosis. Certainly, in addition to optimizing study designs, the present authors aim to intensify quality control measures during the data collection process to minimize missing data and errors.
Conclusion
In conclusion, the present study may demonstrate a positive correlation between admission hyperglycaemia and an escalated hazard of hospital mortality in patients with hypertension and AAD. In these patients, closely monitoring higher BGA seems instrumental in facilitating clinical decision-making and optimizing disease management.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241291742 - Supplemental material for Association between admission hyperglycaemia with in-hospital mortality rate in patients with hypertension and acute aortic dissection
Supplemental material, sj-pdf-1-imr-10.1177_03000605241291742 for Association between admission hyperglycaemia with in-hospital mortality rate in patients with hypertension and acute aortic dissection by Saimire Mutailifu, Qing Zhu, Xintian Cai, Mulalibieke Heizhati, Shasha Liu, Yujie Dang, Ting Wu, Jing Hong, Yue Lin and Nanfang Li in Journal of International Medical Research
Footnotes
Acknowledgements
The Authors would like to thank all collaborators of this study.
Author contributions
Nanfang Li: study conception and supervision. Saimire Mutailifu and Qing Zhu: study protocol design and implementation, including data collection and data analysis, and first draft writing. Xintian Cai and Heizhati Mulalibieke: results analysis and intellectual contributions. Shasha Liu, Yujie Dang, Ting Wu, Hong Jing, and Yue Lin: data collection and curation. All authors discussed the results and approved the final manuscript.
Data availability statement
The datasets used and/or analysed in the current study are available within the manuscript, as additional supplemental material, or from the corresponding author upon reasonable request.
Declaration of conflicting interest
The Authors declare that there are no conflicts of interest.
Funding
This study was supported by a grant from the Youth Science Fund Project of the Natural Science Foundation of the Autonomous Region (2022D01C836) and the Key Science and Technology Special Projects of Xinjiang Uygur Autonomous Region (2022A03012).
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
