Abstract
In this prospective study, we examined the effect of atorvastatin treatment on baroreflex sensitivity (BRS) in subjects with type 2 diabetes. A total of 79 patients with type 2 diabetes with dyslipidaemia were recruited. A total of 46 subjects were enrolled to atorvastatin 10 mg daily and low-fat diet and 33 patients to low-fat diet only. BRS was assessed non-invasively using the sequence method at baseline, 3, 6 and 12 months. Treatment with atorvastatin increased BRS after 12 months (from 6.46 ± 2.79 ms/mmHg to 8.05 ± 4.28 ms/mmHg, p = 0.03), while no effect was seen with low-fat diet. Further sub-analysis according to obesity status showed that BRS increased significantly only in the non-obese group (p = 0.036). A low dose of atorvastatin increased BRS in non-obese subjects with type 2 diabetes and dyslipidaemia after 1-year treatment. This finding emphasizes the beneficial effect of atorvastatin on cardiovascular system, beyond the lipid-lowering effects.
Introduction
The arterial baroreflex system represents an important homeostatic mechanism of the cardiovascular system. The baroreceptors are stretch-activated receptors that regulate short-term changes of arterial blood pressure (BP) and react to large fluctuations of BP. They are mainly embedded in the smooth muscle layers of the carotid sinuses and of the aortic arch. 1 Once systemic arterial pressure is raised, the distention of the arterial wall activates the baroreceptors. Activated baroreceptors send stimulus to the nucleus tractus solitarii of the brainstem that lead to an increase of the parasympathetic activity and a decrease of sympathetic discharge in both the heart and the peripheral blood vessels. These result in slower heart rate, decreased cardiac contractility, decreased peripheral vascular resistance and thus lower arterial BP. 1
Baroreflex sensitivity (BRS) is an index of arterial baroreceptors’ function. Reduced BRS has been associated with fatal ventricular arrhythmias after myocardium infarction 2 and impaired regulation of BP. 3 Recently, new non-invasive methods of estimating ‘spontaneous’ baroreflex have been described. They are based on quantification of heart rate adjustment per unit of spontaneous change of systolic BP (SBP), obtained automatically from computer analysis of non-invasive electrocardiographic (ECG) and arterial pressure wave recordings. 4
BRS is reduced early in the course of both type 1 and type 2 diabetes mellitus, even in the absence of clinically overt autonomic neuropathy.5,6 In addition, BRS has been negatively correlated with serum cholesterol, low-density lipoprotein cholesterol (LDLc) and triglycerides (TG). 7
Inhibitors of 5-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase have been shown to reduce cardiovascular morbidity and mortality in primary and secondary prevention studies far beyond the expected lipid-lowering effect. The reduced mortality rate achieved in large intervention studies suggests a favourable influence of statins on autonomic nervous system. Statins have been shown to reduce sympathetic activity and improve BRS in patients without diabetes with hypertension and hypercholesterolaemia.8,9 However, no studies exist on the potential effect of statins on BRS in subjects with diabetes. The aim of this study was to investigate effect of treatment for 1 year with atorvastatin on BRS in subjects with type 2 diabetes and dyslipidaemia.
Methods
Subjects
Subjects with type 2 diabetes diagnosed according to the criteria recommended by the American Diabetes Association, 10 participated in this prospective study. All patients were recruited consecutively from the outpatient diabetes clinic of our hospital. Inclusion criteria required that they were 45–75 years of age, non-smokers, with serum LDLc > 100 mg/dL and HbA1c ≤ 8.5%. Any lipid-lowering therapy should have been stopped at least 8 weeks prior to enrolment. Exclusive criteria were a history of coronary artery disease, heart failure or cardiac arrhythmia, uncontrolled hypertension, cerebrovascular accident or transient ischaemic attack and symptoms or findings of peripheral arterial disease. We also excluded patients with known hypersensitivity to statins, malignancy, acute liver disease or known hepatic dysfunction (aspartate aminotransferase >2 times the upper normal limits), chronic renal failure, macroalbuminuria (expressed as albumin to creatinine ratio >300 µg/dL), creatine kinase >3 times the upper limits or untreated hypothyroidism [thyroid stimulating hormone (TSH) >5.5 mU/mL]. Patients who consumed more than four alcoholic drinks per day, or who used estrogens, macrolides, systemic steroids, immunosuppressive drugs, erythromycin or antifungal agents were not included. None of the subjects used BP medication or other medication with known effect on autonomic nervous system activity, and none had lost or gained >5% of their usual weight during the 6 months preceding the study. Antihypertensive and hypoglycaemic therapy had to remain stable during the study. All subjects gave their written informed consent before enrolment. The study protocol was approved by the ethics committee of our hospital.
This was an open-label, non-randomized, parallel group, prospective study. A total of 157 subjects were screened; from these, 42 subjects were excluded because they have been exposed to lipid-lowering medications in the past, 12 have been treated with β-adrenergic blockers and 22 were found to have cardiovascular disease or autonomic neuropathy. From the remaining 81 subjects, 48 patients agreed to receive atorvastatin 10 mg daily for 12 months and 46 completed the study (2 patients discontinued the therapy due to myalgia); 33 subjects refused to receive treatment with atorvastatin and served as controls.
Analytical methods
Laboratory determinations were made in the morning in a quiet room with stable temperature 22°C–24°C. Body weight, height, waist and hip circumferences were measured in light clothing; body mass index (BMI) and waist-to-hip ratio (WHR) were calculated. Participants were asked not to take their diabetic and antihypertensive therapy on the morning of each visit. After an overnight fast, venous blood was drawn. Plasma lipids, serum glucose, creatinine and transaminases were measured enzymatically on an automatic analyser. LDLc was estimated using the equation of Friedewald et al. 11 HbA1c was determined using a quantitative assay (DCA Vantage™ Analyzer; Siemens, Deerfield, IL, USA). High-sensitivity C-reactive protein (hsCRP) level determination was performed by fully mechanized automated latex-particle-enhanced immunonephelometric assays on the BN ProSpec nephelometer (Dade Behring, Siemens Healthcare Diagnostics, Liederbach, Germany). The intra- and inter-assay coefficients of variation (CVs) were less than 6% and 7%, respectively. Glomerular filtration rate (GFR) was calculated according to the equation of Cockcroft and Gault. 12
Measurement of BP and BRS
Brachial BP was measured using a semiautomatic BP device with the subjects rested for 5 min. Three measurements were made 1 min apart, and the mean value of the last two measurements was taken as representative of brachial BP. Hypertension was defined as SBP ≥140 mmHg, diastolic BP (DBP) ≥ 90 mmHg or both or use of antihypertensive therapy. All patients underwent three more visits, at 3, 6 and 12 months, during which the same clinical examination, blood tests and BRS estimation were repeated. At each visit, subjects were encouraged to continue with their usual meal plan and physical exercise during the study.
BRS was estimated by the spontaneous sequence method, as described by Parati et al., 13 using the BaroCor System (AtCor Medical, Sydney, NSW, Australia). All patients were examined in the supine position. After application and adjustment of the measuring devices, a period of 30 min was allowed for acclimatization. ECG signal with a three-lead electrocardiogram and beat-to-beat BP were continuously and simultaneously recorded for 20 min. Continuous BP measurements were made using a tonometer (CBM 7000; Colins Medical Instruments Corp., San Antonio, TX, USA) adjusted on the surface of the radial artery of the dominant arm, which enables the derivation non-invasively of the ascending aortic BP waveform via a validated generalized transfer function. 14 Time series of inter-beat (RR) intervals and SBP were analysed by the BaroCor System Software to identify sequences in which SBP and RR intervals increased or decreased concurrently over at least three cardiac cycles. A linear correlation between RR interval and SBP was applied for each sequence, and the regression slope was calculated. The average value of the individual slopes was taken as the measure of BRS and expressed in ms/mmHg. The minimum accepted changes for SBP and RR intervals were equal to or greater than 1 mmHg and 5 ms, respectively. Lag 0 value of central BRS was selected for each subject measurement.
Assessment for cardiac autonomic neuropathy
Cardiac autonomic neuropathy (CAN) was assessed using the cardiovascular function tests proposed by Ewing and Clarke and described in detail previously. 15 Briefly, the heart rate response to slow, deep breathing, Valsalva manoeuvre and to standing up were assessed from ECG recordings of RR intervals using the computer-aided examination and evaluation system VariaCardio TF4 (Medical Research Limited, Leeds, UK). In addition, changes in BP during the handigrip test and after resuming the standing up from the lying positions were measured and evaluated. 15 CAN was diagnosed when two of the five tests were abnormal.
Statistical analysis
Statistical analysis was performed using the SPSS 15.0 statistical package (SPSS, Inc., Chicago, IL, USA). All data were assessed for normal distribution of their values. Parameters are presented as means ± standard deviation (SD), unless indicated otherwise. The comparison of baseline data between the two groups was performed by the paired t-test or the Wilcoxon test. The analysis of variance (ANOVA) for repeated measurements was used to evaluate the effect of atorvastatin and diet therapy on clinical and laboratory values during the study. The Greenhouse–Geisser adjustment was used when the sphericity assumption was not fulfilled. p < 0.05 was considered statistically significant.
Results
The demographic and clinical characteristics of the subjects are presented in Table 1. The two groups were comparable in baseline characteristics. As expected, treatment with atorvastatin for 12 months reduced total cholesterol significantly by 26.55% (p < 0.001), LDLc by 36.15% (p < 0.001) and TG by 11.24% (p = 0.001), while high-density lipoprotein cholesterol (HDLc) levels remained unchanged (p = 0.53; Table 2). In the control group, no significant change in lipids was seen. No significant differences were found during the study in BMI, BP, heart rate, HbA1c, fasting glucose and GFR in either study group (Table 2).
Demographic and clinical characteristics of the study subjects.
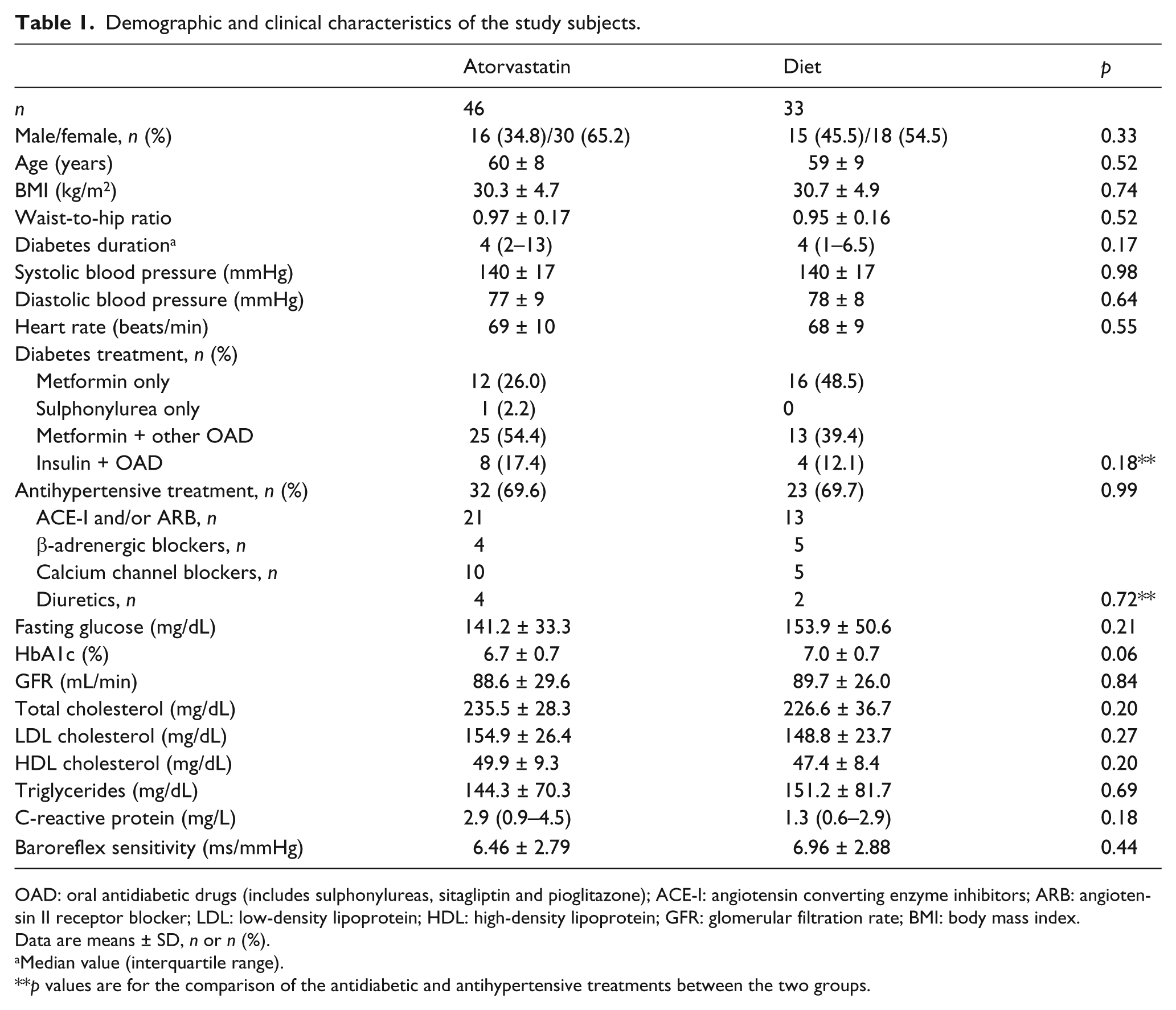
OAD: oral antidiabetic drugs (includes sulphonylureas, sitagliptin and pioglitazone); ACE-I: angiotensin converting enzyme inhibitors; ARB: angiotensin II receptor blocker; LDL: low-density lipoprotein; HDL: high-density lipoprotein; GFR: glomerular filtration rate; BMI: body mass index.
Data are means ± SD, n or n (%).
Median value (interquartile range).
p values are for the comparison of the antidiabetic and antihypertensive treatments between the two groups.
Clinical characteristics of patients in atorvastatin and diet-treated group at baseline, 3, 6 and 12 months.
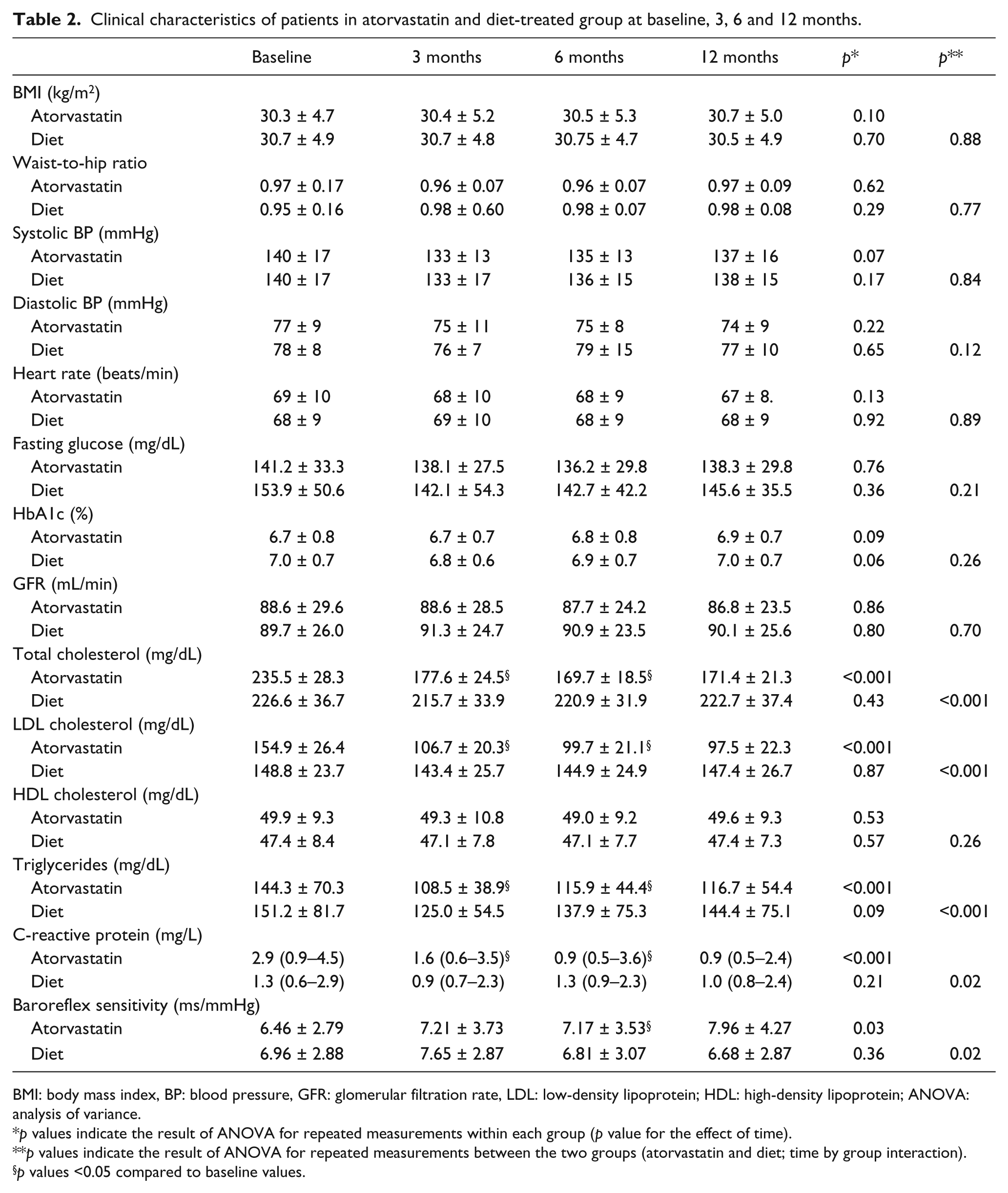
BMI: body mass index, BP: blood pressure, GFR: glomerular filtration rate, LDL: low-density lipoprotein; HDL: high-density lipoprotein; ANOVA: analysis of variance.
p values indicate the result of ANOVA for repeated measurements within each group (p value for the effect of time).
p values indicate the result of ANOVA for repeated measurements between the two groups (atorvastatin and diet; time by group interaction).
p values <0.05 compared to baseline values.
Subjects treated with atorvastatin showed a significant improvement in BRS after 12 months of therapy; this improvement was apparent after 6 months of initiation of therapy (p = 0.03 vs baseline) and was enhanced further at 12 months; no such effect was seen in the control group (Table 2; Figure 1(a)).
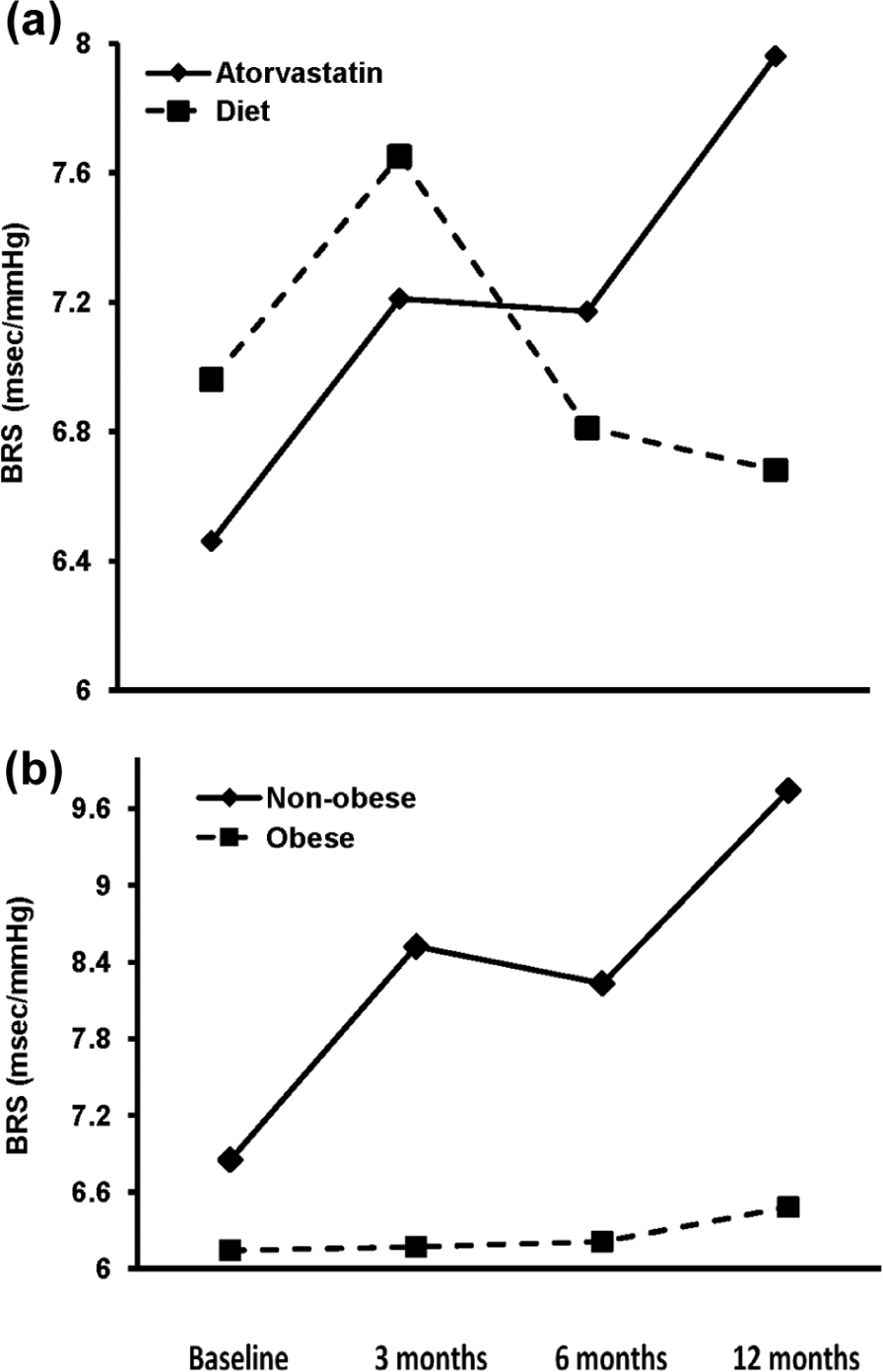
(a) Effect of atorvastatin or diet on BRS after 3, 6 and 12 months and (b) sub-analysis of BRS values in patients treated with atorvastatin according to BMI. Obese: BMI ≥ 30 kg/m2 and non-obese: BMI < 30 kg/m2. (a) Diet group: p value for the effect of time = 0.36; atorvastatin group – p value for the effect of time = 0.03, p value for the group (diet and atorvastatin) effect = 0.04, p value for the time by group (diet and atorvastatin) interaction = 0.02. (b) Non-obese group: p value for the effect of time = 0.03; obese group – p value for the effect of time =0.66, p value for the group (non-obese and obese) effect = 0.03, p value for the time by group (non-obese and obese) interaction = 0.03.
A significant reduction of hsCRP by 41.84% (p < 0.001) was also observed in the atorvastatin group. This decrease was seen at 3 months after initiation of atorvastatin therapy (by 31.25%, p = 0.001 vs baseline) and was maintained at 12 months.
Since obesity is associated with reduced BRS in subjects without diabetes, 14 sub-analysis was performed to examine whether the effect of atorvastatin on BRS was apparent in obese (BMI ≥ 30 kg/m2; n = 25) and non-obese (BMI < 30 kg/m2; n = 21) participants. This analysis showed that BRS improved significantly only in the non-obese group (from 6.84 ± 3.15 ms/mmHg to 9.74 ± 5.29 ms/mmHg, p = 0.03, at 12 months). This effect was apparent at 3 months after initiation of atorvastatin (from 6.84 ± 3.15 ms/mmHg at baseline to 8.52 ± 4.08 ms/mmHg at 3 months, p = 0.03) and was maintained after 6 months therapy (from 6.84 ± 3.15 ms/mmHg at baseline to 8.22 ± 3.77 ms/mmHg at 6 months, p = 0.03). In the obese patients, treatment with atorvastatin did not affect BRS (at baseline: 6.14 ± 2.48 ms/mmHg; at 12 months: 6.47 ± 2.40 ms/mmHg, p = 0.66; Figure 1(b)). The same analysis in the control group showed that BRS did not change during the study, irrespective of the obesity status.
Changes in total cholesterol, LDLc, TG and hsCRP between baseline and 12 months did not correlate with changes in BRS (p > 0.05). Additionally, the median (interquartile range) values of the changes in plasma lipids between baseline and 12 months in the group of subjects who received atorvastatin were not significantly different between lean and obese subjects [total cholesterol: lean −59.0 (−38.5, −78.0) mg/dL and obese −65.0 (−47.5, −82.5) mg/dL, p = 0.47; LDLc: lean −59.0 (−33.5, −78.0) mg/dL and obese −55.0 (−42.0, −77.5) mg/dL, p = 0.72; HDLc: lean 0 (−1.0, 3.0) mg/dL and obese 0 (−1.0, 2.0) mg/dL, p = 0.53; and TG: lean −27.0 (−14.0, −66.5) mg/dL and obese −18.0 (−4.5, −49.5) mg/dL, p = 0.46]. The same was valid for the changes in plasma CRP levels [lean −0.80 (−0.25, −2.03) mg/dL and obese −0.74 (−0.01, −2.87) mg/dL, p = 0.85]. Furthermore, changes in these parameters did not correlate significantly (p > 0.05) with changes in BRS in either group.
Discussion
The novel finding of this study is that treatment with atorvastatin improves BRS in subjects with type 2 diabetes and dyslipidaemia. The clinical significance of BRS estimation in diabetes has emerged over the recent years. Both sympathetic and parasympathetic branches of the baroreflex loop are impaired in diabetic subjects. 16 Diabetic autonomic dysfunction, even subclinical, has been associated with increased mortality. 17 Studies in patients with and without CAN indicate that both time- and frequency-domain measures of spontaneous BRS could allow earlier detection of CAN than the conventional autonomic function tests.5,18 In the early 1980s, Ewing and Clarke proposed five simple non-invasive cardiovascular reflex tests, which have been qualified as specific markers of autonomic nervous system activity. 15 Yet, time- and frequency-domain analyses of BP and heart rate have been shown to be more sensitive for the identification of early autonomic dysfunction in patients with diabetes compared to the classical ‘Ewing’ tests.6,19 Non-invasive methods for the assessment of BRS have gained interest in the last years due to their minimal discomfort of the participants and the low cost and risk. 20 They are based on the combined analysis of spontaneous BP and heart rate fluctuations, evaluating the interaction between ‘spontaneous’ baroreflex function and the daily life modulation of cardiovascular parameters. In this study, we obtained continuous indirect estimation of aortic BP based on the radial artery recordings, as already described. 14
Among statins, atorvastatin and simvastatin have been previously examined for their effects on BRS in humans without diabetes. Administration of 10 mg of atorvastatin for 6 weeks in a small group of 10 men with hypercholesterolaemia resulted in a significant increase of noradrenaline-induced BRS compared to diet or diet plus cholestyramine therapy. 21 In a similar study of 10 male patients with hypertension and untreated hypercholesterolaemia, atorvastatin (20 mg daily) improved BRS significantly after 8 weeks, compared to controls. 9 In another study of 29 non-obese, non-diabetic, normotensive patients with combined hyperlipidaemia, atorvastatin (10 mg daily) increased BRS significantly after 10 weeks and reduced SBP compared to fenofibrate. 22 Simvastatin therapy for 8 weeks in 31 hypertensive subjects with hypercholesterolaemia improved BRS significantly compared to placebo, with no changes being observed in BP. 8 Our prospective study provides the first long-term evidence for the beneficial effect of atorvastatin on BRS in patients with diabetes.
In this study, we found that the changes in BRS between baseline and 12 months did not correlate with changes in plasma lipids or hsCRP, and no significant changes in BP or heart rate were noticed. The pathophysiologic pathway that underlies the effects of HMG-CoA reductase inhibitors on BRS has not been clearly elucidated. Four mechanisms have been proposed to be involved: up-regulation of nitric oxide (NO) production, down-regulation of angiotensin II type-1 (AT1) receptor function, reduction in endothelin-1 and reduction of non-esterified fatty acids (NEFAs).
Nitric oxide is a vasodilator agent synthesized from the amino acid
The inhibitory effect of statins on sympathetic nervous system may also be mediated by the down-regulation of the renin–angiotensin system and of endothelin. Increased LDLc is associated with significant over-expression of AT1 receptors. 25 The AT1 receptors mediate many biological effects of angiotensin II, including sympathetic activation. 26 Blockage of AT1 receptors reduces sympathetic tone in patients with metabolic syndrome and improves arterial baroreflex function. 27 Statins reduce central and peripheral AT1 receptors’ activity, restoring arterial baroreflex control. 28
Endothelin-1 (ET-1) is a powerful vasoconstrictor amino peptide synthesized by vascular endothelial cells. Experimental evidence suggests that ET-1 stimulate central and peripheral sympathetic nervous system activity. 29 Atorvastatin has been shown to reduce significantly the synthesis of ET-1 and the expression of its precursor pre–proET1 messenger RNA (mRNA), 30 leading to down-regulation of the sympathetic tone. The improvement of BRS may also be related to the reduction of the plasma levels of the NEFAs induced by statins. Increased levels of NEFAs have been associated with increased BP and heart rate and increased systemic vascular resistance in humans. 31 An elevation of plasma NEFAs acutely impairs BRS. 32 Atorvastatin has been shown to reduce NEFAs in subjects with type 2 diabetes, 33 providing probably an additional sympatho-inhibitory effect.
A meta-analysis concluded that statins may reduce BP through increasing NO, reducing arterial stiffness and improving endothelial function. 34 However, in accordance with previous studies,8,9 we did not find any significant effect of atorvastatin on either BP or heart rate; thus, the increase of BRS cannot be attributed to changes in these factors.
Our study showed a significant decrease of hsCRP, as soon as 3 months after initiation of atorvastatin, which is in agreement with other studies. 35 High levels of hsCRP have been associated with decreased cardiovascular autonomic function in patients with type 2 diabetes. 36 Serum hsCRP has been positively correlated with arterial stiffness in patients with diabetes. 37 We also found that BRS was significantly and negatively 8 correlated with other metabolic risk factors that influence the elastic properties of arteries, including serum TG and fasting glucose, in line with recent reports.7,14 Autonomic dysfunction has been associated with arterial stiffness in subjects with type 2 diabetes. 38 BRS has also been correlated independently with aortic pulse wave velocity, an index of arterial distensibility. 7 Thus, the decrease of inflammation and the improvement of the elastic properties of the carotid arteries and of the aortic arch, due to the pleiotropic effects of atorvastatin, may account for the higher sensitivity of the embedded baroreceptors and the increase of BRS.
When patients treated with atorvastatin were categorized to obese and non-obese, we found a differential effect of atorvastatin on BRS. Only non-obese subjects showed improvement in BRS. An inverse relationship between obesity and BRS has been constantly reported in the past.14,39 Obesity is characterized by increased sympathetic and reduced parasympathetic nervous system activity. 16 Body weight loss up to 10% results in an increase in parasympathetic and decrease in sympathetic activity in obese subjects. 40 Since no significant change in BMI was observed during this study in either group, it can be hypothesized that the autonomic perturbations associated with obesity are more potent regulators of BRS, blunting the beneficial effect of atorvastatin on baroreceptors function.
Our study has the advantage that, in contrast with previous studies, we enrolled a control group, and the duration of the study was the longest mentioned so far. Yet, limitations should also be mentioned. First, the number of the subjects involved was not large, and the study was not a randomized, double blind, placebo-controlled trial. Second, we studied individuals free of macrovascular complications; therefore, our findings cannot be extrapolated to the total diabetic population. Third, we used a non-invasive method to assess BRS; direct invasive methods, such as injection of vasoactive drugs, may be more accurate, but are impractical in clinical practice. In addition, physical activity may affect BRS. However, data on physical activity were not assessed at baseline or during the study; thus, potential differences in physical activity level among the participants may have confounded the results. Moreover, the improvement in BRS with atorvastatin on BRS was observed after 6 months of therapy. Previous studies showed that BRS increased 10 weeks after initiation of treatment with atorvastatin or simvastatin;8,9 however, none of these studies included diabetic patients, so any comparison should be made with caution.
This study provides the first long-term evidence that treatment with atorvastatin improves BRS in patients with type 2 diabetes and dyslipidaemia. To date, there have been limited studies addressing the effects of statins on baroreflex function. The divergent results observed in obese and non-obese patients suggest that obese subjects who manage to lose extra body weight may have additional cardiovascular benefits from atorvastatin. The underlying mechanisms involved in the improvement in BRS after treatment with atorvastatin in subjects with type 2 diabetes remain to be investigated. Our findings suggest another favourable effect of atorvastatin on BRS in patients with type 2 diabetes.
Footnotes
Declaration of conflicting interests
The authors declare that they have no conflict of interest.
Funding
This study was supported in part by a research grant (S.A.R.G. 70/3/8615) from the National University of Athens.
