Abstract
Aims:
The present study aims to investigate the relationship between cardiac autonomic function (CAF) and glucose variability (GV) and HOMA-IR in subjects with prediabetes and normal glucose tolerance (NGT).
Material and methods:
Ninety-two subjects (59 with prediabetes and 33 with NGT), of mean age 50.3 ± 11.5 years, mean BMI 30.4 ± 6.0 kg/m2, were included in this cross-sectional study. Glucose tolerance was assessed by OGTT according to WHO 2006 criteria. Glucose, HbA1c, insulin, oxLDL, and 3-Nitrotyrosine were measured. CGM was performed with a blinded sensor (FreeStyle Libre Pro). CAF was assessed by ANX-3.0 technology.
Results:
GV indices were increased in prediabetes. CAF was suppressed in subjects with any stage of dysglycemia. The prevalence of cardiac autonomic dysfunction was higher in prediabetes −20.3% as compared to NGT −3.0%, p = 0.028. HOMA-IR [OR 1.5 (95% CI: 1.1–2.1), p = 0.010] and time in target range [OR 0.8 (95% CI: 0.67–0.97), p = 0.021] were found to be predictive variables for impaired CAF. Sympathetic and parasympathetic activity negatively correlated with mean glycemia and GV indices and were independently related to JINDEX in prediabetes (F[1, 47] = 5.76, p = 0.021 and F[1, 47] = 5.94, p = 0.019, respectively); and to time above target range in NGT (F[1, 18] = 4.48, p = 0.049 and F[1, 18] = 4.65, p = 0.046, respectively).
Conclusion:
CAF is declined in prediabetes and seems to be related to GV and HOMA-IR at early stages of dysglycemia.
Introduction
Type 2 diabetes (T2D) is associated with an increased risk for cardiovascular morbidity and mortality. 1 There is an elevated risk of cardiovascular disease even in prediabetes. 2 Impairment of cardiac autonomic function (CAF) is a serious life-threatening complication of diabetes, being a leading independent risk factor for cardiovascular death in diabetes. 3 Data suggest that CAF deterioration exists long before the onset of T2D. It has been observed at very early stages of dysglycemia. 4 Because of its paramount importance for the cardiovascular risk status of subjects with T2D and even with prediabetes, current research is focused on the putative risk factors for CAF impairment at different stages of impaired glucose tolerance.
Chronic hyperglycemia is a major risk factor for CAF deterioration in T2D.5,6 However, improved glycemic levels alone do not show any improvement in CAF in T2D. 7 Therefore, the attention has been drawn to the role of glucose variability (GV). It has been suggested that GV may have a more deleterious effect than sustained hyperglycemia in the development of cardiac autonomic dysfunction in T2D8–13 and in type 1 diabetes.14–16 Available data on this association in a high-risk population with prediabetes are very scarce.
Continuous glucose monitoring (CGM) has become a reliable tool for the measurement of glucose excursions. 17 Data on GV in subjects with early alterations of glycemia have shown that worsening of glucose tolerance is related not only to an increase in mean glucose levels, but also to higher GV indices. 18 These results raise the question of the potential contribution of GV to the development of cardiac autonomic dysfunction and the increased cardiovascular risk in this population.19,20
Thus, the aim of the present study was to assess the relationship between parameters of CAF and some indices of GV, by means of continuous glucose monitoring, and HOMA-IR at early stages of glucose intolerance.
Material and methods
Study design and settings, ethics, informed consent
A total of 92 subjects (20 males), of mean age 50.3 ± 11.5 years, mean BMI 30.4 ± 6.0 kg/m2, divided into two groups – 59 with prediabetes (21 with impaired fasting glucose [IFG], 23 with impaired glucose tolerance [IGT], and 15 with IFG + IGT) and 33 with normal glucose tolerance (NGT), were included in this cross-sectional study.
The study was conducted at the Division of Diabetology, Department of Endocrinology, Medical University – Sofia. This is a public tertiary care center. The participants were recruited from the outpatient clinic within an ongoing screening program for prevention of type 2 diabetes. The procedures were covered by public financing.
All participants received written information about the aim and design of the study and possible risks by participating in the study, and signed informed consent in accordance with the Helsinki Declaration and rules of Good Clinical Practice and the study was approved by the Ethics Committee of the Medical University – Sofia. Informed consent was obtained before any study related activity. All participants were interviewed and screened for the risk of type 2 diabetes based on the evaluation of FINDRISC questionnaire.
Inclusion and exclusion criteria
Subjects were eligible to be included in this study if all the following criteria apply:
Male or female, age from 30 to 65 years.
High estimated risk of developing prediabetes and diabetes based on FINDRISC questionnaire (scores ⩾12 and ⩾10, respectively, as validated for a Bulgarian population), assessed ⩽3 months prior to the day of screening.
Diagnosed with NGT (fasting plasma glucose (PG) < 6.1 mmol/L and 2-h PG < 7.8 mmol/L), with IFG (fasting PG ⩾ 6.1 mmol/L and <7.0 mmol/L and 2-h PG < 7.8 mmol/L), with IGT (fasting PG < 6.1 mmol/L and 2-h PG ⩾ 7.8 mmol/L and <11.1 mmol/L), and with IFG+IGT (fasting PG < 7.0 mmol/L and 2-h PG ⩾ 7.8 mmol/L and <11.1 mmol/L).
eGFR-MDRD ⩾ 45 mL/min/1.73 m2.
Subjects were excluded from the present study if any of the following criteria apply:
Male or female, age <30 years or >65 years.
Treatment with anti-hyperglycemic or anti-obesity medications.
Diagnosed type 1 or type 2 diabetes.
Arrhythmias or taking antiarrhythmic drugs.
History of myocardial infarction, stroke or hospitalization for unstable angina pectoris.
Presence or history of malignant neoplasm within 5 years prior to the day of screening.
Other serious comorbidities.
Pregnancy.
Measurements and definitions of measurements
Metabolic parameters
Anthropometric parameters (height, weight) were measured and BMI was calculated.
Glucose tolerance was assessed by a standard OGTT with 75 g anhydrous glucose after an overnight fast, performed between 8.00 and 9.00 a.m., refraining from eating, drinking coffee, smoking and taking medications, and staying at rest during the test. Glucose tolerance categories were defined according to WHO 2006 criteria.
Fasting and postload plasma glucose (hexokinase enzyme method [Roche Diagnostics]) and serum insulin (electrochemiluminescence method) were measured in all participants. Homeostatic Model Assessment for Insulin Resistance (HOMA-IR) index was calculated using the formula: HOMA-IR =
Oxidative stress parameters
Fasting and postload oxLDL and 3-Nitrotyrosine (ELISA method [Cusabio Biotech Co., Ltd]) were measured in 50 (18 with NGT and 32 with prediabetes) of the participants.
Glucose variability parameters
Continuous glucose monitoring (CGM) was performed with a blinded sensor (FreeStyle Libre Pro, Abbot GmbH & KG) for a mean period of 13.7 ± 1.8 days in routine everyday setting, measuring interstitial glucose concentration by a glucose oxidase method every 15 min, not requiring calibration. 21 Detailed information was provided to the participants on how to stick to their everyday usual lifestyle without any change in dietary regimen and exercise for the period with the sensor in order to obtain data under standard conditions. The following GV parameters were estimated: time in target range defined as glucose concentration between 3.0 and 7.8 mmol/L, standard deviation (SD) defined as the amount of dispersion of a data set, coefficient of variation (CV) defined as SD with correction for the mean glucose, calculated using a computer software (Excel, MicroSoft Office), CONGA (continuous overall net glycemic action), MAGE (mean amplitude of glycemic excursions), MAG (mean absolute glucose), M-value, LBGI (low blood glucose index), HBGI (high blood glucose index), J-index, L-index (lability index), GRADE (glycemic risk assessment in diabetes equation), computed with EasyGV calculator (version 9.0.R2). 22 All GV parameters were calculated on the basis of the raw sensor data on overall measured glucose concentrations for the whole studied period.
Cardiac autonomic function assessment
Cardiac autonomic nervous system function was evaluated with АNX-3.0 autonomic monitoring system (ANSAR Medical Technologies, Inc., Philadelphia, PA) using standard clinical tests: deep breathing, Valsalva maneuver and standing from a seated position. The ANX-3.0 method non-invasively, simultanuously and independently computes sympathetic and parasympathetic activity, applying cardio-respiratory synchronization with concomitant spectral analysis of respiratory activity and heart rate variability. 23 The study was performed 24 h after the last dose of medications affecting autonomic function – antihypertensives, tricyclic antidepressants and SSRIs; while refraining from drinking coffee and smoking 12 h prior to the test; at least 30 min after the last meal; between 8 and 11 a.m.
Confirmed cardiac autonomic dysfunction was defined based on the Ewing’s standard clinical tests as the presence of at least two pathological tests. This means the presence of lower values in two of the tests, calculated by the software and based on the specific age and sex “cut-offs.”
Statistical analyses
Statistical analyses were performed with SPSS versus 23.0. The Kolmogorov Smirnov test of normality was used to determine data distribution. The variables with skewed distribution were analyzed after logarithmic transformation. Descriptive analysis, a single-factor dispersion analysis of variance (ANOVA) with Tamhane’s T2 post hoc analysis, correlation analysis with parametric (Pearson) coefficient and multiple regression analysis with stepwise method were performed for comparison between the groups with different glucose tolerance. Logistic regression analysis with Forward LR method, controlling for age, was performed to determine predictive variables for the presence of cardiac autonomic dysfunction. Principal component analysis was performed to define a principal component variable for sympathetic and parasympathetic activity at rest and during clinical tests. Data are presented as means with standard deviation for the variables with normal distribution and as medians with interquartile ranges for the variables without normal distribution. p-value of < 0.05 (2-sided) was considered statistically significant. After Bonferroni Correction the critical p-value is p = 0.017.
Results
Metabolic and autonomic function parameters in the groups
Main characteristics of the groups according to glucose tolerance are presented on Table 1. Our results show significantly increased glucose variability indices in subjects with prediabetes in comparison to NGT (Table 1). Significantly decreased both sympathetic and parasympathetic tone at baseline and after standing are found in the groups with prediabetes (Table 1).
Main characteristics of the groups according to glucose tolerance – normal glucose tolerance (NGT) and prediabetes, and the subgroups with different categories of prediabetes – impaired fasting glucose (IFG), impaired glucose tolerance (IGT), and IFG + IGT.
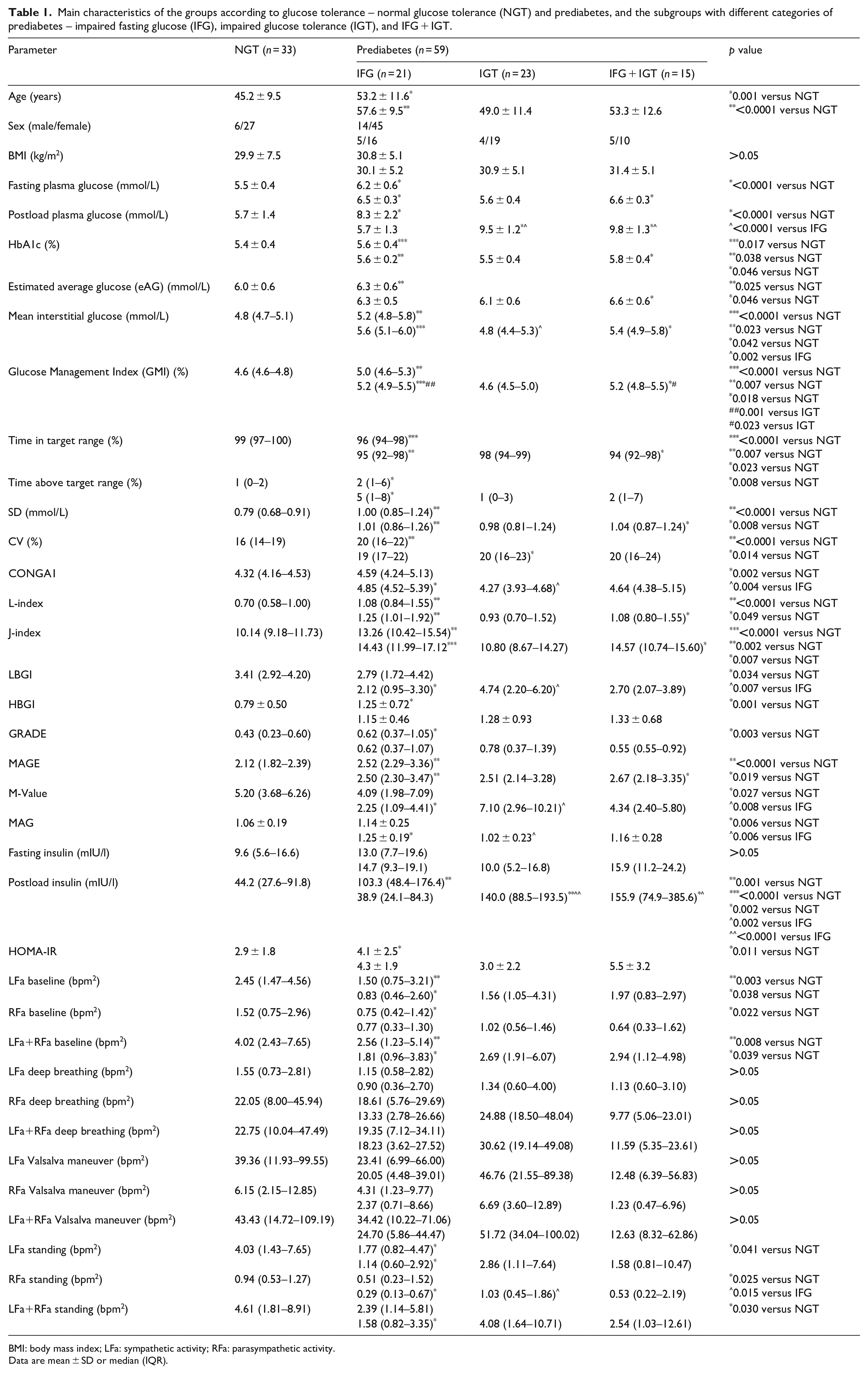
BMI: body mass index; LFa: sympathetic activity; RFa: parasympathetic activity.
Data are mean ± SD or median (IQR).
Prevalence and main determinants of cardiac autonomic dysfunction
The prevalence of confirmed cardiac autonomic dysfunction is 14.1% in the whole cohort, being significantly higher in prediabetes – 20.3% as compared to NGT – 3.0%, p = 0.028. After dividing the participants into groups according to the presence of cardiac autonomic dysfunction, logistic regression analysis with Forward LR method determines HOMA-IR [OR 1.5 (95% CI: 1.1–2.1), p = 0.010] and time in target range [OR 0.8 (95% CI: 0.67–0.97), p = 0.021] as predictive variables controlling for age for the presence of cardiac autonomic dysfunction in the studied cohort.
Relationship between parameters of cardiac autonomic function and mean glucose and GV indices
There is a significant reciprocal correlation between sympathetic and parasympathetic activity and measurements of both mean levels of glycemia, including glycated hemoglobin, estimated average glucose, glucose management index, and mean interstitial glucose; and most of the GV indices, including CONGA1, JINDEX, LI, LBGI, MAG, and M-Value in the studied cohort (Table 2). Both sympathetic and parasympathetic tone are independently related to JINDEX in the whole cohort (F[1, 66] = 5.30, p = 0.025), and in prediabetes (F[1, 47] = 5.76, p = 0.021 for the sympathetic activity and F[1, 47] = 5.94, p = 0.019 for the parasympathetic activity); and to time above target range in NGT (F[1, 18] = 4.48, p = 0.049 for the sympathetic activity and F[1, 18] = 4.65, p = 0.046 for the parasympathetic activity). The multiple correlation coefficients showed that about 11% of the variance of both sympathetic and parasympathetic tone might be accounted for by JINDEX in prediabetes and approximately 21% of the variance of cardiac autonomic function – by time above target range in NGT (Table 2).
Relationship between the parameters of cardiac autonomic function and glycemic indexes.
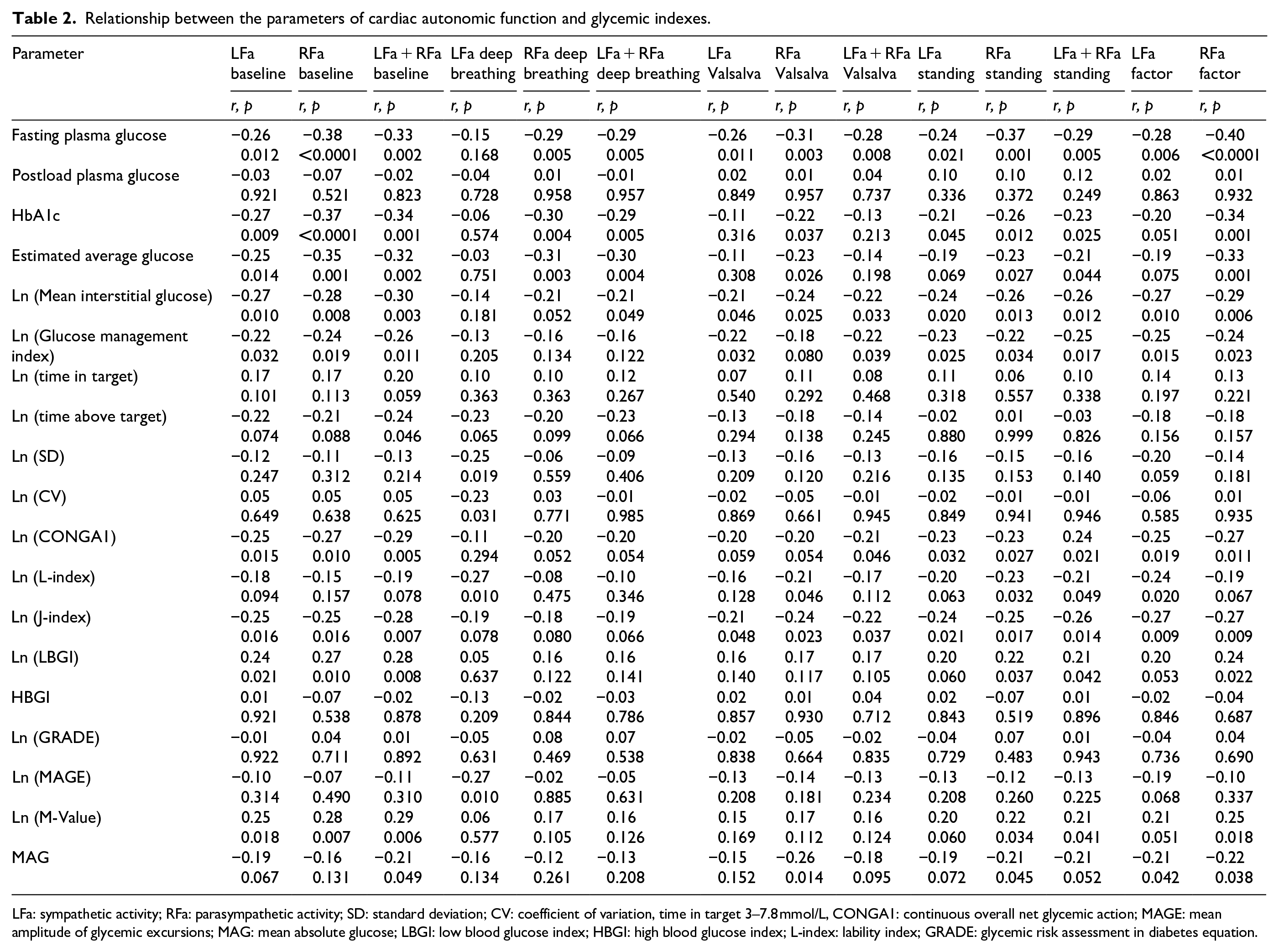
LFa: sympathetic activity; RFa: parasympathetic activity; SD: standard deviation; CV: coefficient of variation, time in target 3–7.8 mmol/L, CONGA1: continuous overall net glycemic action; MAGE: mean amplitude of glycemic excursions; MAG: mean absolute glucose; LBGI: low blood glucose index; HBGI: high blood glucose index; L-index: lability index; GRADE: glycemic risk assessment in diabetes equation.

Variables included in the analysis: SD: standard deviation; CV: coefficient of variation; time in target (3–7.8 mmol/L), time above target (>7.8 mmol/L), CONGA: continuous overall net glycemic action; MAGE: mean amplitude of glycemic excursions; MAG: mean absolute glucose; M-value, LBGI: low blood glucose index; HBGI: high blood glucose index; J-index, L-index: lability index; GRADE: glycemic risk assessment in diabetes equation.
Relationship between parameters of cardiac autonomic function and oxidative stress markers
No significant correlation between cardiac autonomic tone and the assessed markers of oxidative stress is established (Table 3).
Relationship between the parameters of cardiac autonomic function and insulin levels, insulin action index and oxidative stress markers.
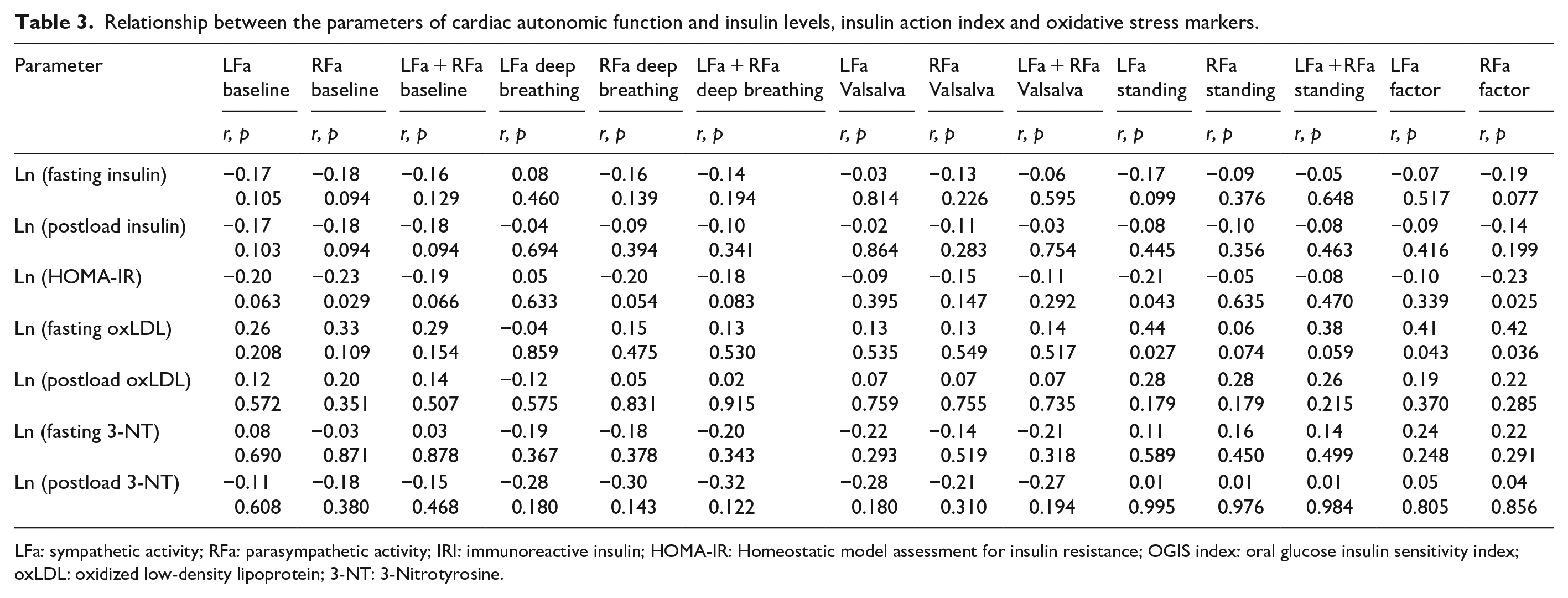
LFa: sympathetic activity; RFa: parasympathetic activity; IRI: immunoreactive insulin; HOMA-IR: Homeostatic model assessment for insulin resistance; OGIS index: oral glucose insulin sensitivity index; oxLDL: oxidized low-density lipoprotein; 3-NT: 3-Nitrotyrosine.
Discussion
Not surprisingly, our results demonstrate higher prevalence of cardiac autonomic dysfunction in prediabetes as compared to NGT and a decline in both sympathetic and parasympathetic activity at rest and after standing in subjects with any alteration of glycemia in comparison to NGT, which is in line with our previously reported data. 4 One of the predictive risk factors for the presence of confirmed cardiac autonomic dysfunction in the prediabetes state seems to be time spent in target range, which for the population without diabetes has been defined as the range of serum glucose levels between 3.0 and 7.8 mmol/L. 24 There is suggestive evidence in the literature for the pivotal role of this relatively new glycemic parameter, derived from the capabilities of the new technologies in diabetes and correlating with HbA1c, in the pathogenesis of chronic complications in T2D.25–27 Based on our findings it might be speculated that time in target range for glycemia is related to long-term complications not only in T2D, but also in prediabetes.
Given that time in target range, rather than some of the indices of GV, is found to be an independent predictor of cardiac autonomic dysfunction, suggests the pivotal role of hyperglycemia but not GV for the presence of autonomic imbalance in this high-risk population. It might be assumed that at this relatively early stage of dysglycemia there are no extreme glucose oscillations and the observed higher amplitudes of glucose excursions probably affect autonomic nerves more mildly than chronic hyperglycemia. Therefore, the effects of GV on autonomic tone should be considered just as an additional risk factor, which could not exert more deleterious effects than chronic hyperglycemia.
Our data demonstrate stronger independent relationship between HOMA-IR and cardiac autonomic dysfunction rather than with slight fluctuations in plasma glucose. Based on our data HOMA-IR is likely to be another risk factor for cardiac autonomic decline at these early stages of glucose intolerance. It has been previously reported that insulin resistance may be responsible for some derangements of the autonomic action in subjects without diabetes. 28 Therefore, there is accumulating evidence for the paramount importance of insulin action in the development of cardiac autonomic dysfunction, even in its mild stages, when still represented asymptomatically. The possible explanation of these findings is probably the inhibitory effect of insulin resistance on the main signaling pathway in the neurons, which results in impaired neurotrophic insulin effects and mitochondrial dysfunction with generation of oxidative stress. This has been supposed to be the leading underlying pathogenic mechanism for the impairments in cardiac autonomic function.
On the other hand, our results show significantly increased GV indices in subjects with prediabetes in comparison to NGT, which has already been reported. 18 Although HbA1c is the gold standard for evaluation of glycemic status, GV parameters provide the means to move beyond HbA1c in the assessment of overall glycemic state. 24 GV seems to be an important marker in addition to OGTT and HbA1c to evaluate glycemia in prediabetes. 18 Literature data have shown its importance for the overall cardio-metabolic risk independently of glucose tolerance. 29 Thus, our findings confirm the risk profile of the prediabetes state.
Though most of the indices of GV significantly differ between the participants with prediabetes and NGT, still it might be difficult to incorporate these data into individual care settings, since the magnitude of the differences is very small. For instance, CV is higher in prediabetes but still within normal reference range. Therefore, to elucidate data interpretation and to incorporate these findings into everyday clinical practice there is a need for reconsideration and definition of “cut-off” values of parameters of GV in prediabetes.
Glycemia, assessed by parameters of mean glucose levels, has proven to be the most robust marker for the presence of cardiac autonomic dysfunction in most huge studies – the Hoorn Study in subjects with diabetes, 30 the ARIC Study in subjects at early stages of glucose dysmetabolism, 31 and even the Framingham Heart Study and another population-based study in subjects with NGT.32,33
The present study addresses the question of whether there is an association between glycemic variability, characterized by the amplitude, frequency, and duration of glucose fluctuations, rather than just mean glycemia levels, and the reduced cardiac autonomic power at the very early stages of dysglycemia. Since the most reliable indices of GV are yet to be determined, especially in prediabetes, the present study evaluates a broad spectrum of GV parameters in order to establish those with higher sensitivity and diagnostic value for the presence of impaired cardiac autonomic function in prediabetes.
Both sympathetic and parasympathetic tone demonstrate a relationship with all indices of mean glycemia and just with some markers of GV. The latter, however, are among the best established parameters of GV. Our results show significant relationship between most of the indices of GV, namely JINDEX, CONGA1, MAG, M-Value and LBGI, and sympathetic and parasympathetic activity in the studied cohort. Both sympathetic and parasympathetic tone have been shown to be independently related to JINDEX in the whole cohort and in prediabetes. There is an independent relationship between both sympathetic and parasympathetic activity and time above target range >7.8 mmol/L in NGT.
Although CV is accepted to be of great importance for GV in diabetes, its significance at the very early stages of dysglycemia needs to be confirmed. Our findings outline JINDEX to be the index with the greatest input for the presence of cardiac autonomic dysfunction in prediabetes. The association between CAF and JINDEX is logical, as this index is based on SD and mean glucose. Thus, it is a measure of both mean glycemia, which has been shown to be of great importance for chronic complications in this high-risk population, and GV. In line with these findings, M-value as another hybrid measure of both mean glucose and GV shows significant association with cardiac autonomic power. On the other hand, other widely used indices of GV – CONGA1 and MAG, which are measurements predominantly of glucose fluctuations, are also found to be related to cardiac autonomic activity. Since LBGI generally estimates the risk of hypoglycemia, it might be speculated that not only in diabetes, but even in prediabetes the presence of low glucose levels is associated with an increased risk of cardiac autonomic imbalance.
NGT group data show independent relationship between the percentage of time above the target range and sympathetic and parasympathetic autonomic function. This means that the currently used “cut-offs” for euglycemia are debatable and even in people with NGT plasma glucose concentrations might exceed the normal ranges from the diagnostic tests.
In accordance with our findings, there is accumulating evidence that in addition to chronic hyperglycemia, increased GV might be an independent risk factor for the development of cardiac autonomic dysfunction. Significant association between CV – the gold standard for assessing GV8,9 and MAGE9,10 and the presence of confirmed cardiac autonomic dysfunction has been reported in T2D. Moreover, even in subjects with mild cardiac autonomic dysfunction, which is often asymptomatic, typical for the prediabetes categories and manifesting with isolated sympathovagal imbalance as measured by heart rate variability indices, there is an association between CAF markers and MAGE34,35 in T2D of different duration, including newly-diagnosed T2D. 10
Regarding type 1 diabetes, it has been reported, that heart rate variability correlates with LBGI 16 and MAGE, 35 and sympathetic and parasympathetic activity correlate with SD and MAG. 8 This is in contrast to DCCT findings, probably because the 7-point daily glucose profile is insufficient to express correctly GV as compared to CGM. 15
In addition to the results from animal models, showing that hypoglycemia influences big motor nerves, whereas hyperglycemia affects small sensory nerves, 36 our results have shown that GV indices, which are predominantly more sensitive to hyperglycemia, affect autonomic nerve fibers in prediabetes and cardiac autonomic activity is linked to time spent above target range even in NGT.
Although oxidative stress has been proposed to be one of the plausible underlying mechanisms,35,37 no association between CAF and oxidative stress markers was observed. Based on these findings it seems that the intimate mechanisms of the relationship between CAF and GV still remain unclear. The hypothesis for chronic elevation in hypothalamus-pituitary-adrenal axis activity in states of increased GV might be another putative mechanism. 38
Limitations
A main disadvantage of the study is the cross-sectional design, which does not allow the evaluation of causative relationship between the studied phenomena. This limits the power of the statistical analysis. Another limitation is that diet is not prespecified which may influence CGM results. Since age is significantly higher in some of the groups with impaired glucose tolerance, this is an additional factor influencing autonomic activity. Last but not least, the estimation of cardiac sympathetic function by ANX 3.0 technology is indirect and there is an increased risk for inaccurate results.
Conclusion
Cardiac autonomic tone is found to be declined in prediabetes, whereas glucose variability is increased in this population. Glucose variability and HOMA-IR are probably additional risk factors for cardiac autonomic function impairment at early stages of glucose intolerance. Establishing a glucose variability risk profile could be important in determining the overall risk in a high-risk non-diabetic population.
Footnotes
Acknowledgements
This work is supported by the Bulgarian Ministry of Education and Science under the National Program for Research “Young Scientists and Postdoctoral Students.”
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study is fully funded by the Medical University – Sofia – Contract no. 9-С/2016 and Contract no. D-220/2018.
