Abstract
Cardiovascular disease is the leading cause of morbidity and mortality in subjects with type 2 diabetes mellitus. Increased aortic stiffness, assessed with the carotid–femoral pulse wave velocity, is an independent risk factor for cardiovascular disease. Statins reduce effectively cardiovascular disease and mortality in high-risk patients. The aim of this prospective non-randomized, observational study was to examine the impact of treatment with either 10 mg atorvastatin plus diet or diet alone on carotid–femoral pulse wave velocity in subjects with type 2 diabetes mellitus and dyslipidaemia. A total of 79 subjects with type 2 diabetes mellitus and dyslipidaemia were included; 46 subjects were treated with atorvastatin 10 mg daily plus diet and 33 were managed by diet alone for 12 months. Carotid–femoral pulse wave velocity and carotid–radial pulse wave velocity were measured using applanation tonometry. In the atorvastatin-treated group, carotid–femoral pulse wave velocity reduced significantly during the study and there was a trend for reduction in the carotid–radial pulse wave velocity. Total cholesterol, low-density lipoprotein cholesterol, triglycerides and C-reactive protein were reduced only in the atorvastatin-treated participants. No significant changes were found in body mass index, blood pressure, heart rate, diabetes control and high-density lipoprotein cholesterol in either study group. Treatment with low-dose atorvastatin for 12 months improves carotid–femoral pulse wave velocity in subjects with type 2 diabetes mellitus and dyslipidaemia.
Introduction
Diabetes mellitus is the most common metabolic disease worldwide with a prevalence of approximately 10% in many countries. 1 The prevalence of cardiovascular disease (CVD) is higher in subjects with type 2 diabetes mellitus (T2DM) in comparison with those without diabetes,2,3 and CVD remains the leading cause of morbidity and mortality in this population.3,4 Dyslipidaemia is a frequent comorbidity of T2DM that increases significantly the risk of CVD events. 5
Carotid–femoral pulse wave velocity (cf-PWV) is a non-invasive method to determine central arterial stiffness that predicts independently CVD morbidity and mortality.6,7 Increased pulse wave velocity (PWV) serves as target organ damage in subjects with hypertension and is included in risk stratification for CVD events. 8 cf-PWV is considered as the ‘gold standard’ method for the assessment of arterial stiffness.6,8
Adult individuals with T2DM and CVD risk factors or atherosclerotic CVD are high-risk patients for CVD events, and current guidelines recommend intensive statin treatment.9,10 Except for their lipid-lowering role, statins are known for their pleiotropic effects, including those on the elastic properties of the arteries such as the possible beneficial impact on aortic wall. 11 With regard to the effect of statins on arterial stiffness, previous studies reported beneficial results in subjects with T2DM;12–16 however, in these studies different methodology and duration of intervention with statins were used. In addition, several studies in non-diabetic population reported conflicted results.17–27 Two recent meta-analyses, examined the impact of statin treatment on PWV and concluded that statin treatment reduced PWV; however, these meta-analyses did not include studies in people with T2DM, and there was significant heterogeneity in the studies analysed.28,29
The main aim of this prospective non-randomized observational study was to examine the impact of treatment with either 10 mg atorvastatin plus diet or diet alone for 12 months on cf-PWV in subjects with T2DM and dyslipidaemia, who were free of clinically apparent macrovascular disease. In addition, we examined the effect of this intervention on carotid–radial pulse wave velocity (cr-PWV).
Methods
Participants
Patients with T2DM were recruited consecutively from the Diabetes Centre of Laiko General Hospital to participate in this prospective study, as described previously. 30 The study was performed between January 2011 and December 2012. The diagnosis of T2DM was based on the criteria of the American Diabetes Association. 31 Inclusion criteria were (1) age of 47–75 years, (2) no-smoking habit, (3) serum low-density lipoprotein cholesterol (LDL-C) > 100 mg/dL, (4) glycated haemoglobin (HbA1c) ⩽ 8.5% and (5) abstain from lipid-lowering therapies for at least 8 weeks before entering the study. Subjects with known macrovascular disease defined as a history of coronary artery disease or stroke or symptoms, and findings of peripheral arterial disease were excluded. Furthermore, patients were excluded from the study if they reported heart failure or cardiac arrhythmia, hypersensitivity to statins, malignancy, acute liver disease or known hepatic dysfunction (aspartate aminotransferase > 2 times the upper normal limits). We additionally excluded individuals with stage 4 or 5 chronic kidney disease [estimated glomerular filtration rate (eGFR) < 30 mL/min/1.73 m2], creatine kinase > 3 times the upper limits or untreated hypothyroidism [thyroid stimulating hormone (TSH) > 5.5 mU/mL]. Patients who consumed more than four alcoholic drinks per day, or who received oestrogens, macrolides, systemic steroids, immunosuppressive drugs, erythromycin or anti-fungal agents or had lost or gained > 5% of their usual weight during the 6 months before entering the study were also excluded. Treatment for diabetes or hypertension had to remain stable during the study.
All subjects gave their written informed consent before entering the study. The ethics committee of Laiko General Hospital approved the study protocol which was conducted according to the Declaration of Helsinki.
Procedures
This was a non-randomized, prospective observational study. A total of 81 individuals were eligible for entering the study. All eligible participants were informed about the procedures of study and the cardiovascular benefit of statins; after a detailed explanation, participants decided on their own whether they would be taking a statin plus diet or only diet for 1 year. All participants were encouraged to follow a Mediterranean diet with instructions given by a dietician. In total, 48 patients agreed to receive atorvastatin 10 mg daily for 12 months (atorvastatin plus diet group) and 46 of them completed the study (2 patients discontinued therapy due to myalgia). In total, 33 subjects decided after their detailed information and their own desire to try with proper diet to control high cholesterol levels that they did not take atorvastatin and they were managed by diet alone (diet alone group). The main reasons the participants chose not to receive atorvastatin treatment were the fear of the side-effects of the medication and their willingness to try to reduce high cholesterol levels by diet.
The study consisted of four visits at 0, 3, 6 and 12 months. Each visit was performed in a quiet room with stable temperature 22°C–24°C. Body weight and height were measured in light clothing. Body mass index (BMI) was calculated in kg/m2. In addition, waist and hip circumferences were measured, and the waist-to-hip ratio was calculated. All subjects abstained from any food or drink apart from water for 12 h before the study and received their medications after the end of the study. Subjects were asked about diabetes duration, anti-diabetic and anti-hypertensive medication. All participants underwent a complete physical examination. Blood pressure was measured using an appropriate cuff size three times at 5-min intervals with the participants in the seated position. The mean value of the three measurements was used in the statistical analysis. Participants were classified as having hypertension when they were on anti-hypertensive treatment or if systolic blood pressure ⩾140 mm Hg and/or diastolic blood pressure ⩾90 mm Hg, according to the guidelines adopted from the European Society of Cardiology. 32
Moreover, venous blood was drawn. Plasma lipids, serum glucose, creatinine and transaminases were measured enzymatically on an automatic analyser. The equation of Friedewald et al. 33 was used to calculate the LDL-C. HbA1c was determined using a quantitative assay (DCA Vantage™ Analyzer; Siemens, Deerfield, IL, USA). The equation of Cockcroft and Gault 34 was used for calculation of the eGFR. High-sensitivity C-reactive protein (hsCRP) level determination was performed by fully mechanized automated latex-particle-enhanced immunonephelometric assays on the BN ProSpec nephelometer (Dade Behring, Siemens Healthcare Diagnostics, Liederbach, Germany). The intra- and inter-assay coefficients of variation (CVs) were less than 6% and 7%, respectively.
Assessment of pulse wave velocity
PWV was measured using a non-invasive device by applanation tonometry (Sphygmocor, AtCor Medical, Sydney, NSW, Australia). The device measures pulse transit time between the common carotid artery and the common femoral artery to determine cf-PWV, as well as pulse transit time between the common carotid artery and the radial artery to determine cr-PWV. The distance measurements were taken with a measuring tape by subtracting the distance from the suprasternal notch to the carotid from the suprasternal notch to the femoral artery at the sensor location for the calculation of cf-PWV. For the calculation of cr-PWV, distances from the suprasternal notch to the carotid and radial artery sampling sites were measured. The same experienced operator who was blinded to the group of the participants performed all measurements.
Statistical analysis
Statistical analysis was performed using the (IBM SPSS software version 22.0 for Windows, Armonk, NY, USA). Data were assessed for normal distribution of their values using the Kolmogorov–Smirnov test. Student’s t-test and the Mann–Whitney U test were used to assess differences in parametric and non-parametric continuous variables between the studied groups; the chi-square test was used for categorical variables.
The comparison of baseline data between the two groups was performed by the paired t-test or the Wilcoxon test. Comparisons of the studies parameters during the different time-points of the study per group were performed using the paired t-test or the Mann–Whitney U test for parametric and non-parametric data, respectively. Correlations between changes in the studied parameters were performed by Spearman’s correlation coefficient. The analysis of variance (ANOVA) for repeated measurements was used to evaluate the effect of time (within subjects’ effect) and type of intervention (atorvastatin plus diet vs diet alone interaction; between-subjects effect) on clinical and laboratory values during the study. The Greenhouse-Geisser adjustment was used when the sphericity assumption was not fulfilled. p values < 0.05 (two-tailed) were considered statistically significant.
Results
The demographic and clinical characteristics of the study participants at baseline are presented in Table 1. There was no significant difference in terms of age, gender, BMI, blood pressure, eGFR and lipid profile between the study groups; there was a trend for higher HbA1c in subjects that were allocated to diet alone group. No significant differences were found in the type of anti-diabetic or anti-hypertensive medications between the two groups (Table 1).
Demographic and clinical characteristics of the study participants at baseline (n = 79).
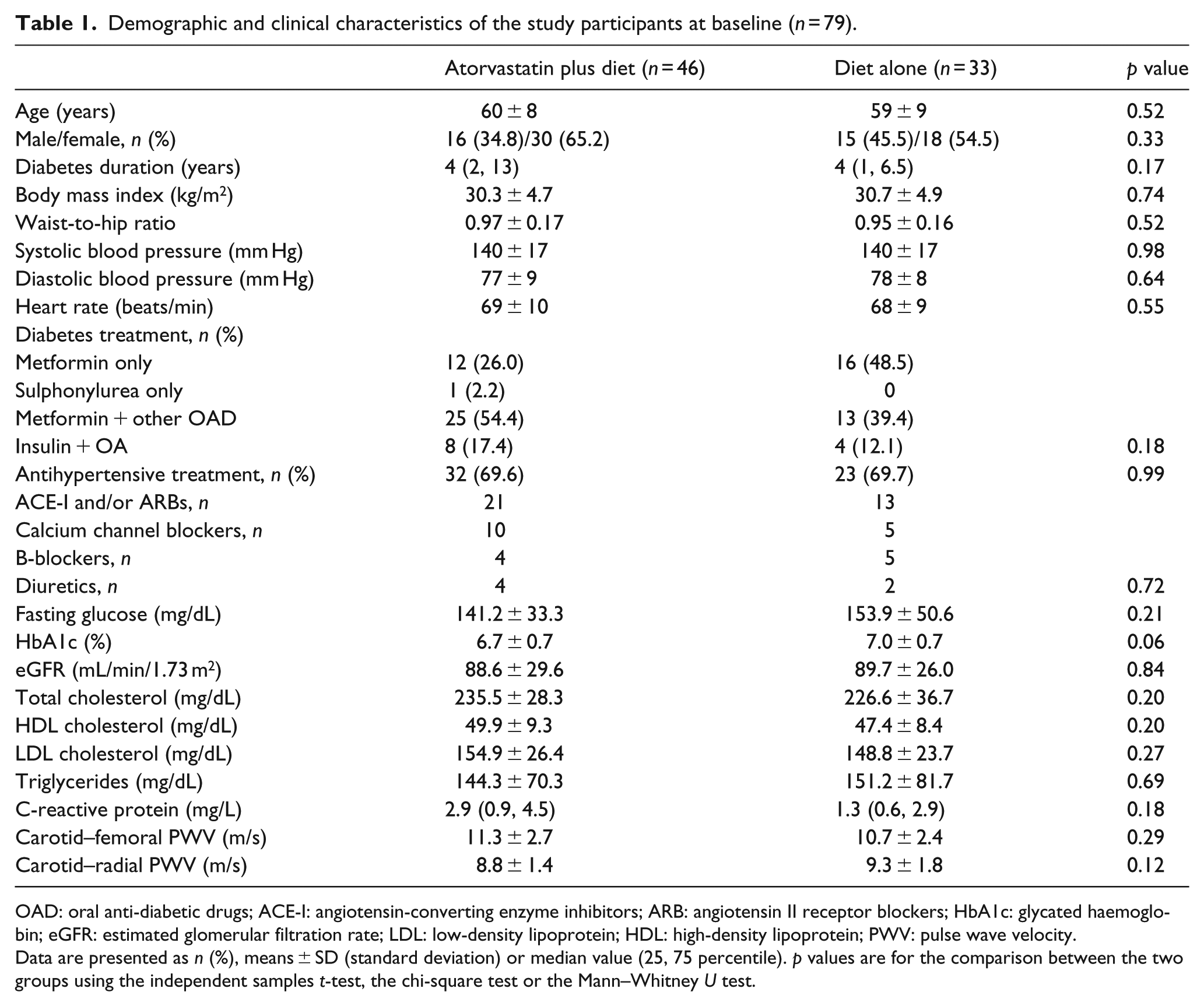
OAD: oral anti-diabetic drugs; ACE-I: angiotensin-converting enzyme inhibitors; ARB: angiotensin II receptor blockers; HbA1c: glycated haemoglobin; eGFR: estimated glomerular filtration rate; LDL: low-density lipoprotein; HDL: high-density lipoprotein; PWV: pulse wave velocity.
Data are presented as n (%), means ± SD (standard deviation) or median value (25, 75 percentile). p values are for the comparison between the two groups using the independent samples t-test, the chi-square test or the Mann–Whitney U test.
cf-PWV reduced significantly during the study period in the group treated with atorvastatin plus diet, while no significant difference was observed in the group managed by diet alone group (p value for the effect of time <0.001 and 0.73, respectively). In addition, during the study cf-PWV was reduced significantly in participants treated with atorvastatin plus diet as compared to the group managed by diet alone [p value for the effect of time by treatment (atorvastatin plus diet vs diet alone) interaction 0.03] (Table 2). The reduction of cf-PWV was observed at 3 months after initiation of treatment with atorvastatin plus diet and was maintained during the study. Treatment with atorvastatin plus diet resulted in a reduction of cf-PWV by 7.52% at 12 months (p < 0.001), while in the group managed by diet alone, cf-PWV increased by 3.74% (p = 0.73) (Figure 1).
Clinical characteristics of patients in atorvastatin and diet-treated group at baseline, 3, 6 and 12 months.
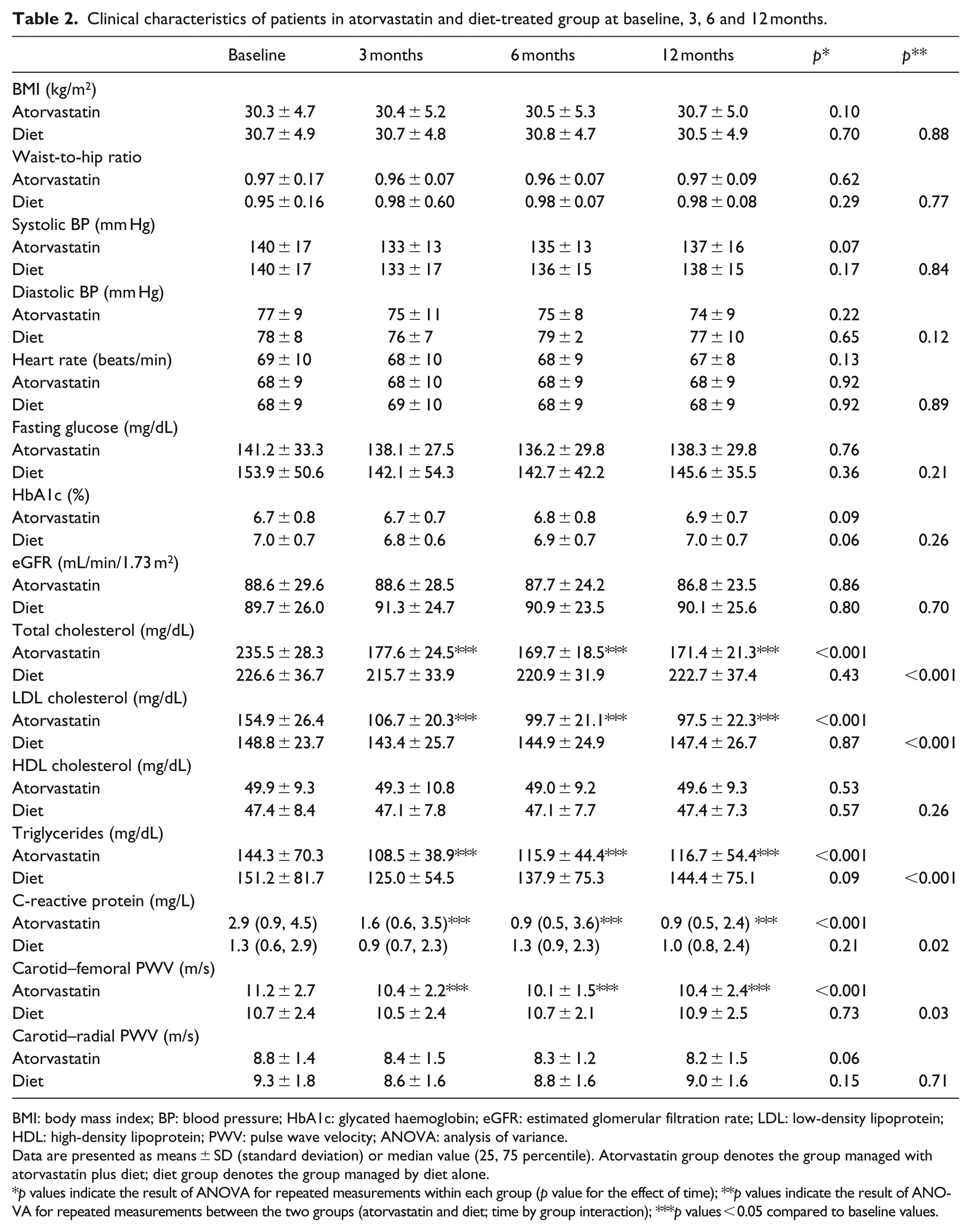
BMI: body mass index; BP: blood pressure; HbA1c: glycated haemoglobin; eGFR: estimated glomerular filtration rate; LDL: low-density lipoprotein; HDL: high-density lipoprotein; PWV: pulse wave velocity; ANOVA: analysis of variance.
Data are presented as means ± SD (standard deviation) or median value (25, 75 percentile). Atorvastatin group denotes the group managed with atorvastatin plus diet; diet group denotes the group managed by diet alone.
p values indicate the result of ANOVA for repeated measurements within each group (p value for the effect of time); **p values indicate the result of ANOVA for repeated measurements between the two groups (atorvastatin and diet; time by group interaction); ***p values < 0.05 compared to baseline values.
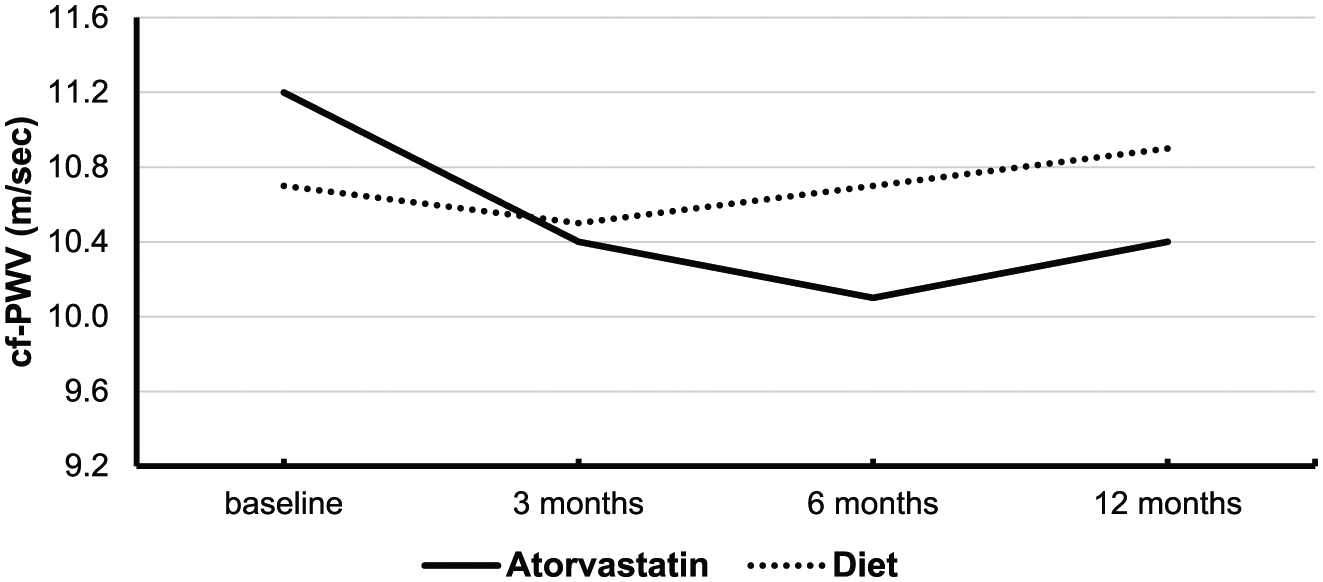
Effect of treatment with atorvastatin plus diet or diet alone on carotid–femoral pulse wave velocity after 3, 6 and 12 months of intervention.
There was a trend for reduction of cr-PWV in the group treated with atorvastatin plus diet, while no significant effect was noticed in the group managed by diet alone (p value for the effect of time 0.06 and 0.15, respectively). In addition, no significant difference was found in cr-PWV between patients receiving atorvastatin plus diet and diet alone [p value for the effect of time by treatment (atorvastatin plus diet vs diet alone) interaction 0.71] (Table 2).
Treatment with atorvastatin plus diet reduced total cholesterol, LDL-C, triglycerides and hsCRP during the study period (all p values for the effect of time < 0.001), while no significant effect was noticed in the group managed by diet alone (p values for the effect of time 0.43, 0.87, 0.09 and 0.21, respectively). Total cholesterol, LDL-C, triglycerides and hsCRP were reduced significantly in participants treated with atorvastatin plus diet as compared to the group managed by diet alone [p values for the effect of time by treatment (atorvastatin plus diet vs diet alone) interaction, <0.001, <0.001, <0.001 and 0.02, respectively] (Table 2). The reduction in these parameters was observed at 3 months after initiation treatment with atorvastatin plus diet and was maintained along the study. Treatment with atorvastatin plus diet resulted in 26.6% reduction of total cholesterol (p < 0.001), 36.2% reduction of LDL-C (p < 0.001), 11.2% reduction of serum triglycerides (p = 0.001) and 41.8% reduction of hsCRP (p < 0.001) at the end of observation period in comparison with the baseline values.
No significant changes were found during the study in BMI, blood pressure, heart rate, HbA1c, fasting glucose, high-density lipoprotein cholesterol (HDL-C) and eGFR in either study group (Table 2).
Changes in either cf-PWV or cr-PWV did not correlate significantly with changes in total cholesterol, LDL-C, triglycerides or hsCRP (all p values > 0.05).
Discussion
The main finding of this non-randomized observational prospective study is that aortic stiffness, assessed by determination of cf-PWV, improves early after treatment with low-dose atorvastatin plus diet, and this effect is maintained up to 1 year in subjects with T2DM and dyslipidaemia who are free of clinically apparent macrovascular disease.
cf-PWV is considered as the ‘gold standard method’ for the study of arterial stiffness in healthy population and in individuals with T2DM.6,35 It is known that patients with T2DM and subjects with hypercholesterolaemia have increased arterial stiffness compared to subjects without diabetes or hypercholesterolaemia.36,37 Increased cf-PWV serves as target organ damage that predicts CVD morbidity and mortality beyond other traditional risk factors; hence, it is recommended for risk stratification in people with hypertension. 8
Increased cf-PWV has a strong impact on cardiac structure and function. 6 Thus, increased aortic stiffening leads to a premature return in heart’s late systole and increases central pulse pressure and systolic blood pressure. 6 As a result, the load of the left ventricle is larger, causing left ventricular hypertrophy, a well-established risk factor for coronary events. 6 In addition, increased cf-PWV is caused by processes such as inflammation, endothelial dysfunction, accumulation of collagen, development of fibrosis and calcification at the arterial wall including coronary arteries and thus increasing the risk for coronary artery disease.38,39 With regard to the cerebral circulation, increased aortic stiffness affects the remodelling of the intracranial and extracranial arteries and endorses carotid wall thickness, development of atherosclerotic plaques and carotid stenosis. 6 Moreover, diabetes per se and diabetes-related factors such as autonomic dysfunction have been associated with increased aortic stiffness beyond the established risk factors for atherosclerosis.40,41
Statins are lipid-lowering drugs that inhibit HMG-CoA reductase that decrease effectively CVD events and mortality. 42 The use of statins is highly recommended to patients with T2DM, since they are stratified as high or very high risk for CVD events.9,10 Statins beyond their lipid-lowering effects have pleiotropic effects on the cardiovascular system. 11 Herein, we demonstrated that aortic stiffness is improved after administration of low-dose atorvastatin plus diet in subjects with T2DM and dyslipidaemia. Possible mechanisms how statins may modulate aortic stiffness involve reduction of sympathetic nervous system activity, anti-oxidative effect, improvement in endothelial function and, in the long-term, structural changes in the arterial wall.43–49 In addition, administration of atorvastatin has shown to attenuate vascular tone and improve endothelial function through increasing the expression of nitric oxide availability.43,44 Similarly, since it is known that hypercholesterolaemia causes an upregulation of vascular angiotensin II type 1 (AT1) receptor expression, 45 statins have been shown to downregulate AT1 receptor in vascular smooth cells, reducing their proliferation. 46 Furthermore, statins inhibit hypoxia-induced endothelin-1 via accelerated degradation of hypoxia-induced factor-1a (HIF-1a) in vascular smooth muscle cells. 47 Another possible mechanism is mediated through the anti-oxidative properties of statins. 48 Moreover, statins cause a rapid reduction of lipid content and the number of smooth muscle cells in the arterial wall, contributing to decreased arterial stiffness. 50
Although there are several previous studies that have examined the effect of statins on PWV, only a few have recruited subjects with T2DM.12–16 Ichihara et al. 12 enrolled participants with diabetes and end-stage renal disease; the group allocated to receive 20 mg of fluvastatin for 6 months had a significant reduction in ankle–brachial PWV in comparison to the group treated with placebo. 12 The number of participants in this study was small (n = 22), and the ankle–brachial PWV was used for the assessment of arterial stiffness. Shinohara et al. 13 enrolled 22 individuals with T2DM and hypercholesterolaemia and treated them with 10 mg of atorvastatin for 6 months. PWV was measured in the heart–brachial, heart–carotid, heart–femoral and femoral–ankle segments; only femoral–ankle PWV showed a reduction during intervention. Moreover, the authors reported that there was a non-significant trend for reduction of the heart–femoral PWV. 13 Mukherjee et al. 14 recruited 71 normotensive and normolipidaemic subjects with T2DM and reported that after 6 months of intervention, the group randomized to receive 10 mg of atorvastatin had significant improvement in brachial–ankle PWV, while no change was found in the placebo group. Davenport et al. 15 enrolled 51 subjects with T2DM and microalbuminuria in an open label study; participants were allocated to receive either 10 or 80 mg of atorvastatin for 12 months. The authors found that the cf-PWV was reduced in both arms, and no difference was noticed between the two groups. Although the participants in the study by Davenport et al. were older and had longer duration of diabetes in comparison with the participants in our study, both came to same conclusion. A recent double-blind trial recruited 40 individuals with T2DM; participants were assigned to either combination therapy of 10 mg of fluvastatin plus valsartan or to placebo for 1 month. 16 It was found that cf-PWV was reduced in the group assigned to the treatment with fluvastatin plus valsartan. Noteworthy, subjects in this study were exclusively male and younger than the individuals in our study.
The divergent results of some studies examining the effect of statin treatment on PWV are probably due to the different methodology used for the assessment of PWV. A few studies used the cf-PWV,15,16 which is considered today as the ‘gold standard’ method,6,8 while others measured PWV in the ankle–brachial segment,12,14 heart–brachial, heart–carotid, heart–femoral or femoral–ankle segments. 13 In addition, the population recruited in these studies was heterogeneous and the duration of intervention was different; therefore, the studies are not comparable.
The impact of statin administration on arterial stiffness has been examined in healthy individuals and in subjects with either hypertension, hypercholesterolaemia, chronic obstructive pulmonary disease, obstructive sleep apnoea, chronic kidney disease or obesity.17–27 The results of these studies were either negative25–27 or they showed a non-significant trend for reduction of PWV.17,18,21 The study with the longest duration was performed by Fassett et al. 18 in 37 patients with chronic kidney disease who were randomly assigned to receive either 10 mg of atorvastatin or placebo for 3 years. The authors reported that the aortic PWV did not deteriorate during the study in subjects treated with atorvastatin, while it increased significantly in the placebo-treated patients; in addition, other central haemodynamic parameters like central pulse pressure and augmentation index did not change significantly in either study group. 18 Another study with relatively long duration was performed by Kanaki et al. 19 They examined the effect of treatment with atorvastatin 10 mg versus placebo for 26 weeks in 50 hypertensive and hypercholesterolaemic patients. They found that treatment with atorvastatin was associated with lower cf-PWV, central aortic systolic blood pressure and pulse pressure at study-end with atorvastatin in comparison with the placebo group. 19 Nevertheless, a double-blind study by Raison et al. 25 that recruited participants with hypertension and hypercholesterolaemia for 12 weeks demonstrated that cf-PWV was increased in both groups treated with either placebo or 10 mg of atorvastatin.
In the light of these conflicted results, two meta-analyses were performed recently.28,29 D’elia et al., 28 using studies that assessed arterial stiffness with the cf-PWV method only, reported a direct association between statin use and decreased arterial stiffness in non-diabetic population. Similarly, another meta-analysis by Upala et al. 29 of studies in non-diabetic subjects on the effect of different statins on aortic PWV reported similar results.
We find only a trend for reduction in cr-PWV in the subjects treated with atorvastatin in our study. The differential effect of statins on various arterial segments may be related to the different properties of the arterial wall. Thus, cf-PWV is a regional measurement of arterial stiffness and provides information about a long segment of the arterial tree that is composed of both elastic proximal segment that contains many collagen and elastin filaments in the tunica media and stiffer more distal arteries. 51 In contrast, medium-sized vessels known as distributing arteries (or muscular arteries) such as in the segment between carotid and radial arteries draws blood from an elastic artery and branch into ‘resistance vessels’ including small arteries and arterioles containing less collagen and elastin and more layers of smooth muscle cells. 49 Previous studies have shown that the stiffness of muscular conduit arteries may decrease with age and in conditions of accelerated vascular ageing, such as in chronic kidney disease and diabetes mellitus possibly as an adaptive mechanism to the increased aortic stiffness.52,53 Indeed, a study in a dialysis cohort found that cr-PWV decreased with time despite a progression of aortic stiffness over a period of 1.2 years. 53 Such differences in the mechanical properties of the arterial tree may have masked the potential effect of treatment with atorvastatin on cr-PWV in our study.
The clinical implications of the results of this study can be summarized as follows. A low-dose atorvastatin reduces aortic stiffness; this effect is seen 3 months after initiation of treatment and is maintained for at least 1 year in subjects with T2DM. The beneficial effect of atorvastatin on the arterial wall strengthens the view of its use in people with T2DM and dyslipidaemia, a population of increased cardiovascular risk for the prevention of cardiovascular events.3–5 Moreover, we found that the favourable effect of atorvastatin on the arterial wall is not related to changes in blood cholesterol or hsCRP concentrations but may be related to the pleiotropic effects of atorvastatin or its effects at the tissue level, regardless of changes in blood of lipids or other biomarkers.27,49,50
The main strength of this study is the long duration of intervention in subjects with T2DM, which is among the longest duration mentioned so far. In addition, a comparative group was used, and hence, random effects of the agent used can be ruled out. There are also several limitations. First, the number of the subjects participated in the study was not large. Second, the study was not randomized or blinded; however, we tried to minimize the potential confounding effect of non-blinding because the person who performed PWV measurements was blind to the treatment arm of the participants. Third, the study was performed in the past when the guidelines for lipids management in subjects with T2DM who were free of clinically apparent CVD were not sο strict as in the recent years. Finally, our findings cannot be extrapolated to the total diabetic population, since studied subjects did not have a history of macrovascular complications.
Conclusion
This study has shown that in subjects with T2DM and hypercholesterolaemia who did not have a history of macrovascular disease, administration of low-dose of atorvastatin (10 mg) plus diet for 12 months improved significantly aortic stiffness.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported, in part, by a research grant (S.A.R.G. 70/3/8615) from the National University of Athens.
