Abstract
Aims:
The present study investigated the effects of lipid-lowering therapy with atorvastatin on skin microvascular function in patients with type 1 diabetes and dyslipidaemia.
Methods:
Twenty patients received daily treatment with atorvastatin 80 mg or placebo during 2 months in a randomised, double-blind, cross-over study. Forearm skin microcirculation was investigated with laser Doppler perfusion imaging during iontophoresis of acetylcholine and sodium nitroprusside to assess endothelium-dependent and endothelium-independent microvascular reactivity, respectively. Various biochemical markers of endothelial function were also investigated.
Results:
Endothelium-dependent microvascular reactivity decreased during atorvastatin (p < 0.001), showing a significant treatment effect compared with placebo (p = 0.04). Atorvastatin treatment was also associated with increased haemoglobin A1C levels from 7.45% to 7.77% (p = 0.008).
Conclusions:
The present study shows impaired endothelium-dependent skin microvascular function during high-dose atorvastatin treatment in patients with type 1 diabetes, thus implicating a risk for deterioration of microvascular function during such therapy in these patients.
Introduction
Statins or 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase inhibitors effectively reduce macrovascular events, while the effects on the microvasculature are unclear. Microvascular dysfunction is common in type 1 diabetes and manifests as retinopathy, nephropathy and neuropathy. In addition to glycaemic control and haemodynamic factors, dyslipidaemia may be important in the progression of diabetic microangiopathy. 1 Although prevention of diabetic microangiopathy through lipid-lowering therapy with statins has been advocated, results from clinical studies have so far not been conclusive.1,2
Skin microcirculation is suitable for non-invasive assessment of microvascular function in a clinical setting, and there is growing evidence that skin microcirculation may reflect microvascular function in other organs.3–6 Disturbances in skin microvascular reactivity, which is used as a marker of skin endothelial function, are found early after onset of type 1 diabetes.7,8 In addition to assessment of vascular dilatation in vivo, various biochemical analyses are used as surrogate markers of endothelial dysfunction. One such marker recently in focus is circulating endothelial microparticles (EMPs), which are small vesicular buds shed from the endothelial cell membrane upon activation or apoptosis. Patients with type 1 diabetes have elevated EMP levels correlating with the degree of microvascular complications. 9
In the present study, we investigated treatment effects of high-dose atorvastatin on skin microvascular function and biomarkers of endothelial function, including circulating levels of EMP in patients with type 1 diabetes and dyslipidaemia. The results of various aspects of haemostasis have been reported elsewhere. 10
Methods
A double-blind, cross-over study with randomisation to receive a daily dose of 80 mg atorvastatin or matched placebo for 2 months was designed (Figure 1). Investigations were performed at start and end of the treatment periods, which were separated by a wash-out period of 2 months.
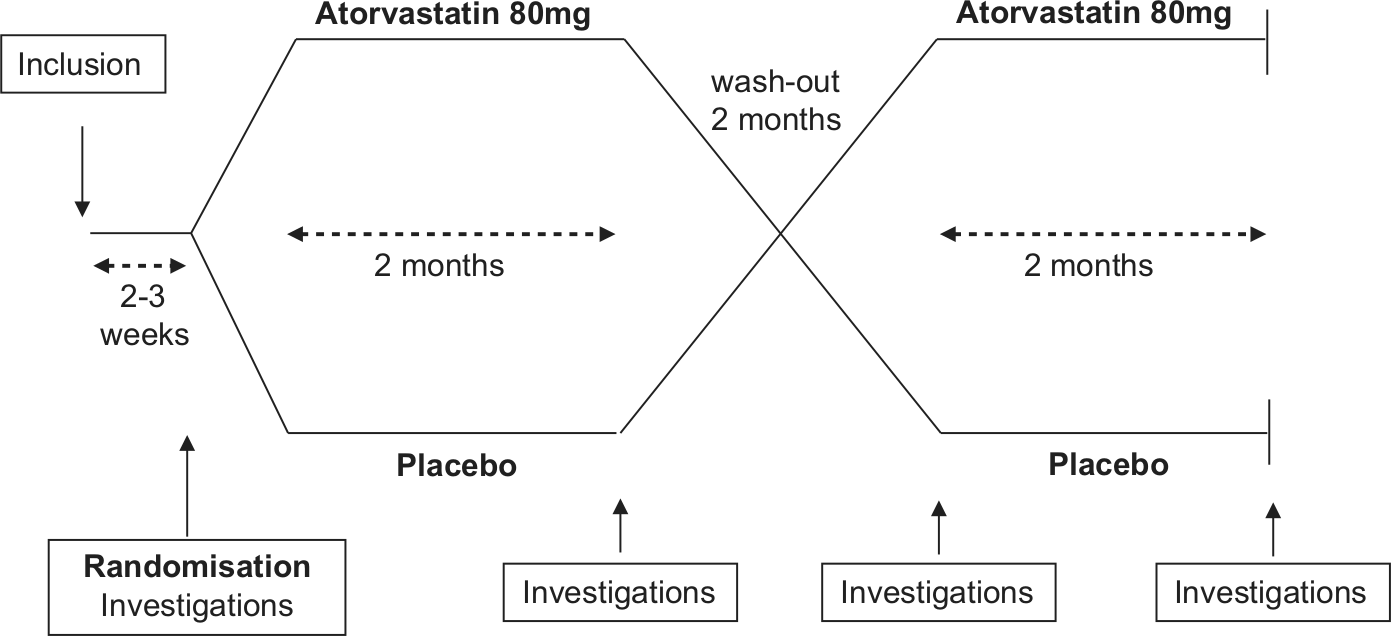
Flow chart showing the cross-over design of the study.
Patients
Twenty patients (10 females) were recruited from the outpatient clinic at the Department of Endocrinology and Diabetology at Danderyd Hospital, Stockholm, Sweden. Eligible criteria for this study were patients aged between 30 and 70 years with type 1 diabetes and elevated levels of low-density lipoprotein (LDL) (>2.5 mmol/L) and/or total cholesterol (>4.5 mmol/L). Patients with a history of macrovascular events were excluded. All patients arrived at the laboratory between 8 a.m. and 9 a.m. after a 10-h fast. Venous blood samples were taken after an acclimatisation period of at least 20 min. Systolic and diastolic arm blood pressures were measured in supine position. Clinical signs of peripheral neuropathy in the feet were investigated through tests of vibration and superficial sensation using a vibration fork (128 Hz) and monofilament (Semmes–Weinstein 5.07), respectively. Prevalence of albuminuria and retinopathy was assessed with urinary dipstick tests (Clinitek®, Bayer HealthCare LLC, Elkhart, IN, USA) and fundoscopic findings, respectively.
Skin microcirculation
Skin microcirculation was investigated through iontophoresis, which is a non-invasive method for drug application across the skin using a small electric current. 11 Acetylcholine (ACh, Sigma–Aldrich AB, Stockholm, Sweden) and sodium nitroprusside (SNP, Hospira, Inc., Lake Forest, IL, USA), diluted in deionised water, were used to investigate endothelium-dependent and endothelium-independent microvascular reactivity. Electrode chambers (LI611 Drug Delivery Electrode Imaging; Perimed, Järfälla, Sweden) were attached to the volar side of the left forearm and filled with a small volume of either ACh (2%) or SNP (2%). A battery-powered iontophoresis controller (Perilont 382b; Perimed) provided a direct current (0.1 mA for 60 s) for drug iontophoresis. ACh was delivered using an anodal charge and SNP with a cathodal charge. Skin microvascular flux before, during and after iontophoresis was measured with laser Doppler perfusion imaging (PeriScan PIM II; Perimed) and expressed in arbitrary unit (AU). Each image has a duration of 36 s and consists of approximately 150 measuring points within a skin area of 11 mm in diameter. Baseline flux was averaged over four images. Microvascular flux was recorded continuously up to 10 and 14 min after iontophoresis of ACh and SNP, respectively. Peak microvascular flux was determined. At our laboratory, mean coefficients of variation (CV) for peak microvascular flux after iontophoresis of ACh and SNP were 11% and 20%, respectively, assessed in seven healthy individuals on three separate days.
EMP
Previously frozen platelet-poor plasma was thawed and centrifuged at 2000 ×g for 20 min at room temperature. The supernatant was then re-centrifuged at 13000 ×g for 2 min at room temperature. EMPs were measured with flow cytometry on a Beckman Gallios instrument (Beckman Coulter, Brea, CA, USA) according to a previously described method. 12 Briefly, a 20 µL sample was incubated for 20 min in darkness with Alexa Fluor® 660 phalloidin (Invitrogen, Paisley, UK), lactadherin-fluorescein isothiocyanate (FITC) (Haematologic Technologies, Essex Junction, VT, USA) and CD144-APC (VE-Cadherin; AH diagnostics, Stockholm, Sweden). The EMP gate was determined using Megamix beads (0.5, 0.9 and 3.0 µm; Biocytex, Marseille, France). EMPs were defined as particles of <1.0 μm in size, negative to phalloidin (cell-fragment marker), positive to lactadherin and CD144. Results are presented as absolute numbers of EMPs [(microparticle counts × standard beads/L)/standard beads counted] (FlowCount; Beckman Coulter, Brea, CA, USA). Mean CV for EMP count assessed in 17 subjects on two separate days was 5% at our laboratory.
Biochemical analyses
Levels of haemoglobin A1C (HbA1C) were determined with the Mono S method using high-performance liquid chromatography (Variant II; Bio-Rad Laboratories, Hercules, CA, USA) with a reference value of less than 5.2%. Lipoproteins were assessed enzymatically with reagents from Synchron LX System(s) (Beckman Coulter, Lismeehan, Co Clare, Ireland). Von Willebrand factor antigen (vWF:ag) levels were measured with enzyme-linked immunosorbent assay (ELISA) (Asserachrom® vWF:ag; Diagnostica Stago, Asnières-sur-Seine, France) with a reference range between 50% and 160%. Enzyme immunoassays (R&D Systems Europe Ltd, Oxon, UK) were used to quantify plasma levels of endothelin-1 and soluble thrombomodulin. Mean ± standard deviation (SD) and intra-assay CV for endothelin-1 and soluble thrombomodulin in 23 healthy subjects investigated at our laboratory were 1.41 ± 0.34 pg/mL (CV of 2.9%) and 3525 ± 601 pg/mL (CV of 1.7%), respectively.
Statistical analyses
Power analyses by a professional statistician of peak microvascular flux responses to ACh iontophoresis showed that, given a power of 80% and a significance level of 0.05 (two sided), 15 patients were sufficient to show a significant difference. The Shapiro–Wilks W test was used to assess conformity with a normal distribution. Variables with a non-normal distribution were transformed logarithmically. Differences between and within the atorvastatin and placebo treatments were investigated with repeated-measures analyses of variance (ANOVAs). Iontophoresis data were analysed using two-way ANOVA assessing interaction of time and treatment on peak flux, as well as repeated-measures ANOVA assessing seven measures for ACh and nine measures for SNP (including baseline), respectively. Correlation between variables was assessed through linear regression analyses. Data are presented as mean values ± SD, 95% confidence intervals for the mean, median with lower–upper quartile values in brackets, or as numbers. A probability (p) of less than 0.05 was considered statistically significant.
Ethical considerations
The protocol of this trial was approved by the local Ethics Committee of Karolinska Hospital and the Swedish Medical Products Agency. Written informed consent was obtained from all the patients.
Results
Patient baseline characteristics are summarised in Table 1. Five patients were treated with continuous subcutaneous insulin infusion, while the others were treated with intermittent doses of short-acting insulin with meals and long-acting insulin analogues once or twice daily. None had previously been treated with lipid-lowering drugs. Four patients had microalbuminuria and were on antihypertensive treatment with angiotensin-converting enzyme inhibitors, and one had prophylactic therapy with low-dose aspirin (75 mg).
Baseline characteristics.
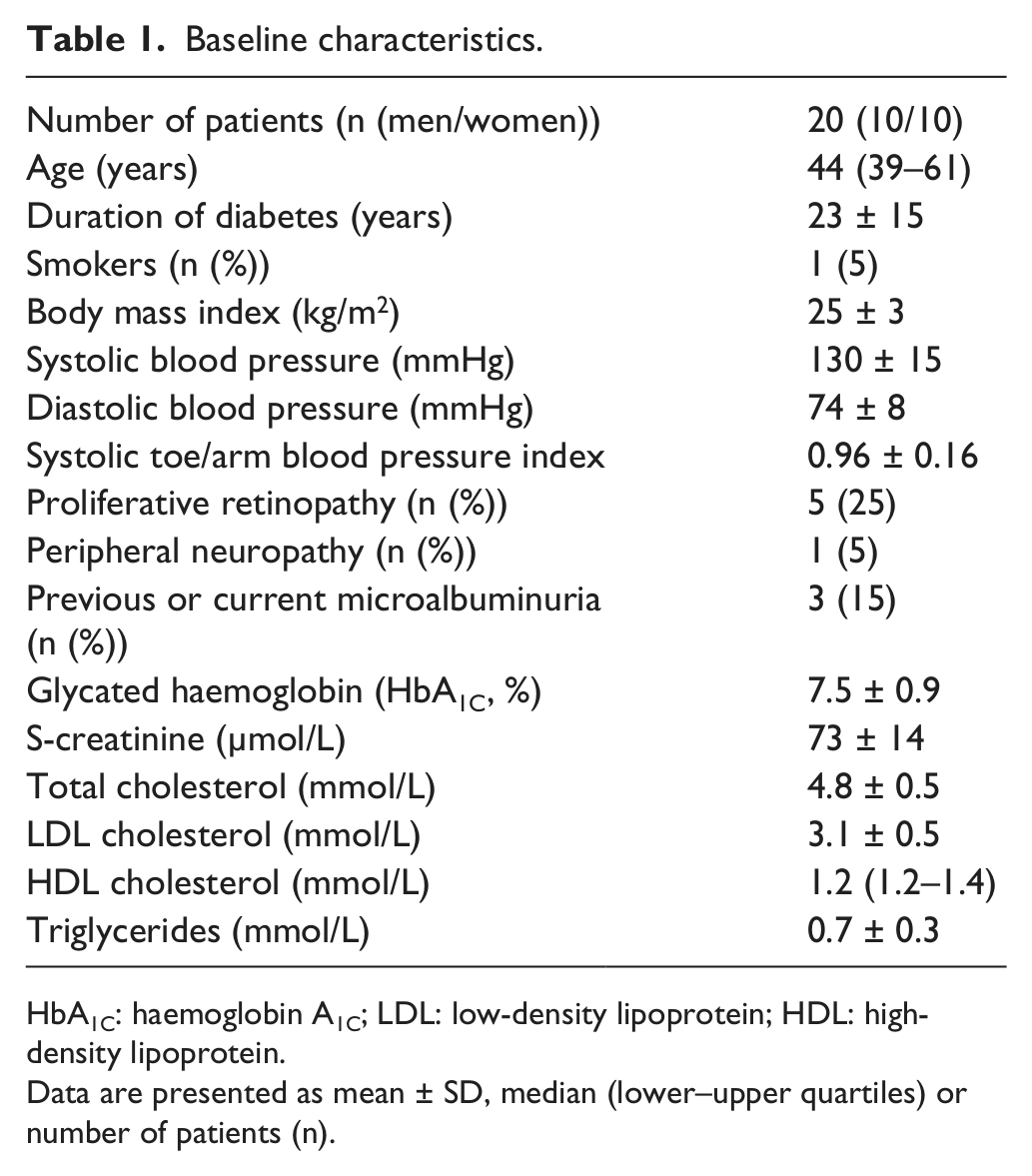
HbA1C: haemoglobin A1C; LDL: low-density lipoprotein; HDL: high-density lipoprotein.
Data are presented as mean ± SD, median (lower–upper quartiles) or number of patients (n).
As expected, atorvastatin treatment significantly reduced the levels of total cholesterol (from 4.86 ± 0.45 to 3.05 ± 0.3 mmol/L), LDL cholesterol [from 3.15 (2.80–3.35) to 1.40 (1.20–1.70) mmol/L] and triglycerides [from 0.72 (0.58–0.97) to 0.46 (0.38–0.68) mmol/L; p < 0.001 for all three variables]. High-density lipoprotein (HDL) cholesterol levels were not significantly changed. HbA1C levels increased from 7.45 ± 0.94% to 7.77 ± 1.08% during atorvastatin therapy (p = 0.008), and a significant treatment effect was seen compared with placebo (p < 0.001). Data on lipid and glycaemic control are reported in an earlier article. 10
Endothelium-dependent (ACh) microvascular flux measured continuously before, during and up to 10 min after iontophoresis was reduced during atorvastatin treatment (p < 0.001; Figure 2), and a significant treatment effect was found compared with placebo (p = 0.04). Similarly, ACh-mediated peak flux was lower during atorvastatin treatment (p = 0.03), showing a significant treatment effect compared with placebo (p = 0.008; Table 2). No significant changes were found in endothelium-independent (SNP) microvascular flux during atorvastatin or placebo treatment (Table 2). No correlations were found between peak values of endothelium-dependent (ACh) microvascular flux and age, diabetes duration, plasma lipids, HbA1C levels or clinical signs of microangiopathy (retinopathy, neuropathy and nephropathy). Baseline flux did not differ between atorvastatin and placebo treatments.
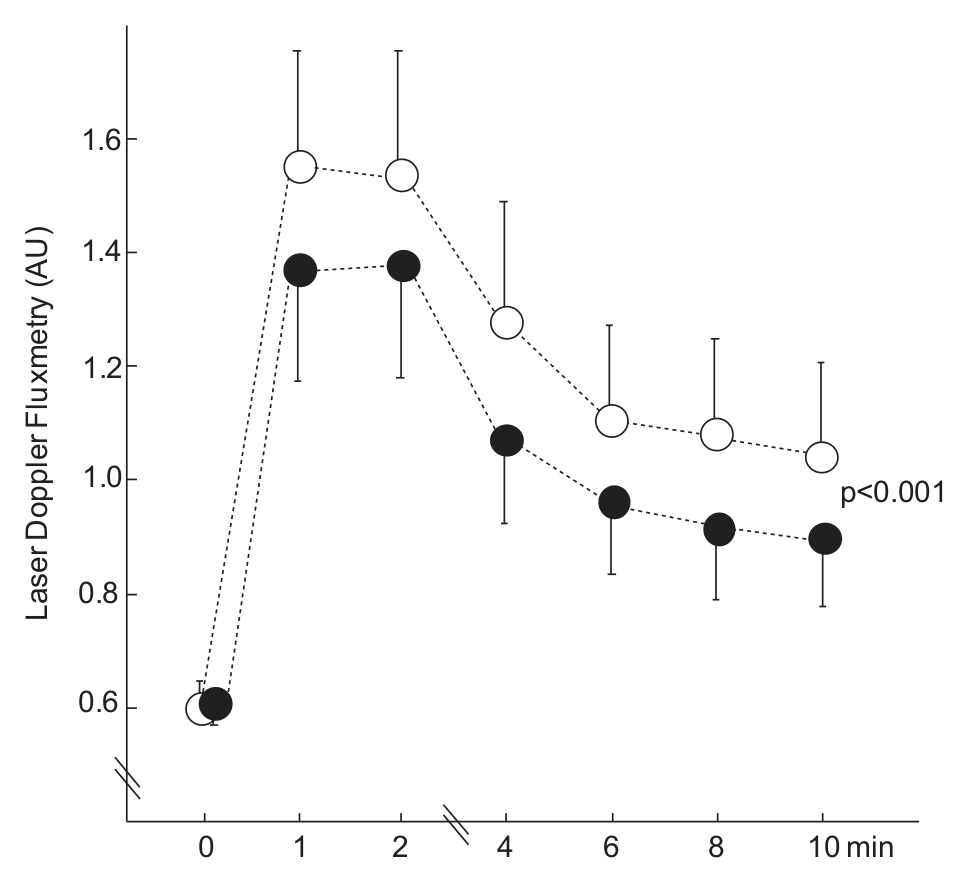
Acetylcholine-mediated skin microvascular reactivity before and after 2 months of atorvastatin treatment.
Results before and after 2 months of treatment with atorvastatin and placebo.
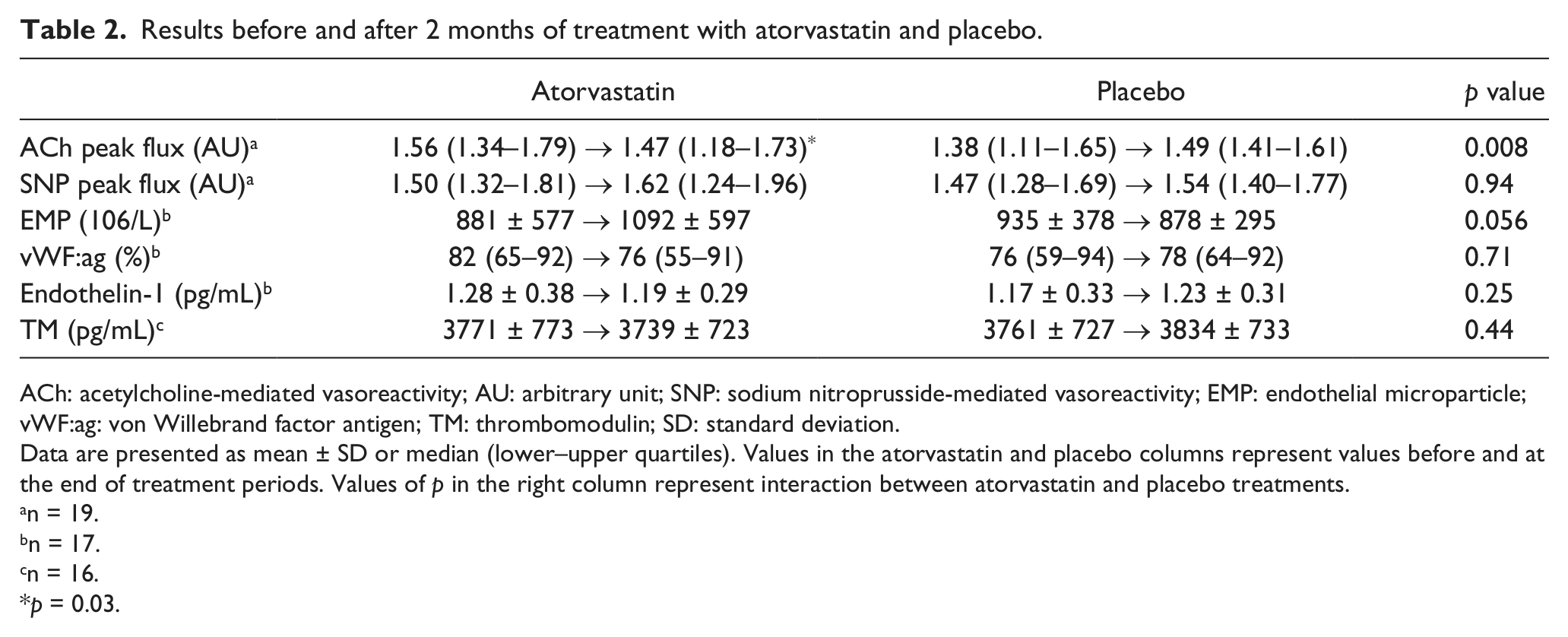
ACh: acetylcholine-mediated vasoreactivity; AU: arbitrary unit; SNP: sodium nitroprusside-mediated vasoreactivity; EMP: endothelial microparticle; vWF:ag: von Willebrand factor antigen; TM: thrombomodulin; SD: standard deviation.
Data are presented as mean ± SD or median (lower–upper quartiles). Values in the atorvastatin and placebo columns represent values before and at the end of treatment periods. Values of p in the right column represent interaction between atorvastatin and placebo treatments.
n = 19.
n = 17.
n = 16.
p = 0.03.
EMP levels tended to increase (p = 0.06) during atorvastatin treatment, and a tendency towards significant treatment effect was found compared with placebo (p = 0.06; Table 2). Plasma levels of vWF:ag, endothelin-1 and thrombomodulin were within the reference values at baseline and not significantly changed during atorvastatin or placebo treatment (Table 2). No correlations were found between endothelium-dependent (ACh) peak microvascular flux and levels of EMPs, vWF:ag, endothelin-1 or thrombomodulin. No carry-over effects were found in any of the analyses.
Discussion
The present study shows novel findings of impaired endothelium-dependent skin microvascular function during high-dose (80 mg) atorvastatin treatment in patients with type 1 diabetes. Atorvastatin therapy was also associated with impaired glycaemic control and a tendency towards increased levels of circulating EMPs. These data may have important clinical implications in patients with type 1 diabetes treated with high-dose statins.
Considering the wide use of statins, remarkably few placebo-controlled studies have investigated the effects on microvascular function. Of note, beneficial effects of statin treatment on kidney function and retinopathy status have been shown in patients with type 2 diabetes,13,14 while studies in patient with type 1 diabetes have failed to show improved microvascular function during statin therapy.15–17 Thus, the impact of statin therapy on the microvasculature in patients with type 1 diabetes remains unclear.
In the present study, skin microvascular reactivity was used to study the effects of atorvastatin treatment on microvascular function in type 1 diabetes. Investigation of skin microcirculation is a reliable and convenient way to assess microvascular function in vivo and may be used as a model for generalised microvascular function.3,7,11,18 However, one has to consider that specific signalling pathways are applied in different microvascular beds. Regulation of skin microcirculation is complex and involves multiple signalling pathways with integrated endothelial, neural and vascular smooth-muscle contributions. Iontophoresis of ACh and SNP is extensively used to assess endothelium-dependent and endothelium-independent skin microvascular function, respectively. 7 ACh causes a localised endothelium-dependent vasodilatation, although the contribution of nitric oxide, prostanoids, hyperpolarising factor and C-fibre nerves in mediating this response remains unclear. SNP is a direct donor of nitric oxide, which bypasses the endothelium and relaxes the vascular smooth-muscle cells. Thus, although the underlying mechanisms are unclear, our results showing reduced endothelium-dependent (ACh) microvascular responses during atorvastatin treatment while the endothelium-independent (SNP) responses remained unchanged indicate impaired endothelial function in skin microcirculation.
In non-diabetic subjects, atorvastatin treatment improved skin microvascular reactivity at lower doses, 19 while higher doses of atorvastatin (20–40 mg) had no influence on skin microvascular responses.20–22 Treatment with simvastatin, which as atorvastatin has lipophilic properties, was in patients with type 2 diabetes associated with impaired endothelium-dependent skin microvascular reactivity. 23 In patients with type 1 diabetes, a small study of nine participants showed improved skin microvascular reactivity during pravastatin treatment. 24 Of note, pravastatin, in contrast to atorvastatin and simvastatin, is a less lipophilic statin associated with positive metabolic effects. 25 Thus, previous studies have shown conflicting results regarding treatment effects of statins on skin microvascular function, and this may in part be explained by the usage of statins with different lipophilic properties at different doses and on different patient categories.
Deterioration of glycaemic control during statin therapy, as observed in the present study, has been in focus during the past few years, since statins reportedly increase the risk of new-onset diabetes. 26 This risk seems more pronounced during high-dose statin treatment. 26 Although the underlying mechanisms are unclear, cholesterol-independent pathways may be involved as statins through inhibition of HMG-CoA reductase not only block the synthesis of cholesterol but also of other mevalonate products, for example, coenzyme Q10 and isoprenoid compounds. Reduced isoprenylation of the small guanosine triphosphatase (GTPase) proteins during statin therapy may be associated with insulin resistance through downregulation of glucose transporter (GLUT) 4 expression and decreased insulin-mediated glucose uptake in adipocytes. 27 The lipophilicity of different statins may be important in this context, since statins with more lipophilic properties, for example atorvastatin, are associated with impaired insulin sensitivity while a hydrophilic statin such as pravastatin reportedly improves insulin sensitivity. 25 Notably, ACh-mediated skin microvascular responses are associated with insulin sensitivity. 28 Hence, we suggest that the impaired glycaemic control during high-dose atorvastatin treatment in the present study might be caused by decreased insulin sensitivity due to deterioration of endothelium-dependent microvascular function.
Circulating EMPs have recently been recognised as a marker of endothelial function, and elevated EMP levels have been reported in cardiovascular diseases. In the present study, EMP levels tended to increase during atorvastatin treatment (p = 0.06; Table 2). This finding resembles the results of a previous study in patients with peripheral arterial occlusive disease. 12 The lack of correlation between EMP levels and endothelium-dependent microvascular reactivity during atorvastatin treatment may in part be explained by the complex regulation of skin microcirculation.
The limitation of our study is that investigation of skin microvascular reactivity was not the primary aim, and the sample size calculation was originally based on haemostatic functions. 10 However, a post hoc power calculation showed that the number of patients exceeded the number needed to investigate endothelium-dependent skin vasoreactivity.
In conclusion, the present study shows impaired endothelium-dependent skin microvascular function during high-dose atorvastatin treatment in patients with type 1 diabetes. Since statins are widely prescribed worldwide and the treatment recommendations are shifting towards more intensive therapy, our findings motivate further investigations on the effects of statins on microvascular function and diabetic microangiopathy.
Footnotes
Acknowledgements
The authors thank Professor emeritus Bengt Fagrell for his valuable comments on this article. The authors also gratefully acknowledge the expert technical assistance of laboratory assistant Ann-Christin Salomonsson and diabetes nurse Sofia Sjöberg.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This study was supported by an unrestricted grant from Pfizer (grant number NRA2581056), who supplied the atorvastatin and placebo tablets. Pfizer had no input into the study design, its execution or interpretation of the findings. Independent financial support was also provided by the Bert von Kantzow Foundation, Diabetesfonden, EndoMet, Karolinska Institutet and through the regional agreement on medical training and clinical research between Stockholm County Council and Karolinska Institutet (these grants were not specific for this study).
