Abstract
Aim:
To define the predictors of long-term mortality in patients with type 2 diabetes mellitus and recent acute coronary syndrome.
Methods and results:
A total of 7226 patients from a randomized trial, testing the effect on cardiovascular outcomes of the dual peroxisome proliferator–activated receptor agonist aleglitazar in patients with type 2 diabetes mellitus and recent acute coronary syndrome (AleCardio trial), were analysed. Median follow-up was 2 years. The independent mortality predictors were defined using Cox regression analysis. The predictive information provided by each variable was calculated as percent of total chi-square of the model. All-cause mortality was 4.0%, with cardiovascular death contributing for 73% of mortality. The mortality prediction model included N-terminal proB-type natriuretic peptide (adjusted hazard ratio = 1.68; 95% confidence interval = 1.51–1.88; 27% of prediction), lack of coronary revascularization (hazard ratio = 2.28; 95% confidence interval = 1.77–2.93; 18% of prediction), age (hazard ratio = 1.04; 95% confidence interval = 1.02–1.05; 15% of prediction), heart rate (hazard ratio = 1.02; 95% confidence interval = 1.01–1.03; 10% of prediction), glycated haemoglobin (hazard ratio = 1.11; 95% confidence interval = 1.03–1.19; 8% of prediction), haemoglobin (hazard ratio = 1.01; 95% confidence interval = 1.00–1.02; 8% of prediction), prior coronary artery bypass (hazard ratio = 1.61; 95% confidence interval = 1.11–2.32; 7% of prediction) and prior myocardial infarction (hazard ratio = 1.40; 95% confidence interval = 1.05–1.87; 6% of prediction).
Conclusion:
In patients with type 2 diabetes mellitus and recent acute coronary syndrome, mortality prediction is largely dominated by markers of cardiac, rather than metabolic, dysfunction.
Introduction
Patients with type 2 diabetes mellitus represent about one-third of patients presenting with acute coronary syndromes (ACS).1–4 They are at higher risk of adverse events and mortality 5 and should, per practice guidelines,1–4 receive specific care in terms of revascularization and drug therapy. In the first few years after an ACS, cardiovascular events represent 70% of all causes of death6,7 as compared to 40% in the general population, 8 in outpatients with diabetes, 9 or cardiovascular risk factors and impaired glucose tolerance 10 and patients hospitalized for other reasons.11–13 Understanding the clinical and laboratory determinants of mortality in this critical time may have important implications for the development of treatment modalities and intensity of follow-up. The objective of this study was to define the predictors of all-cause as well as cardiovascular mortality from a large randomized clinical trial enrolling hospital survivors with type 2 diabetes mellitus and an ACS.
Methods
Study design and patients
The AleCardio (Effect of Aleglitazar on Cardiovascular Outcomes After Acute Coronary Syndrome in Patients With Type 2 Diabetes Mellitus) study was a randomized, double-blind, placebo controlled, multicentre trial (NCT01042769) testing the safety and efficacy of the dual peroxisome proliferator–activated receptor (PPARs) agonist aleglitazar, added to standard medical therapy, in reducing the risk of cardiovascular mortality and morbidity in patients with type 2 diabetes mellitus and a recent ACS event. A detailed description of the study design and results has been published elsewhere.
14
In brief, the trial enrolled 7226 patients at 720 sites in 26 countries between February 2010 and May 2012. Patients were randomized at hospital discharge (or after a screening period of
Outcomes
The primary outcome for this analysis was all-cause mortality assessed over a median follow-up of 2 years. The secondary outcome was cardiovascular mortality defined as sudden death, or death due to acute myocardial infarction, heart failure, stroke and death due to other fully documented cardiovascular causes not included into above categories (e.g. dysrhythmia, pulmonary embolism or other cardiovascular event). There were 224 patients lost to follow-up in the overall cohort (3.1%), whereas an additional 228 (3.2%) withdrew consent. However, all the 7226 patients initially randomized were included in the present analysis.
Statistical analysis
Based on the overall trial results, assigned treatment with either aleglitazar or placebo had no effect on mortality. Therefore, the present analysis comprised the overall population of 7226 patients, irrespective of treatment assignment. The prognostic model was developed using a Cox proportional hazards model stratified by ACS index event (myocardial infarction vs unstable angina). The time-to-event analysis was the time elapsed between randomization and occurrence of the event of interest (death), loss to follow-up or the premature end date of the trial. The reverse Kaplan–Meier method was used to estimate the potential follow-up time.
15
Furthermore, the smoothed hazard estimate was checked to evaluate the course of death risk during the overall time frame. Based on specific literature, we identified a priori 31 candidate variables for prediction (demographic factors, traditional cardiovascular risk factors, comorbidities, laboratory measures and revascularization for the index ACS event). These variables were age, sex, body weight, smoking, race (Asian vs non-Asian), type 2 diabetes duration, medical history (prior myocardial infarction, prior coronary artery bypass graft surgery, history of congestive heart failure, prior stroke and peripheral vascular disease), heart rate and systolic blood pressure at randomization, any coronary revascularization during index admission, left ventricle ejection fraction (LVEF) during index admission, laboratory examinations at randomization [glycated haemoglobin (HbA1c), high-density lipoprotein (HDL) cholesterol, low-density lipoprotein (LDL) cholesterol, serum triglycerides, N-terminal proB-type natriuretic peptide (NT-proBNP), haemoglobin, estimated glomerular filtration rate using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) formula, serum fibrinogen and urine albumin-to-creatinine ratio (UACR)], anti-hyperglycaemic medications (sulfonylureas, insulin and biguanides) and cardiovascular medications at randomization (dual antiplatelet therapy, triple antithrombotic therapy, statins and beta-blockers). The functional form (linear vs nonlinear relations with mortality) was checked for all continuous variables. The test based on Schoenfeld residuals (phtest in Stata) was used to check the proportional hazard assumption. Collinearity was checked and excluded. A final parsimonious model was developed considering covariates readily available in common clinical practice. Meaningful statistical interactions were explored in the multivariable model. Model-adjusted hazard ratios (HRs) were derived along with 95% confidence intervals (CIs). Global measure of model performance
16
was computed in terms of customary between prediction and actual outcome (Royston explained variation with CIs provided using a built-in bootstrap procedure), global fit [Bayesian information criterion (BIC) and Akaike’s information criterion (AIC)] and discrimination (Harrel’s C statistics and Somers’ D coefficient). Furthermore, a post-estimation analysis was performed to evaluate the model with respect to calibration (that defines the ‘reliability’ of the model, referring to the agreement of predicted and observed predictions). The calibration assessment was provided throughout the prognostic index (PI) from the Cox model.
17
It provides only a rank ordering of risk, from which risk groups are created and corresponding survival probabilities are estimated by the Kaplan–Meier method. Indeed, it facilitates the comparison of actual survival probabilities with model-based estimates, without requiring any categorization of continuous variables. To ensure numerical stability, we centre the PI by subtracting its mean. Three prognostic groups from the 25th and 75th centiles of the PI were derived. The smoothed baseline log cumulative-hazard function and the mean survival probabilities were computed and a final graphical check of observed with predicted survival was performed. All discrimination and calibration measures were internally validated and corrected for optimism using internal bootstrap resampling (1.000 bootstrap samples). LVEF evaluation was not protocol-specified in the AleCardio trial. Therefore, it was not measured systematically and missing values were commonly reported, achieving 50% complete data. In view of this and of the inverse linear association existing between NT-proBNP and LVEF, it was not deemed appropriate to adjust for this last variable in the primary analysis. However, due to its prognostic importance after an ACS, it was considered in a sensitivity analysis, after performing a multiple imputation procedure based on the chained equations approaches.18,19 A
Results
All-cause and cardiovascular mortality
The median duration of follow-up was 2 (interquartile range (IQR) = 1.56–2.46) years. At the end of the follow-up, there were 286 deaths (4.0%), of which 210 (73.4%) were from cardiovascular causes, corresponding to an incidence rate (events/100 person-years) of 2.0 (95% CI = 1.8–2.2). The baseline characteristics of the patients, stratified per follow-up vital status, are reported in Table 1.
Baseline characteristics in survivors and non-survivors.
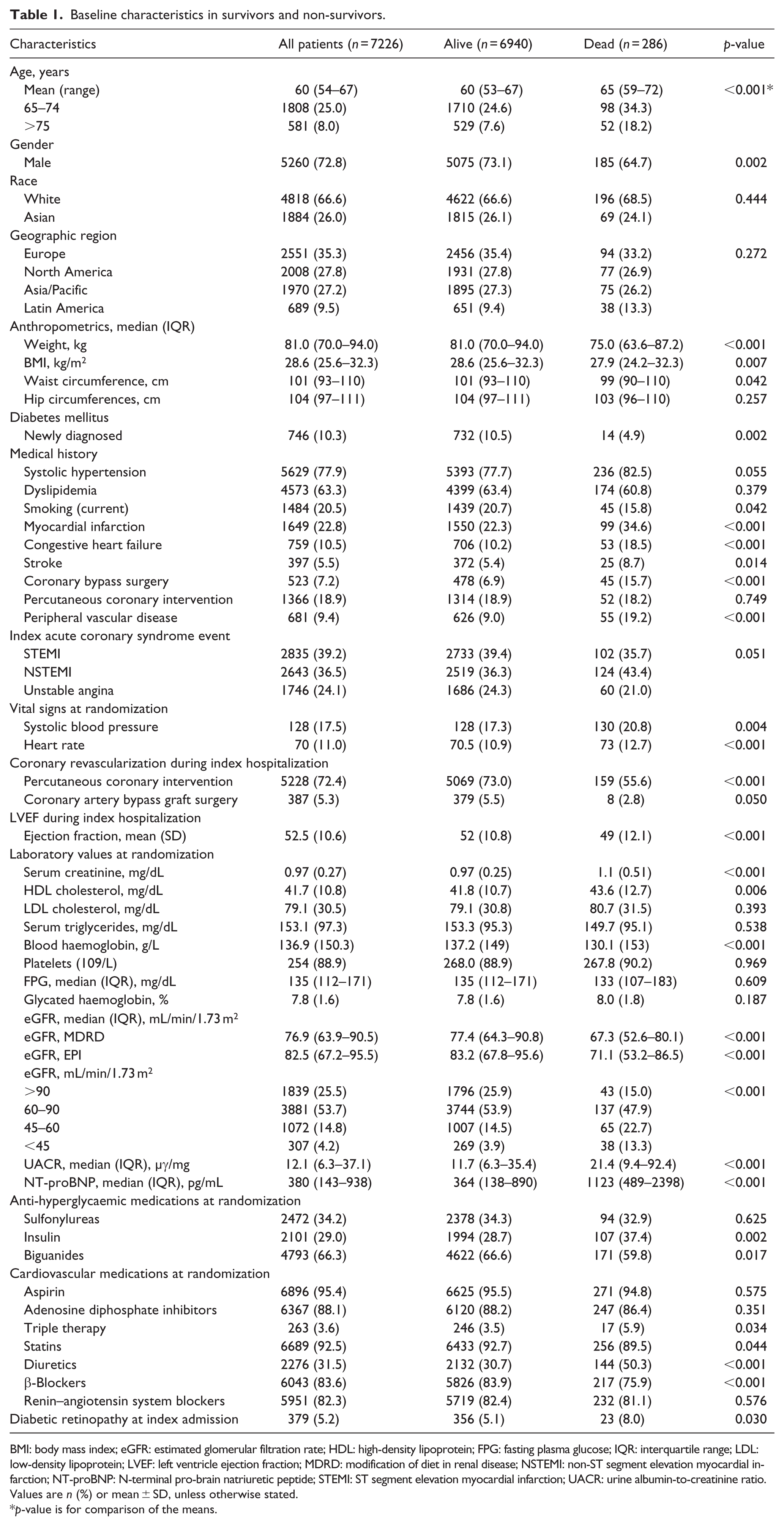
BMI: body mass index; eGFR: estimated glomerular filtration rate; HDL: high-density lipoprotein; FPG: fasting plasma glucose; IQR: interquartile range; LDL: low-density lipoprotein; LVEF: left ventricle ejection fraction; MDRD: modification of diet in renal disease; NSTEMI: non-ST segment elevation myocardial infarction; NT-proBNP: N-terminal pro-brain natriuretic peptide; STEMI: ST segment elevation myocardial infarction; UACR: urine albumin-to-creatinine ratio.
Values are
Multivariable predictive model for all-cause and cardiovascular mortality
Table 2 shows the multivariable predictive model for all-cause mortality, which simultaneously uses all eight risk variables based on the most discriminative power. The multivariable prediction model included NT-proBNP (adjusted HR for the natural log-transformed values = 1.68; 95% CI = 1.51–1.88), lack of coronary revascularization (HR = 2.28; 95% CI = 1.77–2.93), age (1 year increase: HR = 1.04; 95% CI = 1.02–1.05), heart rate (1 point increase: HR = 1.02; 95% CI = 1.01–1.03), HbA1c (1% increase: HR = 1.11; 95% CI = 1.03–1.19), haemoglobin (1 g/dL decrease: HR = 1.01; 95% CI = 1.00–1.02), prior coronary artery bypass (HR = 1.61; 95% CI = 1.11–2.32) and prior myocardial infarction (HR = 1.40; 95% CI = 1.05–1.87). The same model was tested with cardiovascular death as interest outcome and achieved consistent results (Table 3).
Independent predictors of all-cause mortality.
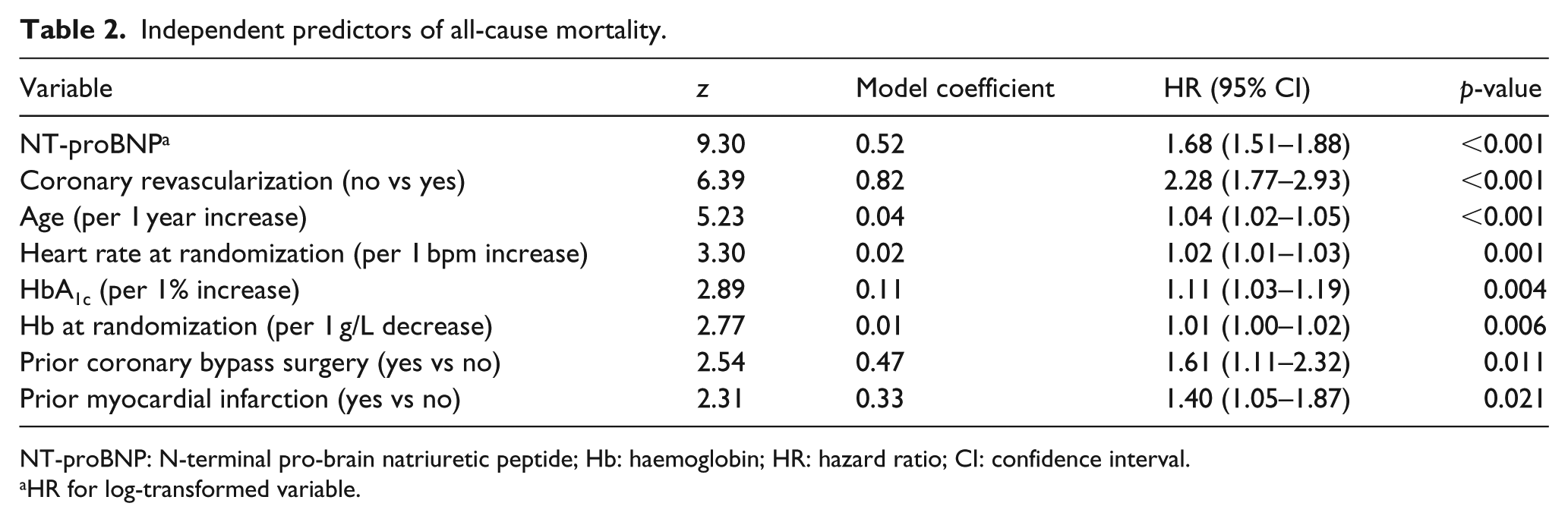
NT-proBNP: N-terminal pro-brain natriuretic peptide; Hb: haemoglobin; HR: hazard ratio; CI: confidence interval.
HR for log-transformed variable.
Independent predictors of cardiovascular mortality.
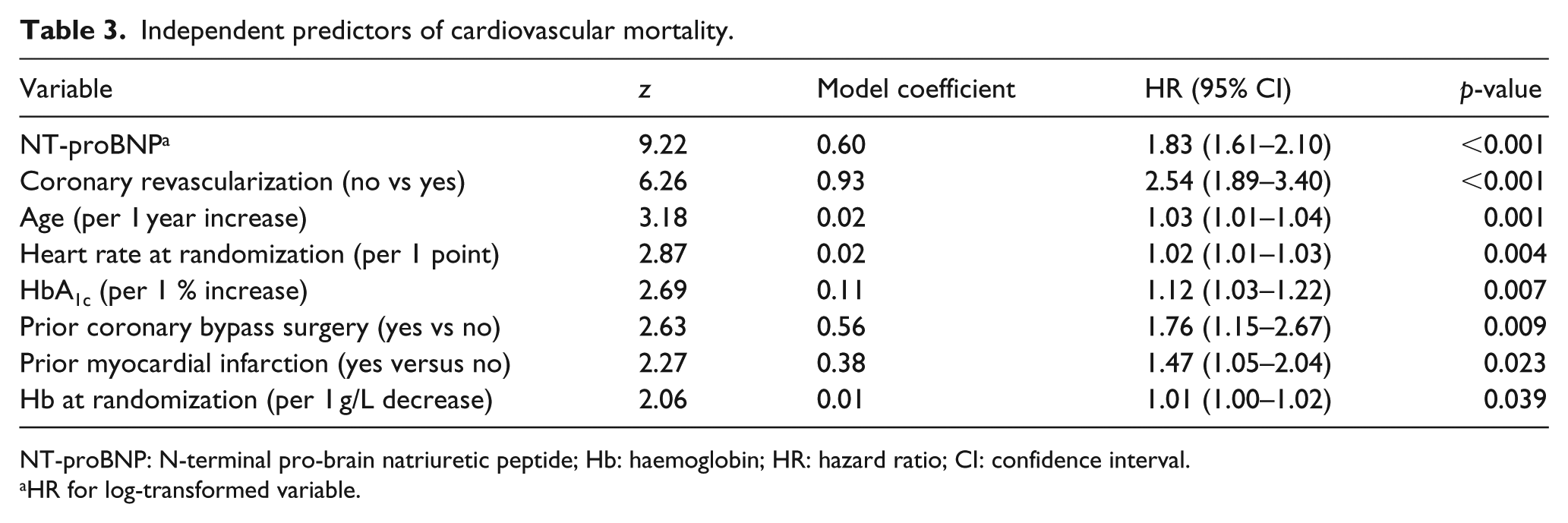
NT-proBNP: N-terminal pro-brain natriuretic peptide; Hb: haemoglobin; HR: hazard ratio; CI: confidence interval.
HR for log-transformed variable.
Predictive information of each variable
The predictive information provided by each variable was calculated as percent of total chi-square of the model. NT-proBNP was the most powerful covariate contributing with 27% and 30% of, respectively, the all-cause and cardiovascular (CV) mortality predictions. Lack of revascularization in the acute phase predicted 18% of all-cause and 20% of CV death. Older age predicted 15% of all-cause and 10% of CV death. Heart rate predicted 10% and 9%, respectively. Higher HbA1c and lower haemoglobin provided an additional 8% of mortality prediction (both all-cause and CV), whereas history of CV disease (prior bypass surgery and/orMI) 13% and 14%, respectively. The informative value of each independent predictor, for both all-cause mortality and cardiovascular mortality, is graphically represented in Figure 1. The final model showed a good global performance. The shrinkage coefficient was 0.97, the Harrell’s C statistics 0.78, the AIC 4196 and BIC 4251, respectively. The predicted versus observed mortality rates, calculated throughout the predictive index, were well matched suggesting a good fit of the developed model. Figure 2 shows the calibration plot between the predicted and observed probabilities. There was a good agreement between the observed and predicted survival probabilities; however, potential underestimation of risk in patients with a higher observed mortality risk was seen. As reported in Supplementary Appendix Table 1, the sensitivity analysis for all-cause mortality including LVEF using multiple imputation showed a prevalent prognostic role of general mortality predictors (HR for 1 year increase of age: 1.05; 95% CI = 1.03–1.06), lack of coronary revascularization (HR = 2.28; 95% CI = 1.79–2.90), comorbidities (HR per 1 g/L decrease haemoglobin: 1.02; 95% CI = 1.01–1.03), cardiac and vascular damage (HR for 1 point increase heart rate 1.02, 95% CI = 1.01–1.03; HR for 1 point decrease of LVEF: 1.03; 95% CI = 1.01–1.04), metabolic status (HR for 1 point increase of HbA1c = 1.12; 95% CI = 1.04–1.20) and cardiovascular history (for prior CABG and myocardial infarction: HR = 1.60; 95% CI = 1.12–2.27 and HR = 1.27; 95% CI = 0.97–1.69, respectively). The sensitivity analysis focused on cardiovascular mortality (Supplementary Appendix Table 2) showed a more prevalent role of lack of effective treatment and cardiovascular damage as prognostic factors.
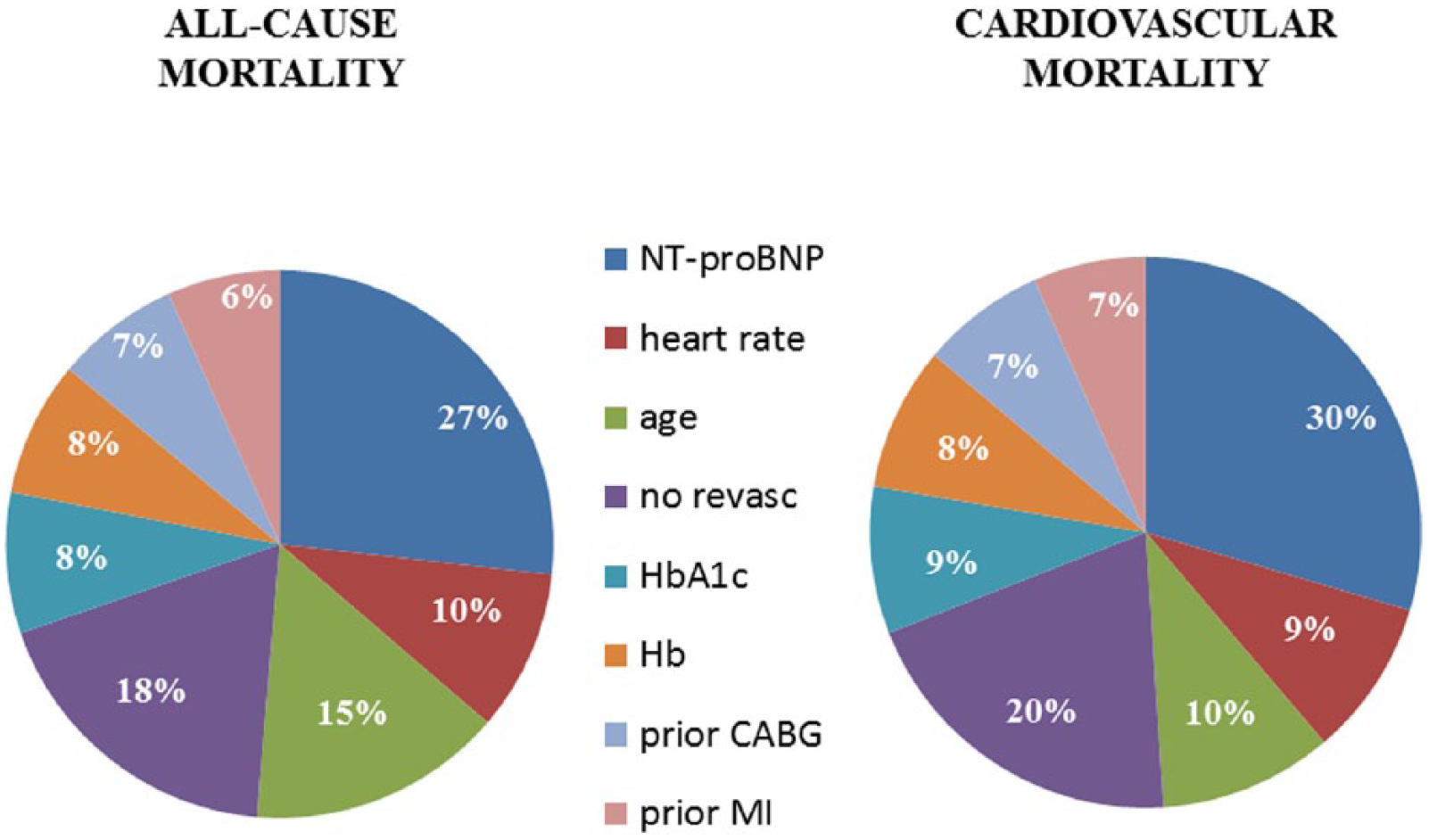
Graphical description of each independent predictor according its informative value for all-cause and cardiovascular mortality calculated as percent of total chi-square of the Cox regression model. Both all-cause mortality and cardiovascular mortality predictions are largely dominated by variables indicative of cardiac damage, whereas variables indicative of metabolic control contribute less.
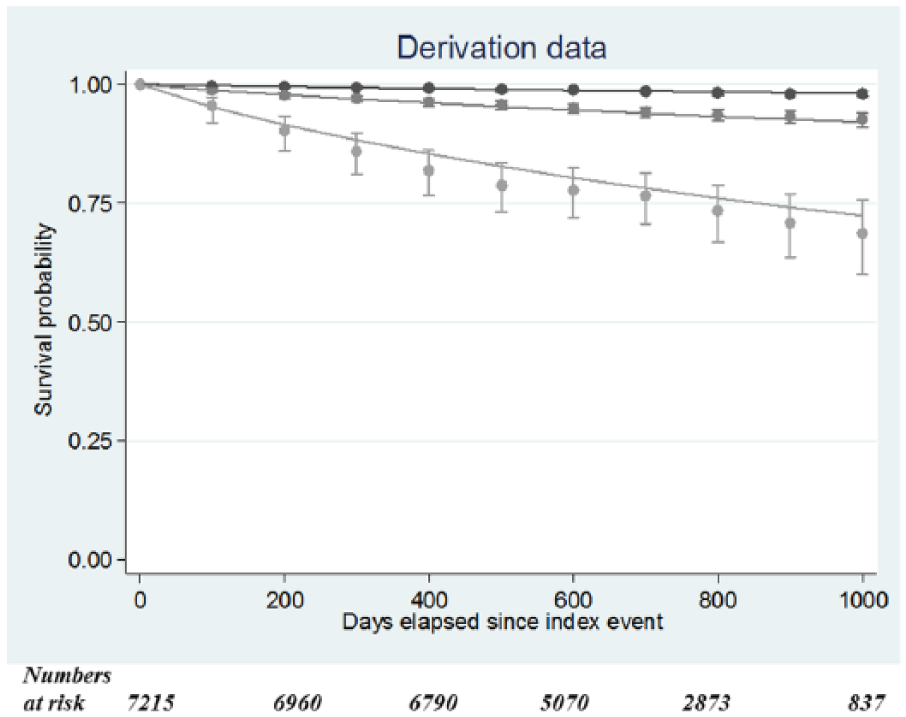
Calibration of the Cox model in the dataset. Smooth lines represent predicted survival probabilities, and scatter plots denote Kaplan–Meier estimates with 95% confidence intervals. Three prognosis groups are plotted. There was a good agreement between the observed and predicted survival probabilities in the groups with lower (darkest line) and intermediate (medium-dark line) observed mortality. However, potential underestimation of risk in patients with a higher observed mortality was seen.
Discussion
Diabetes mellitus and coronary artery disease are powerful predictors of mortality, since most patients with diabetes will die of heart disease, 21 and diabetes is an independent predictor of mortality among ACS patients 22 as well as among those undergoing revascularization. 23
Taking advantage of the study population of a large, prospective, randomized trial among patients with type 2 diabetes mellitus stabilized after an ACS, we identified eight independent predictors of all-cause mortality at a median follow-up of 2 years. These variables were identified a priori and confirmed at univariate analysis of 31 candidate variables selected from a thorough analysis of the specific literature regarding ACS and diabetes and after multiple comparisons of alternative models to maximize the discriminative power of the final set of variables. The eight variables included in the final model represent a logical summary of the most powerful mortality predictors in patients with diabetes and recent ACS. Since cardiovascular mortality accounted for more than 70% of all-cause mortality, it is not surprising that the rankings of total and cardiovascular mortality predictors are almost superimposable.
The identified model clearly shows that mortality is largely dominated by heart failure and lack of revascularization for the index ACS event. Age per se is a powerful mortality predictor across the wide spectrum of ACS and is included in every prognostication model. However, with regard to mortality, age was less powerful than the patient’s burden of cardiovascular disease mirrored by variables such as NT-proBNP and heart rate, and longstanding ischaemic heart disease (prior myocardial infarction and coronary artery bypass grafting).
NT-proBNP turned out to be the most powerful mortality predictor, and it is recommended by current guidelines for the initial diagnosis of heart failure, especially in the non-acute setting, 24 as it was the case in the present patient population. In the AleCardio study, NT-proBNP was systematically measured in the study corelab, whereas the determination of LVEF was not mandatory and, actually, it was available in about 50% of the patients. In a sensitivity analysis after multiple imputation for the missing values, LVEF was identified as almost as powerful as NT-proBNP and these two indexes of left ventricular dysfunction are tightly correlated and both highly predictive of adverse outcomes.
High heart rate upon ACS admission is an intrinsic component in the definition of haemodynamic instability and cardiogenic shock. It is among the most powerful predictors of mortality across the spectrum of ACS,25,26 and thus, a key component of the widely used risk scores for ACS.27,28 An important finding of this study is that also in the post-acute phase, tachycardia keeps its important prognostic value in terms of long-term mortality. This might be due to persisting sympathetic activation or difficult titration of beta blocker therapy due to hypotension, being in both cases a marker of heart failure.
Prior coronary artery bypass grafting and prior myocardial infarction are clearly associated with long-standing and advanced vascular disease and have been shown to predict worse outcomes. 23 It is a common clinical observation that even severe left ventricular dysfunction may not be associated with severe vascular disease, and vice versa. However, when significant cardiac and vascular damage are both present in a patient with diabetes and ACS, they add their independent and powerful prognostic value with regard to mortality.
Low haemoglobin also turned out to be among the most important mortality predictors in the AleCardio study population. Anaemia has been associated with older age and renal dysfunction, and is a recognized predictor of mortality in ACS. 29 However, the fact that in this large study population anaemia emerges as an independent predictor of mortality confirms and extends prior observations in smaller studies in elderly ACS patients 7 in which the correlation between haemoglobin, age and renal function was only weak, and rather limited to the acute ACS phase. 30 Higher levels of HbA1c also emerged as an independent mortality predictor and have been associated with an increased risk of cardiovascular complications.31,32 It should be noted, however, that the informative value of glycaemic dysfunction with regard to mortality in the AleCardio study population is far less important as compared to the value of variables representative of end-organ damage and revascularization. These data are in keeping with the observation that a drug, such as empagliflozin, with only a modest effect on HbA1c, but favourable effects on renal function 33 and, perhaps, myocardial metabolism, 34 has been shown to provide a striking protective effect with regard to cardiovascular mortality and heart failure. 35
Finally, lack of coronary revascularization during index ACS admission was the second most powerful mortality predictor after NT-proBNP, with more than doubling of the mortality risk. The poor outcome of ACS patients treated medically without revascularization has been shown in clinical trials and registries36,37 and may reflect a higher risk profile, lack of significant lesions or lesions not suitable for revascularization, or under-treatment, although the current analysis is prognostic and not etiologic, where confounding by indication may play a role. 38 In the AleCardio study, almost 78% of the study population underwent either percutaneous coronary intervention (72%) or coronary artery bypass graft surgery (5%), reflecting guideline recommendations and current practice in experienced centres, such as those taking part in a phase-III randomized trial of drug development. Despite clear evidence that bypass surgery should be the preferred revascularization modality among patients with diabetes, 39 percutaneous coronary intervention is prominent among ACS patients, particularly in the case of ST segment elevation myocardial infarction (STEMI).
Study limitations
Our study has limitations that may affect generalizability of the results to everyday practice. Overall mortality was low, perhaps reflecting the relatively young age of the study population, with only 8% of patients being older than 75 years, and the exclusion of patients with severe renal dysfunction and heart failure within the previous 12 months. However, this latter feature of the AleCardio population further increases the prognostic impact of indices of left ventricular and vascular dysfunction found in this study.
As in the case of the widely used and guideline-recommended GRACE risk score, 28 we performed an internal validation of our predictive model. Even if an external validation is advocated to determine generalizability to other plausibly related settings, 40 in the case of a specific patient population, such as that with diabetes and recent ACS, it would be disputable to validate a score generated from a randomized trial in a population derived from another randomized trial. External validation should be better performed in a registry population with long-term follow-up, which is not common in clinical practice. Finally, our prediction model does not include all the potential independent predictors of all-cause mortality, but was specifically developed to be parsimonious and to include only variables readily available in current clinical practice.
Conclusion
A meaningful and parsimonious set of clinical and laboratory variables may assist clinicians in risk stratification of patients with diabetes discharged after an ACS. Besides patient age, these variables represent the burden of cardiovascular and other end-organ damage associated to diabetes mellitus. The variables entered in the model are readily available in the common clinical setting and will allow better management of these high-risk patients during their follow-up.
Footnotes
Acknowledgements
Dr Savonitto takes full responsibility for the work as a whole, including the study design, access to data and the decision to submit and publish the manuscript. Dr Morici and Ms Nozza had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Drs Savonitto and Morici are co-first authors, with equal contribution to the manuscript. Savonitto, Morici and Nozza contributed to study concept and design; Savonitto, Morici, Nozza, Cosentino, Cavallini, Eijkemans, Stähli, Schrieks, Heerspink, Malmberg, Schwartz, Ryden, Tardif and Grobbee contributed to acquisition, analysis or interpretation of data; Savonitto and Morici contributed to drafting of the manuscript; Cosentino, Perrone Filardi, Murena, Morocutti, Ferri, Cavallini, Eijkemans, Stähli, Schrieks, Toyama, Lambers Heerspink, Malmberg, Schwartz, Lincoff, Ryden, Tardif and Grobbee contributed to critical revision of the manuscript for important intellectual content; Morici and Nozza contributed to statistical analysis; Tardif, Lincoff and Grobbee obtained funding; Tardif, Lincoff and Grobbee contributed to study supervision.
Declaration of conflicting interests
The AleCardio trial was sponsored by F. Hoffmann-La Roche (Basel, Switzerland); the sponsor had no role in the present analysis, data interpretation or drafting of the manuscript, nor in the decision to submit the manuscript for publication. Dr Savonitto reports grants and personal fees from Eli Lilly, Novartis, Iroko, Daiichi Sankyo and Pfizer, outside the submitted work. Ms Nozza is Senior Biostatistician at the Montreal Heart Institute Coordinating Center, receives salary from her institution, but received no specific or additional compensation for her contribution. Dr Schwartz reports receiving research grants from Anthera, Resverlogix, Roche and Sanofi-Aventis through his institution. Dr Lincoff reports receiving research grants from AstraZeneca, CSL Laboratories, Eli Lilly, Regado, Roche, Takeda and Vivus through his institution. Dr Ryden reports receiving research grants from AFA Insurance Company, Bayer AG, Karolinska Institutet Funds, Roche, the Swedish Diabetes Association and the Swedish Heart Lung Foundation through his institution; has received honoraria from expert committee or steering committee service from AstraZeneca, Bristol-Myers Squibb, Roche and Sanofi-Aventis. Dr Tardif reports receiving research grants from Amarin, Cerenis, Eli Lilly, Isis, Merck, Pfizer, Roche, Sanofi-Aventis and Servier through his institution and has received honoraria from Cerenis and Servier. Dr Grobbee reports receiving research grants from Roche, Novartis and Pfizer through his institution; has received consultancy fees from Julius Clinical; and has received consultancy fees from Roche to his institution. All other authors have reported that they have no relationships relevant to the contents of this paper to disclose.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
