Abstract
Background:
A randomised, double-blind, placebo-controlled study evaluated lipid- and glucose-lowering effects of colesevelam in patients with prediabetes and primary hyperlipidaemia. We report the effect of colesevelam on lipoprotein particle concentration and particle size (determined by nuclear magnetic resonance spectroscopy) in these patients.
Methods:
Adults with prediabetes (World Health Organization criteria), low-density lipoprotein cholesterol (LDL-C) ≥ 100 mg/dL (≥2.6 mmol/L) and triglycerides < 500 mg/dL (<5.6 mmol/L) were randomised to colesevelam 3.75 g/day or placebo for 16 weeks. The intent-to-treat population comprised 103 colesevelam and 106 placebo recipients.
Results:
At the end of the study, mean reduction from baseline in total LDL particle concentration was significantly greater with colesevelam versus placebo (mean treatment difference: −113 nmol/L; p = 0.02). Increases in total very low-density lipoprotein particle concentration (VLDL-P) and high-density lipoprotein particle concentration (HDL-P) did not differ significantly between the groups; however, with colesevelam versus placebo, there were significantly (p < 0.05) greater increases in large and medium VLDL-P and large HDL-P and reductions in small VLDL-P. Mean size increases were significantly greater with colesevelam for VLDL (mean treatment difference: 5.3 nm; p < 0.0001) and HDL (0.1 nm; p = 0.002).
Conclusions:
Colesevelam improved the overall atherogenic lipoprotein profile in adults with prediabetes and primary hyperlipidaemia, despite potentially less favourable changes in VLDL particles.
Keywords
Introduction
Cardiovascular risk is increased in patients with prediabetes. A meta-analysis of studies that evaluated the risk of cardiovascular disease in patients with prediabetes found that moderate increases in cardiovascular risk were associated with impaired fasting glucose, based on fasting plasma glucose (FPG) levels of 110–125 mg/dL [summary estimate of relative risk, 1.20; 95% confidence interval (CI) = 1.12–1.28] or 100–125 mg/dL (summary estimate of relative risk, 1.18; 95% CI = 1.09–1.28), and with impaired glucose tolerance (summary estimate of relative risk, 1.20; 95% CI = 1.07–1.34). 1 In part, this is because insulin resistance is associated with dyslipidaemia, characterised by low high-density lipoprotein cholesterol (HDL-C) levels and elevated levels of triglyceride-rich lipoproteins; there may or may not be elevation of low-density lipoprotein cholesterol (LDL-C) levels, but there is commonly an overabundance of smaller, denser LDL particles, along with larger very low-density lipoprotein (VLDL) and smaller HDL particles. 2
Although reducing LDL-C is considered the main objective of lipid-lowering therapy to reduce cardiovascular risk,3,4 studies have demonstrated that lipoprotein subfractions may provide more information on cardiovascular risk than the standard lipid profile. Thus, increased LDL particle concentration (LDL-P) is an important predictor of cardiovascular risk5–13 and may be a better predictor of cardiovascular risk than LDL-C.7,9,11,13,14 There is conflicting information regarding whether LDL particle size predicts cardiovascular risk,5–9,13,15,16 and noted associations may be merely due to its relationship with LDL-P.3,11,16 Reduced HDL particle concentration (HDL-P) has also been associated with increased cardiovascular risk in multivariate models that include major risk factors and LDL-P.17,18 It has been suggested that smaller VLDL particles and their catabolic product, intermediate-density lipoprotein (IDL) particles, could contribute substantially to atherosclerosis because of their ability to enter the subendothelial space, 3 but larger VLDL particles are usually unable to penetrate the endothelium to enter the vascular wall, so their atherogenic potential is unclear.
The bile acid sequestrant colesevelam hydrochloride is approved both to reduce LDL-C levels in adults with primary hyperlipidaemia and to improve glycaemic control in adults with type 2 diabetes mellitus (T2DM). 19 Colesevelam reduces LDL-C levels by binding to bile acids in the intestine and preventing their reabsorption, resulting in depletion of the bile acid pool, which leads to increased conversion of cholesterol to bile acids (via upregulation of cholesterol 7-α-hydroxylase), in turn resulting in increased clearance of LDL-C from the blood. In addition to reducing LDL-C, colesevelam increases HDL-C and may slightly increase triglyceride levels. 20 Furthermore, colesevelam significantly reduced LDL-P and increased LDL size in adults with primary hyperlipidaemia 21 and reduced LDL-P (mostly due to an effect on small LDL-P) when added to metformin and/or sulphonylurea therapy in adults with T2DM. 22 A clinical study evaluated the lipid- and glucose-lowering effect of colesevelam in patients with prediabetes and primary hyperlipidaemia; 23 a separate arm of the study evaluated the combination of colesevelam with metformin in patients with T2DM. 24 Here, we report the effect of colesevelam on the concentration and size of various lipoprotein particle subclasses in patients with prediabetes and primary hyperlipidaemia from that study.
Methods
A 16-week, randomised, double-blind, placebo-controlled, parallel-group study was conducted at 16 sites in the United States, 10 in Mexico, 7 in Colombia and 5 in India. Full details of this study, including the design, inclusion/exclusion criteria and statistical methods, have been published previously.23,25 Briefly, adults diagnosed with prediabetes [based on World Health Organization criteria: glucose ≥ 140 and < 200 mg/dL (≥7.8 and <11.1 mmol/L) 2-hour post-oral glucose tolerance test, and FPG ≥ 110 and ≤ 125 mg/dL (≥6.1 and ≤6.9 mmol/L)], 26 with LDL-C ≥ 100 mg/dL (≥2.6 mmol/L) and with triglycerides < 500 mg/dL (<5.6 mmol/L) were eligible. Use of lipid-altering drugs [3-hydroxy-3-methylglutaryl (HMG)-CoA reductase inhibitors (statins), fibrates, niacin and ezetimibe] was permitted, provided a stable dose had been maintained for ≥3 months and dosage changes were not anticipated.
Patients were randomised to receive colesevelam 3.75 g/day or placebo for 16 weeks of double-blind treatment (Figure 1). Colesevelam or placebo was taken either once daily (six tablets with the evening meal) or twice daily (three tablets each with the noon and evening meals), based on patient preference; however, the selected dosing schedule was to be maintained for the study duration.
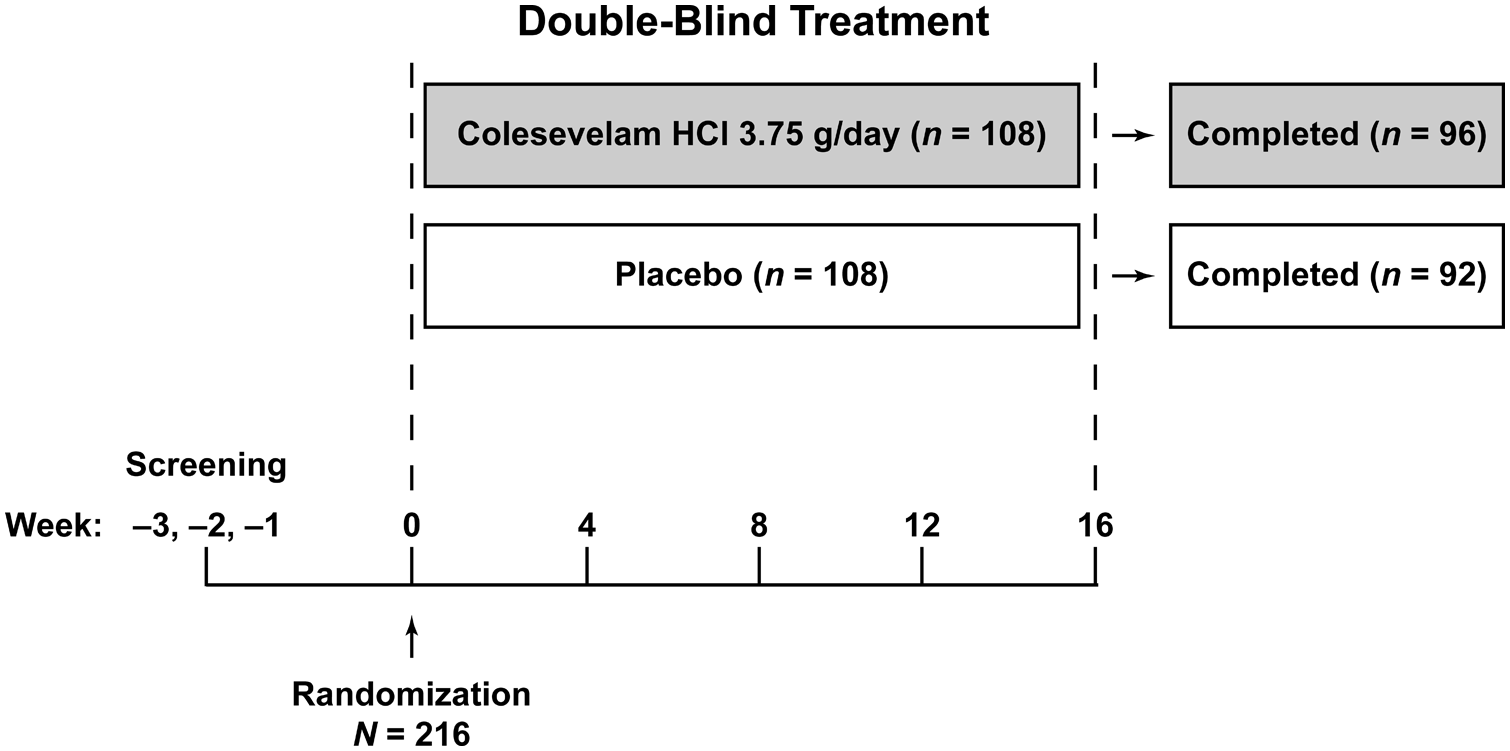
Study design.
The primary efficacy variable was the change in LDL-C from baseline to Week 16 with last (post-baseline) observation carried forward (LOCF) imputation. Secondary efficacy variables, which were also evaluated using Week 16 LOCF analyses, included change and percent change in lipids, apolipoproteins (apo) and lipoprotein particle concentration and change in lipoprotein particle size, FPG level and haemoglobin A1C value. Safety variables included adverse events. The LDL-C level was calculated using the Friedewald equation for patients with triglycerides < 400 mg/dL (<4.5 mmol/L) and using the Lipid Research Clinics Beta-Quantification method 27 for patients with triglycerides of 400–500 mg/dL (4.5–5.6 mmol/L) (inclusive). The method of LDL-C level determination used at screening was maintained throughout the study, regardless of changes in triglyceride levels. Nuclear magnetic resonance spectroscopy was used to determine VLDL, LDL and HDL sizes: large/chylomicron (estimated subclass diameter range >60 nm), medium (35–60 nm) and small VLDL particle concentration (VLDL-P) (27–35 nm); large (21.2–23 nm) and small LDL-P (18–21.2 nm) and large (8.8–13 nm), medium (8.2–8.8 nm) and small (7.3–8.2 nm) HDL-P.28,29 The concentration of particles was then calculated.
Statistical methods
Efficacy analyses were carried out with the intent-to-treat population, which included all randomised patients who had received at least one dose of the study medication and had a baseline and at least one post-baseline efficacy variable measurement. An analysis of covariance model was used, which included treatment and country as fixed effects and the baseline value as a continuous covariate. The treatment difference was expressed as the difference between the least squares mean change for each treatment arm.
Results
Demographic and baseline characteristics
In total, 216 patients were randomised to receive either colesevelam (n = 108) or placebo (n = 108). As previously reported, demographic and baseline characteristics were similar between the treatment groups. 23 The majority of patients were Hispanic (74%) and had been previously diagnosed with prediabetes (77%). At baseline, 6 patients (6%) in the colesevelam group and 10 (9%) in the placebo group were receiving statin therapy. Seven patients (7%) and three patients (3%) in the colesevelam and placebo groups, respectively, were receiving fibrate therapy at baseline. One patient assigned to colesevelam who did not take study medication or have any post-baseline efficacy measurements was excluded from the safety population. A total of five patients assigned to colesevelam and two assigned to placebo who did not have any post-baseline efficacy measurements were excluded from the intent-to-treat population; thus, the intent-to-treat population comprised 103 colesevelam and 106 placebo recipients.
Efficacy
Treatment with colesevelam, compared with placebo, resulted in a significant mean reduction in LDL-C [mean treatment difference: −21.1 mg/dL (−0.55 mmol/L; −15.6%), primary endpoint], total cholesterol [−16.6 mg/dL (−0.43 mmol/L; −7.2%)], non-HDL-C [−16.0 mg/dL (−0.41 mmol/L; −9.1%)] and apoB [−9.0 mg/dL (−0.09 g/L; −8.1%)] and a significant median increase in triglycerides [median treatment difference: 27.0 mg/dL (0.30 mmol/L; 14.3%); 95% CI = 10.0–44.5] from baseline at Week 16 (p < 0.001 for all). 23 The mean change in HDL-C [mean treatment difference: −0.6 mg/dL (−0.01 mmol/L; −0.5%)] and apoA-I [2.3 mg/dL (0.02 g/L; 1.8%)] was not significant for colesevelam compared with placebo at Week 16. In addition, patients who received colesevelam, compared with placebo, had a significantly greater median reduction from baseline in FPG (median treatment difference: −2.0 mg/dL; p = 0.02) and mean reduction from baseline in haemoglobin A1C level (mean treatment difference: −0.10%; p = 0.02) at Week 16. 23
Nuclear Magnetic Resonance Findings
LDL
Colesevelam treatment did not result in a significant mean [± standard error (SE)] change from baseline in overall LDL size compared with placebo at Week 16 (0.1 ± 0.07 vs 0.2 ± 0.06 nm; mean treatment difference: −0.1 ± 0.09 nm; Figure 2). At Week 16, a significantly greater reduction from baseline in total LDL-P was seen with colesevelam compared with placebo, resulting in a mean treatment difference of −113 nmol/L (p = 0.02; Figure 3), which corresponded to a mean percentage change from baseline of −7.2% (p = 0.01; Figure 4). Large LDL-P was increased and small LDL-P was decreased from baseline in both treatment groups at Week 16; however, the treatment differences were not significant (Table 1).
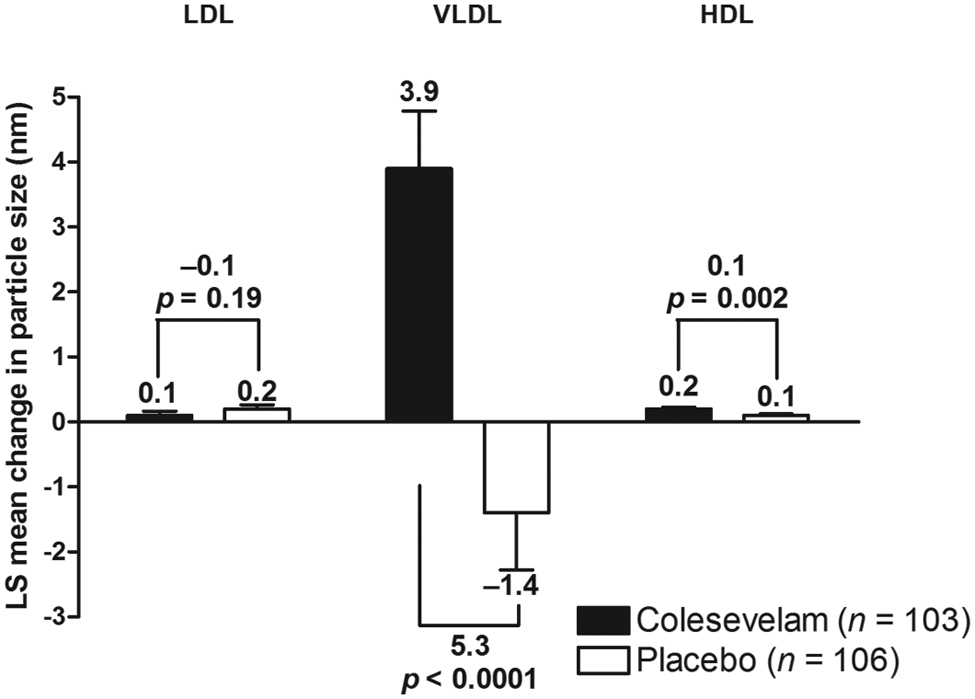
Mean change from baseline in LDL, VLDL and HDL particle sizes in patients with prediabetes and primary hyperlipidaemia.
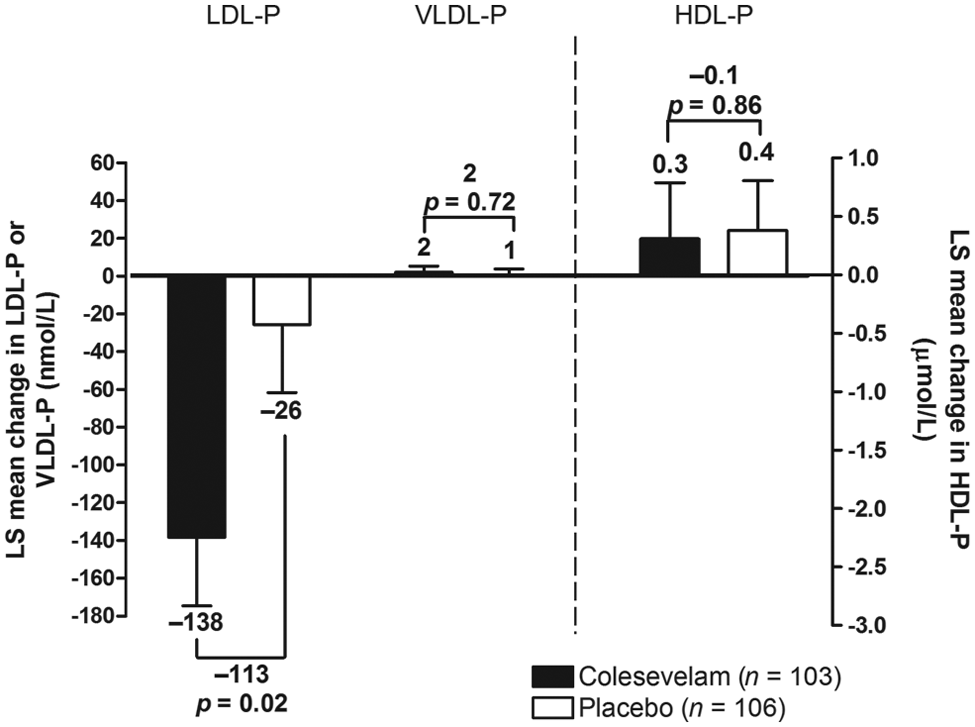
Mean change from baseline in total LDL-P, VLDL-P and HDL-P in patients with prediabetes and primary hyperlipidaemia.
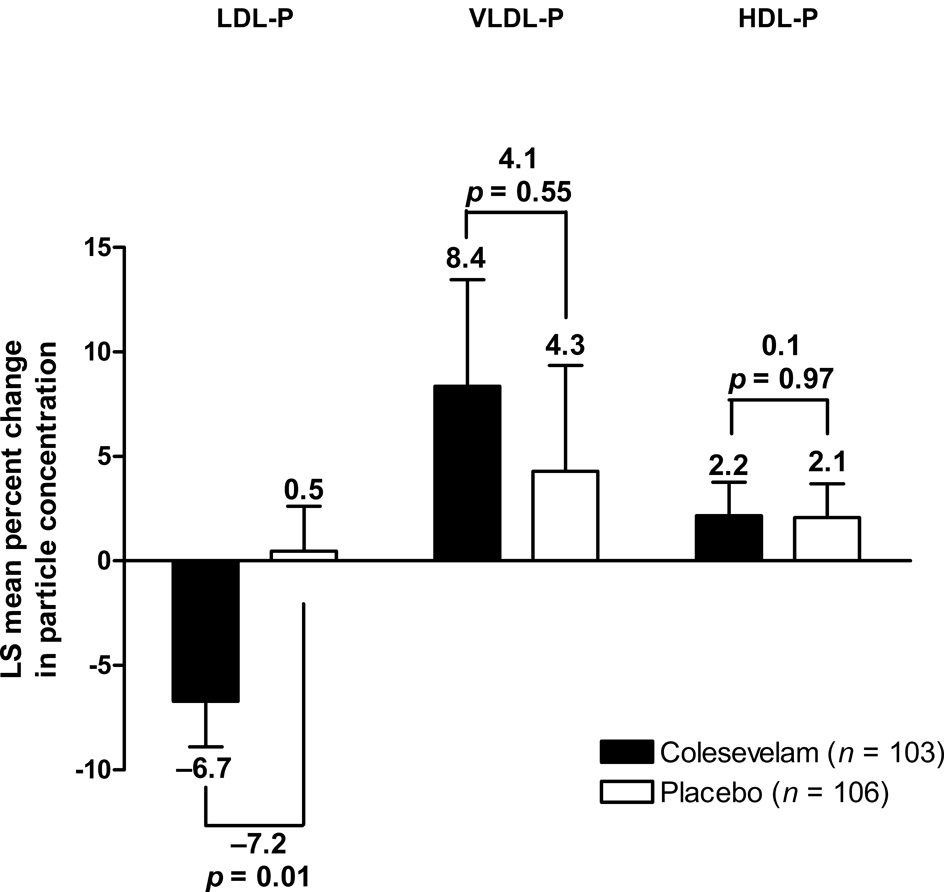
Mean percent change from baseline in total LDL-P, VLDL-P and HDL-P in patients with prediabetes and primary hyperlipidaemia.
Lipoprotein particle subclass concentrations at baseline and Week 16 (intent-to-treat population).
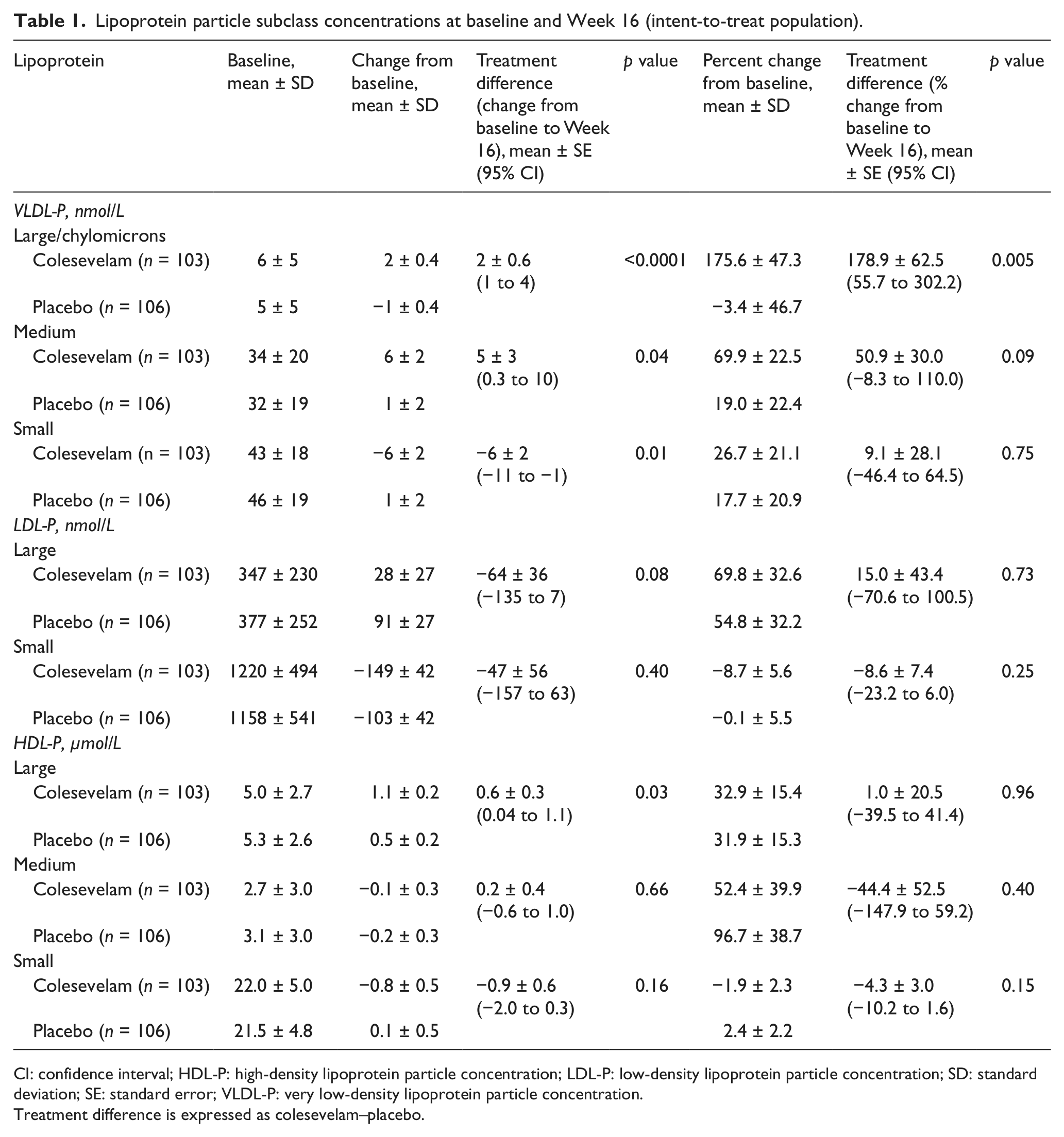
CI: confidence interval; HDL-P: high-density lipoprotein particle concentration; LDL-P: low-density lipoprotein particle concentration; SD: standard deviation; SE: standard error; VLDL-P: very low-density lipoprotein particle concentration.
Treatment difference is expressed as colesevelam–placebo.
VLDL
Colesevelam led to a significant mean (± SE) increase from baseline in overall VLDL size compared with placebo at Week 16 (3.9 ± 0.9 vs −1.4 ± 0.9 nm; mean treatment difference: 5.3 ± 1.2 nm; p < 0.0001; Figure 2). Total VLDL-P increased from baseline with both colesevelam and placebo treatment at Week 16; the mean treatment difference was not significant in terms of absolute change (2 nmol/L; Figure 3) or percentage change (4.1%; Figure 4) from baseline. At Week 16, large and medium VLDL-P were significantly increased from baseline with colesevelam compared with placebo (p < 0.05 for both), whereas small VLDL-P decreased from baseline with colesevelam and increased from baseline with placebo, resulting in a significant mean treatment difference at Week 16 (p = 0.01; Table 1). The treatment difference for percentage change was only significant for large VLDL-P (+178.9% ± 62.5%; p = 0.005; Table 1).
HDL
HDL size showed a mean (± SE) increase from baseline with both colesevelam and placebo treatment (0.2 ± 0.02 and 0.1 ± 0.02 nm, respectively), but the increase was greater in the colesevelam group, resulting in a significant mean treatment difference of 0.1 ± 0.03 nm at Week 16 (p = 0.002; Figure 2). Total HDL-P increased from baseline in both the colesevelam and placebo groups at Week 16; however, the mean treatment difference was not significant in terms of absolute change (−0.1 µmol/L; Figure 3) or percentage change (0.1%; Table 1) from baseline. Large HDL-P increased from baseline with both colesevelam and placebo, resulting in a significantly greater mean absolute increase with colesevelam compared with placebo at Week 16 (p = 0.03), although the treatment difference was not significant for percentage change (Table 1). There was no significant treatment difference for absolute or percentage change from baseline in medium or small HDL-P at Week 16 (Table 1).
Safety
Colesevelam treatment was well tolerated in patients with prediabetes and primary hyperlipidaemia. 23 Overall, 120 patients reported an adverse event [colesevelam (n = 57); placebo (n = 63)]. Adverse events considered by the investigator to be drug-related (possibly, probably or definitely) were reported by 18 patients in the colesevelam group and 12 in the placebo group. Gastrointestinal drug-related adverse events (constipation, diarrhoea and dyspepsia) were most common, occurring in 14 colesevelam recipients and 5 placebo recipients. Serious adverse events were reported by two patients in the colesevelam group [erectile dysfunction (n = 1); polycystic ovaries (n = 1)] and two in the placebo group [cholelithiasis (n = 1); cystocele (n = 1)]; however, none of these events were considered by investigators to be study drug-related or led to study withdrawal. One patient from each treatment group reported hypoglycaemia.
Discussion
In this study, colesevelam significantly improved the LDL and HDL profiles, including reductions in LDL-P, in adults with prediabetes and primary hyperlipidaemia. Such changes are generally considered to reduce cardiovascular risk; however, the effect of colesevelam on cardiovascular morbidity and mortality has not been determined. These results are consistent with previous studies demonstrating significant reductions in LDL-P with colesevelam monotherapy in adults with primary hyperlipidaemia 21 and with colesevelam in combination with metformin and/or sulphonylurea therapy in adults with T2DM. 22 They further suggest that most of the LDL-P reduction is due to a change in small LDL-P, given that the magnitude of the reduction in small LDL-P and increase in large LDL-P was numerically greater in patients receiving colesevelam, compared with placebo. The reduction in small LDL-P occurred despite the fact that colesevelam increased triglycerides and VLDL-P. This study did not show a significant change in overall LDL size.
Despite the absence of an effect of colesevelam on HDL-C in this study, significant increases were seen in overall HDL size and the proportion of large HDL. Evidence suggests that increased HDL-P and size are associated with reduced cardiovascular risk.17,30
Colesevelam increased total VLDL-P and size, mostly due to an increase in medium-sized, and to a lesser extent, large VLDL-P, but decreased small VLDL-P. Increased large VLDL-P is typical of the insulin-resistant state and is thought to drive the formation of small LDL and small HDL particles, which are associated with increased cardiovascular disease risk. Interestingly, despite this effect of colesevelam on VLDL-P, there was no accompanying increase in either small LDL-P or small HDL-P. Thus, the effects on small LDL-P and small HDL-P were comparable to those seen in other studies with statins, fibrates and niacin, in the absence of increases in VLDL-P.31,32 Therefore, it is possible that the hypertriglyceridaemic effect of colesevelam is relatively benign. In addition, the fact that colesevelam led to a reduction in small VLDL may be a benefit, as it is known that small VLDL may gain access to the subendothelial space, which is less accessible to larger VLDL.
Study limitations include the following: randomisation was not stratified by sex, ethnicity or country and the study population consisted mostly of Hispanic females, which could limit the overall generalisability of these results. However, given the lack of clinical trial data in non-Caucasian populations, these data serve to expand our knowledge in patients of different ethnicity. In addition, the majority of patients were not receiving statin treatment, which is recommended to reduce the increased cardiovascular risk associated with prediabetes.
In conclusion, colesevelam improved the atherogenic lipoprotein profile by significantly reducing LDL-P in adults with prediabetes and primary hyperlipidaemia. In addition to reducing LDL-C levels and LDL-P, colesevelam induced favourable changes in the quality of HDL, LDL and small VLDL particles despite potentially less favourable changes in large and medium VLDL particles.
Key messages
Lipoprotein particle concentrations, and perhaps also size, are an important predictor of cardiovascular risk.
Treatment with colesevelam, compared with placebo, resulted in a significantly greater reduction from baseline in total LDL-P, which may be largely due to a reduction in small LDL-P.
Colesevelam treatment led to a favourable increase in concentrations of large HDL particles accompanied by an overall increase in HDL size, despite no change in HDL-C.
Total, large and medium VLDL-P and VLDL size increased, while small VLDL-P decreased, with colesevelam.
Footnotes
Acknowledgements
Medical writing services and editorial assistance were provided by Karen Stauffer, PhD, Lucy Whitehouse and Sushma Soni of inScience Communications, Springer Healthcare. The statistical analyses were undertaken by a contract research organization (Kendle) and by statisticians at Daiichi Sankyo, Inc., and the academic authors had full access to the data.
Conflict of interest
Dr Goldberg has received research grants from Abbott Laboratories, GlaxoSmithKline and Roche.
Dr Rosenson has participated in advisory boards for Abbott Laboratories, Amgen Inc., AstraZeneca, Hoffmann-La Roche Inc., LipoScience, Inc. and sanofi-aventis and owns stock in LipoScience, Inc. Dr Rosenson’s institution has received research grants from Amgen Inc. and Hoffman-La Roche Inc.
Dr Hernandez-Triana has served on scientific advisory boards for Eli Lilly and Company, Novo Nordisk, Pfizer Inc and Roche and received consulting fees and research grants from Boehringer Ingelheim, Bristol-Myers Squibb, GlaxoSmithKline, Johnson & Johnson, MSD, Novartis and sanofi-aventis. Dr Hernandez-Triana and his immediate family do not have ownership interest and/or stocks in any pharmaceutical or device company.
Drs Jones and Misir are employed by Daiichi Sankyo, Inc., the study sponsor.
Funding
This study was funded by Daiichi Sankyo, Inc.
ClinicalTrials.gov identifier: NCT00570739
