Abstract
The lectin-like oxidized low-density lipoprotein receptor-1 (LOX-1) has gained attention for its pro-inflammatory potential in atherogenesis. This study evaluates LOX-1 receptor modulation in the presence of an atheroprotective cytokine, interleukin-10 (IL-10). Both oxidized low-density lipoprotein (oxLDL) and IL-10 stimulated LOX-1 cell surface expression on THP-1 macrophages. However, our study demonstrates differential roles of oxLDL and IL-10 on LOX-1 functionality. Seemingly, oxLDL-induced LOX-1 promoted pro-inflammatory signalling by increasing intracellular NO, a substrate for pro-inflammatory peroxynitrite. In contrast, IL-10-induced LOX-1 facilitated scavenging of extracellular oxLDL without any effect on pro-inflammatory signalling. The atheroprotective effects of IL-10 were demonstrated by both facilitation of cellular oxLDL uptake and expression of LOXIN, an atheroprotective haplotype of the LOX-1 gene. Thus, increased expression of IL-10 may help to attenuate the risk of atherosclerosis developed by pro-inflammatory signal(s) generated through the interaction of oxLDL with its cognate receptor LOX-1 on macrophages.
Keywords
Introduction
Resistance to clearance and persistence in blood vessels promote chemical oxidation of existing low-density lipoprotein (LDL) particles by dissolved oxygen and other oxidizing entities such as myeloperoxidase, lipoxygenases and oxidative stress.1,2 Arterial lipoprotein modification generates chemically reactive lipid peroxidation and aldehyde products such as malondialdehyde (MDA) and 4-hydroxynonenal. Ongoing lipid peroxidation also triggers inflammatory reactions in vascular cells to accelerate internalization of lipoprotein-derived lipids, eventually leading to cell death. 3 Modified lipoproteins such as oxidized LDL (oxLDL) and acetylated LDL escape detection by native LDL receptors and trigger recognition by families of macrophage receptors capable of unregulated uptake. This in turn leads to intracellular accumulation of lipoprotein-derived cholesterol and cholesterol esters, characteristic of foam cells.
oxLDL has been reported to facilitate the formation of more oxLDL and other pro-inflammatory cytokines by a vicious cycle mediated through a specific receptor known as the lectin-like oxidized LDL receptor-1 (LOX-1).4,5 Vascular endothelial cells have been demonstrated to utilize this LOX-1 receptor to degrade oxLDL.
6
Recently, a novel dynamin-2-mediated pathway for the internalization of oxLDL via LOX-1 has also been demonstrated.
7
Studies have demonstrated that the binding of oxLDL to LOX-1 increases intracellular reactive oxygen species (ROS) including superoxide anion (
Interleukin-10 (IL-10) is well known for its prototypical anti-inflammatory property,9,10 and the latest report has supported its therapeutic potential in atherosclerosis. 11 IL-10 has been shown to stabilize atherosclerotic plaques by inducing lipid accumulation in oxLDL-stimulated macrophages. 11 This study also highlights the atheroprotective effect of IL-10 from the perspective of LOX-1-mediated clearance of extracellular oxLDL by monocyte-derived THP-1 macrophages.
Materials and methods
Cell culture
Human monocyte leukaemia cell line, THP-1, was cultured in Roswell Park Memorial Institute (RPMI) 1640 medium containing 10% fetal calf serum (FCS), 100 units of penicillin, streptomycin and amphotericin B at 37°C in a 5% CO2 environment. For differentiation into macrophages, the monocytes were plated in RPMI containing 10% FCS in the presence of 100 nM of phorbol 12-myristate 13-acetate (PMA), without antibiotic/antimycotic for 24 h.
Isolation of LDL
LDL was isolated from pooled human plasma [procured from the blood bank facility at All India Institute of Medical Sciences (AIIMS), New Delhi, after institutional ethics committee clearance] by density gradient ultracentrifugation. 12
Dil-oxLDL labelling
oxLDL was incubated with l,l′-dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine perchlorate (Dil) at 300 µg of Dil/mg oxLDL protein for 18 h/37°C in dark. The labelled fraction was isolated following ultracentrifugation, 12 dialyzed at 4°C and filter-sterilized. The protein content was determined, 13 and Dil-oxLDL was stored in dark vials under nitrogen gas until use.
Immunoblot analysis
Cells were lysed, and 50 µg of protein was resolved in 12% sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE) after brief denaturation under standard conditions. The proteins were trans-blotted onto a nitrocellulose membrane at 60 V/5 h in an ice bath. Following blocking with 4% bovine serum albumin (BSA) for 2 h, the membranes were incubated overnight with rabbit anti-LOX-1 (1:2000), mouse anti-IL-10 (1:1500) or mouse anti-glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (1:1000) primary antibody in 2% BSA at 4°C. Following washing, the primary antibodies were detected with secondary goat anti-rabbit-horseradish peroxidase (HRP) (1:3000) or goat anti-mouse-HRP (1:5000) conjugate in 2% BSA for 2 h at room temperature (RT). The blots were developed with enhanced chemiluminescence (ECL) kit. The band intensity was determined densitometrically by Alpha Imager EC gel documentation system, CA, USA gel documentation system and expressed in terms of percentage integrated density value (%IDV).
Reverse transcriptase and semi-quantitative polymerase chain reaction
A measure of 1000 ng of RNA was reverse transcribed into complementary DNA (cDNA) using RevertAid Reverse Transcriptase enzyme (Fermentas, New Delhi, India) and random hexamer primers. Semi-quantitative polymerase chain reaction (PCR) was carried out to amplify all three transcript variants (TRVs) of LOX-1 individually from cDNA using LOXTRV primers (FP – CAGATCTCAGCCCGGCAACAAGC; RP – GGAGACAGCGCCTCGGACTCTA). The LOXTRV primers were designed in an exon-specific manner to pick up all three splice variants of LOX-1 under common amplification conditions with different amplicon lengths owing to the respective lacking exon (namely, TRV1-456 bp, TRV2-316 bp and TRV3-340 bp). Amplification was carried out in 20 µL volume for 35 cycles – denaturation at 95°C/15 s, annealing at 62°C/45 s and extension at 72°C/45 s.
Quantitative real-time PCR
Quantitative real-time PCR (qRT-PCR) was performed using Power SYBR® Green (2X) Master Mix on a 7500 SDS real-time PCR machine (Applied Biosystems, Inc., CA, USA). Acquisition and analysis were carried out using SDS-V2.04 software. The sequence of inducible nitric oxide synthase (iNOS) primers used was as follows: FP – ACAAGCCTACCCCTCCAGAT and RP – TCCCGTCAGTTGGTAGGTTC. Amplification was carried out in 20 µL volume for 40 cycles – hot start (hold at 50°C/2 min and 95°C/10 min), denaturation at 95°C/15 s, annealing at 60°C/1 min and extension at 72°C/30 s.
Cell surface expression of LOX-1 protein by fluorescence-assisted cell sorting analysis
The 1 × 106 THP-1 monocytes/well were differentiated with 100 nM of PMA in a six-well plate format into macrophages, which were washed and incubated with media containing either oxLDL and/or IL-10. The cells were scraped off in ice-cold 10 mM of phosphate-buffered saline (PBS) containing 2% BSA and 0.1% sodium azide and centrifuged at 2000 × g for 5 min. The pellets were incubated with human anti-LOX-1 mouse IgG2B antibody (specific for the C-terminal extracellular domain of LOX-1; 1:200) in 1% BSA/1 h. A parallel IgG2B isotype control was included to negate non-specific interactions. The cells were then washed and incubated with anti-mouse IgG-fluorescein conjugated goat F(ab′)2 for 1 h/4°C. The cells were washed and resuspended in 2% paraformaldehyde and acquired for fluorescence-assisted cell sorting (FACS; BD FACSCanto instrument with FACSDiva acquisition software) against an untreated control.
Dil-oxLDL uptake assay
The functional activity of LOX-1 was explored on the basis of its ability to bind and internalize oxLDL. Following pretreatment of THP-1 macrophages with varying concentrations of IL-10 for 19 h, 20 µg/mL of Dil-oxLDL was added to the system, and the cells were incubated in dark under standard growth conditions for 5 h. Furthermore, the cells were washed with incomplete RPMI 1640 medium and incubated with dextran sulphate buffer [10 mg/mL of dextran sulphate, 50 mM of NaCl and 10 mM of 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES)] for 1 h at 4°C to remove cell membrane–bound Dil. A volume of 1 mL of isopropanol was added per well, and the system was incubated for 25 min at RT with constant mixing. The isopropanol was collected and centrifuged for 10 min at 5000 × g, and fluorescence was read at 520 nm of excitation and 574 nm of emission wavelengths in a spectrofluorimeter (Bio-Rad Technologies, PA, USA). A standard curve using ‘Dil’ in isopropanol was made from the same Dil stock used to tag oxLDL. Parallel readings of the standards and tests were taken in order to avoid major fluctuations in voltage associated with spectrofluorimetry. Also, for every batch of Dil-oxLDL prepared, a standard of 5 µg/mL of Dil-oxLDL in isopropanol was taken. The amount of Dil internalized was determined from the standard curve. The standard Dil-oxLDL was read using the same standard curve. The amount of ‘Dil’ determined from the graph was tabulated against the Dil-oxLDL standard and represented as Dil-oxLDL internalized per milligram of oxLDL protein.
DAF-FM-DA assay for nitric oxide
Assay for intracellular nitric oxide (NO) generation was performed with an NO-specific fluorogenic probe 4-amino-5-methylamino-2′-7′-difluorofluorescein diacetate (DAF-FM-DA)14,15 that gets oxidized to form a highly fluorescent (up to 160-fold) triazole product.
PMA-differentiated THP-1 macrophages were incubated in phenol red deficient growth medium with/without cytokine challenge. Pretreatment with IL-10 was carried out for 24 h, and oxLDL was added for the last 5 h. Macrophages treated with 5 ng/mL of interferon-gamma (IFN-γ) served as positive control for NO generation; 30 min before the end of incubation, 5 µM of DAF-FM-DA was added to the system and cells were incubated in dark. Untreated THP-1 macrophages incubated with DAF-FM-DA were used for baseline fluorescence. Thereafter, the media with DAF-FM-DA was replaced with serum-free RPMI. Following 30-min incubation in dark, change in fluorescence was monitored using FLUO-star OMEGA-ELISA reader at 485 nm of excitation and 515 nm of emission wavelengths.
MTT assay
PMA-differentiated THP-1 macrophages (6.25 × 104 cells/well in a 96-well format) were incubated with 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) for 4 h. Following this, the medium was replaced with 200 µL of dimethyl sulphoxide (DMSO) and kept for 5 min at RT. Absorbance was measured at 503 nm in FLUO-star OMEGA-ELISA reader, and the amount of MTT formed was calculated on the basis of MTT-Formazan standard curve.
Confocal microscopy
PMA-differentiated THP-1 macrophages were seeded onto sterile cover glasses in a 12-well plate format and incubated with/without IL-10 (5 ng/mL) for 24 h. In total, 20 µg of Dil-oxLDL was added during the last 5 h of the cytokine treatment. Live cell staining was performed with anti-LOX-1 antibody in 1% BSA (1:200) for 1 h at 4°C. Following washing with ice-cold 10 mM of PBS (containing 1% BSA without detergent), cells were incubated with fluorescein-conjugated (green) secondary antibody in 1% BSA (1:200) for 30 min at 4°C. Following fixation with 2% paraformaldehyde, the cover glasses were mounted onto glass slides in 10 mM of PBS:glycerol (1:1) and immediately analysed by confocal microscopy. Nuclear staining was carried out just before fixation with 5 µg/mL of Hoechst 33258 stain for 30 min at RT.
Statistical analysis
Comparison of means was carried out by one-way analysis of variance (ANOVA) followed by Dunnett’s multiple comparison (between three or more groups) or two-tailed Student’s t-test (between two groups).
Materials
PMA; 1,1,3,3-tetramethoxypropane; Dil; DAF-FM-DA; MTT; Formazan and Hoechst 33258 were purchased from Sigma, St Louis, Missouri, USA, and THP-1 monocyte leukaemia cell line was procured from NCCS, Ganeshkhind, Pune, India. Thiobarbituric acid (TBA) was obtained from LOBA Chemie Pvt. Ltd, India. Power SYBR green (2X) master mix was procured from Applied Biosystems, Inc, CA, USA, Mouse-monoclonal IgG2B LOX-1 antibody; IgG-fluorescein conjugated goat F(ab′)2 and IgG2B isotype antibody were procured from R&D Systems, Inc., Minneapolis, MN, USA. IL-10 and IFN-γ cytokines were procured from PeproTech, Inc., Rocky Hill, NJ, USA. All primary and HRP-conjugated secondary antibodies and Western blotting Luminol reagent kit were obtained from Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA. Nitrocellulose membrane (0.2 µm pore size) was purchased from Whatman GmbH, Dassel, Germany.
Results
Basal parameters of the study
PMA-mediated monocyte differentiation and LOX-1 expression
Although LOX-1 expression is negligible in monocytes, their differentiation into macrophages stimulates its expression. 4 PMA is known to induce monocyte differentiation into macrophages (adherent morphology) 16 with an increased expression of LOX-1. 17 A volume of 100 nM of PMA has been optimally used for THP-1 cells to be transformed into macrophages. 17 In order to determine the time required by 100 nM of PMA to induce optimal expression of LOX-1 protein, we treated THP-1 cells (~3.5 × 106 cells/90-mm plate) with 100 nM of PMA for different durations. Immunoblot analysis showed significant expression of LOX-1 by 12 h and an optimal plateau from 24 h onwards. Hence, we used 100 nM of PMA for 24 h throughout our study to maintain a baseline LOX-1 expression.
Preparation and standardization of ligand (oxLDL)
LDL was isolated from pooled human plasma and subjected to copper-mediated oxidation. The degree of oxidation was determined by evaluating the MDA content and the net charge of the preparation by REM determination (supplementary material: Figure S1). In our study, we found that an oxLDL preparation with an MDA content of 30–40 nmol/mg protein and a relative electrophoretic mobility (REM) of ~1.48 elicited significant LOX-1 expression (Figure 1).
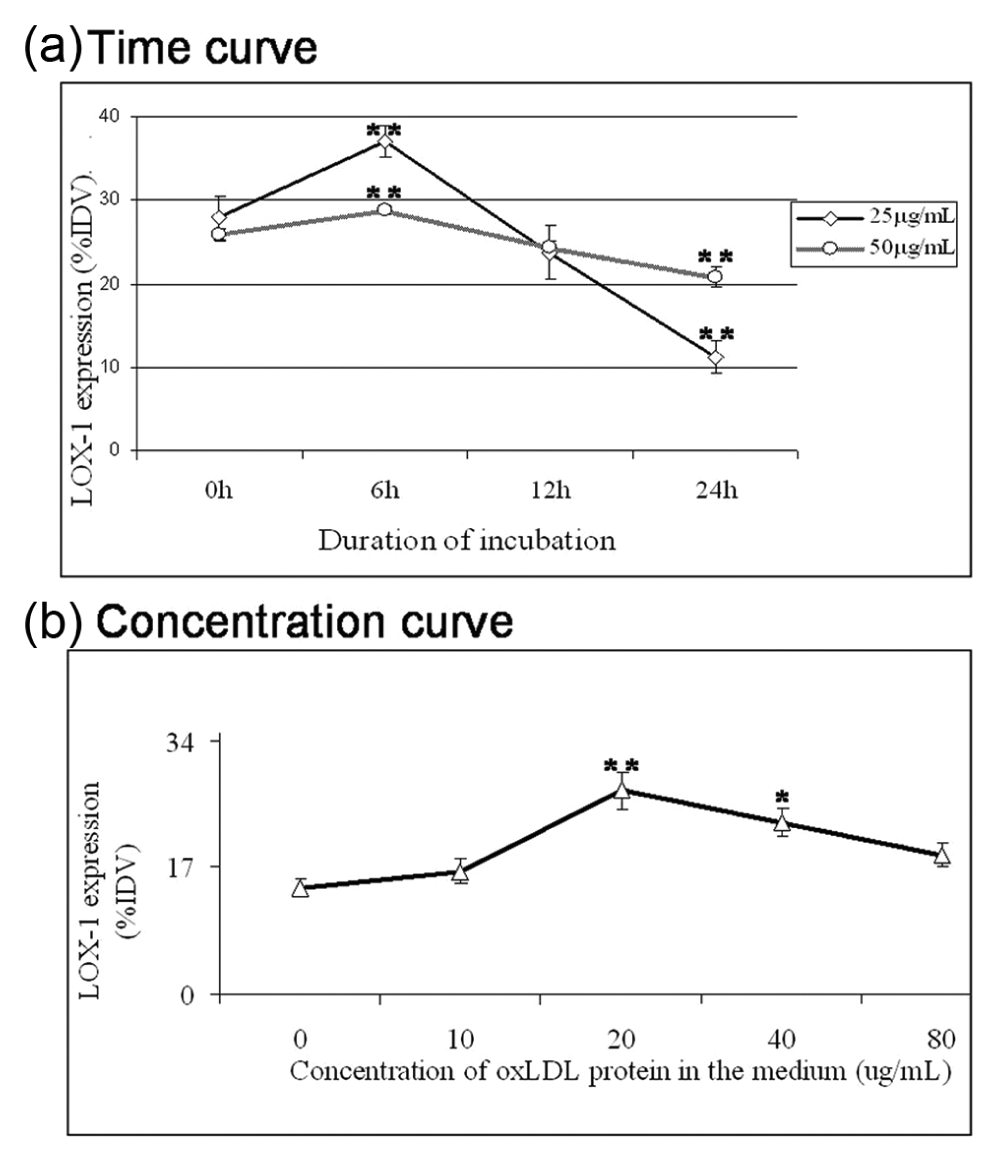
The effect of oxLDL on LOX-1 expression: (a) the optimal time of LOX-1 expression by Western blot in response to oxLDL (25 and 50 µg/mL) treatment. Maximal LOX-1 expression was observed within 6 h. (b) The optimal concentration of oxLDL (20 µg/mL) for maximal LOX-1 expression. PMA-differentiated THP-1 macrophages were incubated with varying concentrations of oxLDL for 5 h.
Standardization of oxLDL concentration used in this study for optimal LOX-1 expression
In order to determine an optimum duration for incubation with oxLDL, we used arbitrary concentrations of oxLDL protein and determined LOX-1 protein expression by immunoblotting. As seen in Figure 1(a), since there was no detectable expression of LOX-1 protein beyond 6 h, we chose 5 h as the exposure duration. Following this, experiments involving 5 h incubation with various concentrations of oxLDL showed optimal LOX-1 expression only at 20 µg/mL of oxLDL in the medium (Figure 1(b)). The concentration (20 µg/mL of oxLDL) and duration (5 h) of incubation with oxLDL for maximal LOX-1 expression was confirmed by using three different oxLDL preparations made in the laboratory.
Effect of IL-10 on LOX-1 expression
IL-10 has been reported to be atheroprotective for its role in the formation of foam cells. After incubation with various concentrations of the cytokine for 24 h, THP-1 macrophages showed peak expression of LOX-1 at 5-ng/mL IL-10 (Figure 2(a)). The LOX-1 expression after incubation with 5 ng/mL of IL-10 for 24 h was found to be similar to that observed with 20 µg/mL of oxLDL for 5 h (Figure 2(b)). Interestingly, a significant decrease in LOX-1 expression was observed when 20-µg/mL oxLDL was added during the last 5 h of 24 h incubation period with 5 ng/mL of IL-10 (Figure 2(b)). This phenomenon led us to explore the effect of IL-10 on the functional activity of LOX-1, as previous reports have suggested that IL-10 induces cellular internalization of oxLDL and foam cell formation. 11
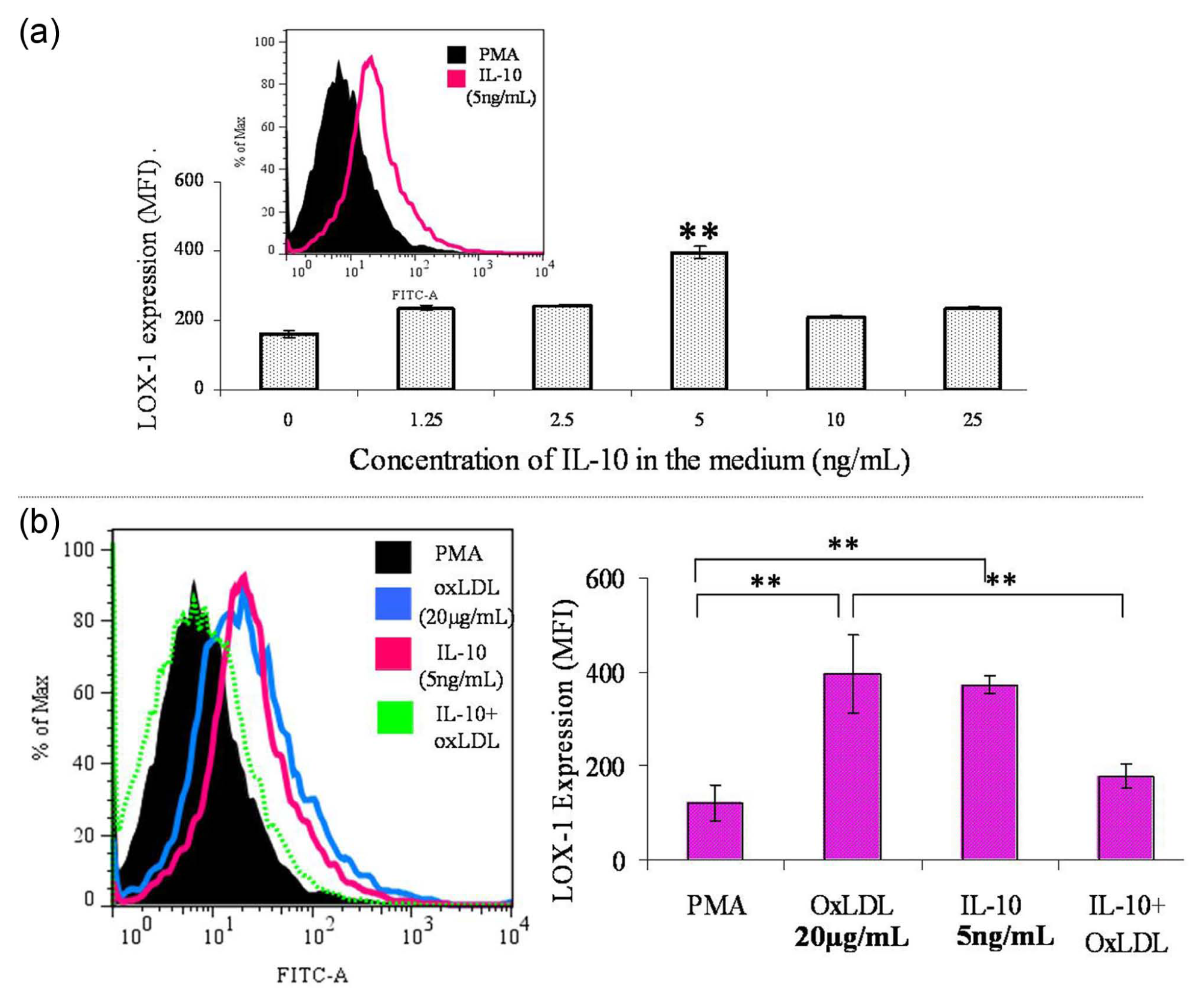
Effect of oxLDL and IL-10 on LOX-1 expression. (a) The effect of increasing concentrations of IL-10 on LOX-1 expression by FACS analysis. PMA-differentiated THP-1 macrophages were incubated with varying concentrations of IL-10 for 24 h under standard growth conditions. Optimal expression of LOX-1 was found at 5 ng/mL of IL-10. The data represent MFI ± SD of four independent experiments. Comparison of means was done by one-way ANOVA followed by Dunnett’s multiple comparison; *p < 0.05. (b) Expression of LOX-1 protein by FACS analysis. The histogram gives a representative picture of LOX-1 expression in response to oxLDL, IL-10 and IL-10 with oxLDL as compared to PMA baseline. All data are represented as MFI ± SD, and statistical analysis was done by unpaired two-tailed Student’s t-test; *p < 0.01; n = 4.
Functional activity of LOX-1 (Dil-oxLDL uptake)
We explored the functional aspect of LOX-1 by monitoring the uptake profile of Dil-oxLDL in the presence of varying concentrations of IL-10 in the medium. A volume of 20 µg/mL of Dil-oxLDL was added in the last 5 h of pretreatment with varying concentrations of IL-10 (24 h). The incubated THP-1 macrophages showed a significant increase in uptake of oxLDL at 5 ng/mL of IL-10 (Figure 3(a)). To confirm the significance of this concentration, we carried out an uptake curve with varying concentrations of oxLDL in the presence and absence of 5 ng/mL of IL-10. Clearly, there was a significant increase in Dil-oxLDL uptake at all concentrations in the presence of IL-10 (Figure 3(b)). Confocal microscopy with Dil-oxLDL also demonstrated increased intracellular oxLDL (red) in the presence of IL-10 (Figure 3(c)). To confirm whether this oxLDL uptake was mediated through LOX-1, we performed confocal microscopy for the existing LOX-1 receptors [green fluorescein isothiocyanate (FITC) tagged] and Dil-oxLDL (red) on macrophage surface (Figure 3(d)). The yellow colour showing co-localization of LOX-1 and Dil-oxLDL mostly existed on cell membrane in the absence of IL-10 (panel i) and in the cytoplasm in the presence of IL-10 (panel ii).
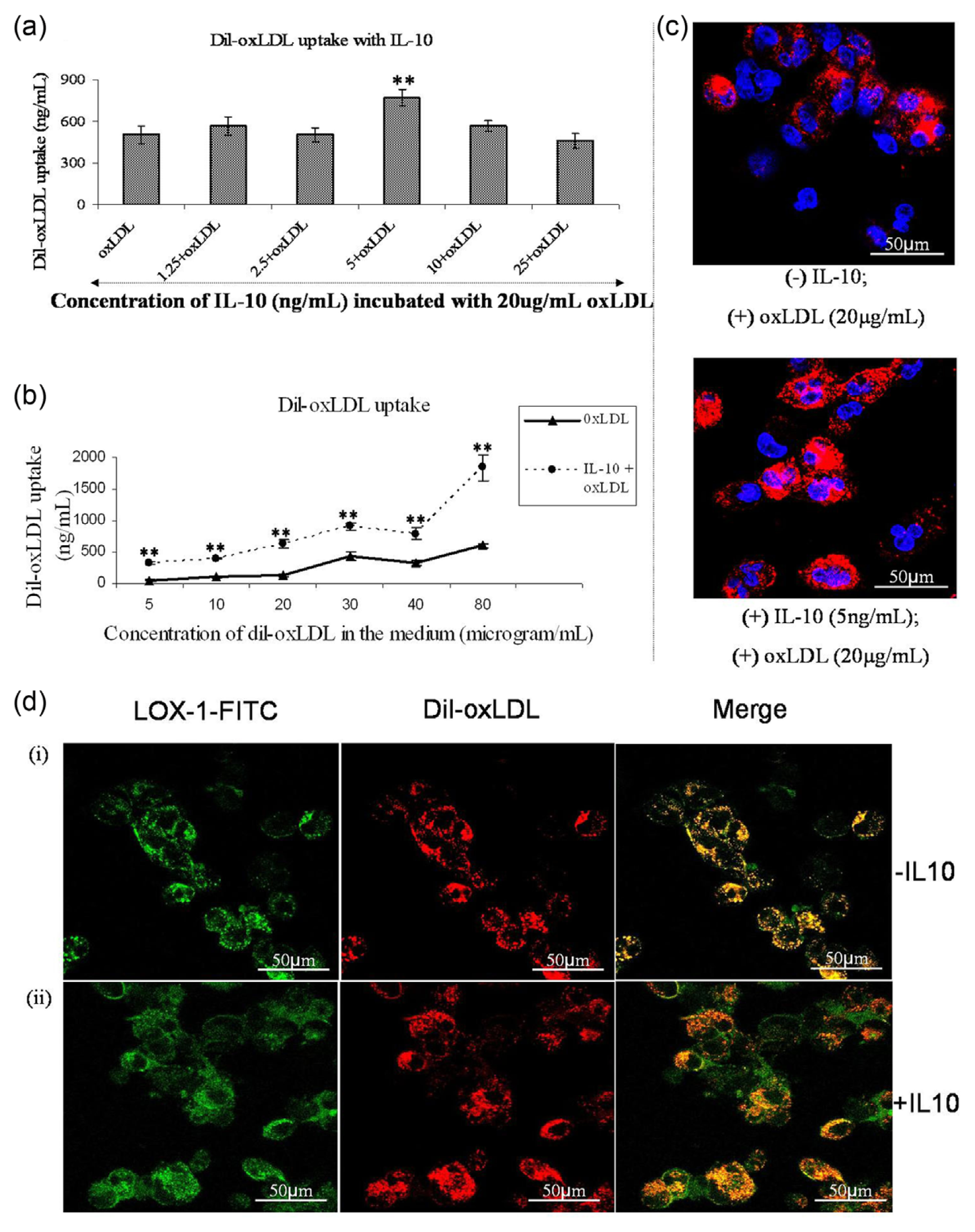
Cellular uptake of oxLDL. (a) The effect of IL-10 on oxLDL uptake. PMA-stimulated THP-1 macrophages were incubated with different concentrations of IL-10 for 24 h along with 20 µg/mL of Dil-oxLDL in the last 5 h of incubation. At 5-ng/mL concentration, IL-10 stimulated significant uptake of oxLDL. The data represent mean ± SD of three experimental repeats, comparison of means was done by one-way ANOVA followed by Dunnett’s multiple comparison; *p < 0.05. (b) The uptake of Dil-tagged oxLDL in the presence and absence of IL-10. In 24 h incubation period with IL-10 (5 ng/mL), varying concentrations of Dil-oxLDL was added in the last 5 h. There was a biphasic response in the uptake of oxLDL. IL-10 enhanced the uptake significantly. All values represent mean ± SD of three experimental repeats. The uptake values determined from standard curve of Dil and a 5 µg/mL of Dil-oxLDL standard. Statistical analysis was carried out by unpaired two-tailed Student’s t-test; *p < 0.05. (c) Images of confocal microscopy following challenge with Dil-labelled oxLDL (red) with/without IL-10 pretreatment. The nuclei have been stained with Hoechst 33258 (blue). It is depictive of enhanced Dil-oxLDL internalization in the presence of IL-10. (d) The co-localization of Dil-oxLDL with LOX-1 in the absence (panel i) and presence (panel ii) of 5 ng/mL of IL-10 by confocal microscopy. Co-localization (yellow) of oxLDL (red) with LOX-1 (green) on membrane surface without of IL-10 and their entry into the cells (yellow) with IL-10.
iNOS expression and NO production
Fixation of in vitro metabolic study duration (pulse–chase study)
In order to understand the effect of oxLDL on iNOS expression based on its signal advancement with respect to time, we designed an in vitro metabolic satire by first treating differentiated THP-1 macrophages with oxLDL (pulse) and then withdrawing the ligand and monitoring its remnant, time-dependent effects (chase). For this, we first had to fix the maximum duration up to which no significant loss of cell viability was observed.
PMA-differentiated macrophages once seeded cannot proliferate. Trypan blue exclusion (data not shown) in pulse–chase study with oxLDL showed that THP-1 macrophages remained viable up to 72 h. MTT assay carried out in the pulse–chase set-up to reaffirm the cell viability also did not show any major change in cell viability (Figure 4(a)).
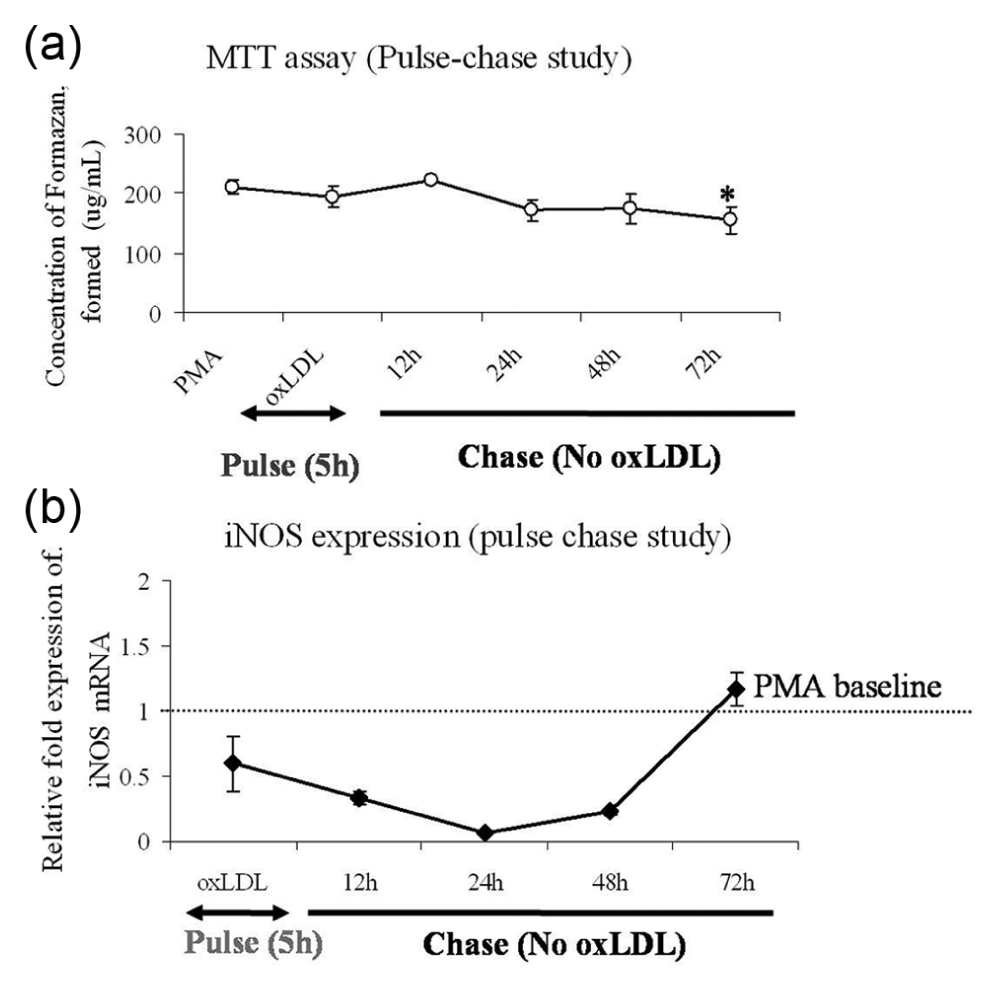
Pulse–chase study. (a) MTT assay showed hardly any change in cell viability during pulse–chase time with only a marginal decline at 72 h. All data are represented as mean ± SD of four experimental repeats. Comparison of means was done by one-way ANOVA followed by Dunnett’s multiple comparison; *p < 0.05. (b) iNOS expression: the relative fold mRNA expression of iNOS mRNA is shown during the course of the pulse–chase study. oxLDL pulse had an immediate inhibitory effect on iNOS mRNA, even below PMA baseline, having only mild elevation at 72 h. The data are represented as mean ± SD of the relative fold expression (2−ΔΔCT) of LOX-1 mRNA expressed as ‘X’ fold over PMA baseline calculated against 18srRNA internal control and carried out in triplicates.
A 5-h pulse with 20 µg/mL of oxLDL inhibited iNOS messenger RNA (mRNA) expression (Figure 4(b)); 72 h chase without any oxLDL in the medium recovered the mRNA level up to baseline (PMA-induced expression in THP-1 macrophages). Induction of iNOS is important for NO synthesis in macrophages. Previous reports have shown that the interaction of oxLDL with LOX-1 reduces intracellular NO concentration by converting it to peroxynitrite (ONOO−), which promotes NF-κB activation and a pro-atherogenic response. 18 However, as shown in Figure 5, oxLDL demonstrated a stimulatory effect on intracellular NO pool (DAF-FM fluorescence) as compared to PMA baseline. IFN-γ was used as a positive control for NO generation. Ironically, a dual role of oxLDL is apparent from the existing report and our data. On the one hand, oxLDL inhibits iNOS, and on the other hand, it increases intracellular NO concentration to promote pro-inflammatory signalling by facilitating peroxynitrite formation. We believe that a probable underlying mechanism could be the induction of endothelial NOS (eNOS) activity in macrophages. This is also in concordance with a previous report that has demonstrated the existence of eNOS [or constitutive NOS (cNOS)] in macrophages. 19 The presence of IL-10 alone or in combination with oxLDL did not show any significant change in the intracellular NO pool as compared to that induced by oxLDL alone levels. This shows that IL-10 is not involved in intracellular NO synthesis and hence is not a partner to LOX-1-mediated induction of pro-atherogenic modulators.
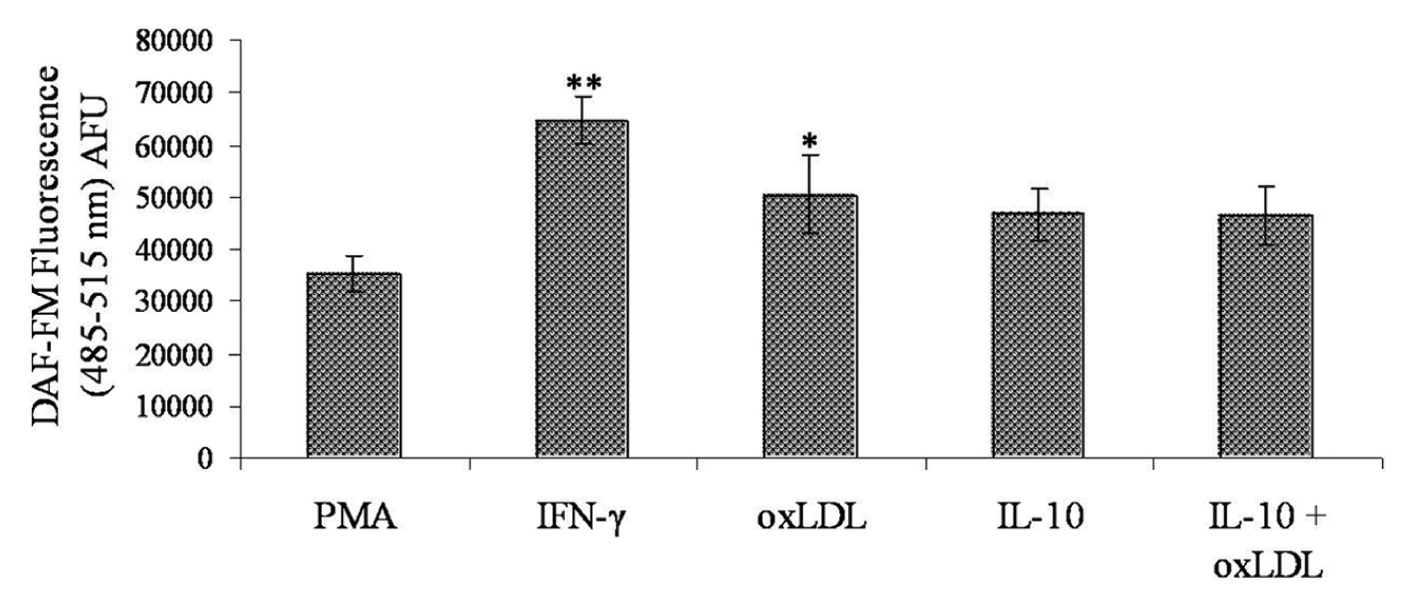
IFN-γ induction was used as a positive control for NO generation. All values were background corrected and determined against PMA baseline. IL-10 with/without oxLDL showed no significant change in NO generation. All data are represented as mean ± SD of four experimental repeats. Comparison of means was carried out by one-way analysis of variance followed by Dunnett’s multiple comparison; *p < 0.05.
Effect of IL-10 and oxLDL on OLR1 gene (gene for LOX-1) transcripts
Recent reports have shown the association of OLR1 gene variants in myocardial infarction. 20 Figure 6(a) depicts the three spliced variants, TRV 1, TRV 2 and TRV 3, at transcriptional (mRNA) and protein level in THP-1 macrophages. The nucleotide sequence determination of the component bands (TRV 1 and TRV 3 were sequenced) also matched with their distinct identities (data not shown). TRV 3 (lacking exon 5) has been shown to be the ‘non-risk’ haplotype of LOX-1 gene and named as LOXIN by Mango et al. 20 It is also evident from our experiments that the presence of IL-10 with atherogenic oxLDL increased the expression of the atheroprotective TRV 3 (LOXIN) in THP-1 macrophages (Figure 6(b)), hence reducing the LOXTRV 1/LOXTRV 3 ratio in the presence of IL-10 alone or in combination with oxLDL.
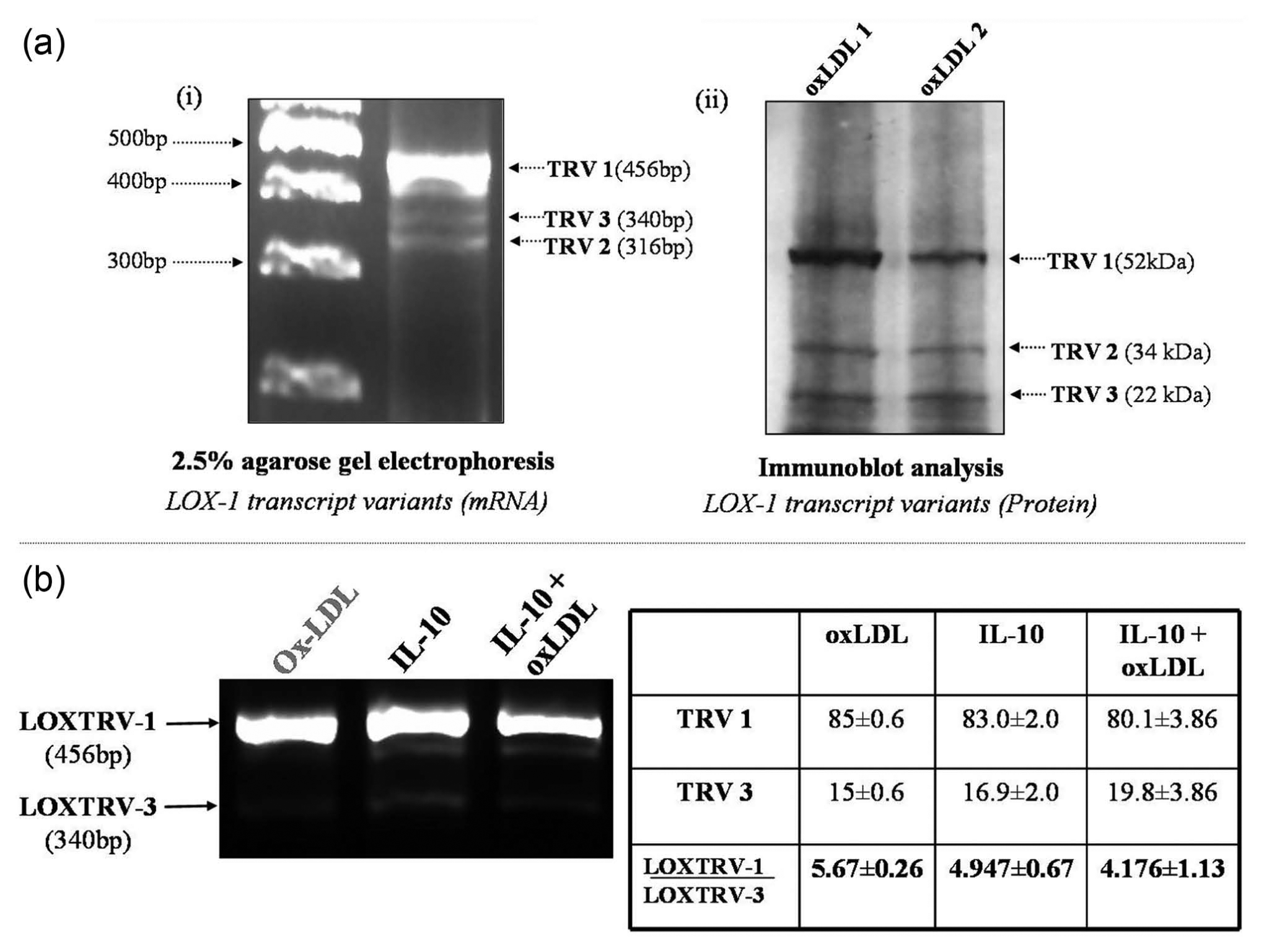
Expression of LOX-1 transcript variants. (a-i) Expression of LOX-1 transcript variants with LOXTRV primers. As shown in the image, the TRV 1 (all six exons) or the mature transcript was very highly expressed in response to oxLDL, while TRV 2 (exon 4 lacking) and TRV 3 (LOXIN, exon 5 lacking) were both minimally expressed. (a-ii) Depiction of (a-i) by immunoblot analysis using antibody against the C-terminal lectin-like domain present in all three variants. Lanes 1 and 2 represent treated specimens from two different batches of oxLDL preparations, namely, oxLDL 1 and oxLDL 2. (b) Agarose gel electrophoresis of PCR-amplified product using LOXTRV (LOX-1 transcript variant primers). Cells were incubated either with oxLDL/5 h or IL-10/24 h or IL-10 pretreatment for 19 h followed by the addition of oxLDL for 5 h. Three experimental repeats were carried out and the ratio of LOXTRV-1/LOXTRV-3 was determined densitometrically (as shown in the table). As compared to oxLDL, the LOXTRV-3 expression was increased in response to IL-10 alone. Combination of oxLDL and IL-10 further enhanced LOXTRV-3 expression.
Discussion
IL-10 has been thought to act via anti-apoptotic pathways to induce plausible protective mechanisms in the atherogenic arena. 11 LOX-1, a receptor for oxLDL, has been shown to be stimulated by pro-inflammatory signals, but remains under-evaluated for its role in response to anti-inflammatory stimuli. We used 100 nM PMA treatment for 24 h in our study to differentiate THP-1 monocyte into macrophages, as PMA is known to induce a basal LOX-1 expression in macrophages. These expressed LOX-1 receptors actively react with the moderately oxLDL used in our study, without producing any significant cell death.
Only a partial antagonistic effect of 100 nM of PMA on cellular expression of IL-10 was apparent during our study (Figure S2). Incubation with oxLDL (20 µg/mL for 5 h) was found to be most suitable for maximal cell surface expression of LOX-1 protein, comparable to that induced by 5 ng/mL of IL-10 (Figures 1 and 2). Although both oxLDL (20 µg/mL) and IL-10 (5 ng/mL) induced LOX-1 expression to a similar extent, IL-10 interestingly decreased cell surface expression of LOX-1 protein in the presence of oxLDL (Figure 2(b)). In fact, IL-10 at a concentration of 5 ng/mL in the culture medium was found to be atheroprotective by enhancing cellular uptake of oxLDL (Figure 3(a)). It is also apparent from Figure 3(b) that in the presence of this concentration of IL-10, a significant increase in uptake was maintained at all concentrations of oxLDL. Furthermore, the IL-10-induced cellular uptake of oxLDL was found to be LOX-1 mediated as evident from confocal microscopy using two fluorescent labels, fluorescein-linked anti-LOX-1 antibody (green) and Dil-oxLDL (red) (Figure 3(d)). The resulting yellow colour demonstrates the interaction of oxLDL with LOX-1. Its existence in cell cytoplasm (Figure 3(c)) provided evidence for cellular uptake of oxLDL.
oxLDL demonstrated its atherogenicity by inhibiting iNOS transcription (Figure 4(b)), which should have ideally reduced intracellular NO levels in macrophages. However, paradoxically, there was an increase in intracellular NO concentration (Figure 5), which serves as a substrate for pro-inflammatory peroxynitrite (ONOO−). 21 This was interesting, since one of the chief ROS known to be induced in endothelial cells is NO and induction of eNOS activity in endothelial cells by oxLDL has already been demonstrated. 22 However, in our study, unexpectedly, the predominant iNOS mRNA in macrophages was notably inhibited by oxLDL. This, along with the increase in NO radical itself suggested the induction of an NO source independent of iNOS in the presence of oxLDL. Previous studies have demonstrated the presence of trace amounts of eNOS/cNOS mRNA 19 in macrophages. So, our results suggests that when LOX-1 signalling takes priority, oxLDL probably induces eNOS in macrophages as well, a phenomenon previously reported only in endothelial cells.
Recently, it has been shown that LOXIN, a TRV (TRV3, 22 kDa) of LOX-1, reduced the risk of myocardial infarction, 20 while the other variants of LOX-1 gene, namely, TRV1 and TRV2 did not. 23 Since IL-10 has so far demonstrated an atheroprotective response, we wanted to evaluate the status of the atheroprotective LOXIN in response to IL-10 treatment. We found that IL-10 increased the expression of LOXIN and hence reduced the ratio of TRV1/TRV3 in THP-1 macrophages (Figure 6).
Overall, our study shows that oxLDL exhibits its atherogenicity by increasing intracellular NO concentration, a substrate for pro-inflammatory ‘peroxynitrite (ONOO−)’. Earlier reports have shown that the intracellular increase in ONOO− (reaction product of NO and superoxide anion) is mediated by the interaction of oxLDL with LOX-1.4,24 Since oxLDL inhibited iNOS in our study, the results suggest an alternative source for NO generation by oxLDL. The probable effecter here could be the constitutive/endothelial NOS (cNOS), which has been reported to be present in macrophages of physiological origin as well as in cell line–derived macrophages. 19 Alternatively, IL-10 is atheroprotective by committing LOX-1 receptor towards scavenging of extracellular oxLDL, rather than signalling.
While the maintenance of cell surface expression of LOX-1 is probably taken as a common responsibility by both oxLDL and IL-10, the differential regulation of receptor functionality (signalling/uptake) was evident. Interaction of oxLDL with LOX-1 induced downstream pro-inflammatory signalling cascade, whereas IL-10 reduced the atherogenic profile of the extracellular environment by facilitating clearance of accumulating oxLDL particles via LOX-1-mediated cellular uptake (probably followed by lysosomal digestion). Hence, IL-10 behaves as an athero-protectant by suppressing oxLDL-triggered LOX-1 signalling. In addition, IL-10 also promotes expression of the non-risk LOX-1 splice variant ‘LOXIN (or TRV3)’, an atheroprotective competent for the atherogenic, namely, TRV1 and TRV2 LOX-1 variants.
Footnotes
Conflict of interest statement
None to declare.
Funding
This research was funded by Department of Biotechnology, New Delhi, Government of India (BT/PR7678/MED/14/1061/2006), and A.A. received research fellowship from CSIR, New Delhi, Government of India (9/6(0408)2010).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
