Abstract
Glucose in the glomerular ultrafiltrate is actively reabsorbed by sodium glucose transporters (SGLT) in the proximal tubule. The SGLT2 protein is a high capacity molecule responsible for the majority of glucose reuptake with pharmacological inhibition, resulting in the loss of about 80g of glucose in the urine each day. About a dozen inhibitors of SGLT2 have entered clinical development, and the first has recently been submitted for registration with the United States Food and Drug Administration. The rationale for the clinical evaluation of these agents is their beneficial effects on glycaemia, blood pressure and body weight. No adequately powered trial has yet determined the effects of an SGLT2 inhibitor on either macrovascular or microvascular outcomes, although a number of large-scale trials are now ongoing. Evidence that will define the overall balance of benefits and risks of this new drug class is anticipated within the next 5 years.
Background
Phlorizin was isolated in 1835 from the bark of apple trees by French chemists. 1 Bitter in flavour, it was tested as a treatment for fevers, infectious diseases and malaria. It was subsequently discovered that high doses of phlorizin caused glycosuria, and that chronic administration in dogs produced signs such as polydipsia and polyuria, typical of those observed in humans with diabetes. 2 In the 1970s the location of the active-transport system responsible for glucose reabsorption was identified as the kidney tubule and there was a resurgence of interest in phlorizin as a possible mechanism for glucose control. Two renal sodium glucose transporters were subsequently identified: SGLT1, a low capacity, high affinity transporter located in the loop of Henle and the distal tubule, and SGLT2, a high capacity, low affinity transporter found in the proximal tubule. 3 Animal studies confirmed the effects of phlorizin on these transporters 4 but clinical development stalled due to poor absorption and unacceptable gastro-intestinal side effects, 1 the latter being a consequence of inhibition of the actions of SGLT1 proteins present in the small intestine. Further support for the targeting of SGLT2 was provided by studies of the clinical course of a rare, inherited deficiency of SGLT2 which results in affected individuals excreting about 100g of urinary glucose per day. The observation that this genetically determined renal glycosuria produced no significant short- or long-term adverse effects, served to further enhance interest in the clinical potential of compounds inhibiting SGLT2. 5
The first selective inhibitor of SGLT2, T-1095, was developed by Tanabe Seiyaku and Johnson & Johnson. 6 Administration in diabetic animal models increased urinary glucose excretion and reduced hyperglycaemia 7 but T-1095, like other early inhibitors, had a short half-life, poor bioavailability and did not proceed to clinical development. 8 Work on these compounds persisted, however, because there was a strong patho-physiological rationale for anticipating clinical benefit. In patients with diabetes, elevated blood glucose levels mean that the glucose load filtered by the glomerulus is increased, with altered renal glucose handling. 9 The observed increase in maximum tubular glucose transport is attributed primarily to up-regulation of SGLT2, 10 with the expectation that inhibitors of SGLT2 should induce a greater level of urinary glucose excretion in patients with diabetes compared with individuals with normal glucose metabolism. 11 The agents were also postulated to deliver other benefits consequent upon the effects of the mild osmotic diuresis (blood pressure lowering) and the substantive caloric loss through the glycosuria (weight reduction). The first SGLT2 inhibitor was recently submitted to the United States Food and Drug Administration (US FDA) for marketing approval 12 and there are a number of other compounds under investigation in large-scale research programs.
SGLT2 inhibitors under development
In an effort to systematically identify all SGLT2 inhibitors that have entered clinical development, we completed an electronic search of Medline, The Cochrane Central Register of Controlled Trials (CENTRAL) and clinicaltrials.gov, supplemented by a hand search of recent endocrine conference abstracts and a review of pharmaceutical company websites. The search was completed in July 2011 using the term ‘sodium-glucose transporter 2’ in addition to keyword and text word searches of all known compounds inhibiting SGLT2. The search identified a series of orally active SGLT2 inhibitors at various stages of clinical development (Table 1). The three compounds with the most advanced programs were dapagliflozin, canagliflozin and empagliflozin with all having substantial selectivity for SGLT2 over SGLT1.13,14
SGLT2 inhibitors and latest phase of development.

The search also revealed a large number of completed and ongoing trials of SGLT2 inhibitors being done with the goal of defining the effects of the compounds on intermediate cardiovascular markers and cardiovascular safety. 15 Published clinical trial data are available for dapagliflozin, canagliflozin and empagliflozin, although the majority of publicly available data are for the former and are contained within the registration documents submitted to the US FDA. 12 Completed studies have evaluated the effects of SGLT2 inhibitors as initial mono-therapy, initial combination therapy and as add-on therapy to a range of different agents (metformin, sulfonylureas, insulin, thiazolidinediones and dipeptidyl peptidase-4 inhibitors) as well as examining the effects of the compounds in special populations such as those with renal impairment or established cardiovascular disease.
Effects of SGLT2 inhibitors on clinical measures
Glycaemia endpoints
Completed trials of SGLT2 inhibitors have evaluated effects on a range of different measures of glucose control including the change in glycated haemoglobin (HbA1c), fasting and postprandial glucose and the proportion of patients achieving targets. Benefits have been observed for both fasting and postprandial glucose measures, as well as for HbA1c. For dapagliflozin, the fall in fasting glucose varied with dose, ranging from about 0.8mmol/L with 2.5mg dapagliflozin to between 1.2 and 4.7mmol/L with 10mg dapagliflozin. 12 Reductions in blood glucose were also achieved when dapagliflozin was used in combination with other oral hypoglycaemic agents and insulin. Mean percentage decreases in HbA1c were 0.6% with a dapagliflozin dose of 2.5mg, and between 0.8% and 2.7% with a 10mg dose. Canagliflozin has been reported to produce reductions in fasting glucose ranging from 0.9 to 2.1mmol/L with daily doses between 50mg and 600mg. The corresponding reduction in HbA1c was 0.9% at a dose of 300mg daily.16–18 Empagliflozin, the other SGLT2 inhibitor for which a substantial volume of data are available, also showed significant decreases in fasting glucose when used alone 19 or in combination with metformin, 20 with reported reductions in blood glucose of 1.7mmol/L and HbA1c of 0.63% at a daily dose of 100mg. 19 As would be anticipated, significantly greater reductions in HbA1c were achieved in patients with higher starting HbA1c levels.12,21,22
The risk of hypoglycaemia with SGLT2 inhibitors appears to be low and much lower than comparison treatment with a sulphonylurea. 12 Relatively lower rates of hypoglycaemia might be expected because the agents have a mode of action independent of insulin, produce only moderate reductions in blood glucose, and the urinary excretion of glucose is determined in part by blood glucose concentrations themselves, providing for a natural brake on the amount of glucose lost. 23
Body weight
Patients receiving SGLT2 inhibitors consistently experience weight reduction. Early decreases in weight may represent fluid lost due to the osmotic diuretic effect of the agents, 24 whereas incremental weight loss over subsequent weeks is likely due to caloric loss. The glucose excreted in the urine as a consequence of SGLT2 inhibition equates to about 200–300 calories each day. 24 Weight loss of 2–3kg has been demonstrated in 12 week trials of dapagliflozin, 24 canagliflozin 17 and empagliflozin, 19 as well as in a 52 week trial of dapagliflozin when used as an add-on to metformin therapy. 25 A trial of dapagliflozin has also demonstrated a significant reduction in waist circumference which would be consistent with a reduction in fat mass. 21 Data from longer-term trials demonstrate sustained weight loss with dapagliflozin as add-on to metformin therapy 26 and also when compared with glipizide 27 for up to 2 years. Further longer-term clinical trials of SGLT2 inhibitors, with body composition assessments, are required to fully understand the weight loss changes and to define their durability. However, initial studies using magnetic resonance imaging and dual energy X-ray absorbtiometry (DEXA) scanning suggest that dapagliflozin reduces both total body fat and abdominal fat. 12
Blood pressure
Another favourable effect of SGLT2 inhibitors on the determinants of cardiovascular risk is a fall in blood pressure. Chronic osmotic diuresis caused by glycosuria would be anticipated to reduce blood pressure, and dose-related increases in 24 hour urinary volumes of between 107 and 470mL have been reported.23,24 Reductions in systolic blood pressure of up to 5mmHg have been described in trials of dapagliflozin both when used as add-on therapy 28 or when used alone. 24 Canagliflozin has similarly been shown to significantly reduce systolic blood pressure. 29 Changes in diastolic blood pressure have been smaller and inconsistently reported by the individual trials, probably reflecting the lesser statistical power to detect effects on diastolic measures. 23 The blood pressure reductions achieved are comparable to those seen with the administration of established blood pressure lowering agents in high-risk patient groups and would be anticipated to afford significant vascular protection, particularly against the risks of stroke and heart failure. There has been no large excess in the rates of hypotension or syncope in the individual trials reported to date,21,22,24 although some excess is apparent in the combined datasets. 12
Blood lipids
To date, no SGLT2 inhibitor has demonstrated a substantive effect on blood lipid levels. Small increases in HDL cholesterol (1.8–4.4% dapagliflozin vs. 0.4% placebo) and small reductions in triglycerides (–2.4% to –6.2% dapagliflozin vs. 2.1% placebo) 12 are probably attributable to the weight reduction achieved with the compound. 21
Renal function
Increases in blood urea nitrogen have been identified 21 but there is no evidence that SGLT2 inhibitors cause serious complications attributable to this effect. Likewise, transient abnormalities of serum sodium and potassium, in some studies 21 but not others, 22 and elevations in serum creatinine concentration 24 have not been associated with significant clinical complications. 22 Small reductions in estimated glomerular filtration occur shortly after commencement of therapy, with measures returning to normal after a few weeks. Effects on urinary protein remain to be established by ongoing studies. Dapagliflozin was observed to be significantly less effective at reducing HbA1c in patients with significant renal impairment which might be anticipated on the basis of the renal mechanism by which the compound has its glucose lowering effect. 12 The longer-term effects of reduced glucose reabsorbtion in the kidney, and particularly the renal tubule, remain to be established but may be important given the central role of the tubules in progressive loss of kidney function.
Major vascular outcomes
The currently limited clinical trial experience with SGLT2 inhibitors means that there are few data to precisely or reliably describe the effects of these agents on stroke, heart attack and other serious vascular complications. The data for dapagliflozin submitted to the US FDA are the most comprehensive available and reported a hazard ratio of 0.67 (95% confidence interval 0.42–1.08) for a composite endpoint comprised of vascular death, non-fatal stroke, non-fatal myocardial infarction and hospitalised angina. 12 A series of large ongoing trials (Table 2) will accrue a significant number of cardiovascular safety outcomes for canagliflozin and empagliflozin over the next few years. Trials powered to detect possible cardiovascular protective effects are also underway with the canagliflozin cardiovascular assessment study (CANVAS) having recruited 4330 patients to phase I of the study, with a possible phase II that would significantly expand the trial population. Likewise, a large trial of empagliflozin is currently recruiting towards a planned enrolment of 12,500, with the goal of testing whether the compound protects against the risk of serious cardiovascular complications.
Ongoing and completed phase III trials of SGLT2 inhibitors with n>1000.*
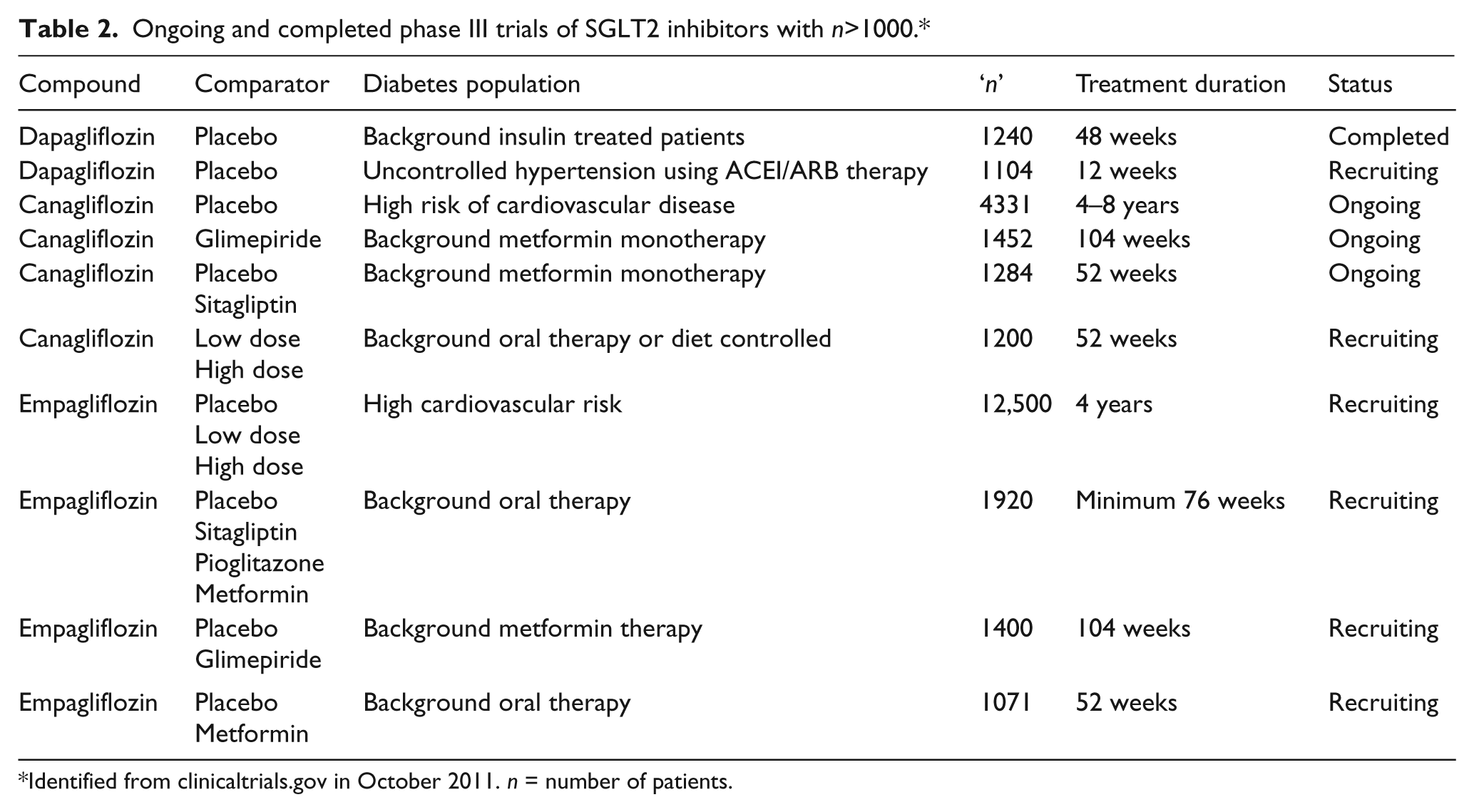
Identified from clinicaltrials.gov in October 2011. n = number of patients.
Adverse effects and safety
The key side effect associated with inhibition of SGLT2 appears to be non-sexually transmitted, predominantly mycotic, genito-urinary infection attributed to the elevated glucose levels in the urine. The rates of adverse genito-urinary events rise with the duration of follow-up such that an excess of between 4 and 8% has been reported in actively treated patients compared with the placebo. 12 In women this is mostly mycotic vulvo-vaginitis and in men mostly mycotic balanitis. Rates of balanitis in men are several-fold less than rates of vulvo-vaginitis in women, and in both sexes infections are usually resolved by standard therapies. Urinary tract infections are less common but there is a clear excess in comparison to the placebo with infective organisms typical of those seen in the general population. 12
The diuresis associated with inhibition of the SGLT2 transporter leads to small mean elevations in hematocrit, 23 but clinically significant abnormalities have not been detected in trials completed to date, 21 and there has been no reported excess of venous thrombo-embolic events. However, the limited total exposure data available, to date, means that the effects on most body systems have not reliably been established. The US FDA in its review of dapagliflozin noted a non-significant numerical excess in breast and bladder cancers, with the latter potentially due to an ascertainment bias related to the treatment of drug-related urinary symptoms. 12 The full safety profile of these agents remains to be determined in ongoing trials, although the mostly uncomplicated course of patients with the rare genetic deficiency of SGLT2 30 provides some reassurance about the probable safety of chronic elevation of urinary glucose through inhibition of SGLT2.
Discussion
The modulation of renal glucose handling through the inhibition of SGLT2 appears to have significant potential as a strategy for the prevention of vascular complications in patients with diabetes. While recent trials of other glucose lowering regimens have failed to show definitive protective effects for macrovascular outcomes, there have been clear benefits for microvascular complications. With SGLT2 inhibitors significantly improving blood pressure and weight as well as glycaemic indicators, there is a strong rationale for expecting that these compounds will protect not only against microvascular complications but also against macrovascular disease. Furthermore, because the action of SGLT2 inhibitors depends neither on the secretion of insulin nor on the sensitivity of tissues to the effects of insulin, 23 the agent should be active across the spectrum of diabetes, from newly diagnosed to long-standing disease, including extremes of insulin resistance and β-cell dysfunction, as well as in type 1 diabetes.23,31
However, while the rationale for beneficial effects of SGLT2 inhibitors is strong, definitive evidence that precisely and reliably defines the effects of these agents on vascular events, along with a clear understanding of their safety profile, will be a prerequisite for their widespread uptake in clinical practice. Recent experience with drugs for the management of diabetes has served to highlight the importance of such data early in the development program because only with this information can an adequate and timely understanding of the overall balance of benefits and risks be arrived at. The withdrawal of rosiglitazone after more than a decade of widespread use because of probable adverse effects on vascular events, 32 and more recent concerns about possible carcinogenic effects of pioglitazone, 33 have highlighted the shortcomings of registration programs based solely upon intermediate indicators.
In the United States, as a direct consequence of these challenges, the FDA has provided guidance on the evidence required for the initial and ongoing registration of new compounds for the management of diabetes, and other agencies are taking similar approaches. 34 The guidance makes specific recommendations about the type of safety data needed at different stages of program development and is more demanding than existing requirements. In particular, the guidance recommends that companies exclude a major adverse effect of the agent on vascular outcomes prior to registration by demonstrating that it is not likely to elevate vascular risk by 80% or more. This is a requirement met by dapagliflozin, the first agent submitted for regulatory review, and this provides some reassurance for other compounds in development. Subsequently, a hazard of 30% or greater must be excluded for maintenance of registration. These levels of reassurance are entirely arbitrary and these requirements will not assure the safety or efficacy of compounds entering the market. However, in conjunction with post-marketing surveillance programs, compounds with large adverse effects should now be identified earlier than they might otherwise have been.
The magnitude of the vascular protection that might be afforded by SGLT2 inhibitors can be projected on the basis of current knowledge about the effects on intermediate outcomes and their associations with vascular disease. For example, a 0.8% reduction in HbA1c might, on the basis of meta-analyses of randomised trials, be anticipated to reduce coronary risk by about 8%. 35 Likewise, a 4mmHg reduction in systolic blood pressure would provide at least the same level of protection. 36 Jointly, these effects would be expected to reduce vascular risk by about 15%, and this might be a reasonable effect size upon which to estimate the sample size for trials of SGLT2 inhibitors designed to explore possible protective effects on vascular outcomes. Cardiovascular benefits associated with weight reduction are likely to act mostly through changes to blood pressure and glucose levels, although it is possible that other independent mechanisms might add to the protection provided.
Against the potential cardiovascular benefits of SGLT2 inhibitors it will be necessary to balance the adverse effects. Mycotic genito-urinary infection appears to be the primary side effect with clearly elevated levels of risk in both men and women. 12 If vascular protection were proved, then infective complications that are mostly superficial in nature and controlled with standard therapies would probably be a tolerable downside of treatment. If vascular protection is not demonstrated then the balance of benefit and risk might be perceived quite differently. Reported imbalances in selected cancer outcomes observed within the development program for dapagliflozin were based on a very small numbers of events, 12 and while an ongoing focus of attention, require the collation of much more data over a much longer time period before meaningful conclusions can be drawn.
The exploration of SGLT2 inhibitors as another strategy for the management of diabetes reflects the many and diverse patho-physiological processes associated with the condition. While there are already multiple pharmacological compounds available to treat the different stages of the disease, 37 control of hyperglycaemia remains suboptimal for most and premature death and disability ensues for many. 38 Furthermore, progressive decline in β-cell dysfunction usually leads to intensification of therapy 39 with greater risks of side effects such as hypoglycaemia, weight gain, gastrointestinal disturbances and peripheral oedema. 40 The need for novel agents, with new modes of action therefore remains a clinical and public health priority and a focus of efforts in the field of drug discovery. With the majority of patients with type 2 diabetes being overweight or obese, 41 and with excess weight being a major contributor to the development of insulin resistance and impaired glucose tolerance, 42 the beneficial effects of SGLT2 inhibitors on weight will be significantly to their advantage. Weight loss is difficult to achieve in this patient population with mainstays of therapy such as sulfonylureas and insulin both associated with weight gain.43,44
The magnitude of the global diabetes problem is the primary driver behind these efforts to identify new treatment modalities. With some 285 million people worldwide having diabetes and this figure anticipated to rise to 440 million by 2030, 45 diabetes is a leading contributor to premature death in most high-income countries and a fast-growing epidemic in low- and middle-income regions. 45 Against this background, there is an urgent need for new treatments that can ameliorate the adverse health problems caused. Clear evidence from definitive studies of SGLT2 inhibitors that precisely and reliably quantify the effects of these agents on major health risks will be a significant step forward.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
