Abstract
Introduction
Fluctuating hyperglycaemia has been suggested to be an independent risk factor for the development of macrovascular complications in diabetes. Increased oxidative stress resulting from an overproduction of reactive oxygen species (ROS) has been suggested as the underlying mechanism.1,2 Recently, reports have shown poor regulation of several glycolytic genes and transport mechanism during fluctuating compared to sustained hyperglycaemia,3,4 and this lack of adaptability may cause oxidative stress. However, the mechanisms by which glucose fluctuations may impair macrovascular function remain unclear and call for better defined animal models. This study examined the effect of fluctuating versus sustained high or low levels of glucose on markers of systemic oxidative stress and hepatic glucose metabolism in catheterised rats.
Materials and Methods
Thirty male Sprague–Dawley rats (400–430 g; Taconic, Ejby, Denmark), were housed under controlled conditions (temperature, 22 ± 2°C; humidity, 50 ± 20%; light, 12 h) with water and chow (Altromin GmBH, Lage, Germany) ad libitum. Animals were catheterised (S-50-HL; Tygon, Saint Gobain, UK) in the jugular vein for infusion and in the carotid artery for blood sampling and treated with Baytril (Bayer AG, Leverkusen, Germany) and Rimadyl (Pfizer A/S, Ballerup, Denmark) for 3 days post-surgery. The animals were randomised into four groups (n = 7–8) and were connected to an Accusampler (DiLab, Lund, Sweden) for automated blood sampling and glucose infusion (50% glucose; Fresenius Kabi AG, Bad Homburg, Germany) and received either a continuously high (CHG; 4.0 ml/kg/h), continuously low (CLG; 1.2 ml/kg/h) or pulsating (FLU; 0–2 min, 30 ml/kg/h; 2–30 min, 4.0 ml/kg/h, i.e. nine daily infusions separated by 2.5 h) glucose infusion for 72 h. Controls received a constant saline infusion (VEH, 1.2 ml/kg/h). Blood was collected in K3-EDTA coated tubes and centrifuged (4°C, 5000 rpm, 3 min). Plasma malondialdehyde (MDA) and ascorbate oxidation ratio (AOR, i.e. the % of dehydroascorbic acid of total ascorbate) were measured daily as biomarkers of oxidative stress. Plasma protein was used to correct for haemodilution. Plasma glucose and insulin were analysed throughout the study covering the entire circadian rhythm. After 72 h, animals were anaesthetised with isofluran and liver was excised and preserved in liquid nitrogen. Animals were euthanised by intra-cardiac injection of pentobarbital (100 µl; 200 mg/ml). Plasma glucose was analysed by glucose oxidase methodology (EBIO Plus; Eppendorf AG, Hamburg, Germany). Plasma insulin was analysed by ELISA (Chrystal Chemistry, Downers Grove, IL, USA). Plasma MDA and AOR were analysed as described previously.5,6 Liver triglycerides and glycogen were determined in homogenised tissue (sodium acetate 0.15 mol/l and 0.75% Triton X-100, pH = 4.9). Homogenate was boiled (2 min), cooled (0°C) and liver TG determined on the supernatant (Cat#11488872; Roche, Basel, Switzerland). For glycogen, liver homogenate was incubated (20°C/24 h) with amyloglycosidase before analysing total glucose.
Statistical analyses were carried out by SAS JMP software (version 8.1; SAS institute, Cary, NC, USA). Data were analysed by ANOVA with random effect using group and time as factors. In the case of significance, an appropriate post hoc test was performed.
Results
Baseline measurements were not different between groups. Hyperglycaemia (18 mmol/l) and hyperinsulinaemia (1500 pmol/l) were immediately manifested in the CHG group and maintained throughout the study. The FLU group showed regular oscillation of glucose (16–18 mmol/l) and insulin (1300 pmol/l) at peak level and returned to normal levels between pulses. The CLG group displayed slight increases in both glucose (8 mmol/l) and insulin (300 pmol/l) throughout the study.
Both plasma AOR and MDA were significantly affected by the intervention (Table 1). FLU had 50% higher plasma MDA over the entire time course compared to controls (p < 0.05) and 40% higher than CLG (p < 0.05). FLU showed a significant increases at time points 48 and 72 h (p < 0.05 vs. VEH, both cases) whereas CHG was only affected at 72 h (p < 0.05 vs. VEH). FLU also showed 50% increased plasma AOR vs. VEH (p < 0.05).
Biochemical measurements of oxidative stress (malondialdehyde and ascorbate oxidation ratio) in plasma and liver triglycerides/glycogen in rats subjected to various glucose infusion regimens
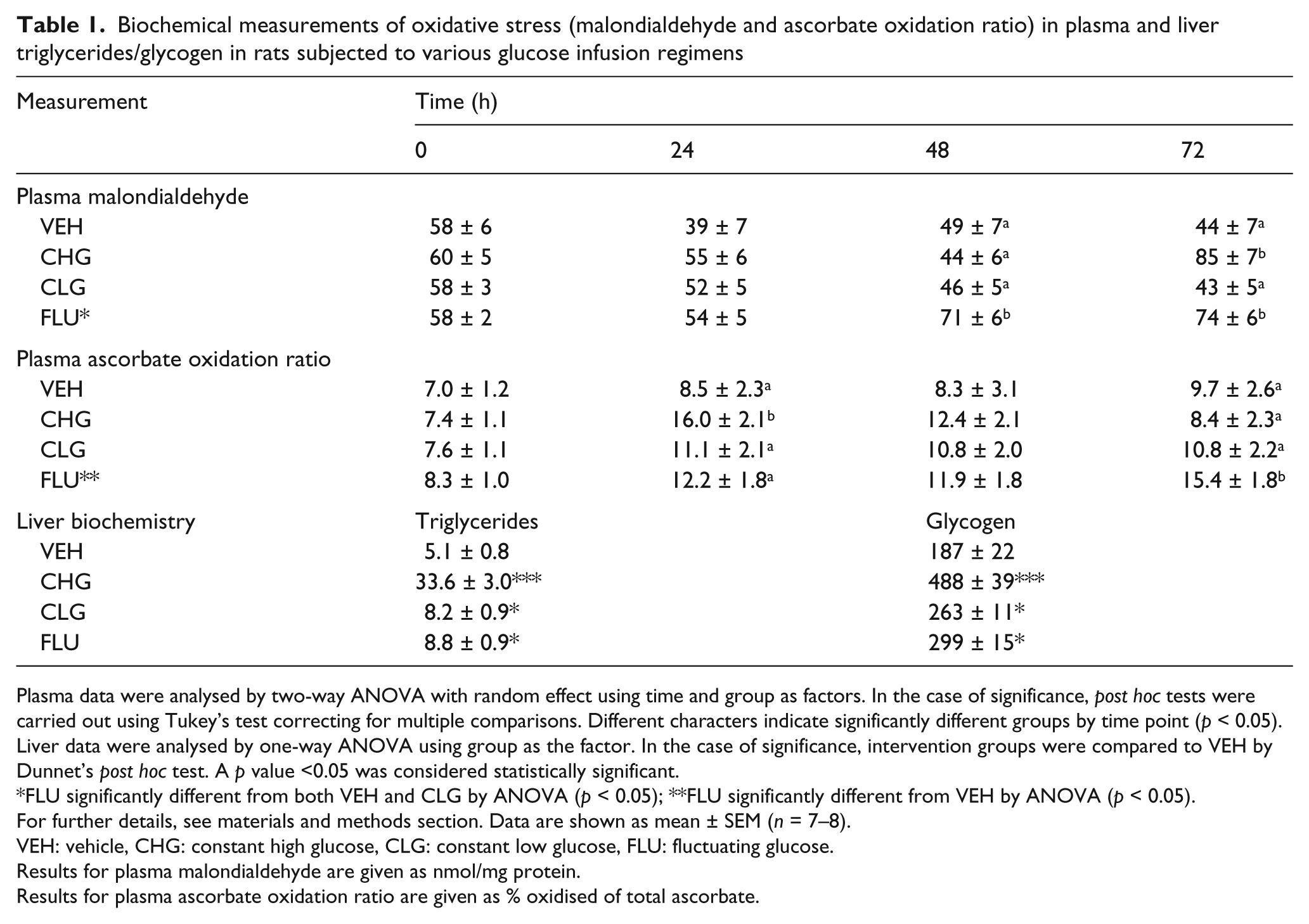
Plasma data were analysed by two-way ANOVA with random effect using time and group as factors. In the case of significance, post hoc tests were carried out using Tukey’s test correcting for multiple comparisons. Different characters indicate significantly different groups by time point (p < 0.05).
Liver data were analysed by one-way ANOVA using group as the factor. In the case of significance, intervention groups were compared to VEH by Dunnet’s post hoc test. A p value <0.05 was considered statistically significant.
FLU significantly different from both VEH and CLG by ANOVA (p < 0.05); **FLU significantly different from VEH by ANOVA (p < 0.05).
For further details, see materials and methods section. Data are shown as mean ± SEM (n = 7–8).
VEH: vehicle, CHG: constant high glucose, CLG: constant low glucose, FLU: fluctuating glucose.
Results for plasma malondialdehyde are given as nmol/mg protein.
Results for plasma ascorbate oxidation ratio are given as % oxidised of total ascorbate.
In liver (Table 1), CHG showed 2.5-fold increased GLY and 6-fold increased TG (p < 0.0001 vs. VEH; both cases), whereas FLU and CLG showed only moderate changes in GLY and TG (p < 0.05 vs. VEH; both cases).
Discussion
Oxidative damage to lipids is considered an important factor in the progression of macrovascular complications in diabetes. 7 Increased plasma MDA was found in both CHG and FLU. Notably, this effect was earlier and more pronounced in FLU than in CHG. Also, FLU had a higher AOR showing that the capacity to recycle ascorbate is overwhelmed by its oxidation rate, indicating increased oxidative stress. Oxidative stress has been shown to correlate with glycaemic exposure, 8 but we found this relationship to be more complex since FLU received only one third of the glucose amount given to CHG animals. Thus, oxidative stress apparently also depends on the glycaemic profile. In agreement, human and animal studies have previously shown fluctuating glucose to impair vascular function presumably due to increased oxidative stress.1,2
Other mechanisms independent of glycaemic exposure may cause increased oxidative stress. Cultured endothelial cells subjected to sustained hyperglycaemia down-regulated glucose transport protein 1 (GLUT 1) expression whereas short-term hyperglycaemia had no effect on GLUT 1 expression or activity. 4 This attenuated metabolic adaptability during repetitive hyperglycaemia may, hypothetically, leave the endothelium exposed to excess glucose influx resulting in oxidative stress. Moreover, a potential role of insulin in the lack of adaptability must be considered as well. A recent study, where rats were chronically infused with insulin, glucose or both found that insulin improved endothelial function through its hyperglycaemia reducing action. 9 Insulin increases expression and activity of GLUTs and lipogenic enzymes particularly in liver and thereby hepatic glucose uptake. 10 This action may exert an indirect protection of the vascular endothelium during sustained high-glucose exposure. In FLU animals, insulin protection may be absent due to the short-term hyperinsulinaemia observed there. However, additional studies evaluating oxidative stress and glucose disposition in insulin sensitive versus non-sensitive tissues during hyperinsulinaemia are needed to support this hypothesis.
Conclusion
Our study shows that compared to sustained hyperglycaemia, fluctuating glucose induces earlier signs of lipid oxidation and redox imbalance despite a lower total glycaemic exposure. This supports the hypothesis that poorly controlled hyperglycaemia in diabetic patients, regardless of mean glucose levels, may be an independent risk factor for the development of diabetic complications.
Footnotes
Acknowledgements
Annie B. Kristensen, Sarah Zouhtane, Annette Rosendal and Linda Buch-Rasmussen are thanked for their excellent technical assistance.
This study was supported, in part, by The Danish In Vivo Pharmacology PhD Program and Novo Nordisk A/S.
The authors declare there are no conflicts of interest.
