Abstract
The cured rubber-made bladder used for curing the green tire because, it is thermally stable, and has low gas permeability. The bladder is made with the cured butyl rubber as a base rubber under a phenolic curing system. To save the resources and also, the environmental protection, it should be de-crosslink to gum rubber. A commercial source waste bladder rubber compounded with different de-crosslinking agents, including TMTD, DPDS, DCBS, MBTS, MBT, and TBBS. To do de-crosslinking, the prepared compounds were heated in a laboratory heating oven at different temperatures. The de-crosslinking parameters including the sol fraction and de-crosslinking percent measured using traditional rubber swollen tests. The Fourier transform infrared spectroscopy (FTIR) graphs showed the existence of the phenolic resin in the structure of waste bladder butyl rubber powder. Efficient de-crosslinking was observed for all studied de-crosslinking agents. It evidenced by the scanning electron microscope micrographs and also, the measured crosslink densities of the reclaimed rubbers. The highest and the lowest de-crosslinking percents belonged to the compounds with 4 phr DPDS in recipe at 220

Introduction
Among traditional cured rubbers, butyl rubber (IIR) has enough importance in industrial and daily used rubber articles. 1 It shows excellent resistance against chemicals, moisture, and steam and gas permeability. Also, the phenolic-cured butyl rubber demonstrates outstanding heat stability at high temperatures. 2 The above-mentioned properties made the butyl rubber an ideal rubber for manufacturing the inner liner of the tubeless tyres. The butyl rubber was categorized as a synthetic rubber which was synthesized by Thomas and Sparks in 1937. Isoprene (cis- 2-methyl-1,3-butadiene) and isobutylene (2-methylpropene) are monomers for synthesizing the butyl rubber through cold cationic copolymerization. The polymerization carries out with the aid of a Lewis catalyst in an organic solvent. Amount of isoprene in the structure of the copolymer is low and varies between 1 to 3%. The isoprene makes the unsaturation centres for the crosslinking (curing) of the rubber. The uncured rubber has no industrial use. There are three main curing techniques, sulphur or vulcanization, phenolic or resin, and peroxide crosslinking. 3 The final property of a rubber article depends on its curing technique. 4 Typically, cured rubbers with the phenolic and peroxide techniques demonstrate outstanding heat resistance because of the formed stable C-C bonds between the rubber macromolecules after curing.
The thermally stable and low gas permeability are two distinguished properties of the phenolic cured IIR-made bladder. 5 This rubber article used for curing the tire. After assembling the several un-cured rubber-made parts of a tire (green tire), i.e., tread, carcass, bead, etc., the bladder put inside of the green tire and the whole assemble cures inside of a metal-made mold using high-pressure steam. The phenolic curing of rubber may accomplish by different techniques and materials, i.e., allyl phenol–formaldehyde novolac, bisoxazoline–phenolics, polybenzoxazines, phenol–epoxy systems, phenolic resins with phenyl maleimide functions, pendant phenol functional linear polymers, propargyl ether functional phenolics, and phenolic resins with phenyl ethynyl groups. 6
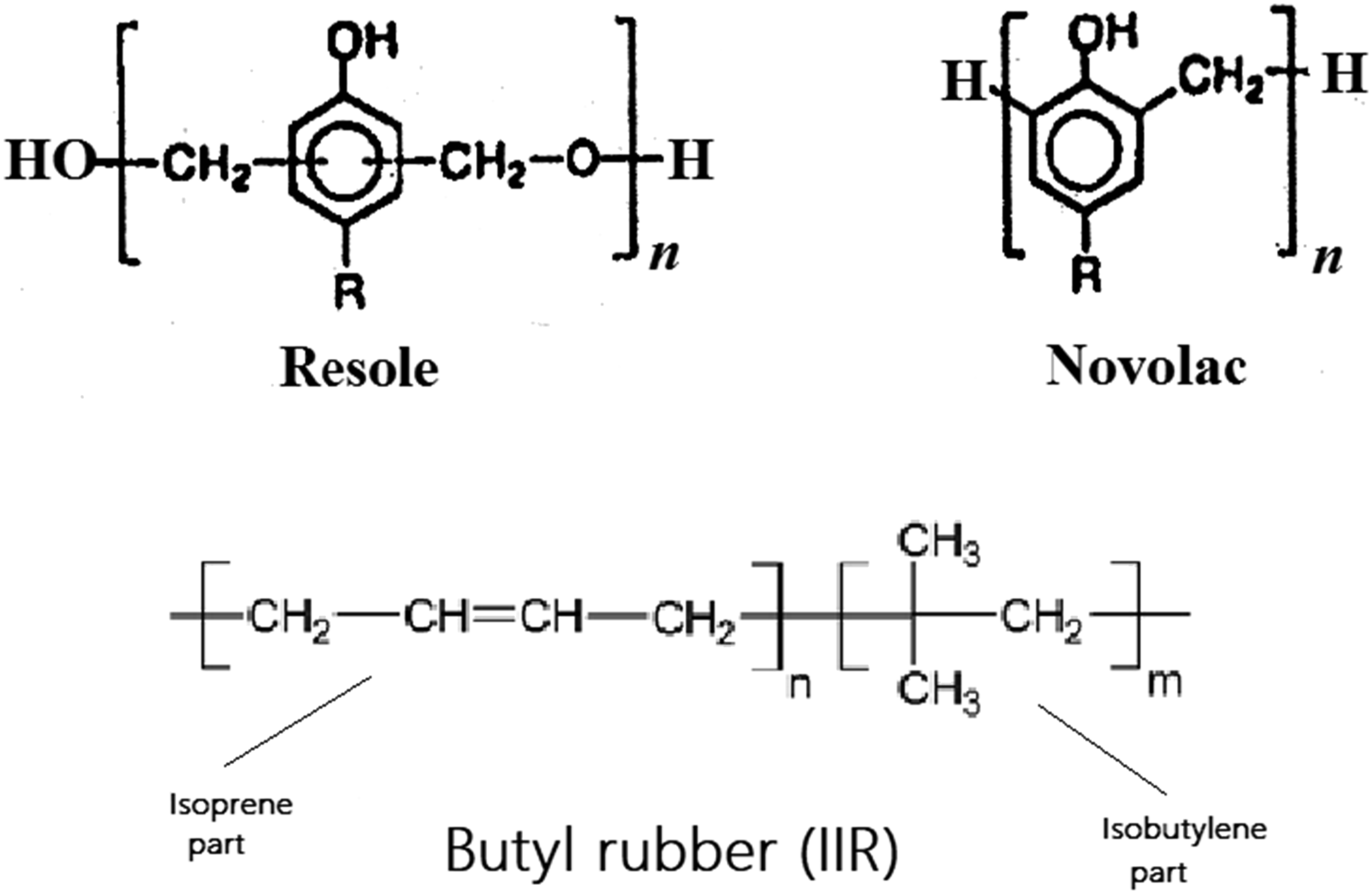
Scheme 1 shows the traditionally used phenolic (phenol-formaldehyde) resins for the cured rubber-made bladder. As observed, the methylene groups and di-methylene ether connect the phenolic units in Novolac and Resole, respectively. Also, Scheme 2 represents the known Chroman mechanism of curing.
7
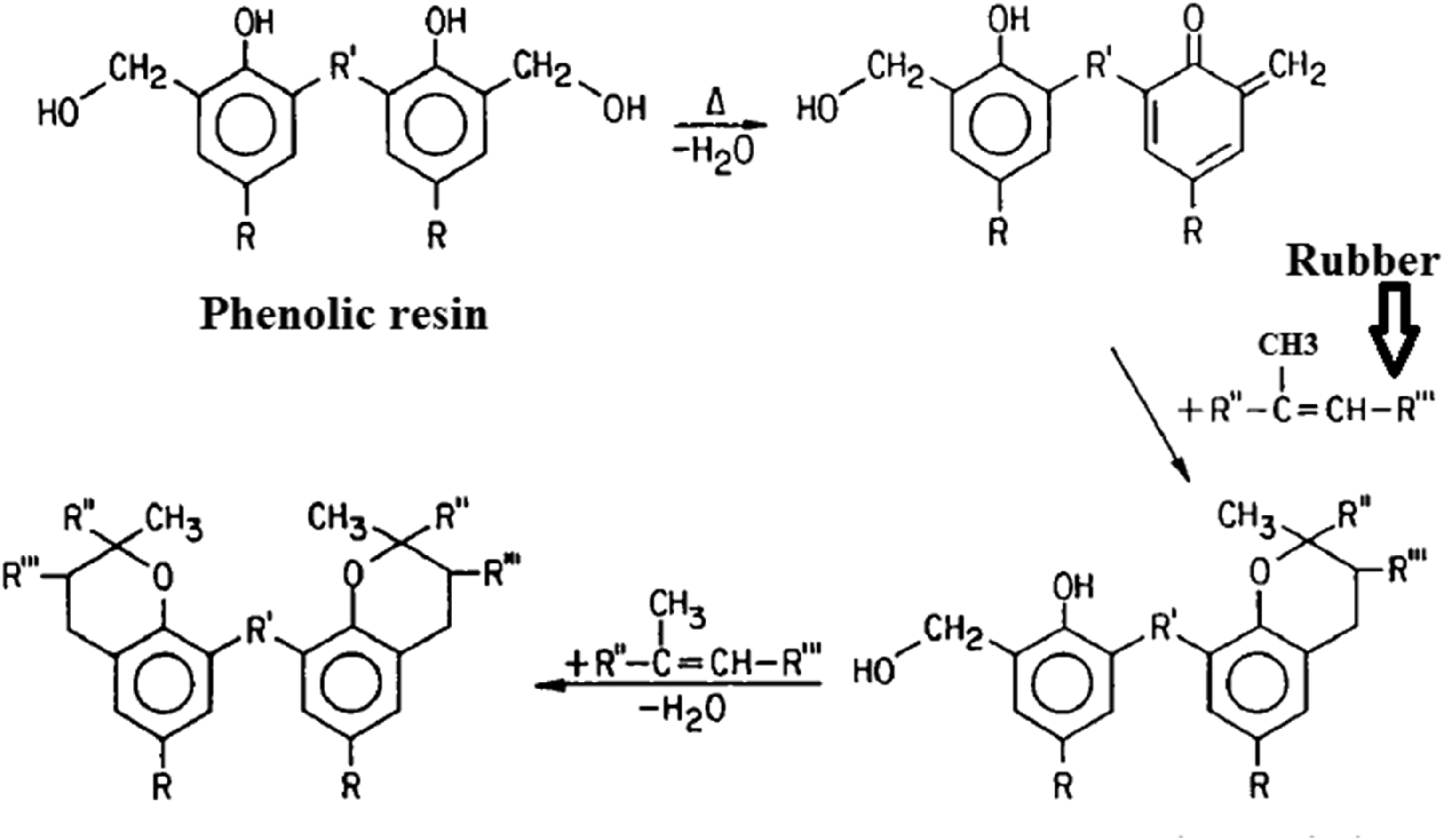
Based on the above-mentioned mechanism, a bridge made between phenolic resin and rubber macromolecules resulted in a three-dimensional network. However, alternative mechanism may involved which named the cationic reaction mechanism.
8
Here, the reactions proceed via a cationic mechanism with benzylic cations as reactive species, adding to the least sterically hindered olefinic C-atom. As observed, there is a distinguished difference between the phenolic curing with the other traditional rubber curing, sulphur and peroxide crosslinking. Here in phenolic curing, the unsaturated double bond in butyl rubber (isoprene part) attacks by the phenolic resin. Hence, the cured rubber has fewer double bonds when compared with the uncured rubber. Butyl rubber (IIR) and traditional phenolic (phenol-formaldehyde) resins, Resole and Novolac. Chroman mechanism of rubber curing by phenolic resin.
7
The same as all polymeric articles, the life service of the cured rubber-made bladder reaches to end, and it enters the waste stream. There are three distinguished reasons for recycling the mentioned article, protection of the environment, saving raw material resources, and earning money. The main strategies for the recycling of polymeric wastes are re-using, mechanical, chemical, and energy recovery which are reputed as primary, secondary, tertiary, and quarter, respectively. 9 In chemical recycling, the polymeric waste decomposes either to initially made monomers, and or light valuable hydrocarbons. The former and the latter may use for producing new polymers and the feedstuff of petrochemical plants. In this study, the chemical recycling refers to the break or de-crosslinking of the phenolic bridges between the butyl rubber macromolecules. The de-crosslink (reclaim) butyl rubber individually or in combination with virgin rubber may re-cure to produce new rubber-made articles.
To reach to a high-quality reclaim rubber, only the crosslinks should be broken during the de-crosslinking process.10,11 In practice, the scission of the main rubber chains takes place during the de-crosslinking process, which reduces the quality of the reclaimed rubber. Several techniques have already been introduced for cured rubber de-crosslinking,12,13 including microwave and ultrasonic irradiation, shear forcing (mechanical), thermal, and chemical. The latter may use individually or in combination with the left techniques. In chemical technique, the de-crosslinking takes place with the aid of a chemical, named de-crosslinking agent. However, the above-mentioned techniques have some advantages and also disadvantages. The de-crosslinking of the butyl rubber has already been done by the researchers using some of the mentioned techniques, i.e., Polvoy et al., 14 Karmanova, 15 Liliane et al., 16 Khavarnia et al., 1 Kuan Jiang et al., 17 Tao Zhang el al., 5 Vagizova et al., 18 and Feng et al.19,20 They reclaimed the sulphur (vulcanized) and peroxide-cured butyl rubber.
Based on the author’s best knowledge, there are few published papers for reclaiming the phenolic-cured butyl rubber. Zhang et al. 5 studied a mechanical technique using a twin screw extruder. The radiation technique was used by Scagliusi 21 and Karmanova 22 with the help of their colleagues. As observed, the techniques used and de-crosslinking agents were limited, and no one used a thermo-chemical technique. In this study, a combination of two techniques, thermal and chemical used to de-crosslink the waste bladder butyl rubber powder. The de-crosslinking process was carried out inside a heating oven as a thermal source using different de-crosslinking agents (chemicals) at designated temperatures.
Experimental
Materials: De-crosslinking agents and waste rubber powder
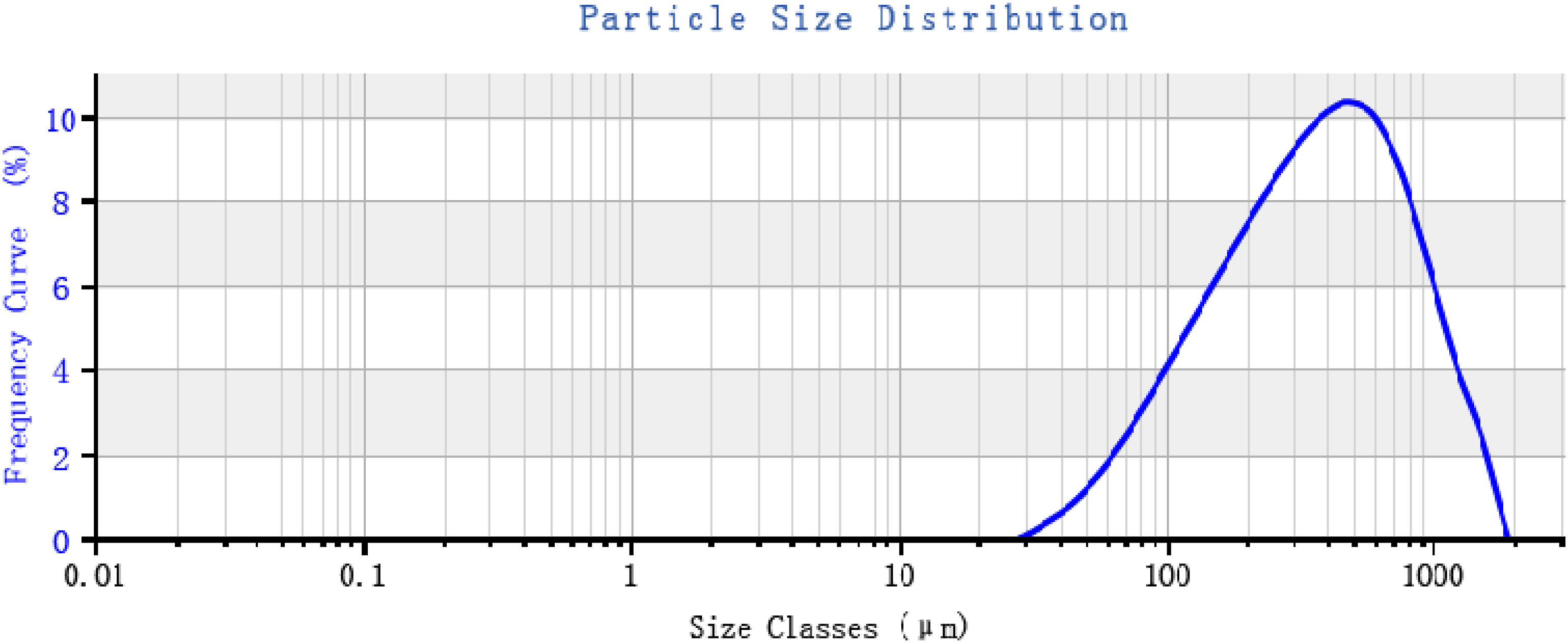
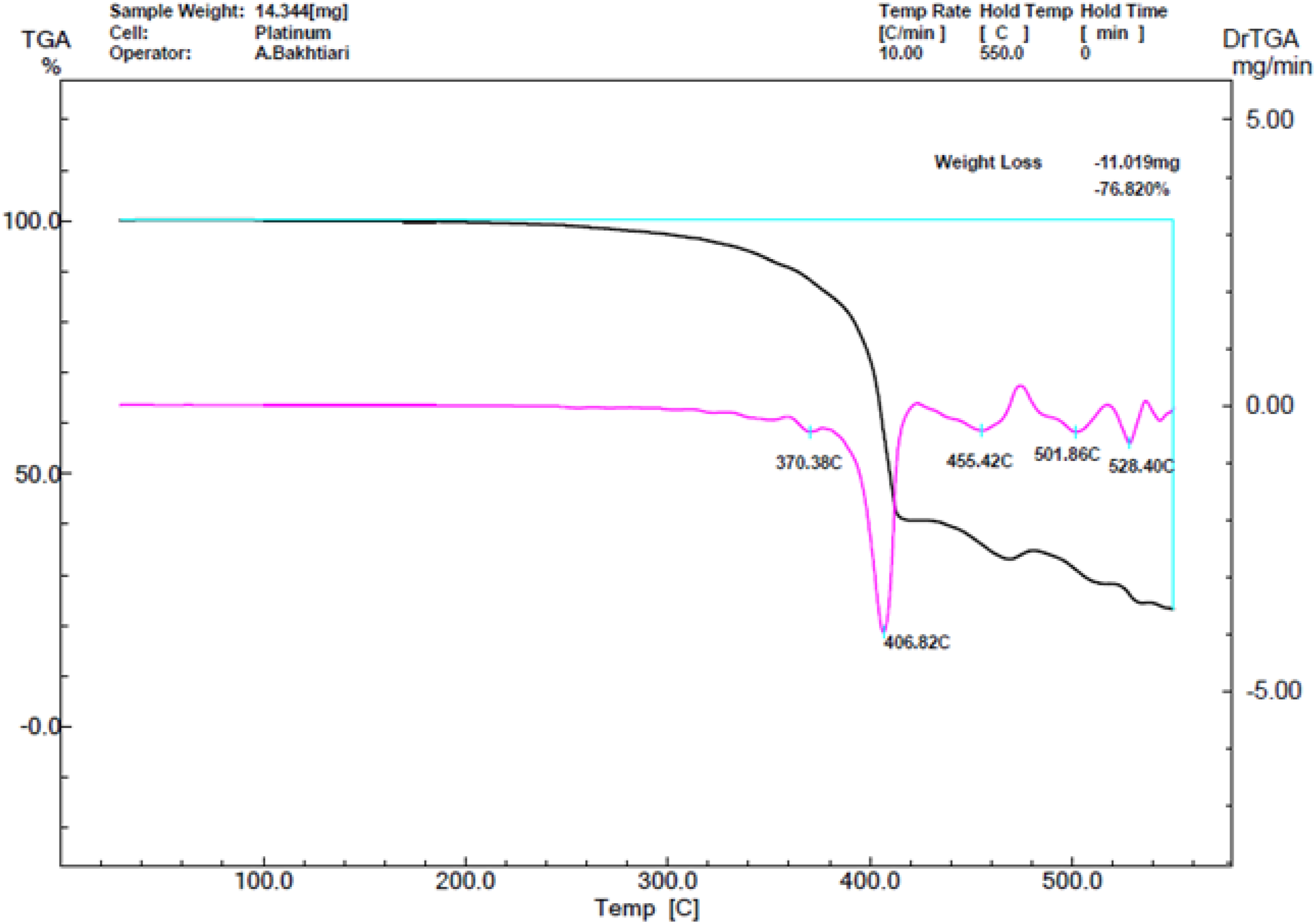
The Yazd rubber industrial complex, Yazd, Iran supplied the waste rubber powder. It was the phenolic-cured butyl rubber used for manufacturing the rubber-made tire curing bladder. The powder size distribution (Figure 1) was assessed by a laser diffraction particle size analyser (Mastersized3000, Malvern Panalytical, UK). A thermo gravimetric analysis (TGA) instrument (STA 1500, Scinco Co. Ltd., South Korea) was employed to assess the chemical composition of the rubber powder. The TGA was performed in an inert (nitrogen) atmosphere below 400°C. As soon as the temperature increased to 400°C, the atmosphere changed by the air to evaluate the content of the carbon black in the rubber powder. The results were represented and summarized in Figure 2 and Table 1. The processing aid (aromatic oil, Fariman Co.,Iran) with a boiling point of 270°C supplied by the local market. Tetramethylthiuram disulfide (Perkacit TMTD, Flexsys, Belgium), Diphenyl disulfide (DPDS, Sigma-Aldrich, Germany), Benzothiazyl-2-Dicyclohexyl Sulfenamide (DCBS, Acro Chem,USA), N- tert-butyl-benzothiazole sulphonamide (TBBS, Western reserve chemicals, USA), Benzothiazole disulfide (MBTS, NOCIL Limited, India), and 2-mercaptobenzothiazole (MBT, NOCIL Limited, India) used as de-crosslinking agents. The cured rubber made waste bladder powder size distribution. The particles dispersed in the water before size distribution measurement. The thermo gravimetric analysis (TGA) curves for the cured rubber made bladder waste powder. Physical properties and composition of the used cured rubber made waste bladder powder. aCLD was calculated using the Flory-Rehner equation.
1

De-crosslinking method
In the first stage, the de-crosslinking agent dissolved in aromatic oil with the aid of heat at 40°C for about 15 min to facilitate the agent’s homogeneous dispersion in the rubber matrix. Subsequently, it was added to the waste rubber powder in a litre glass beaker and allowed the rubber powder to soak in the treated oil in an oven for 72 h at 80°C. This procedure guaranteed the maximum penetration of the oil in the rubber powder. In the next stage and for de-crosslinking of the rubber compound, the beaker put inside a laboratory heating oven for 5 min at the selected temperatures, 180, 200, 220, 240, and 250°C. The used heating oven (TEHRAN sterilizator, Tehran, Iran) had 30 L capacity. It was equipped with a temperature regulator to control the inside temperature of the oven. Finally, the treated compounds were processed and sheeted on a two-roll mill with cylinders length and diameter of 32 and 14 cm, respectively, at 45°C for 1 min to produce rubber sheets with a thickness of 3 mm for further tests.
The morphology and identification of the rubber compounds and measurement of the de-crosslinking parameters
The attenuated total reflectance Fourier transform infrared spectroscopy (ATR-FTIR, AVATAR 370FT-IR, Termo Nicolet, USA) with a resolution of 4
5 g of the formed rubber sheet by two-roll mill extracted first in acetone for 48 h using a Soxhlet apparatus to remove low-molecular-weight polar substances such as curative residues and accelerator by-products. After this initial extraction, the sample was immediately subjected to a second extraction for 72 h using THF to remove constituents such as oil, un-crosslinked polymer residues or even soluble polymer released from the network due to de-crosslinking. After that, the sample was dried in a vacuum oven until reaching to constant mass. Subsequently, rubber swelling tests were performed to determine the crosslink density of the rubber compounds. To do the rubber swelling test, the dried rubber compound (square sheet with 3 mm thickness) obtained from the previous stage was placed inside a tapped glassware filled with 500 mL toluene at ambient temperature and allowed to reach the ultimate weight. After weighing the swollen gel, it was dried for 72 h at ambient temperature to remove the solvent. Each experiment was repeated three times, and the median value reported.
The crosslink density (CLD) then calculated using the Flory–Rehner equation
1
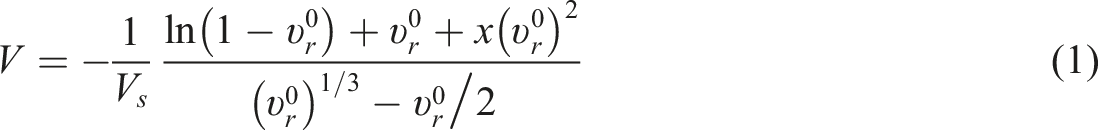

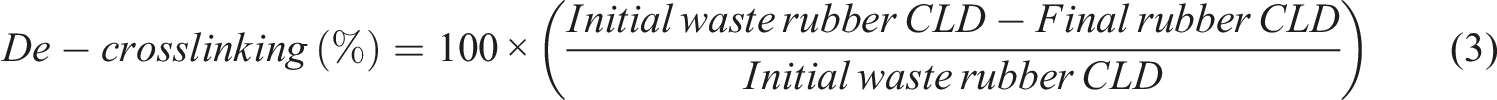
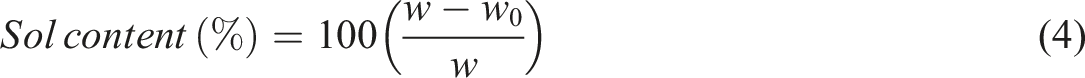
The relationship between sol fraction and CLD may express by Horikx analysis.
1
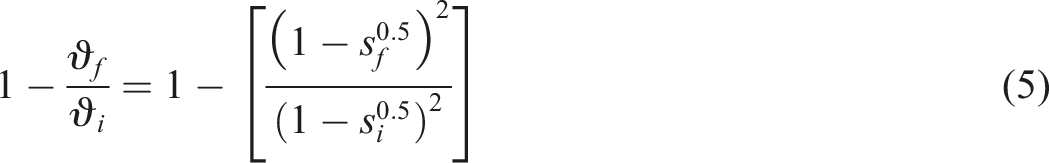
The Horikx equation normally used to characterise network structures undergoing oxidative degradation, devulcanization or recycling. The relative decrease in CLD when main chain scission occurs
22
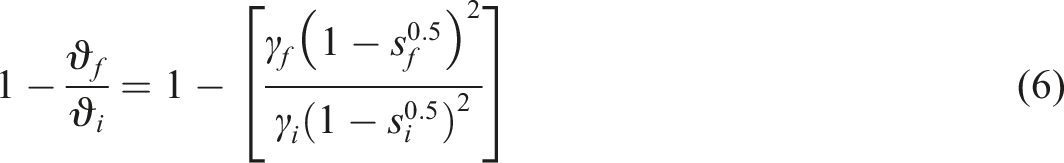

By substituting the
The surfaces of the de-crosslink rubber compounds were assessed by an LEO 1530 VP field emission gun scanning electron microscope (SEM). Small pieces of the rubber samples were obtained under a liquid nitrogen atmosphere, with approximate dimensions of 10 by 10 mm coated with gold and subsequently photographed in the SEM. The obtained SEM micrographs used for the morphology study of the rubber compounds.
Results and discussion
The composition of the used waste butyl rubber
The used waste butyl rubber was the waste rubber-made bladder. It was used for curing the tire in the mould. The de-crosslinking efficiency depends on the size distribution and mean size of the rubber powder particles remarkably. 12 For particles with a greater diameter than 0.5 cm, the hot spots appear on the rubber surface during oven heating. The above-mentioned phenomena make a temperature profile in the bulk of the sample resulting in heterogeneous de-crosslinking of the rubber sample. To prevent mentioned difficulty, the waste rubber was ground to produce a powder with a median size of 371 microns (Figure 1). As observed, 25, 43, 64, and 86 volume % of the waste rubber particles had diameters less than 183, 293, 469, and 750 microns, respectively. Also, the minimum and the maximum particle diameter were 28 and 1518 microns, respectively.
Table 1 depicts the composition of the waste butyl rubber powder obtained from TGA curves (Figure 2). As shown, the light hydrocarbons and oil content degraded between 160 to 340°C with about 6% mass loss of the rubber sample. Also, the rubber began to degrade from 340°C and ended at 455°C just before changing the atmosphere from an inert gas (nitrogen) to air. The mass loss for rubber was 54%. The attributed mass loss for carbon black (filler) was 17% for temperatures between 455 to 545°C. The left mass beyond 545°C belonged to unburned residues, including minerals with 23% mass loss.
The Fourier transform infrared spectroscopy (FTIR) graphs analysis was used to identify the existence of the phenolic resin in the structure of the waste bladder butyl rubber powder (Figure 3). As shown, the appeared peaks at 3412, 1539, 1621, 1180, and 715 The FTIR graph for the used waste butyl rubber powder.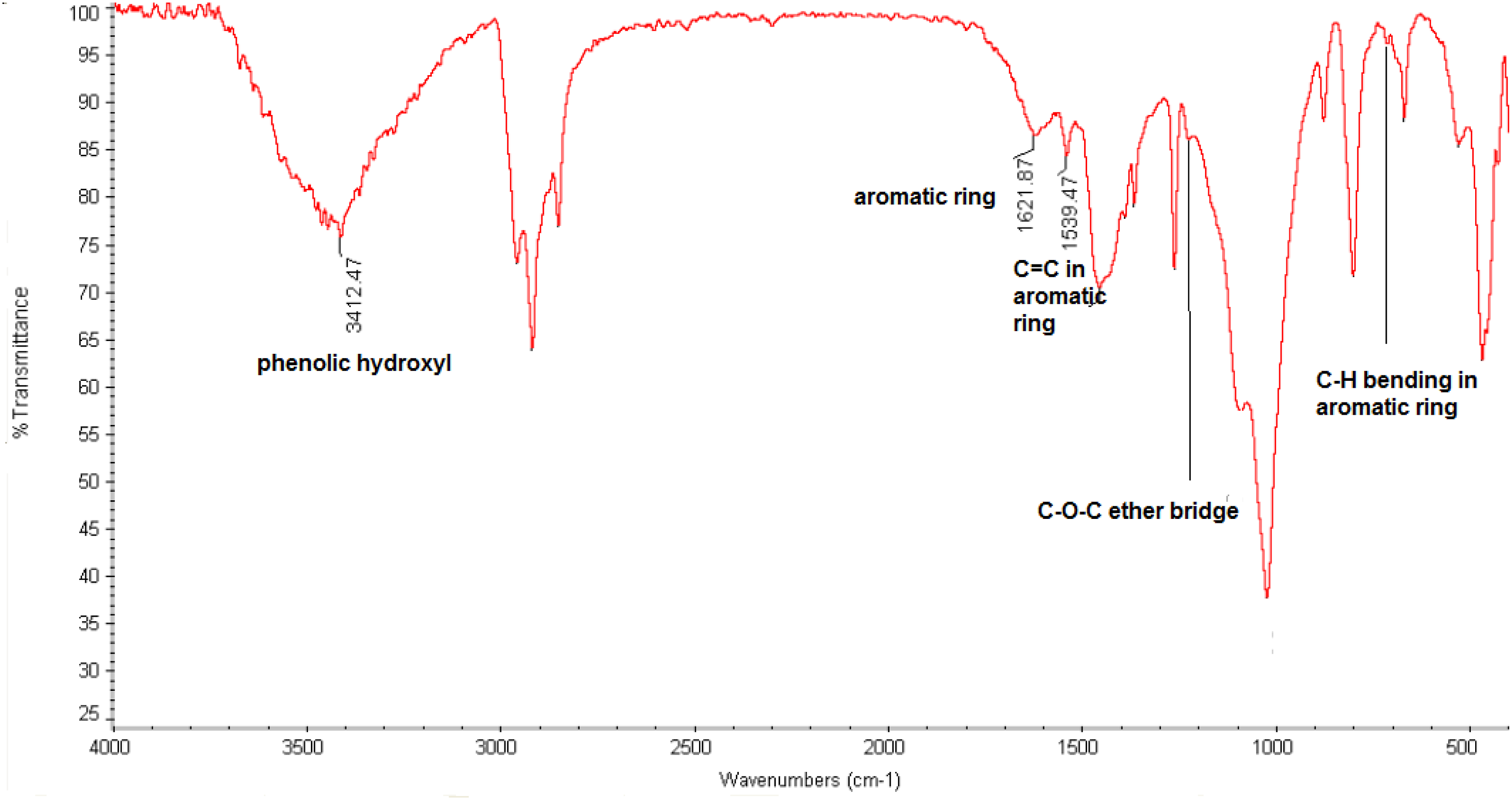
De-crosslinking of the waste butyl rubber powder
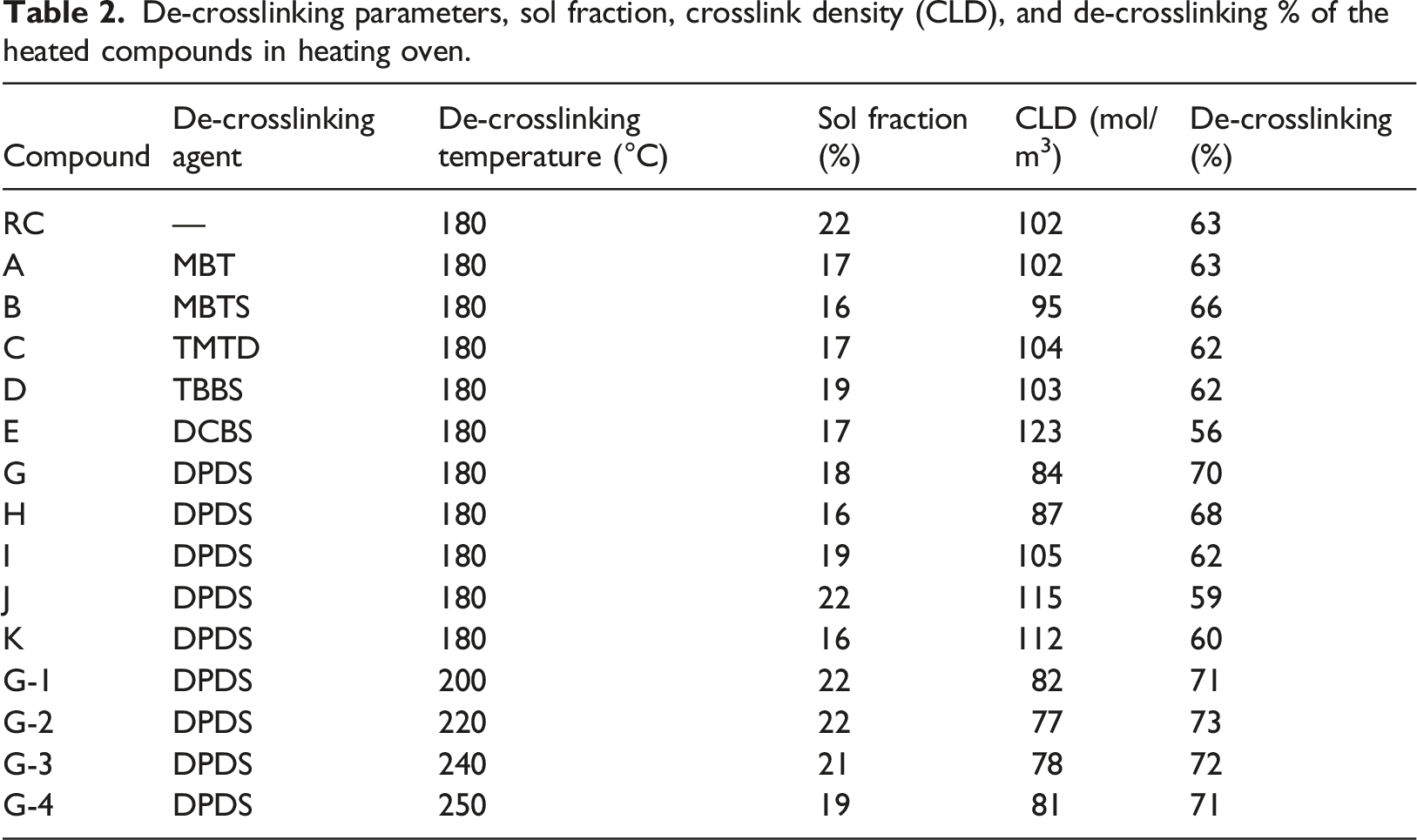
Selected de-crosslinking agents are known as routine curing accelerators in the rubber industry. This is true in conventional curing temperatures, i.e., 160–180°C. However, for higher temperatures, they may act as de-crosslinking agents and accelerate the reverse crosslinking reactions. During the heating of the waste rubber in the heating oven, the apparent state of the rubber powder changed from a moderately rigid solid particle to a bulky state. It was good evidence for the de-crosslinking of the rubber powder. It conformed with the SEM micrographs in Figure 4 and also the measured de-crosslinking % ‘s of the treated compounds (Table 2). The SEM micrographs showing the interior of the (a) Waste butyl rubber powder (b) Reference compound, RC (c) Compound E with the lowest de-crosslinking % (d) Compound G-2 with the highest de-crosslinking %. De-crosslinking parameters, sol fraction, crosslink density (CLD), and de-crosslinking % of the heated compounds in heating oven.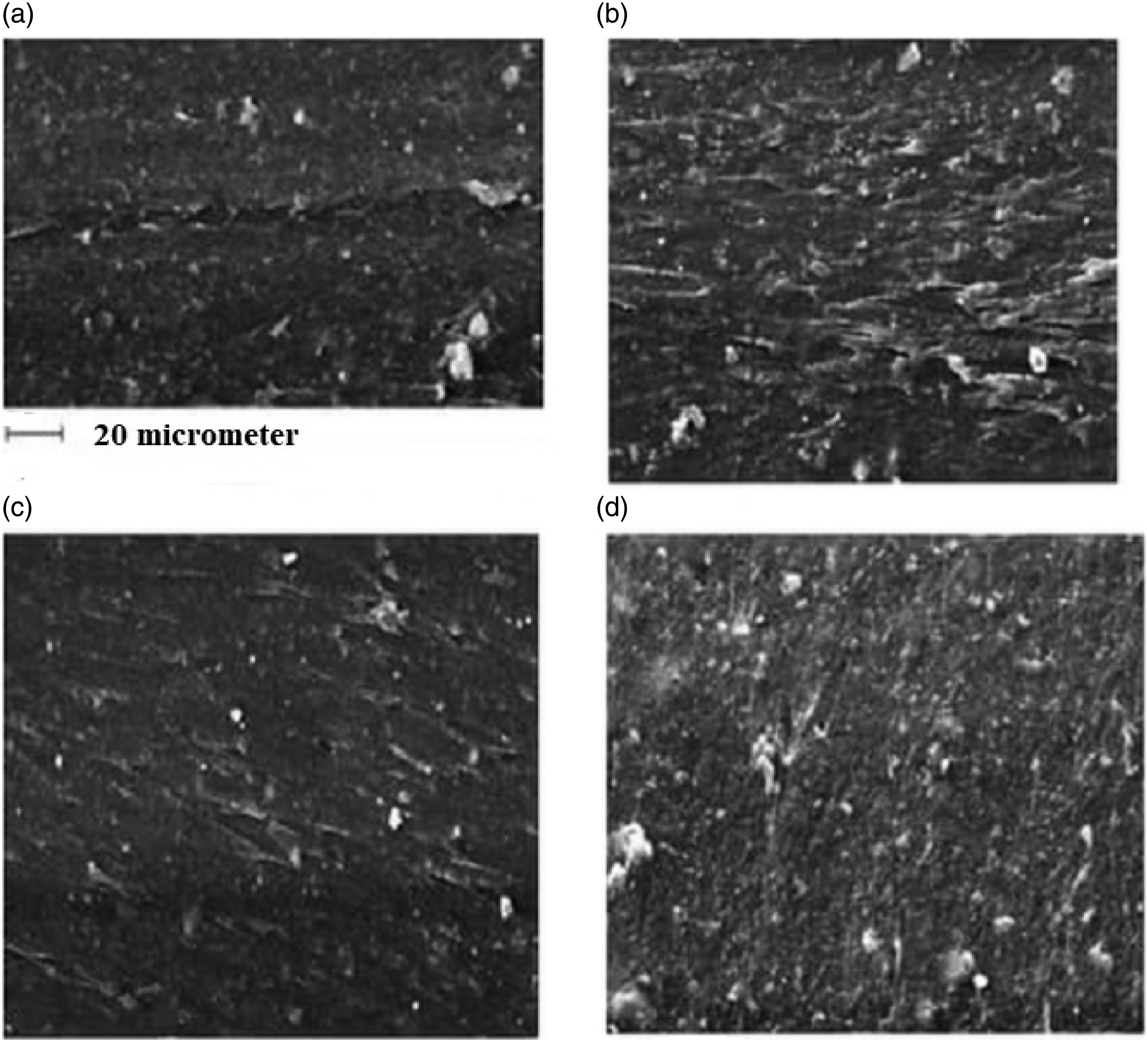
The compound’s formulation for de-crosslinking of the waste butyl rubber powder. De-crosslinking took place inside of heating oven for 5 min at 180
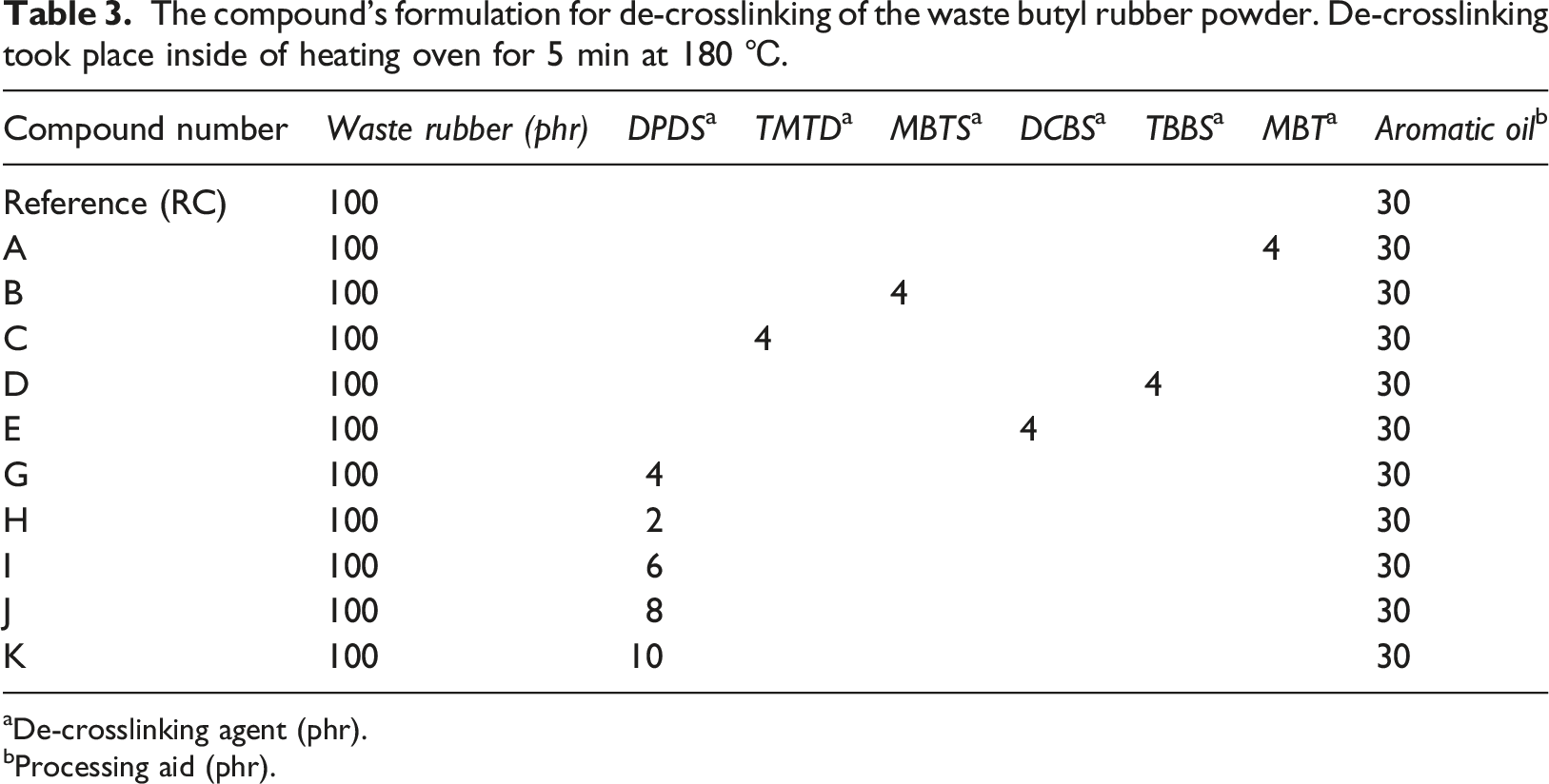
aDe-crosslinking agent (phr).
bProcessing aid (phr).
The sol fractions versus relative reduction in crosslink densities (CLD’s) or (
Table 3 shows the Compounds G (DPDS) and E (DCBS) with 70 and 56% had the highest and the lowest de-crosslinking % among studied de-crosslinking agents, respectively. Interestingly, the de-crosslinking % of Compound E was even lower than the reference compound (RC) with a value of 63%. It concluded that DCBS as a delayed action sulfenamide agent acted as a retardant for the de-crosslinking process. The de-crosslinking %‘s of the left compounds ranged between 62% for Compound D with TBBS to 66% for Compound B with MBTS. Usually, the CLD reduction accompanies with the increase in rubber sol fraction (%).
1
However, it is not a general rule and it depends on the ratio of macromolecular main chain to crosslinks scission.23,24 As an illustration, the sol fraction and CLD for the RC were 102
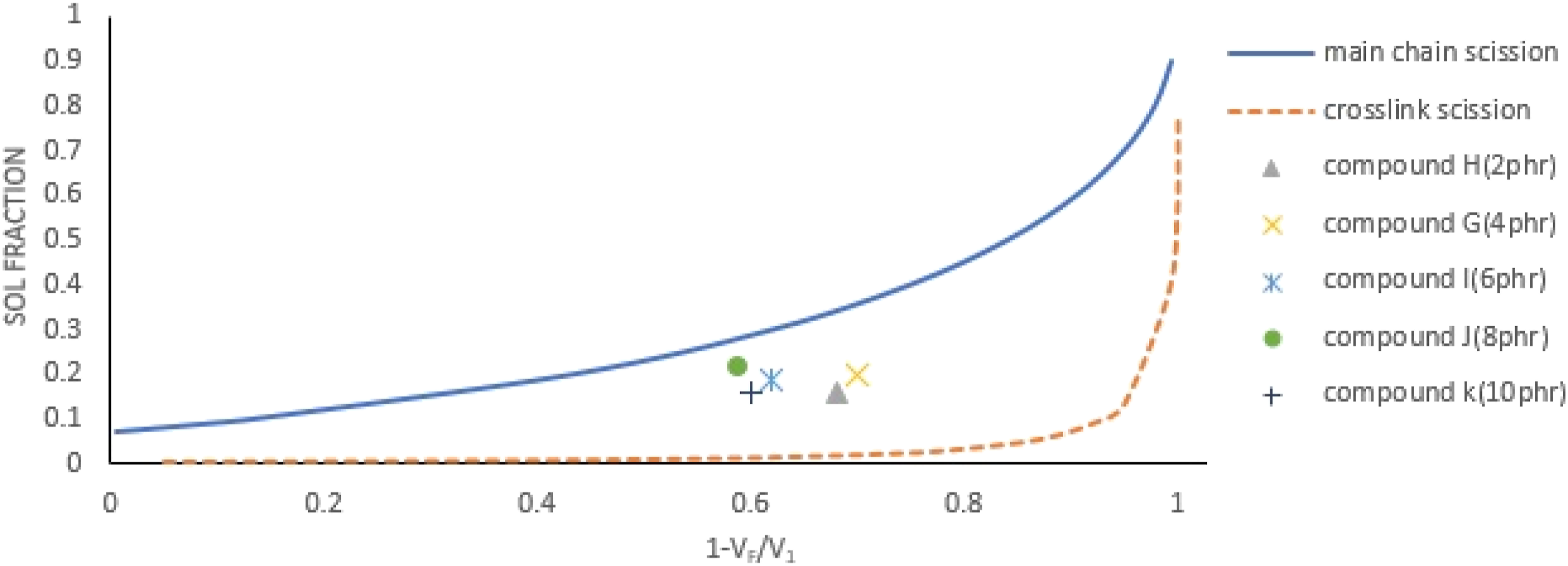
The relative decrease in CLD versus the sol fraction of the compounds with different amounts of DPDS, 2, 4, 6, 8, and 10 phr (Compounds H, G, I, J, and K, Table 3) was depicted in Figure 5. As shown, together crosslink and main chain scission were observed for all compounds with different amounts of DPDS in the formulation. However, the highest and the lowest crosslink scission happened for Compounds H and G, and J, respectively. Also, increasing the DPDS reduced the de-crosslinking % from 68-–70% for Compounds H and G with 2 and 4 phr to 59–60% for Compounds J and K with 8 and 10 phr DPDS in formulations. As a result, increasing the DPDS in compound’s formulation had no benefit for de-crosslinking process. The relative decrease in CLD versus the sol fraction of Compounds A to G and reference Compound. De-crosslinking took place inside of heating oven with at 180 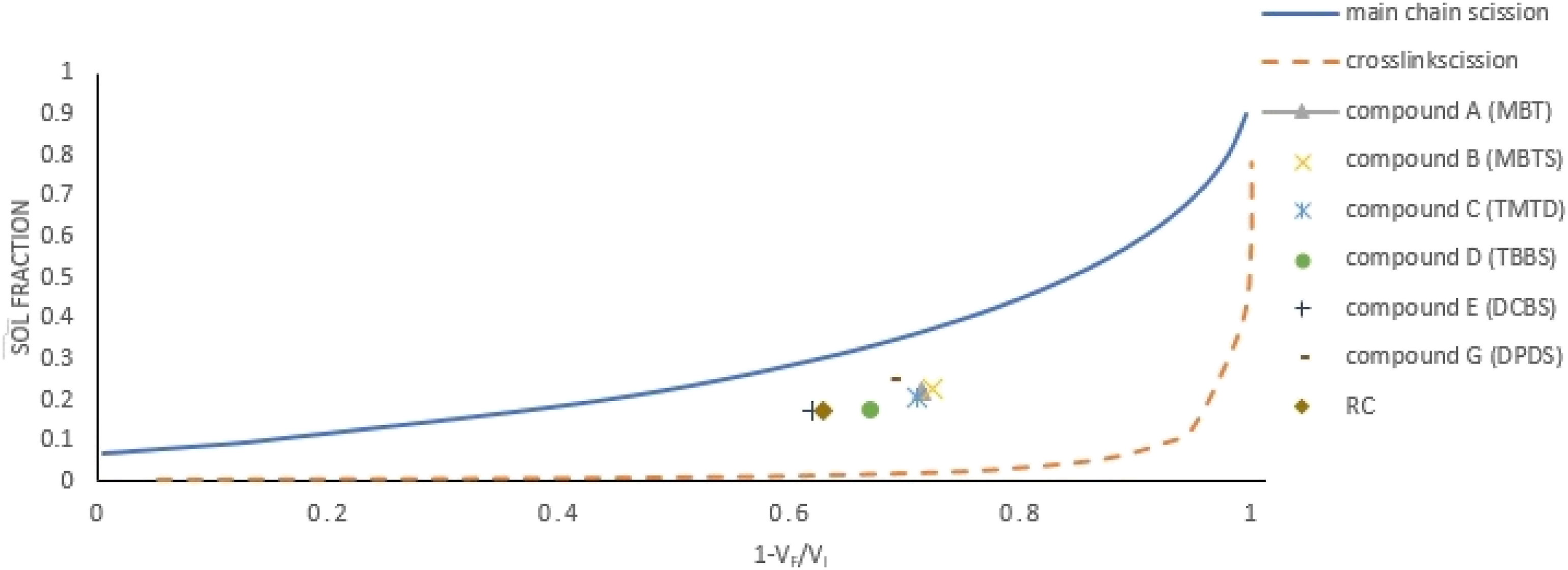
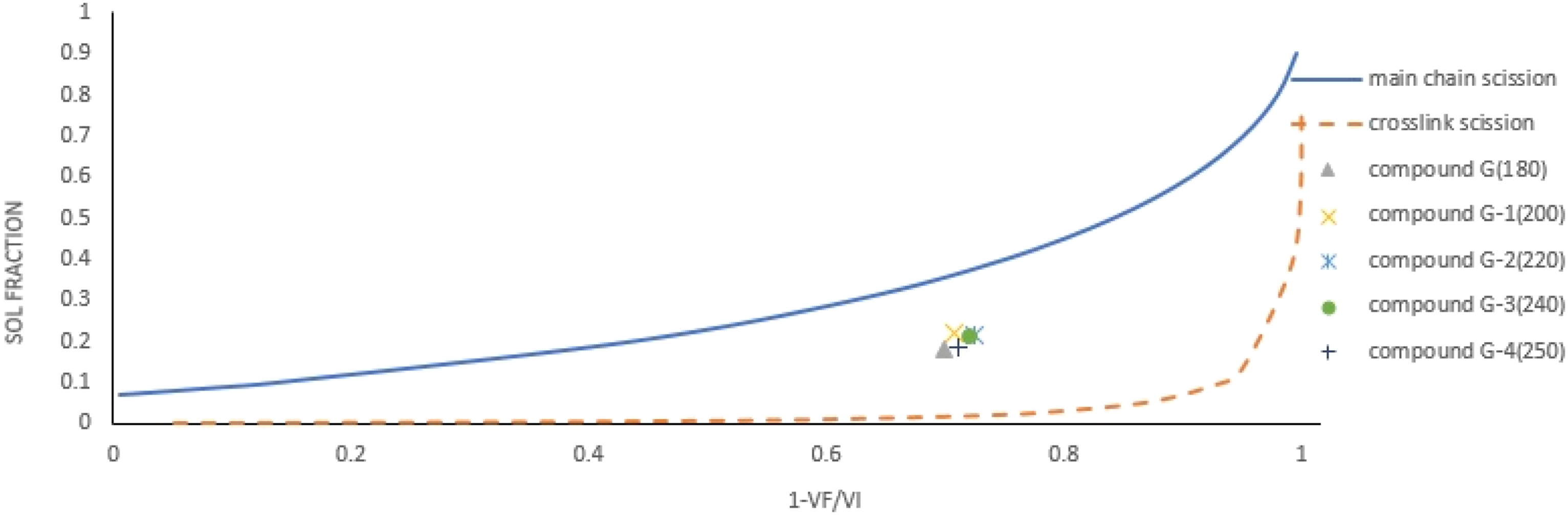
Table 3 shows the de-crosslinking parameters for selected compounds with heated in oven at selected temperatures 180, 200, 220, 240, and 250 Proposed de-crosslinking mechanism. The relative decrease in CLD versus the sol fraction of the compounds with different amounts of DPDS. De-crosslinking took place inside of heating oven at 180 The relative decrease in CLD versus the sol fraction of the compounds G and G-1 to G-4. De-crosslinking took place inside of heating oven at different temperatures.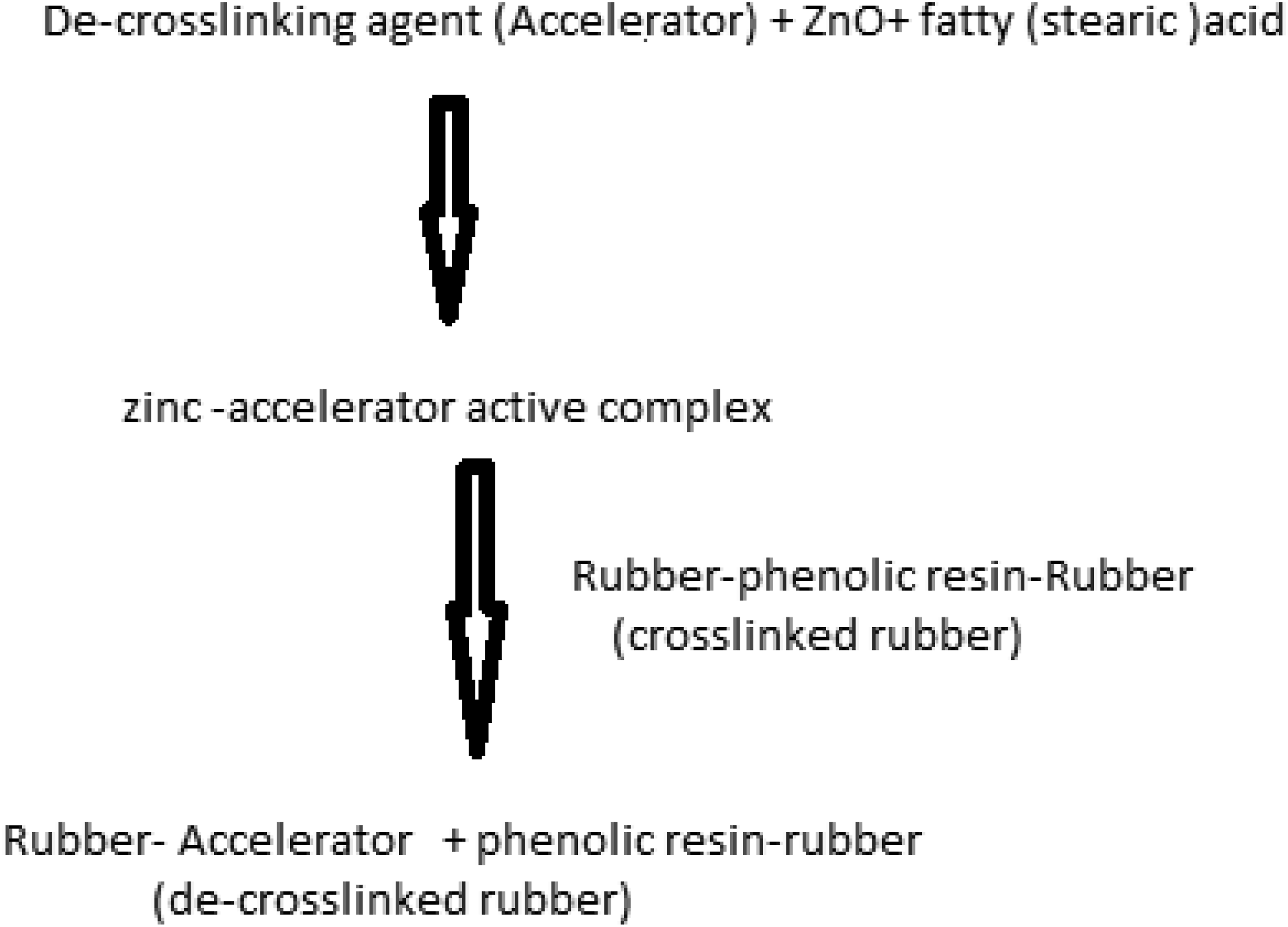
Conclusions
The Fourier transform infrared spectroscopy (FTIR) graphs were suitable evidences to prove the existence of phenolic resin in the structure of the used waste bladder butyl rubber powder. Efficient de-crosslinking was observed for all studied de-crosslinking agents. It evidenced by the SEM micrographs and also the measured crosslink densities of the reclaimed rubbers. The highest and the lowest de-crosslinking %‘s belonged to the compounds with 4 phr DPDS in recipe at 220
Footnotes
Acknowledgements
The authors sincerely thank the staffs of the polymer chemistry laboratory located at faculty of science, Ferdowsi University of Mashhad for their sincere cooperation. Approval no. 3/53624.
Author Contributions
Zahra Miranian, Nadia Ostad Movahed and Saeed Ostad Movahed designed the experiments. Dr. Saeed Ostad Movahed prepared the manuscript with contributions from all co-authors. The authors applied the SDC approach for the sequence of authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
