Abstract
Blend of acrylonitrile butadiene/isoprene-isobutene rubber (NBR/IIR) with different compositions was prepared. Scanning electron microscopic observations confirmed the compatibilization of bromo isoprene-isobutene rubber on NBR/IIR blend. Each rubber blend was loaded with different ratios of N-326 (high-abrasion furnace (HAF)): N-774 (semi-reinforcing furnace) carbon blacks. Current–voltage curves, joule electrical heating, and temperature-dependent electrical conductivity of the prepared blends were measured. The electrical and thermal properties of the blends were enhanced upon the increase of N-326 (HAF) content.
Introduction
Polymer composites doped with carbon black have attracted growing interest due to their potential use in various applications. 1 They can be utilized for electrical heating and devices that convert electrical energy into thermal energy. Fillers are introduced into rubbery polymers for many varied reasons, more generally to help in tailoring the physical and/or chemical properties of the rubber for various applications where flexibility is an important parameter. 2 The concentration, particle size, and structure of the carbon black are essential for controlling the electrical and thermal properties. 3,4 The incorporation of carbon black has been reported to form a carrier path in the insulating rubber matrix. 5 An effective electrical conductivity of carbon black/polymer has been achieved at relatively high concentration of carbon black. 6 The temperature dependence of the rubber electrical properties has been utilized in various polymer applications.
When the filler distribution results in the formation of a segregated structure, the electrical conductivity is enhanced as compared to a random distribution of filler in the polymeric matrix. 7 The negative temperature coefficient of conductivity (NTCC) of polymer is a parameter that determines the efficiency of self-controlled heater and current limiters. 8,9 However, a polymer with positive temperature coefficient of conductivity (PTCC) is needed for electromagnetic radiation shielding. 10,11
Blends of nitrile butyl rubber (NBR) with polymers have been investigated by Hofmann. 12 The instability of physical properties of butyl rubber (isoprene-isobutene rubber (IIR)) is a serious obstacle to its use as heating element applications. 9 This problem has been overcome by the addition of uncross-linked layers of NBR to the IIR. The observed T-peel adhesion between the NBR and IIR is rate and temperature dependent. 13 KP Sau et al. 14 found that rubber blends having differences in polarity are very useful in achieving suitable conductivity since the distribution of carbon black at the interference of two rubbers gives rise to high conductivity. NBR has good electrical properties due to polar C≡N groups, 15 while butyl rubber (IIR) possess good thermal properties. This combination was known to enhance the thermal resistance of NBR.
In this work, we prepared NBR/IIR rubber blends and doped them with N-326 and N-774 carbon blacks in an attempt to enhance the entire electrical and thermal properties. N-326 and N-774 were selected as dopants in this study because they differ greatly in their particle size, surface area, and surface conditions of carbon particles. This large variation is expected to modify the electrical and thermal properties of the blend. These properties are investigated by measuring the current–voltage curves, electric heating behavior, and temperature dependence of electrical conductivity.
Experimental part
Rubber of acrylonitrile butadiene/butyl rubber (NBR/IIR) blends were manufactured at (50/50) and (25/75), respectively. The rubber blend was reinforced with N-326 (high-abrasion furnace (HAF) black) and N-774 (semi reinforcing furnace black). The percentage of N-326 to N-774 was selected as (0:100), (25:75), (50:50), (75:25), and (100:0). The particle size, surface area, and pH of different carbon black fillers used are listed in Table 1. 16 The ingredients of each blend are listed in Table 2. Mastication and mixing were carried out on a two-roll mill (length 0.3 m, radius 0.15 m, speed of slow roll 18 r min−1, and gear ratio 1.4) was used. The compounded rubbers were compression molded into cylinders of 1 × 10−4 m2 area and 0.01 m in height. The vulcanization process was carried out using special home-made mold in an electrically heated hydraulic press at a fixed temperature of 153°C under a pressure of 300 kPa for 30 min. Two brass electrodes were embedded inside the cubic sample at distance 0.04 m and length 0.025 m during the vulcanization process. Suitable electrical circuit was used for measuring Joule heating and direct current conductivity measurements, suitable cell for current–voltage (I–V) measurements, and Quanta FEG 250 scanning electron microscope (SEM; Taiwan) for structural investigation.
Physical and chemical properties of different carbon black fillers used. 16

HAF: high-abrasion furnace; SRF: semi-reinforcing furnace.
The compositions of rubber blend NBR/IIR with different types of carbon black.

phr: part per hundred parts of rubber by weight; BIIR: bromo isoprene-isobutene rubber; MBTS:dibenzthiazyl disulphide; DOP: dioctyl phthalate; DPG: diphenyl guanidine; NBR: nitrile butyl rubber; IIR: isoprene-isobutene rubber.
Results and discussion
Scanning electron microscopy
Pure vulcanized (50NBR/50IIR) rubber blends without and with compatibilizer were investigated using Quanta FEG 250 SEM type. The surface morphology of the previous blend without compatibilizer is shown in Figure 1(a).

(a) SEM micrographs of pure vulcanized (50NBR/50IIR) rubber blend without compatibilizer (BIIR). (b). SEM micrographs of (50NBR/50IIR) rubber blend with compatibilizer (BIIR). SEM: scanning electron microscopic; NBR: nitrile butyl rubber; IIR: isoprene-isobutene rubber; BIIR: bromo isoprene-isobutene rubber.
The SEM micrograph shows that low degree of compatibility and no enough miscibility between the main components of blend and the sample were continuous in nature associated with the phase separation. So the SEM micrograph shows two different phases inside the blend as a result of the difference in polarity between the pair rubber components of the blend.
Figure 1(b) shows the (50NBR/50IIR) blend containing bromo isoprene-isobutene rubber (BIIR) as a compatibilizer. It is evident that the phase separation appears to be diminished to a greater extent and the dispersed IIR phase was seen more evenly dispersed in the NBR phase. So the surface morphology of the prepared blend appeared as continuous phase. 17
I–V characteristics
Figure 2 shows I–V curves of the (50NBR/50IIR) rubber blend with different ratios of N-326:N-774 carbon black fillers. It is shown that the curves display ohmic behavior. The current density was controlled by thermally generated electric field, across the gap between the conductive particle agglomerates. 9 With the increase of the electric fields, the behavior of I–V changes from linear to nonlinear. The nonlinearity of the curves was attributed to the joule heating effect, which increased the sample temperature and increased the current. 18 From Figure 2, it is observed that a low electric current passing through the samples that contain high content of N-774 carbon black is in contrast to the higher current observed in the samples that have higher content of N-326 carbon black fillers. This related to the nature of N-326 carbon black that improves the aggregations inside the rubber matrix due to its small particle size, which agglomerate themselves forming conduction network structure and hence small interspacing distances between carbon black particles, resulting in notable increment in electric current.
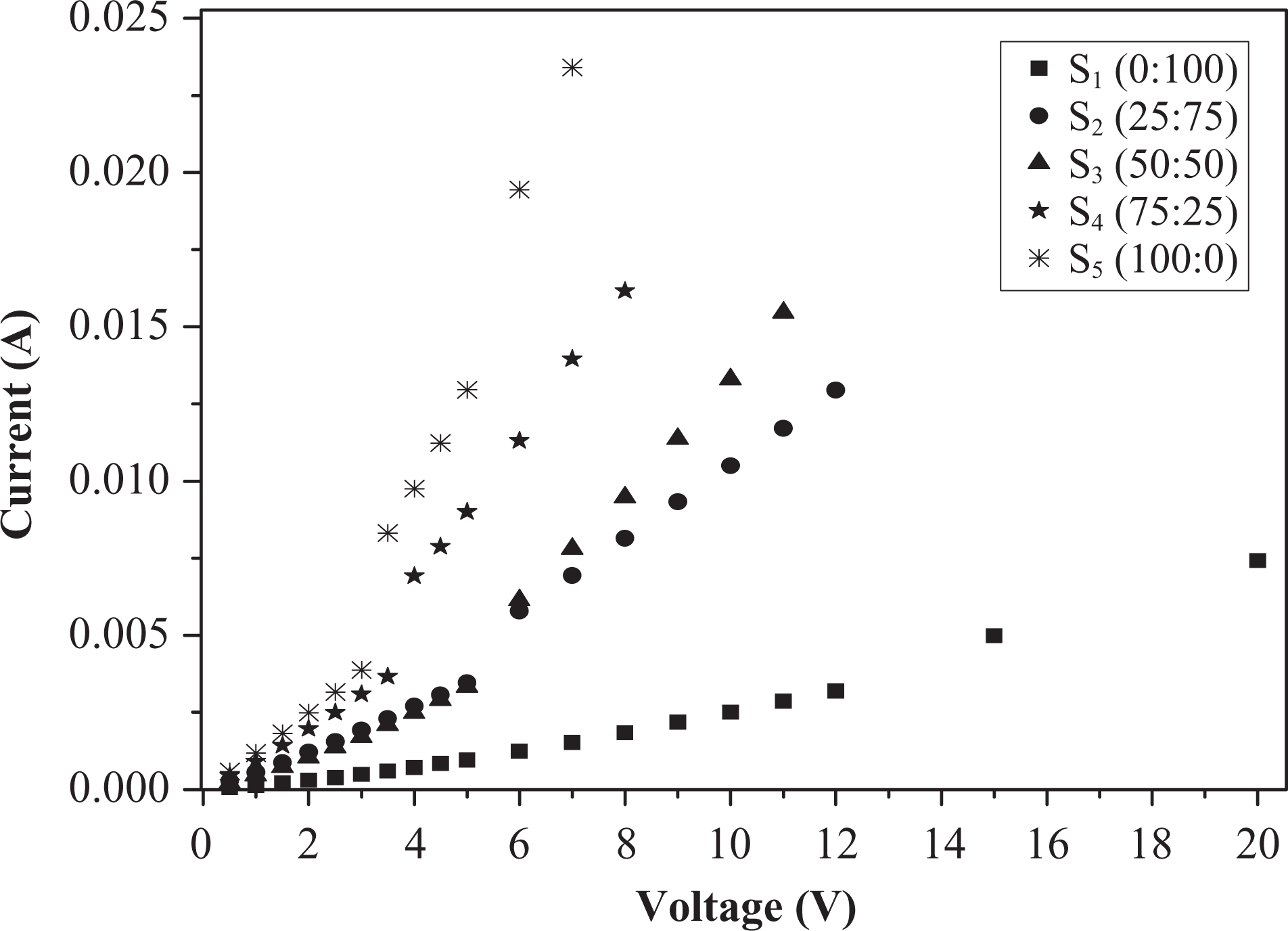
I–V relation for (50NBR/50IIR) rubber blend loaded with different ratios of N-326:N-774. I–V: current–voltage; NBR: nitrile butyl rubber; IIR: isoprene-isobutene rubber.
The nonohmic behavior could be fitted to an empirical formula of the form: 8

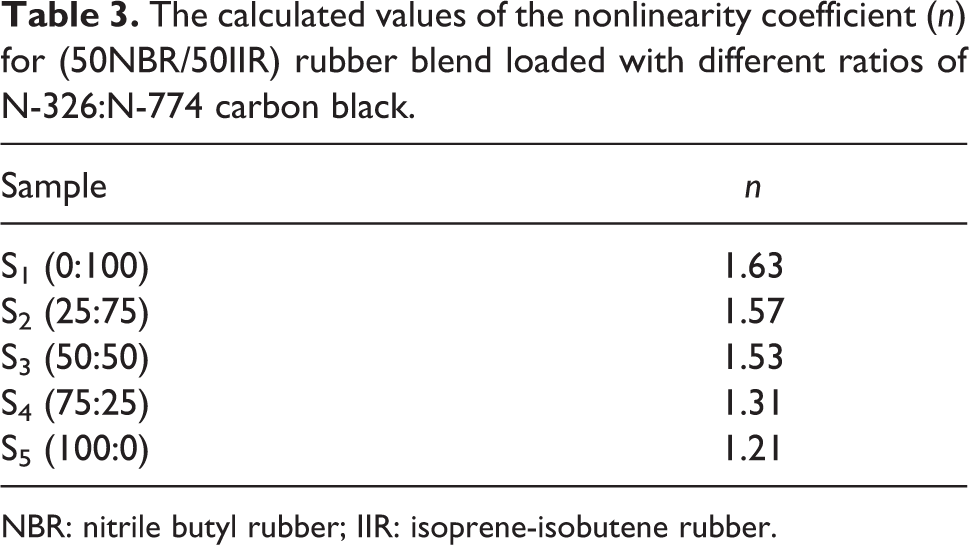
where k is a constant and n is the nonlinearity coefficient. The values of n were calculated and are tabulated in Table 3. It was noticed that n deviated slightly from the ohmic behavior. Also from the table, the value of n decreases with increasing N-326 HAF carbon black, and this reflects that N-326 improves the thermal stability of the microstructure core of the rubber matrix.
The calculated values of the nonlinearity coefficient (n) for (50NBR/50IIR) rubber blend loaded with different ratios of N-326:N-774 carbon black.
NBR: nitrile butyl rubber; IIR: isoprene-isobutene rubber.
Joule heating behavior
A constant power of 2 W was applied on (50NBR/50IIR) samples and the evolution of temperature with time was investigated as shown in Figure 3. We observed no significant effect on the temperature–time curves using different concentrations of NBR and IIR.
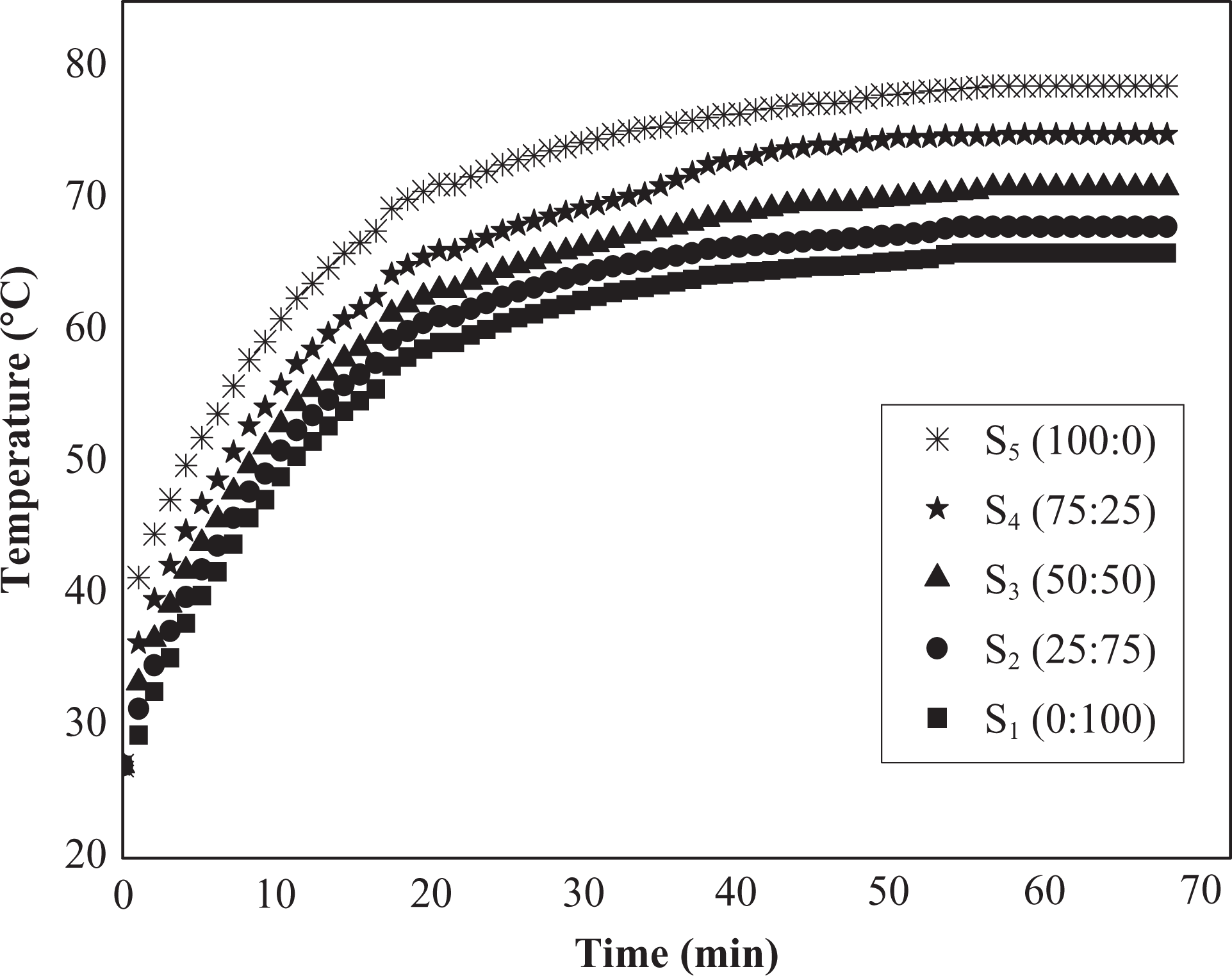
Time-dependent temperature changes at constant electric power (P = 2 W) for (50NBR/50IIR) rubber blend, respectively, loaded with different N-326:N-774 carbon black ratios. NBR: nitrile butyl rubber; IIR: isoprene-isobutene rubber.
The temperature of the blend rises slowly with time until a maximum temperature is attained and maintained afterward constant over time. This behavior is linked to the fact that the applied electric power is effectively dissipated as heat. This trend was similarly observed for all composites. The maximum temperature attained at a given power was higher for the blend with higher carbon black filler N-326 HAF content as shown in Figure 3. The changes of temperature with time can be classified in two regions, namely, the heating region and the maximum temperature region. In the heating region, the temperature–time evolution can be expressed as: 9

where T 0, T m, T t, and τ g are the initial temperature, maximum temperature, an arbitrary temperature at time t, and growth time constant, respectively.
Figure 4 represents the variations of τ g value for the plotted blend in Figure 3 at constant electric power (P = 2 W). It is clear that the value of τ g decreases with the increase of N-326 content and vice versa. The low value of τ g for samples that contain high ratio of N-326 carbon black fillers means the samples have rapid temperature response to the applied powers. The maximum temperature region, on the one hand, displays a constant temperature. According to the principle of energy conservation, the heat gain by electric power is equal to the heat loss by radiation and convection. 3
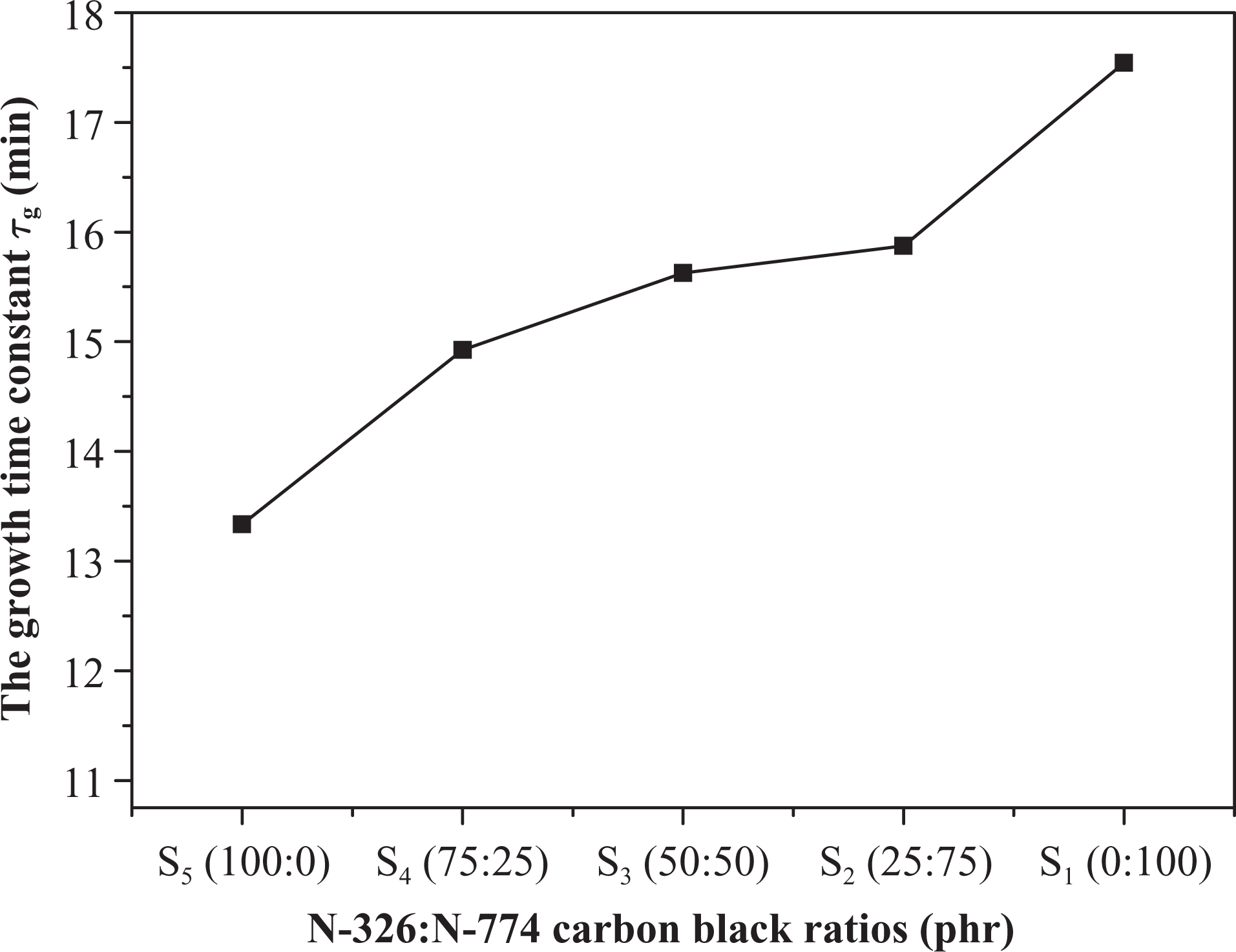
Illustration of the τ g versus types of carbon black ratios relation for (50NBR/50IIR) blend. τ g: growth time constant; NBR: nitrile butyl rubber; IIR: isoprene-isobutene rubber.
Figure 5 displays time-dependent temperature changes at different constant power for (50NBR/50IIR) blend loaded with (50:50) N-326:N-774 carbon blacks. It is clear that, as the applied power increases, the maximum temperature increases, consequently the τ g value will decrease. It was found that, the maximum temperature was quadratically increased with the increment of the applied power, as shown in Figure 5.
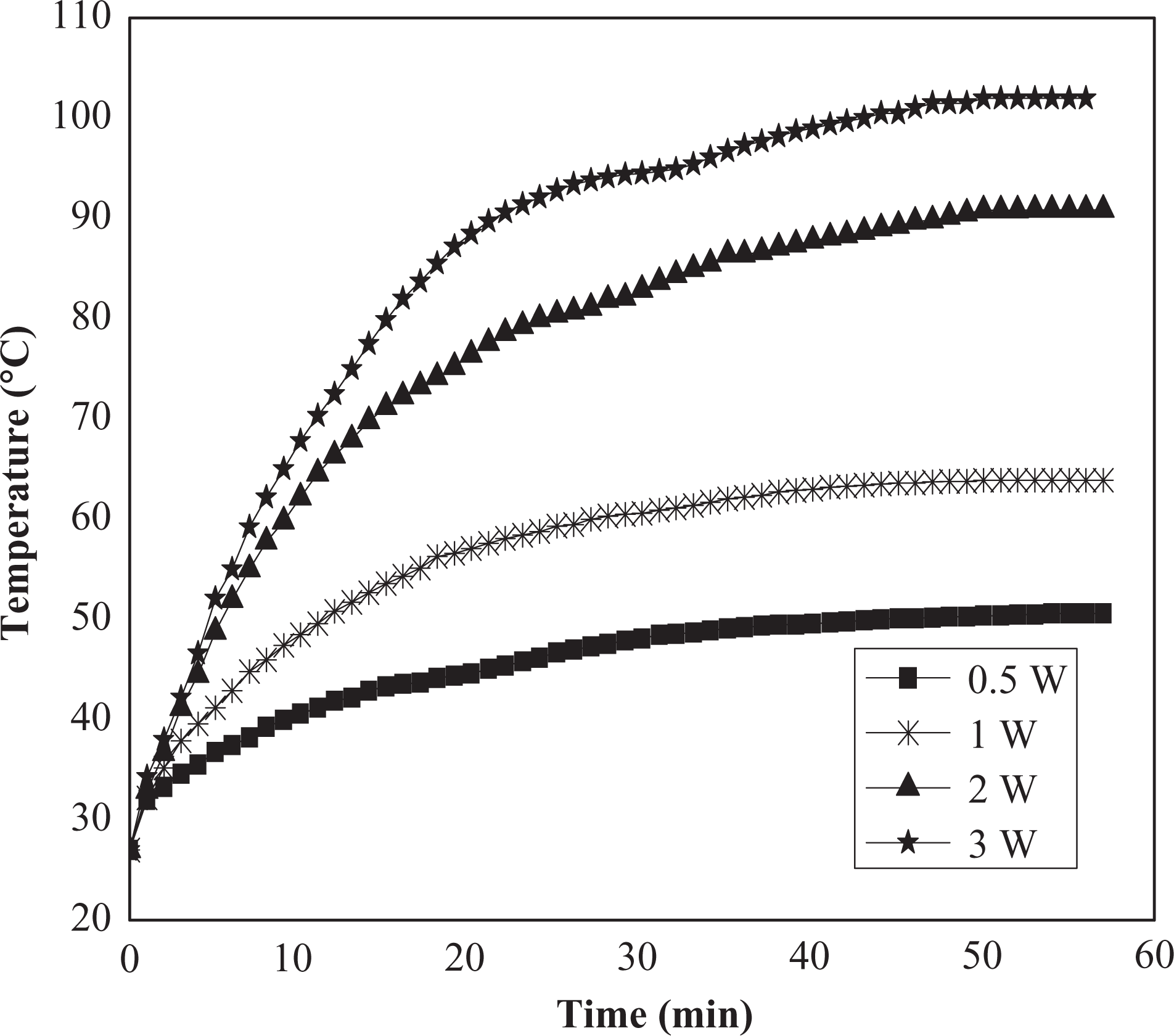
Time-dependent temperature changes at different constant power for (50NBR/50IIR) blend loaded with (50:50) N-326:N-774 carbon black. NBR: nitrile butyl rubber; IIR: isoprene-isobutene rubber.
Temperature dependence of electrical conductivity
Figure 6 illustrates the temperature dependence of electrical conductivity ln σ of (50NBR/50IIR) rubber blend loaded with different carbon black ratios of N-326:N-774. There is a notable increment in the electrical conductivity of blends that contain higher ratio of N-326 carbon black filler in comparison with other blends that contain lower ratio, this is related to the large surface (small particle size) of N-326, which fills the free volume inside the rubber matrix and decreases the conductive aggregate–aggregate interspacing distance, the later creating conductive path channels that lead to an increase in the electrical conductivity. 19
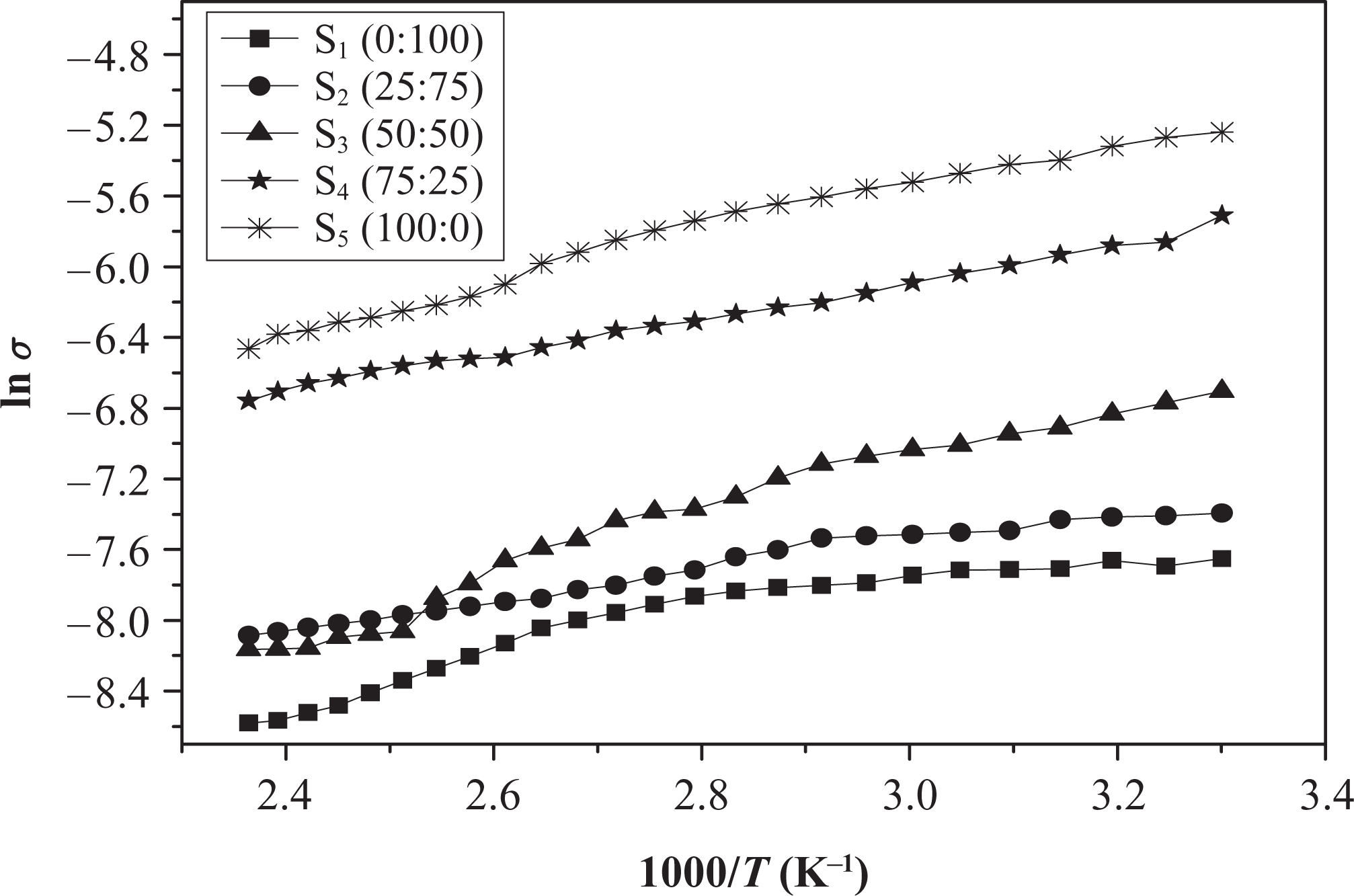
Illustration of ln σ versus 1000/T variations for (50NBR/50IIR) rubber blend loaded with different N-326:N-774 carbon black ratios. NBR: nitrile butyl rubber; IIR: isoprene-isobutene rubber.
It was noticed that at a relatively low temperature range, the conductivity is slightly dependent on temperature. This may be attributed to the direct contact of conductive aggregates that resist the breakage, as the rubber is thermally expanded. 18 At a moderate temperature, the conductivity decreases with increasing temperatures, which is probably a result of breakdown structure of conductive phases due to thermal expansion of the rubber matrix. Indeed, the rise of temperature will cause an appreciable increase in the separation distance between conductive phases, and charge carriers will be scattering at rubber layers between the conductive phases that lead to decrease in conductivity.
It was found that, this blend exhibits NTCC behavior where the electrical conductivity decreases with temperature. This is probably a result of breakdown structure of conductive phases due to thermal expansion of the rubber matrix. 2 In the other words, the rise of temperature causes an appreciable increase in the separation distance between conductive phases, and the dimensions of continuous conductive paths decrease, resulting in a decrease in conductivity of the blends.
Thereupon, the temperature dependence of conductivity could be expressed by a simple empirical form
18
:
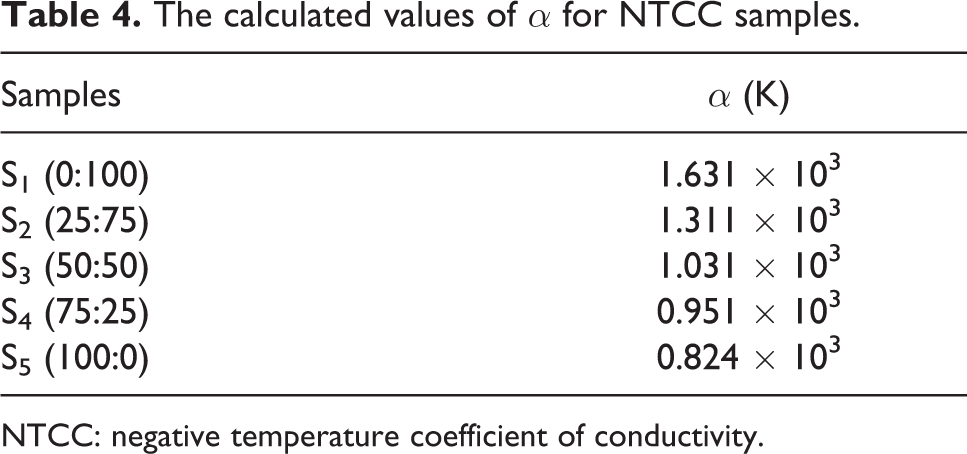
where α is the characteristic parameter and σ 0 is a constant. The values of α for samples (S1 to S5) in Figure 6 were calculated and are listed in Table 4. It was found that, by increasing N-326 carbon black ratio, the values of α decrease.
The calculated values of α for NTCC samples.
NTCC: negative temperature coefficient of conductivity.
During the experimental work, we found that such blends exhibit the NTCC and PTCC behavior at the same time. The (25NBR/75IIR) and (75NBR/25IIR) (results aren’t shown) rubber blends loaded with different carbon black ratios exhibit the NTCC changes abruptly to PTCC behavior. Figure 7 shows NTCC and PTCC behavior for (25NBR/75IIR) rubber blend loaded with (50:50) N-326:N-774 carbon black ratio. This figure shows that, as the temperature of sample increases, the conductivity decreases until a certain temperature, namely the critical temperature (T C), and the NTCC behavior changes abruptly to PTCC. Two possible explanations may be evoked for this change.
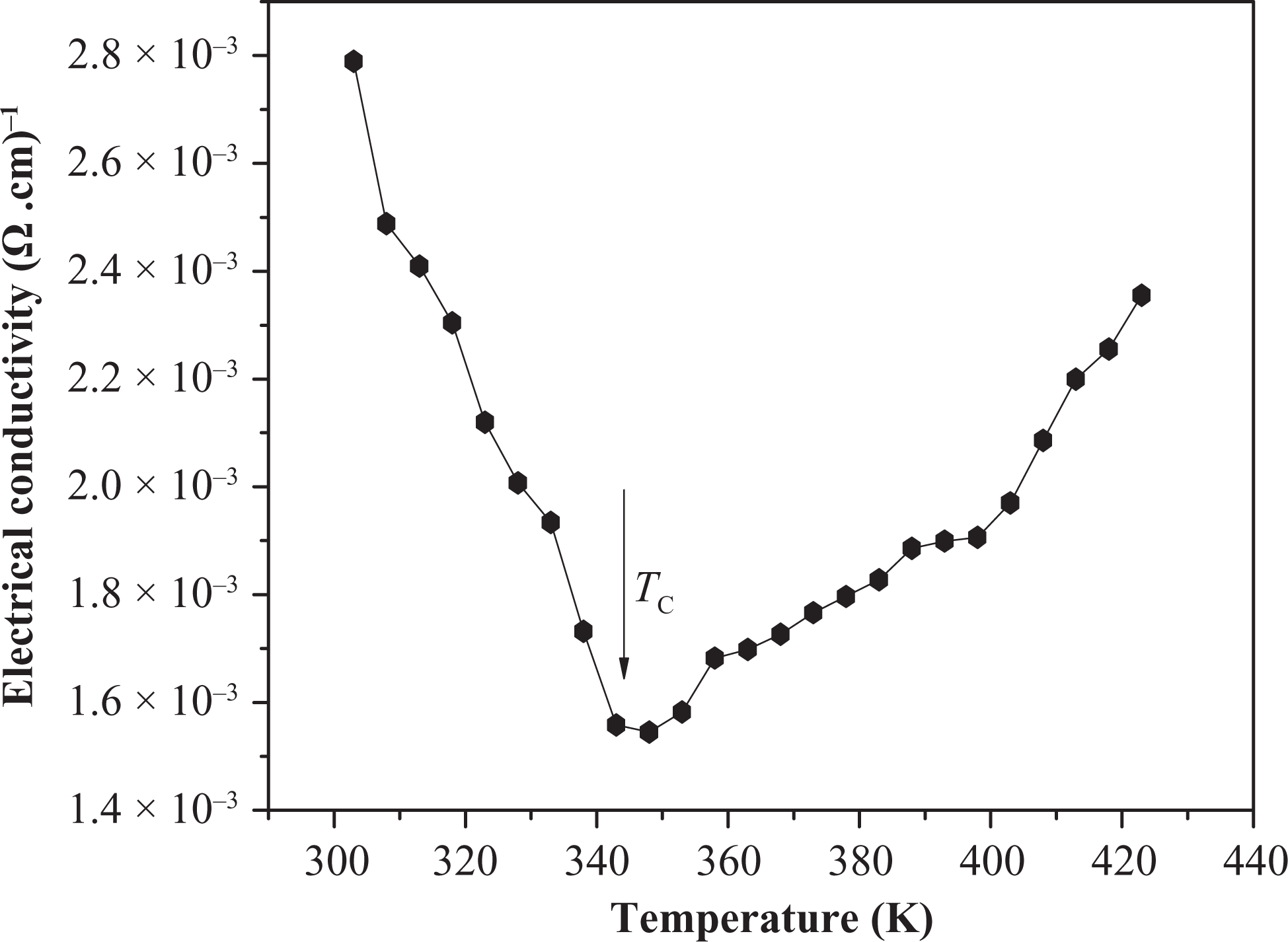
Illustration of NTCC and PTCC behavior of electrical conductivity for (25NBR/75IIR) rubber blend loaded with (50:50) N-326:N-774 carbon black ratio. NTCC: negative temperature coefficient of conductivity; PTCC: positive temperature coefficient of conductivity; NBR: nitrile butyl rubber; IIR: isoprene-isobutene rubber.
First, the conductive particles at T C have a tendency to agglomerate because of van der Waals forces and covalent bonds among some particles. Also, as at T C viscosity of the blends is being very high, the diffusion of conductive particles increases where they align to form new conducting networks, resulting in an abrupt increase in conductivity.
Second, at T C, the interparticle distance between conductive aggregates becomes so large that simultaneously the intrinsic conduction begins to appear due to the carriers of the carbon black fillers (i.e. thermal activation conduction becomes the governing mechanism). 18
Conclusion
SEM image also supports the evidence for the compatibility of NBR/IIR blend and the homogeneity observed after the addition of BIIR as a compatibilizer. N-326 (HAF) carbon black improves the thermal stability and microstructure core of the (50NBR/50IIR) rubber blend. Also, as its ratio increases, the electrical conductivity and electric heating behavior are improved. Most of the prepared blends exhibit NTCC, in comparison with other (25NBR/75IIR) rubber blend loaded with (50:50) N-326:N-774 that exhibits the combination of positive and negative coefficients. The NTCC and PTCC behavior of our blend enables it to be employed as efficient heaters.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
