Abstract
The exceptional low permeability to oxygen and water of PVDC and the retention of barrier properties to oxygen even in humid conditions explain its success for packaging in food industry as barrier layer in Polyolefins (PO) multilayer films, whose contribution to the reduction of food waste is largely recognized. Several chemical recycling technologies have emerged in the recent years that, together with methodologies based on separation of layers by the use of solvents, will strongly increase in a near future the extent of recycling of multilayer films, including the PVDC-containing ones. At the same time, it is generally accepted that for several years these technologies will be complemented by the now dominating mechanical recycling. A lack of detailed study in the literature and a general confusion between very different PVDC grades, with and without internal stabilization, led to the idea that the dehydrochlorination of PVDC would cause a low quality of the recyclate obtained by mechanical recycling of PO in presence of PVDC and a corrosion risk for the equipment. In this study, the effect of a stabilized PVDC grade developed for coextrusion, at concentration typical of real life PO mix for mechanical recycling and in suitable condition, has been investigated in a pilot scale extruder and corrosion tests were performed. In conclusion, quantities of PVDC to be found in PO streams for mechanical recycling are compatible with a PO recyclate of excellent characteristics, even at extrusion temperatures as high as 220°C, with no damage to the PO structure, with only minimum discoloration, that can be mitigated by relatively low quantities of additives of general use in PO extrusion, and with no risk of corrosion of the equipment.
Keywords
Introduction
The use and demand of plastic materials, due to the variety of available polymers with excellent properties for many applications and to the generally more favorable economical and lightweight characteristics compared to other materials, have been growing in an extraordinary way since the beginning of large-scale production in the 1950 and so their contribution to solid waste generation. 1 In particular, the largest market is packaging where a strong impulse is due to the success of single-use containers. 2 Multilayer films’ success arises from the combination of characteristics of the different polymers, in particular the presence of a barrier layer allows the preservation of food or pharmaceutical drugs characteristics. 3 On the other hand, this complexity can lead to big challenges for recycling technologies. 4 A general pressure towards reduction of plastic waste has led to different initiatives worldwide aimed to promote sustainability of packaging industry. The promotion of sustainable packaging is part of the European Green Deal. In Europe, the CEFLEX consortium 5 is a collaborative initiative representing the entire value chain of flexible packaging with the aim to develop guidelines for a circular economy. While new flexible packaging needs to be designed suitable for collection, sorting and recycling, it is clear that, in the case of food preservation, no compromise can be accepted towards a reduction of quality, safety and shelf-life, that will lead to increased food waste having a dramatic environmental footprint on its part.4,6 In 2016 only 16% of polymeric materials were collected for recycling while 40% were sent to landfills and 25% incinerated globally, so that the remaining 19% was dispersed in the environment. European countries are leading towards increasing recycling rates and, in 2018, 29.1 million tons of post-consumer plastic waste were collected in Europe, but still less than a third were finally recycled. 7 Mechanical recycling, or secondary recycling, consists of sorting of polymer waste streams, reduction of polymer waste size, followed by extrusion and it is the principal process suitable for flexible packaging, mainly constituted of polyolefins, in particular polyethylene grades and copolymers. While treatment of pure mono-material stream is relatively straightforward, mixture of plastics including multilayers of different compositions and complexity can lead to challenges in terms of processing conditions and final quality of the recycled materials.
The main application of polyvinylidene chloride (PVDC) in flexible films is its use as a barrier layer in multilayer shrink bags for meat and cheese packaging. These coextruded polyethylene (PE)/PVDC multilayer shrink films typically contain 10–25% w/w PVDC and can include ethylene vinyl acetate (EVA) up to 50% w/w. The Solvay product is PVDC from suspension polymerization of the IXAN® family, 8 a stabilized grade developed for coextrusion.
There are other multilayer barrier films for food on the market, containing PVDC introduced with coatings on BoPP substrates, where Solvay PVDC is of the Diofan® family 9 from emulsion polymerization, but the market is smaller and they contain a lower w/w % of PVDC. Barrier films for pharmaceutical applications are also based on coatings from the Diofan® family. Diofan® PVDC, being developed for coatings and not for extrusion, has no stabilization.
The main degradation mechanism of PVDC is through dehydrochlorination (DHC) 10 with HCl release and formation of conjugated double bonds revealed by discoloration of the resin. PVDC grades developed for coextrusion such as Solvay Ixan® include internal stabilizers with the scope to inhibit dehydrochlorination and to facilitate extrusion process.
In the field of plastic recycling, the reputation of PVDC is generally associated with PVC so that presence of PVDC and PVC in multilayer films is generally considered to “render package non recyclable” per definition of Association of Plastic Recyclers, APR 11 as an example with specific reference to Mechanical Recycling only.
Different approaches for the extrusion of PVDC-containing blends with the addition of external stabilizers of different nature are reported in patent literatures. For PVDC/PO the use of a combination of an organic phosphite or phosphonite and a metal salt of a fatty acid 12 is reported while in 13 a mixture of organic phosphite or phosphonite, Zn and Ca salts of fatty acids and polyols are proposed. PVDC/PVC from pharmaceutical packaging can also be stabilized with acid scavengers of the series of metal oxydes, metal hydroxides or metal carbonates. 14
Most of the additives proposed for PVDC have already been explored for PVC stabilization where a more extensive literature is available. 15
Ca and Zn thermal stabilizers have been used with acrylic acid-low density polyethylene (LDPE-g-AA) graft copolymer as compatibilizer, and linear low density polyethylene (LLDPE) as modifier to obtain from recycled LDPE/PVDC composite films blends with excellent properties. 16 The use of chlorinated polyethylene (CPE) as a compatibilizer was proposed to obtain blends from waste LDPE/PVDC/LDPE composite film in presence of Ca-Zn stabilizer as well. 17
Other approaches of recycling PVDC from multilayer films based on separation of the layers by using a solvent, 18 as well as chemical recycling methods via pyrolysis and gasification 19 are being explored by Solvay and will contribute in the future to increase the extent of PVDC recycling.
As all the innovative recycling technologies introduced in the market will coexist for years with the more diffused mechanically recycling approach, this study aims to investigate the mechanical recycling process of PVDC/PO blends present in PO streams in real life concentrations and conditions and possible methods to mitigate potential negative effects induced by the presence of PVDC, including the use of relatively low quantity of stabilizers already of general use in PO extrusion. A particular attention to evaluate if a corrosion risk is present in these conditions is a peculiar feature of this study.
The recycling process has been conducted mixing a virgin or even an already processed PVDC (in order to simulate the consumption of internal stabilizer occurring during coextrusion), at concentration expected to be found in the stream of post-consumer flexible packaging, and LLDPE grades suitable for shrink films production. Increasingly challenging conditions were tested. The results demonstrated that, with this quantity of PVDC, the recycled LLDPE blends have excellent characteristics, even at extrusion temperatures as high as 220°C, with no damage to the PO structure, with a minimum discoloration, that can be mitigated by additives that act as acid scavengers and of general use in PO extrusion, and with no risk of corrosion of the equipment.
Materials and methods
Materials
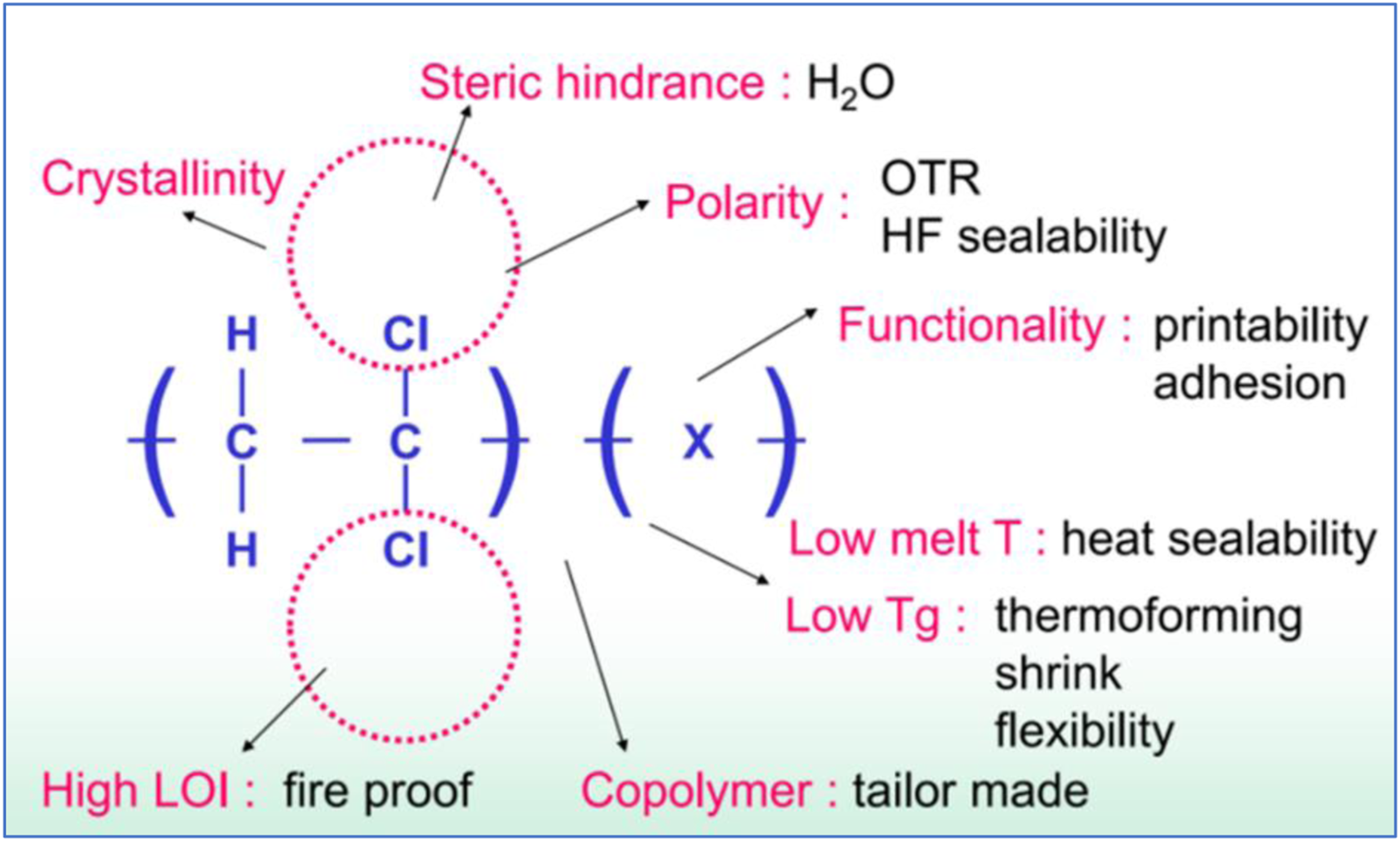
In Figure 1, PVDC structure and properties are outlined.
20
The presence of the comonomer induces an increase of thermal stability versus the homopolymer, interrupting the unstable polyvinylidenchloride chain
21
but also imparts or improve other properties such as adhesion, printability, heat sealability, flexibility, shrink, thermoforming. At the same time, a reduction of barrier properties is observed at increasing co-monomer content so a compromise has to be found. This study is focused on multilayer shrink bags recycling. PVDC content is typically 15–25% w/w of the total structure. Examples of a structure with 5 layers is reported in Figure 2. PVDC structure and related properties.
20
Comonomer X = various alkyl (meth)acrylates, vinyl chloride (VC), acrylonitrile (AN). Example of a 5-layers multilayer film: transmission optical microscopy image of a cross-section.
20

Knowing the market penetration of PVDC in flexible packaging (Figure 3) a dilution factor has been calculated and taken into account for this study. Considering 8,4 MTon/y of flexible packaging market (constituted mainly of polyolefins, principally PE) as reported by CEFLEX
5
and ∼10 kTon/y of PVDC in EU market as Solvay estimation in 2019, ∼0.12% PVDC is expected to be found on average in Polyolefin (PO) post-consumer waste from flexible packaging if completely not sorted. Introducing a safety factor, we propose the following dilution factor argument: typical “maximum average concentration” of 0.15% w/w PVDC is considered representative of composition of mixed plastic streams from polyolefins flexible packaging. In addition, to take into accounts spikes of concentrations in certain streams, tests at 1% w/w PVDC were also performed. These concentration ranges are realistic and in agreement with typical chlorine (from PVDC and/or PVC) concentration in feedstock of post-consumer plastic mix from packaging streams collected by several companies involved in mechanical and chemical recycling (data shared to Solvay in private communications by recycling companies). European market distribution of plastic (mainly polyolefin-based) packaging
5
to be compared with PVDC European market penetration in 2019 from Solvay internal data.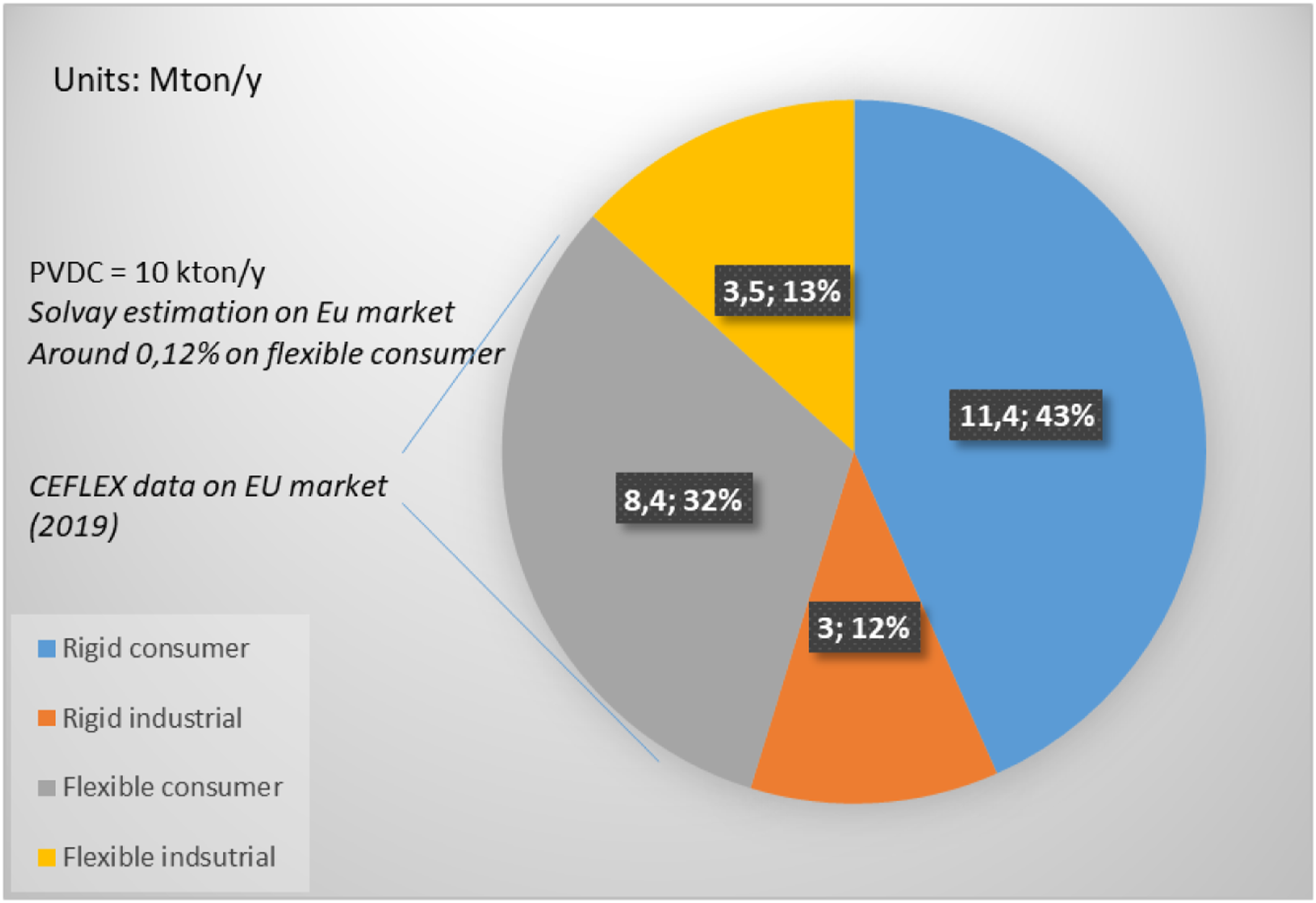
IXAN® PV 910, a commercial PVDC grade from Solvay, was chosen as representative of a grade developed for coextrusion with an internal stabilizer, obtained by suspension polymerization. 8 To simulate the reduced stability after coextrusion, a reprocessed PVDC (r-PVDC) has been prepared from pellets of virgin PVDC IXAN® PV 910 (see Preparation of pre-processed PVDC in Methods session).
As representative Polyolefin, linear low-density polyethylene (LLDPE) has been chosen, given its wide use for food contact applications, with the following main properties: • Melt Flow Rate (190°C/2.16 kg) in the range 1–3 g/10 min (Test method ISO 1133) • Density between 0.900-0.925 g/cm3 (ISO 1183) • Melting point in the range 110–125°C (UNI EN ISO 11,357-3:2018).
More specifically, the following LLDPE from Versalis have been selected, with increasing recycling challenges with respect to PVDC, such as processing temperature and decreasing MFI: LLDPE CLEARFLEX® CLD0 (LLDPE1) with MFI = 3, density = 0.900 g/cm3, melting temperature = 115°C and LLDPE FLEXIRENE® FG20 F (LLDPE2) with MFI = 1, density = 0.918 g/cm3, melting temperature = 120°C.
LLDPE1 was easily processed @180°C due to relatively higher MFI, while LLDPE2 was processed @220°C to simulate more severe recycling conditions.
For stabilization towards dehydrochlorination of PVDC during extrusion, the addition of synthetic Hydrotalcite22,24 DHT-4A®2 (from now on indicated also as Hydrotalcite or DHT-4A2), produced by Kisuma, 25 demonstrated a particular efficiency and a peculiar mechanism (see Results). In these conditions additives as CaO (Reagent plus grade, Sigma Aldrich), typically used as de-hydrant in PO extrusions, as well as CaCO3 (Socal® 312, Imerys) were found to have similar effects at concentration less than 0.5% with 0.15% r-PVDC and less than 1% with 1% r-PVDC (data not reported).
Methods
Preparation of pre-processed PVDC
In order to simulate the reduced stability after the processing to form multilayer structure by coextrusion, a reprocessed PVDC (r-PVDC) has been prepared from pellets of virgin PVDC IXAN® PV 910.
A Brabender extruder with a 30 mm barrel with 3 heating zones and a mild screw and a diameter of the die of 6 mm was used with these conditions: • Barrel temperature: 148°C/165°C/174°C - Die temperature: 178°C • Screw Speed: 10rpm.
Strands of ∼1 m length were obtained and stored at room temperature for 3 days to allow PVDC crystallization to more easily pelletize the strands. Another storage for 24 h at 40°C allowed pushing PVDC crystallization.
Cryogenic grinding
LLDPE and r-PVDC pellets, mixed with dry-ice, were both grinded to powder with a Retsch ZM 200 Ultra Centrifugal Mill (https://www.retsch.com/products/milling/rotor-mills/zm-200/function-features/), equipped with 1 mm sieves in order to obtain a particle size distribution centered around 0.4–0.6 mm. Afterwards the coarse powder was dryed at 80°C for 4h to remove the water condensate during the milling process.
Dry blend and pelletization
Dry blends were prepared in order to obtain a final 0.15% w/w or 1% w/w PVDC and the target concentrations of stabilizer (always reported in % w/w) in LLDPE matrix and mixed in a rotating drums system.
Temperature profiles for LLDPE1 (low T) and LLDPE2 (High T) blends extrusion.

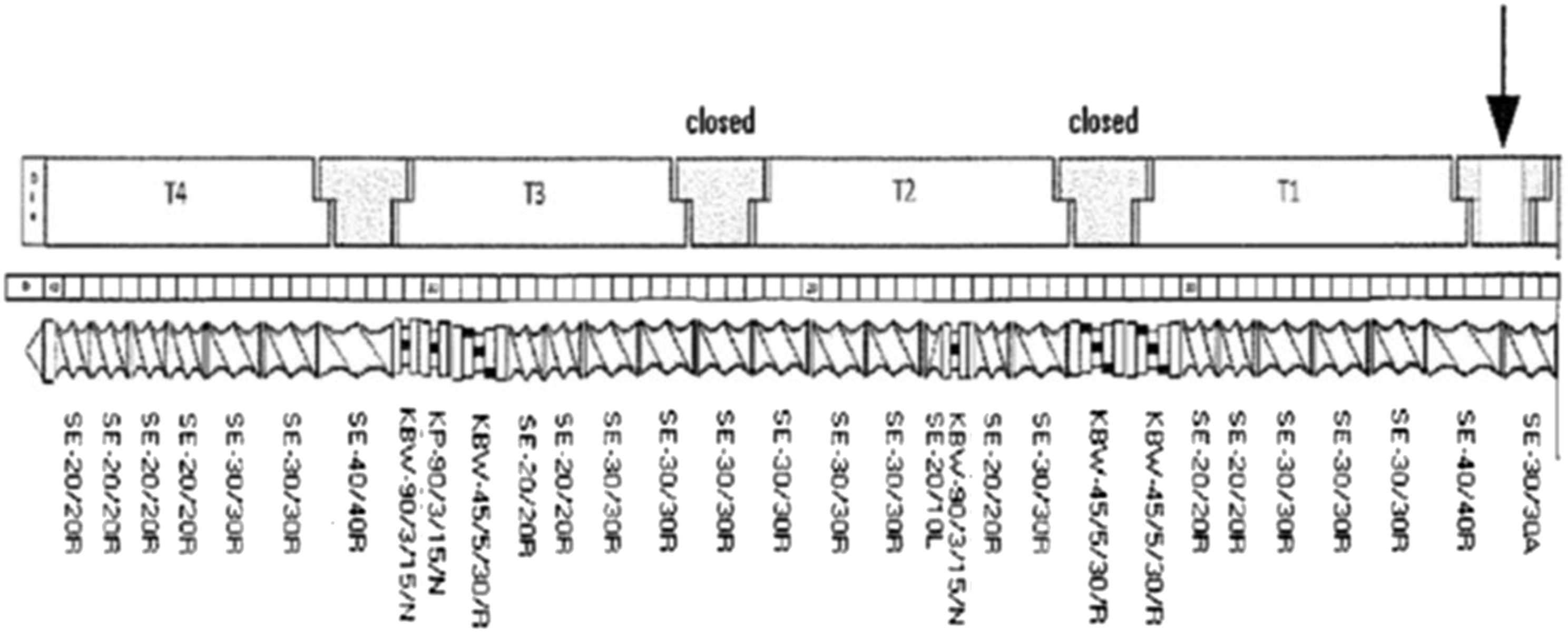
BRABENDER® TSE 20/40 extruder screw profile.
Mild screw profiles were used (Figure 4) and an optimized screw speed of 100 r/min was used to have a good output and a balance between shear stress and residence time.
Optical and tensile properties
Plates of dimensions 13 cm × 13 cm and 0.6 mm thickness were obtained by compression molding of the pellets @ 170°C. A Kapton® film was used in order to obtain a smooth and regular surface. With this kind of thickness, differences in optical properties between blends and reference materials are highlighted and the sensitivity to the formation of regions with dehydrochlorinated PVDC affecting both color and light transmission is amplified. With sample of lower thickness less differences in color and total transmission are expected, so the results we obtained are conservative in comparison to real cases, where the recyclate material is used to produce thin films from a few tens of μm to around 100–200 μm.
Total Transmission was measured on compression molded plates for 0.15% and 1% PVDC containing blends according to ASTM D1003 with a BYK Haze-Gard plus instrument. Total Transmission (TT) percent of retention versus pure LLDPE reference samples were calculated.
Delta Color (ΔE) was measured on compression molded plates of 0.15% and 1% PVDC containing blends versus pure LLDPE reference samples according to ASTM E313 with a BYK Gardner Color view colorimeter.
The tensile properties stress at break and strain at break were measured on compression molded plates according to ASTM D638 type V with an Instron 5965 Universal testing machine. Tensile properties percent of retention versus plates obtained with pure LLDPE reference samples were calculated.
Corrosion tests
Recyclers of polyolefins streams typically use extruders made in alloys not resistant to acids. To test if the quantity of HCl released, due a partial dehydrochlorination of PVDC, could induce a corrosion risk, a corrosion study was performed in the more challenging conditions tested in our experiments, that is at 220°C with LLDPE2, the grade with lower MFI and processed in higher shear stress conditions in comparison to LLDPE1. Pellets of pure LLDPE2 reference, blends of LLDPE2 containing 0.15% or 1% w/w without and with the addition of stabilizers were introduced at the bottom of glass sealed containers.
Polished specimens of Carbon Steel alloy, having a particularly low resistance to corrosion from HCl, were introduced both in the immersed phase (to simulate corrosion in the melt phase in contact with the extruder), that is inside the mass of the pellets, and in the gas phase (to simulate corrosion in vent lines) as can be seen in Figure 5. Corrosion test setup - C-steel plates in gas phase and immersed in the pellets to simulate long-term effect on vent lines and extruder respectively.
A cyclic test was set up with target temperature 220°C for 14 days, alternating 1 day @220°C and 1 day @60°C. This kind of test is particularly challenging, as the reduction of temperature at 60°C induces a condensation of the fumes that could initiate a corrosion process, especially in the gas phase. Corrosion rates were determined by measuring difference in weight before and after the tests and expressed in loss of thickness per unit of time (μm/y) as the surfaces of the metal plates corroded uniformly across the area exposed.
Water content
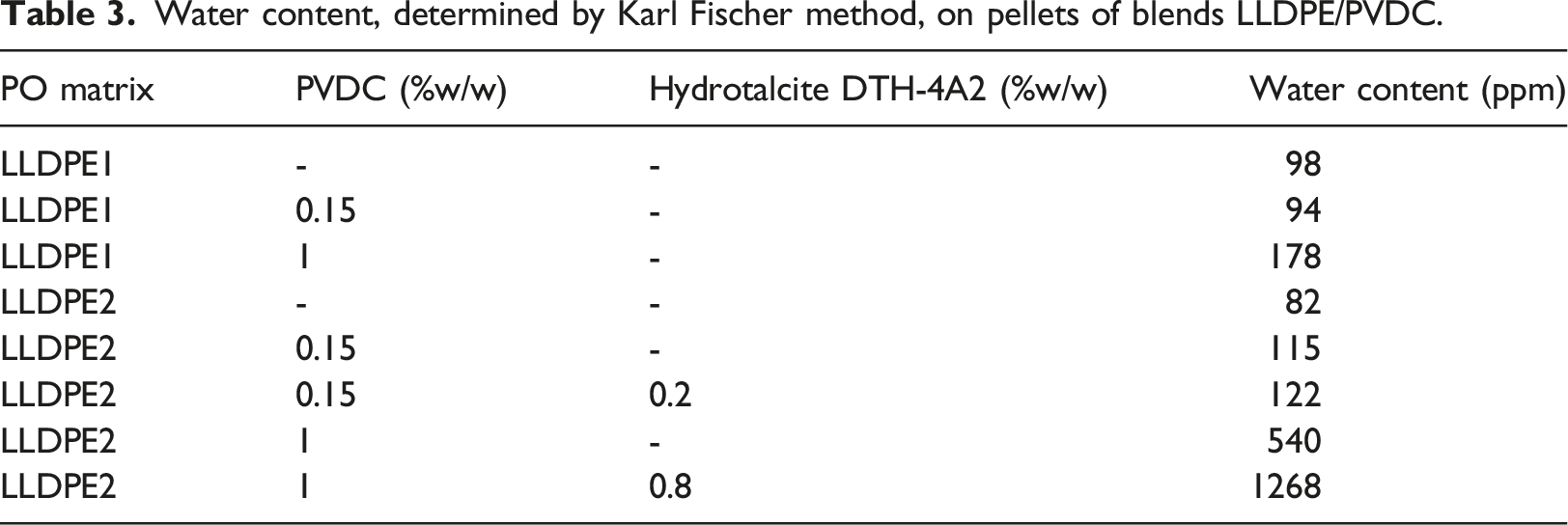
In the extrusion of blends, the extrudate was cooled in water bath and then pelletized. As no drying step was included, we measured the water content in the pellets. Humidity is in fact of great importance in corrosion tests, considering that humid HCl is more corrosive than dry HCl on steel alloys. Water content was measured by Karl Fischer methods by means of a Mettler C30 coulometric titrator equipped with a Stromboli oven. Stripping temperature was set at 135°C.
Dehydrochlorination test
The extent of dehydrochlorination (DHC) of PVDC was measured by means of 895 Professional PVC Thermomat by Metrohm. Specimen of blends (0.5 g) were introduced in the reaction vessels and the release of HCl at a fixed temperature was transferred by a stream of nitrogen into the measuring cell filled with distilled water, where it was detected by conductivity measurement. Three-fold repetitions for each sample were performed.
Results
Appearance of PVDC/PE Pellets
For LLDPE1, processed in the milder conditions using the lower T profile, there was negligible evidence of PVDC degradation as can been observed on the pellets of the blends barely distinguishable from the virgin LLDPE1, even in presence of 1% virgin PVDC (Figure 6). PVDC dehydrochlorination should be witnessed by a discoloration, linked to the formation of conjugated double bonds.
21
Photo of pellets of virgin LLDPE1 and of blends PVDC/LLDPE1 obtained by extrusion at 180°C.
For LLDPE2 processed at 220°C, a slight discoloration towards yellow color, coming from partial degradation of PVDC, appears at 0.15% r-PVDC and more at 1% r-PVDC, but the addition of stabilizers acting as acid scavenger were particularly effective in minimizing this effect (see Figure 7 and below). Photo of pellets of virgin LLDPE2 and of blends PVDC/LLDPE2 obtained by extrusion at 220°C with and without addition of Hydrotalcite DHT-4A2).
Optical properties on compression molded plates
Optical properties on 0.6 mm plates for PVDC/LLDPE1 blends obtained by extrusion at 180°C and PVDC/LLDPE2 blends obtained by extrusion at 220°C.
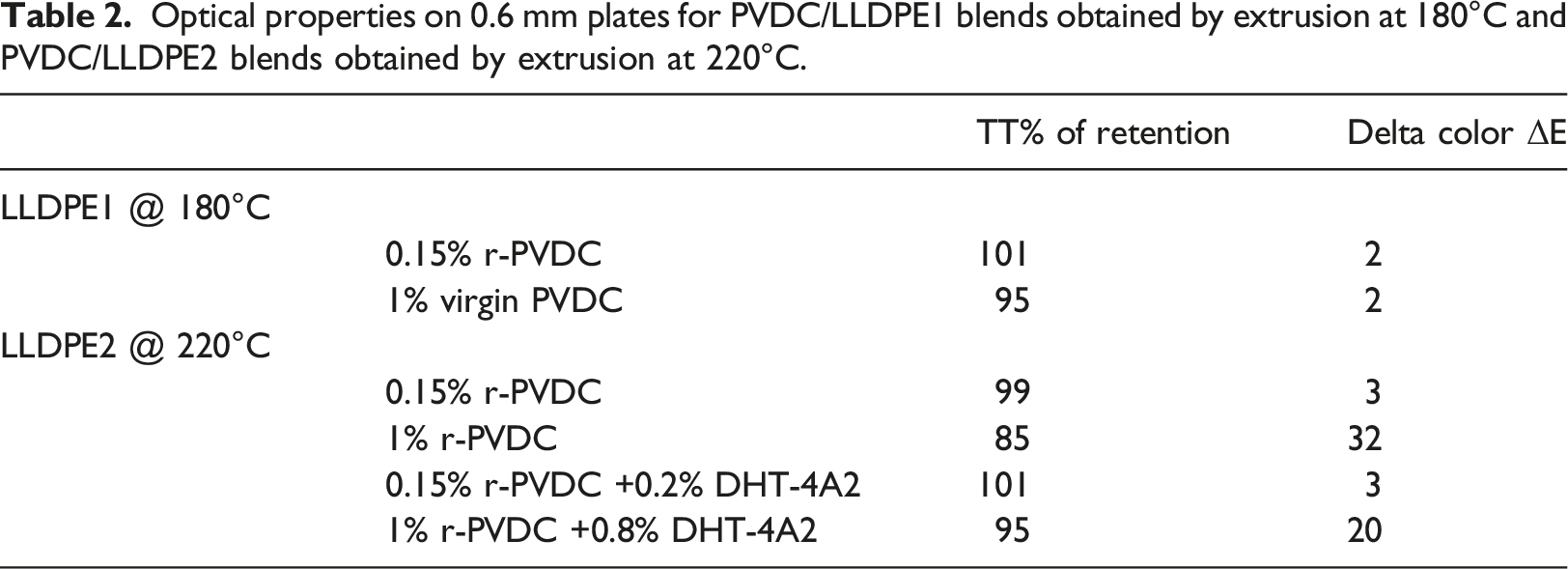
For LLDPE1 @180°C, retention of Optical properties is excellent up to 1% v-PVDC. Retention of TT is 95% and ΔE vs reference LLDPE1 is 2. This color trend is similar to the one observable by visual inspection of the pellets as shown in Figure 6.
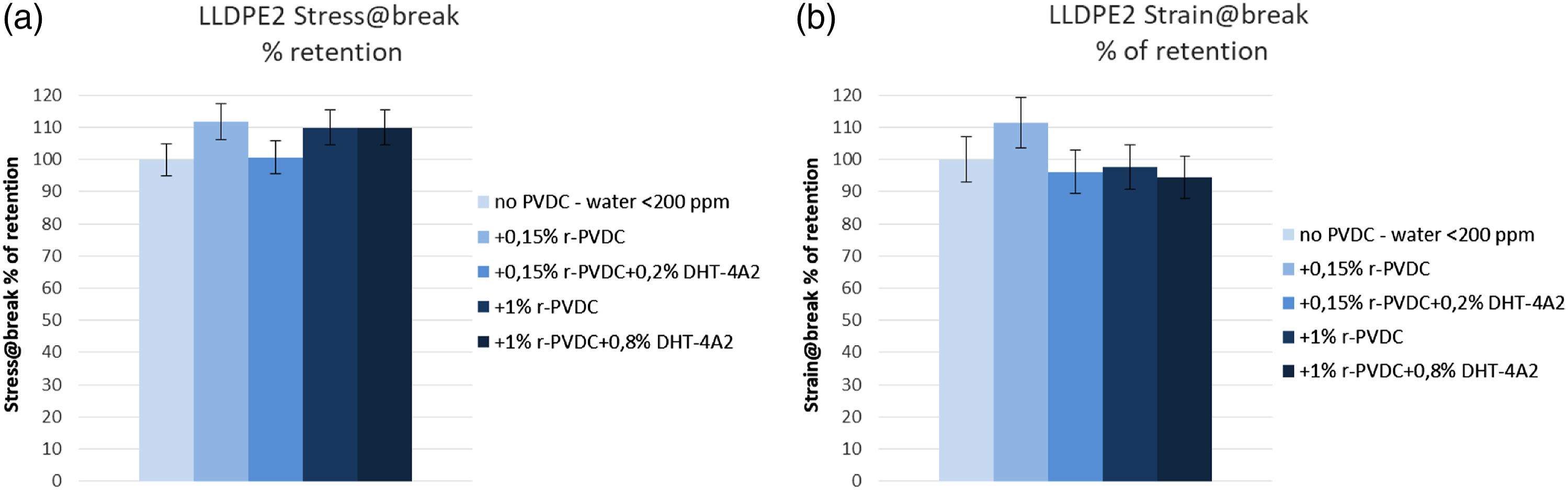
For LLDPE2 blends extruded at 220°C, retention of optical properties is excellent at 0.15% r-PVDC: TT retention is 98% and ΔE = 3. With 1% r-PVDC, TT retention moves to 85% and ΔE to 32 respectively, but Hydrotalcite is effective to bring TT retention to 95% and reduce ΔE to 20. These measurements represent quantitatively the qualitative trend observable by visual inspection of the pellets as shown in Figure 7. We tested other stabilizers acting as acid scavenger and of extended use in polyolefin processing, such as CaO (widely used as dehydrating agent) and CaCO3, (a popular filler for PO) and we obtained similar results in term of stabilization at the same concentration shown for Hydrotalcite. The latter is particularly interesting, being a basic compound, but having a peculiar different scavenging mechanism for the HCl acidity, through an anion exchange mechanism reported below in corrosion test section, and being able to absorb a small amount of water in its structure. 22
Tensile properties on compression molded plates
The retentions of the tensile properties were excellent for both LLDPE1 (Figure 8) and LLDPE2 (Figure 9) blends, always higher than 80% for all samples and typically in the range 85–110%. No general trend is evident with r-PVDC concentration or addition of stabilizer, witnessing the stability of the LLDPE matrices in all conditions and no influence of PVDC dehydrochlorination. Tensile properties on 0.6 mm plates for PVDC/LLDPE1 blends obtained by extrusion at 180°C. (a) Percent of retention of Stress at break versus virgin LLDPE1; (b) Percent of retention of Strain at break versus virgin LLDPE1. Tensile properties on 0.6 mm plates for PVDC/LLDPE2 blends obtained by extrusion at 220°C. (a) Percent of retention of Stress at break versus virgin LLDPE2; (b) Percent of retention of Strain at break versus virgin LLDPE2.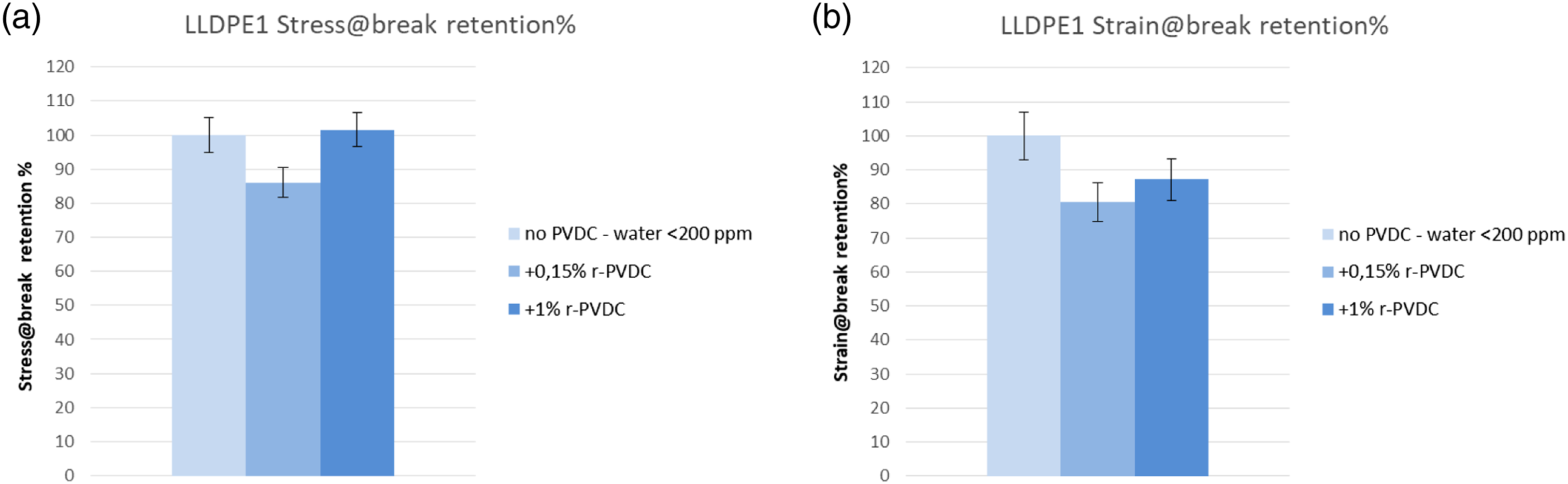
Water content on blends
Water content, determined by Karl Fischer method, on pellets of blends LLDPE/PVDC.
Corrosion tests on blends
Resistance to corrosion of metals/alloys in function of corrosion rate.

In Figures 10 and 11 are reported photographs of Carbon Steel specimens after cyclic testing at 220°C in molten (immersed) and gas phase respectively with 0, 0.15 and 1% w/w PVDC and after cleaning in citric acid. Photo of Carbon Steel specimens after cyclic tests at 220°C immersed in molten phase of LLDPE2 blends with: (a) 0% w/w PVDC; (b) 0.15% w/w PVDC; (c) 1% w/w PVDC. Photo of Carbon Steel specimens after cyclic tests at 220°C exposed to gas phase of LLDPE2 blends with: (a) 0% w/w PVDC; (b) 0.15% w/w PVDC; (c) 1% w/w PVDC.

A slight color change is visible only in plate immersed in molten phase of 1%w/w PVDC blend (Figure 10). No visible signs of corrosion are evident and no indication of a pitting mechanism was observed, so that a uniform corrosion mechanism can be hypothesized.
In Figures 12 and 13 are reported the corrosion rates measured in dynamic tests with LLDPE2 at 220°C in molten (immersed) and gas phase respectively. Corrosion rate after cyclic tests at 220°C measured in plates immersed in molten phase, simulating corrosion risk inside the extruder. (a) r-PVDC 0.15% in blend with LLDPE2 without and with 0.2% of Hydrotalcite; (b) r-PVDC 1% in blend with LLDPE2 without and with 0.8% of Hydrotalcite. Corrosion rate after cyclic tests at 220°C measured in plates in gas phase phase, simulating corrosion risk in the vent line. (a) r-PVDC 0.15% in blend with LLDPE2 without and with 0.2% of Hydrotalcite; (b) r-PVDC 1% in blend with LLDPE2 without and with 0.8% of Hydrotalcite.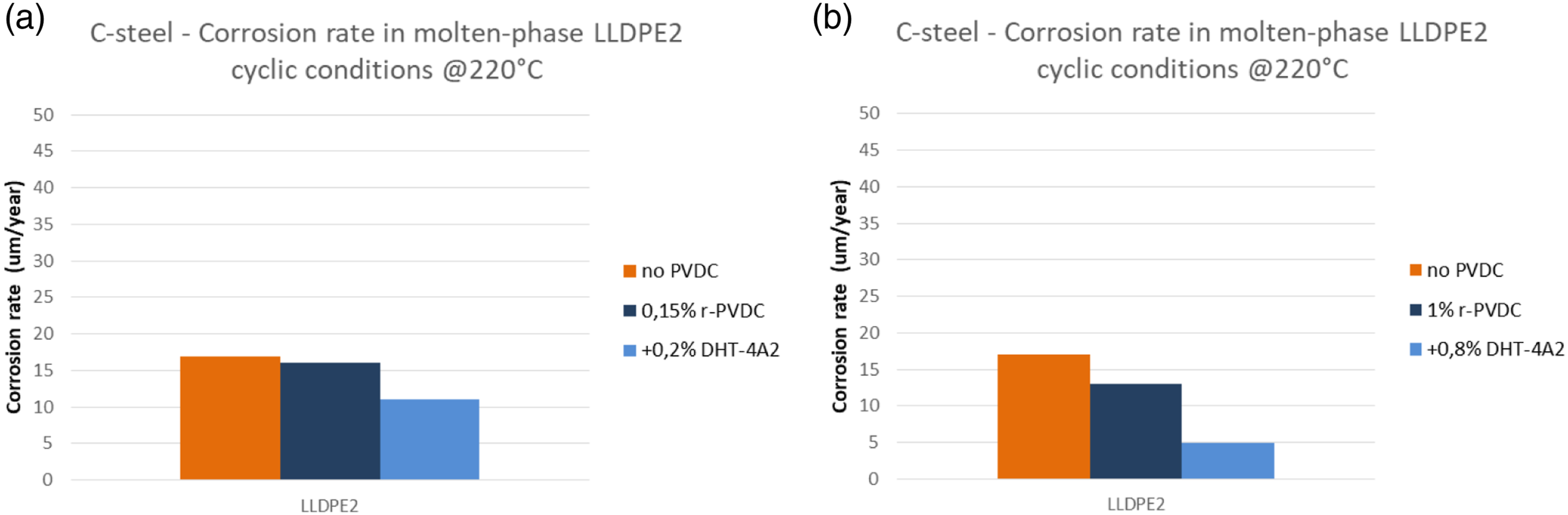
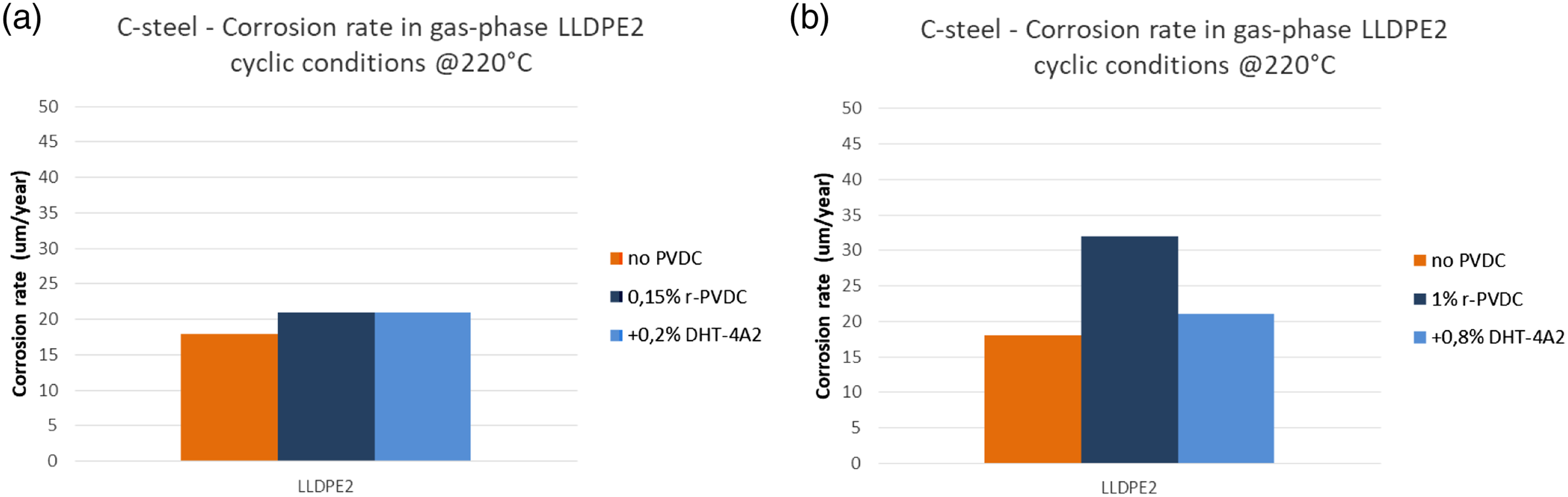
As can be seen a ‘blank’ value of corrosion rate for pure LLPDE2 was measured of around 20 μm/y.
In presence of r-PVDC up to 1%, corrosion rates were measured of the same order of magnitude and still much lower than the threshold of 100 μm/y, indicating that corrosion risks are minimum and not influenced in significant way by PVDC presence. As expected a slightly higher rate was measured in gas phase, where the condensation of fumes can be more effective. A particular attention to the aspiration of the vent lines will further reduce any risk.
Considering the water content in the blends (Table 3) due to the absence of a drying step in the process we followed, we can say that if a drying step is introduced that is able to reduce water content to a value <500 ppm or more preferably to <300 ppm, as can be easily reached, the corrosion risks will be kept even lower. Some effect of stabilization can be seen, especially at 1% r-PVDC by using the Hydrotalcite, which is in fact used in polyolefin industry for its ability to capture Cl traces present as residuals of polymerization catalyst. The particularly efficient mechanism is based on an ion exchange between chloride ions and the carbonate ions present in the synthetic hydrotalcite structure as can be seen in Figure 14.
22
Scavenging mechanism of HCl in hydrotalcite structures from reference.
22
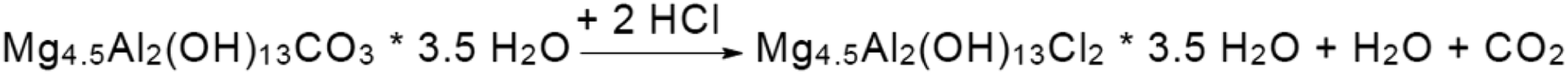
Effect of HCl scavengers: dehydrochlorination test
The effect of HCl scavengers was quantitatively determined using the DHC test described in Methods section.
Two typical curves are shown in Figure 15 for two samples, which are different only Water conductivity variation as a function of time by heating at 200°C two identical compounds, except for the content of CaCO3.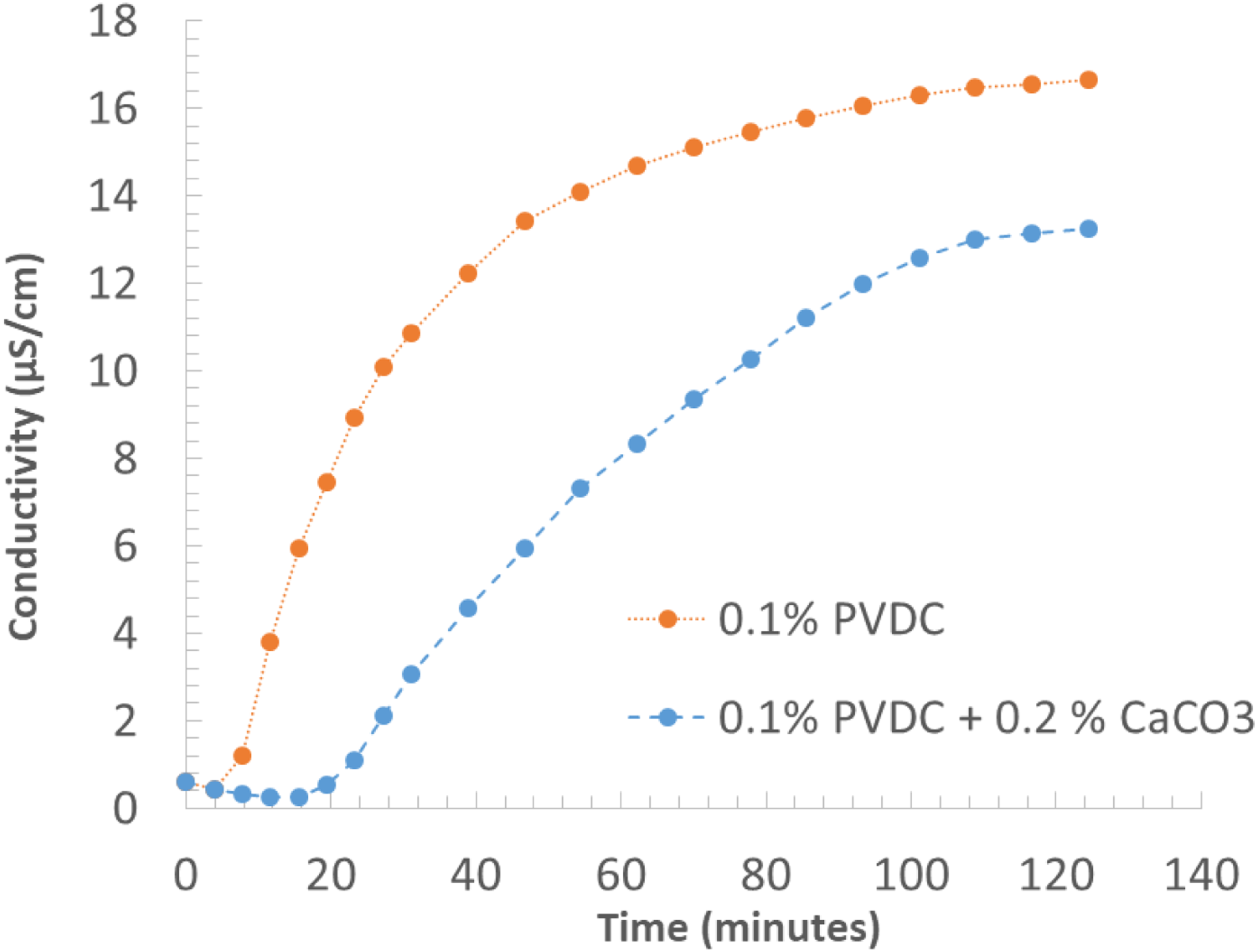
for the content of CaCO3 as acid scavenger. Both the curves have similar shape and they show an initial induction time with no increase of conductivity, followed by a steep increase and then by a final “plateau” region. The effect of the presence of CaCO3 is evident in all the curves globally reducing the overall amount of released HCl. Moreover it is quite significant that the induction time, estimated graphically as the intercept with the time axis of the tangent to the curve in the initial steep conductivity increases, becomes much longer than for neat PE + PVDC compounds, exceeding the typical residence time in an extruder. The methodology, based on conductivity measurements, is particularly sensitive and allows the determination of the very beginning of the dehydrochlorination process.
The effect of the temperature and of the acid scavenger are summarized in Figure 16. It is quite evident that though the induction time decreases with temperature, in the presence of CaCO3, it remains larger than 8 min even at 220°C. Dependence of the induction with temperature for a neat PE + PVDC compound and for the same compounds containing Calcium Carbonate as acid scavenger.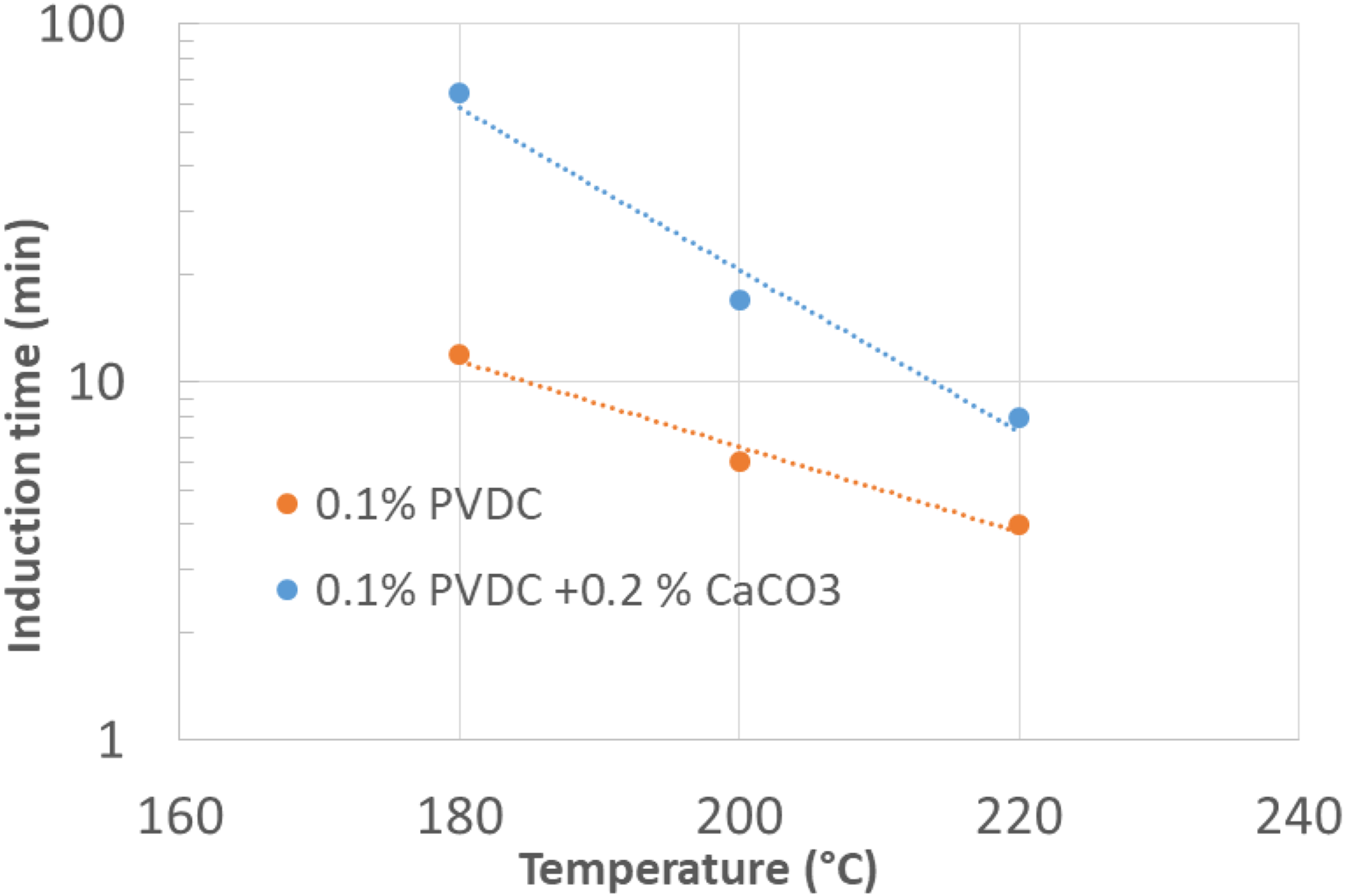
Discussion
Based on the results obtained some conclusions can be drawn. The retentions of tensile properties for all the blends were excellent, demonstrating the absence of any degradation of the LLDPE structure.
The influence of r-PVDC on optical properties (total transmission retention and color difference) of the blends, measured in conservative conditions versus real life films for packaging, i.e. with plates having relative high thickness of 0.6 mm, was negligible in the milder conditions, that is at 180°C with the more easily processable (higher MFI) LLDPE1 grade. In more challenging conditions, 220°C and higher shear with LLDPE2 of lower MFI, some measurable loss of transmission and a discoloration towards yellow color were detected, witnessing some dehydrochlorination of PVDC. Discoloration can be minimized by additions of fillers acting as acid scavenger and of common use in processing of PO, such us CaO, CaCO3 or synthetic Hydrotalcite DHT-4A2. The quantity sufficient to have an appreciable effect is of the order of 80–100% the quantity of PVDC present in the blends. Additionally, these additives delay the onset of dehydrochlorination and the overall amount of HCl which can interact with extruder barrels and screws.
A particularly challenging dynamic corrosion tests on C-steel plates with the LLDPE2 blends, lasting for 14 days and alternating 1 day at 220°C and 1 day at 60°C, showed corrosion rates less than 50% of the threshold (100 μm/y) which is considered as relevant to assess the beginning of the corrosion risk.
In conclusion, our study demonstrated that, up to 220°C, the mechanical recycling of PO in the presence of PVDC content normally included in the streams, allows to obtain a recycled material having very good properties and no evidence of corrosion risk in these conditions was shown by dynamic corrosion tests.
Footnotes
Acknowledgements
Authors warmly thank Dr. Laurent Diguet and Daniel Alves-Fernandez for the corrosion tests and Dr. Jerome Vinas for dehydrochlorination test measurements.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the Solvay Specialty Polymers Italy SpA and Solvay SA.
