Abstract
This research focuses on utilization of kapok husk (KH) as a natural filler in linear low-density polyethylene (LLDPE) ecocomposites. The effect of KH content and coupling agent on tensile properties, thermal properties, water absorption behavior, and morphology of ecocomposites were studied. The addition of KH had increased tensile modulus and water absorption of ecocomposites, whereas tensile strength and elongation at break decreased. However, the polyethylene-grafted acrylic acid (PEAA) was used as a polymeric-coupling agent to enhance the properties of LLDPE/KH ecocomposites. The incorporation of PEAA improved the tensile strength, tensile modulus, crystallinity, and thermal stability as well as reduced the water absorption of LLDPE/KH ecocomposites. The improvement of those properties was caused by the enhanced interfacial bonding, which was evidenced by scanning electron microscopy. The Fourier transmission infrared spectra also confirmed the presence of ester linkage between PEAA and KH.
Keywords
Introduction
In Malaysia, kapok (Ceiba pentandra) tree is heavily cultivated to obtain the kapok cotton and seed. In kapok industry, the kapok cotton is used to fill pillows and mattress, and kapok seeds are processed to obtain seed oil that has similar properties to cotton seed oil. However, the kapok husk (KH) is the outer hard shell of kapok fruit and most of it discarded as waste material. The resource of KH is readily abundant but it does not have any economic value; thus, the research to comprehensively utilize KH as a filler for plastic materials is underway.
Nowadays, ecocomposite materials made from agricultural waste and plastic material have garnered interest among researchers and industries ecological and economic advantage. 1 –4 In recent years, there are numerous eco-friendly products made from ecocomposite materials that have been successfully marketed. The most well-known example, such as IKEA injection molded furniture using wood flour-filled polypropylene composite material. 1,5 In Malaysia, eco-tableware made from rice husk-filled plastic ecocomposites had been marketed by Melsom Biodegradable Enterprise. 4,6 Currently, the developments of linear low-density polyethylene (LLDPE)/KH ecocomposites have potential to produce eco-friendly packaging product in our university (as displayed in Figure 1).

Eco-friendly packaging product from LLDPE/KH ecocomposites. LLDPE: linear low-density polyethylene; KH: kapok husk.
Commonly, the poor filler dispersion and weak filler–matrix adhesion are always the main issues during compounding of natural filler and plastic material. 1–2,7 –10 Usually, reactive copolymer is used as a coupling agent in the production of natural filler-based ecocomposites, such as grafted polymer with maleic anhydride 6,11 –18 or acrylic acid. 19 –21 Those coupling agents are highly reactive to hydroxyl group on natural filler at high processing temperature. The reaction between filler and coupling agent generated a covalent linkage, which allowed the long polymer chains to attach on the filler surface, providing a strong interfacial bonding between natural filler and matrix. Many researchers reported that the addition of polyethylene (PE)-grafted acrylic acid (PEAA) had significantly enhanced the properties of ecocomposites with palm kernel shell17–18 and wood flour. 21
Recently, the utilization of KH in ecocomposites has not been fully explored. Against this background, the current research has been undertaken to utilize KH as a natural filler in LLDPE ecocomposites. The present study is to evaluate the effect of PEAA on the tensile, thermal, water absorption, and morphology properties of LLDPE/KH ecocomposites.
Experimental
Materials
The KH was collected from kapok plantation, Perlis, Malaysia. First, the KH was dried in a circulated air flow oven at 80°C for 24 h. Then, the KH was crushed and ground into fine powder. The average particle size of KH was 80 µm, which was analyzed using Malvem Particle Size Analyzer Instrument (Italy). The LLDPE with grade of LL0220SR (melt flow index value: 2 g 10 min−1 at 190°C) was supplied by PT Titan Petrokimia Nusantara (Indonesia). The PEAA was supplied by Sigma Aldrich (Penang, Malaysia). The composition of PEAA was 80 wt% PE and 20 wt% acrylic acid.
Compounding and molding procedures
The LLDPE/KH ecocomposites were prepared using Z-Blaze mixer (MCN ELEC Co., Taiwan) with counter-rotating mode at 160°C and 50 r min−1. The LLDPE/KH biocomposites were compounded according to the formulation given in Table 1. The compounding sequences involved were as follows: (i) the LLDPE resin was transferred into a compounding chamber for 7 min until it fully melted and (ii) the KH was incorporated to melt LLDPE and compounded for 8 min. The total compounding time used was 15 min. All the LLDPE/KH compounds weremade into thin sheets with 1 mm thickness using hotpress machine model GT 7014A (Gotech, Taiwan). The molding cycles involved were as follows: (i) the compound was preheated at 160°C for 4 min; (ii) the compound was compressed under pressure at 100 kgf cm−2 for 1 min; and (iii) cooling under similar pressure for 5 min. Next, the LLDPE/KH ecocomposite sheets were cut into tensile bars using a dumbbell cutter with dimensions according to ASTM D638 type IV. 22
Formulation of LLDPE/KH ecocomposites.
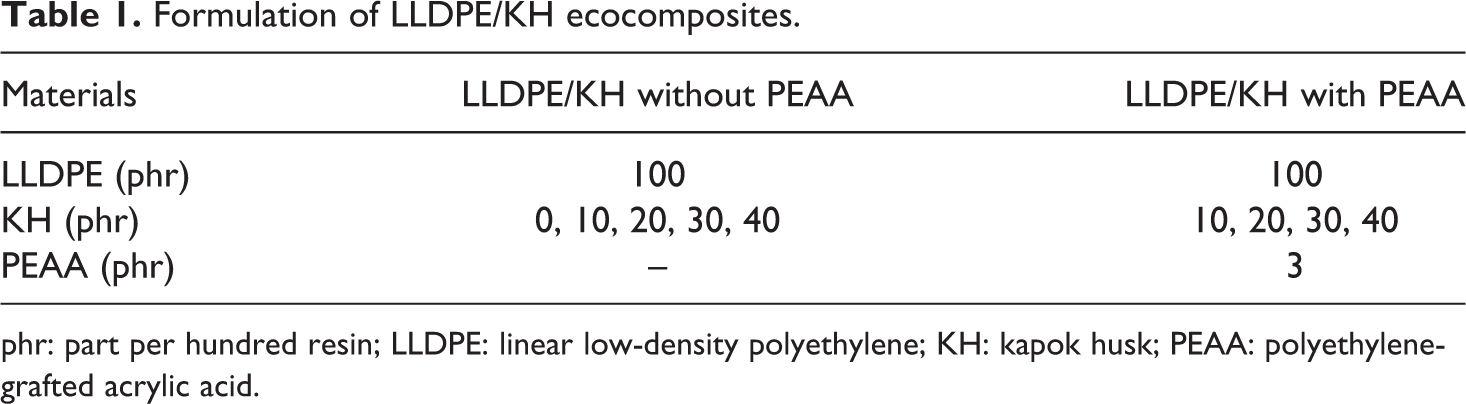
phr: part per hundred resin; LLDPE: linear low-density polyethylene; KH: kapok husk; PEAA: polyethylene-grafted acrylic acid.
Tensile testing
The tensile testing was carried out using an Instron Universal Testing machine (model 5566; Norwood, Massachusetts, USA). The test was performed at 23 ± 2°C with crosshead speed of 30 mm min−1 according to ASTM D638. 22 The values for tensile properties were the mean of measurements performed on seven samples.
Water absorption
The samples of LLDPE/KH ecocomposites with dimension 30 × 25 × 1 mm3 were immersed in distilled water at room temperature (25°C). The water absorption was determined by weighing the sample at regular intervals. A Mettler Balance Model AJ150 (Columbus, Ohio, USA) with precision of ±1 mg was used. The percentage of water absorption (moisture content (M t)) was calculated by the formulation given below:

where W d and W n are original dry weight and weight after exposure, respectively.
Morphological analysis
The tensile fracture surfaces of the LLDPE/KH ecocomposites were sputter coated with a thin layer of palladium. The samples were analyzed using a scanning electron microscope (SEM) model JEOL JSM-6460 LA (Japan) at 15 keV.
FTIR spectroscopy
PerkinElmer Spectrum Fourier transmission infrared (FTIR; Model Paragon 1000, Germany) was used to characterize functional groups in LLDPE/KH biocomposites with and without PEAA. The attenuated total reflectance method was used. The specimens were recorded with four scans in the frequency range of 4000–650 cm−1 with a resolution of 4 cm−1.
DSC analysis
Differential scanning calorimetric (DSC) analysis was evaluated using a DSC Q10 (Research Instruments, Lebanon, Oregon, USA). The LLDPE/KH ecocomposites were cut into small pieces and placed in close aluminum pan with the weight in the range of 7 ± 2 mg. The specimen was heated from 30°C to 150°C with the heating rate of 10°C min−1 under nitrogen environment. The nitrogen gas flow rate was 50 ml min−1. The degree of crystallinity of ecocomposite (X c) can be evaluated from DSC data by the following equation:

where ΔH
f was the heat fusion of the ecocomposites and
Thermogravimetric analysis
Thermogravimetric analysis (TGA) was evaluated using TGA Pyris Diamond PerkinElmer apparatus. The sample was cut into small pieces about 7 ± 2 mg in weight and was placed into a platinum crucible. Then, the sample was heated from 30°C to 700°C at a heating rate of 10°C min−1 under nitrogen atmosphere condition with the nitrogen gas flow rate of 50 ml min−1.
Results and discussion
Tensile properties
Figure 2 illustrates the effect of KH content and PEAA on tensile strength of LLDPE/KH ecocomposites. The incorporation of KH decreased the tensile strength of LLDPE/KH ecocomposites. This result trend is similar to that of previous study on ecocomposites with particle natural filler. 1 –4,6 –8 Generally, natural filler in form of particles has low aspect ratio and its efficiency of stress transfer is poor. 1,7 Therefore, the addition of KH reduced the tensile strength of LLDPE matrix. Moreover, the weak interfacial bonding between hydrophilic KH and hydrophobic LLDPE matrix also assigned to poor stress transfer between filler–matrix. However, the LLDPE/KH ecocomposites with PEAA exhibited higher tensile strength than LLDPE/KH ecocomposites without PEAA. The increases of tensile strength of ecocomposites are due to the coupling effect of PEAA. The PEAA probably covalently bonded or hydrogen bonded to KH surface and enhanced the interfacial bonding between the filler and thematrix. 17–18
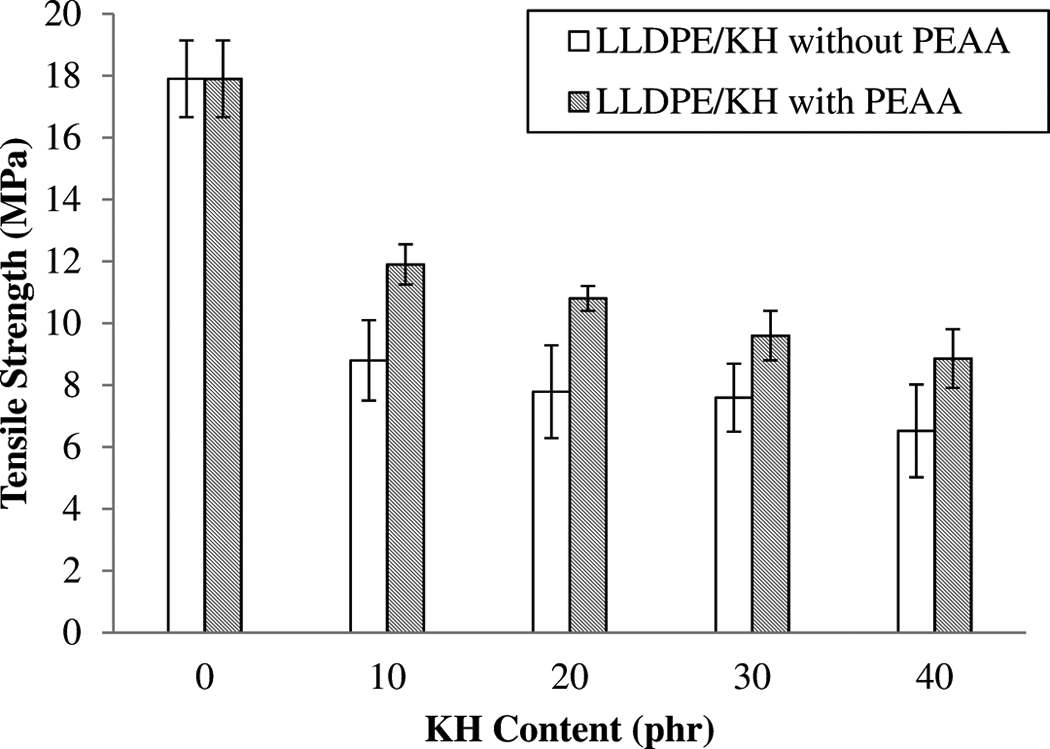
The effect of PEAA on tensile strength of LLDPE/KH ecocomposites. LLDPE: linear low-density polyethylene; KH: kapok husk; PEAA: polyethylene-grafted acrylic acid.
The elongation at break of LLDPE/KH ecocomposites with and without PEAA is shown in Figure 3. It can be observed that the elongation at break dramatically dropped due to the addition of KH. This can be explained by the presence of KH particles as stress concentrator, which might initiate the micro-crack when the stress was applied. Thus, the LLDPE/KH ecocomposites are easily fractured after a little elongation at break. This was a common observation that was also found in previous study and other researchers. 11–12,24,25 Nevertheless, the addition of PEAA further decreased the elongation at break of LLDPE/KH ecocomposites. This is because the PEAA enhanced the interfacial interaction between KH and LLDPE matrix, which generated a stronger interface bonding that inherent the polymer chain mobility. As a result, the ductility of LLDPE matrix reduced.
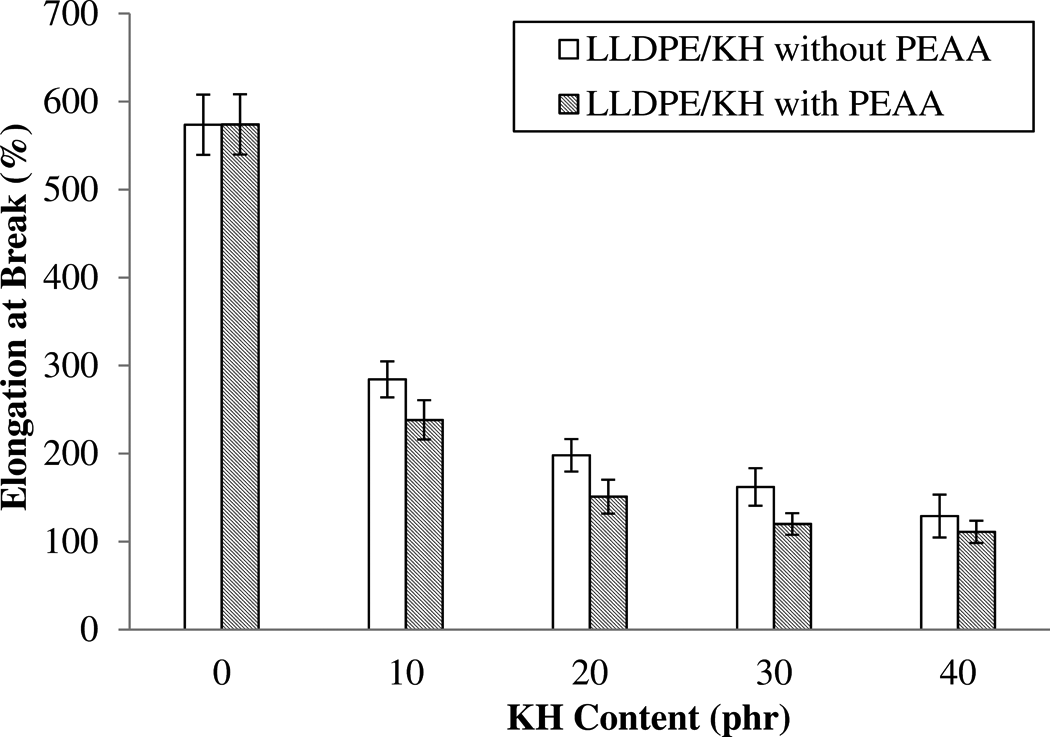
The effect of PEAA on elongation at break of LLDPE/KH ecocomposites. LLDPE: linear low-density polyethylene; KH: kapok husk; PEAA: polyethylene-grafted acrylic acid.
Alternatively, the increases in amount of KH content caused an increase in tensile modulus of LLDPE/KH ecocomposites (as shown in Figure 4). The tensile modulus probably increased with KH content because of the filler being more rigid than the matrix, which attributed to stiffer and rigid ecocomposites. A similar result was also reported by other researchers. 13–14,17,18 Consequently, the tensile modulus of LLDPE/KH ecocomposites was increased by the addition of PEAA. The increment of tensile modulus is due to the improvement of interfacial bonding between KH and LLDPE matrix. Salmah et al. 18 also reported that the tensile modulus of palm kernel shell-filled low-density PE ecocomposites was enhanced by PEAA.
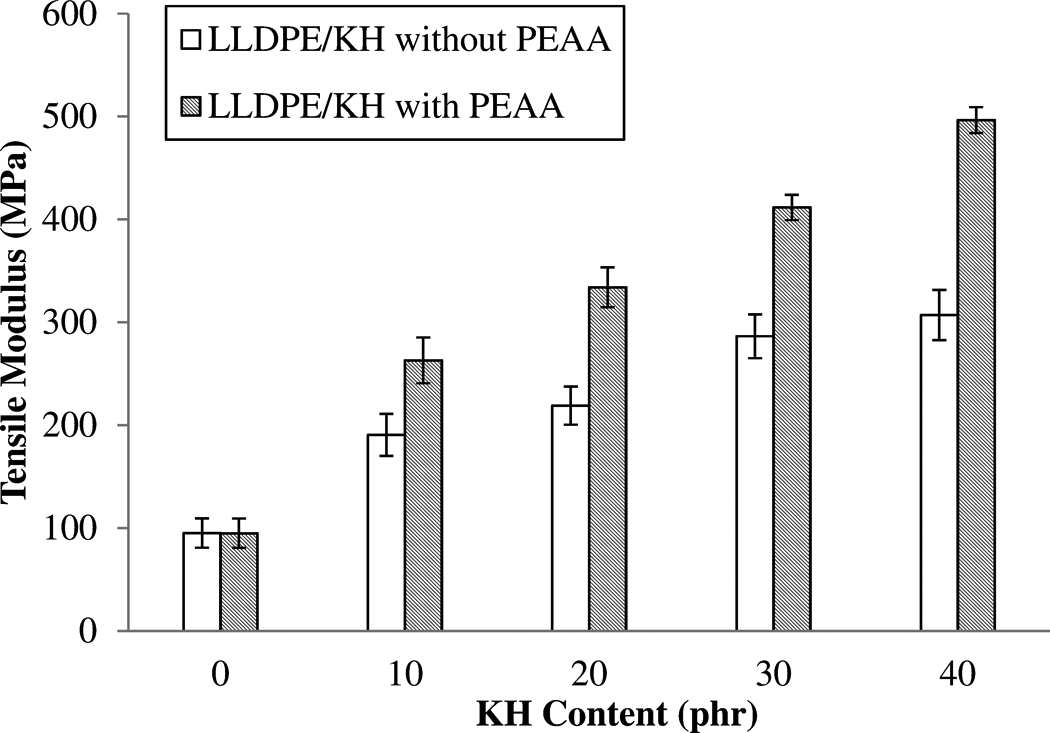
The effect of PEAA on tensile modulus of LLDPE/KH ecocomposites. LLDPE: linear low-density polyethylene; KH: kapok husk; PEAA: polyethylene-grafted acrylic acid.
Morphological properties
The SEM micrograph of tensile fracture surface of LLDPE/KH ecocomposites with PEAA at 20 and 40 part per hundred resin (phr) of KH content is illustrated in Figure 5(a) and (b), respectively. The SEM micrographs show the detachment of KH and filler pull out from matrix. This indicated poor filler–matrix interaction between KH and LLDPE matrix. Figure 5(c) and (d) illustrates the SEM micrograph of LLDPE/KH ecocomposites with PEAA at 20 and 40 phr of KH content, respectively. The SEM micrographs showed that the KH was embedded in the matrix and less filler pull out can be observed. It can be seen that the KH bonded with PEAA had better wetting with LLDPE matrix. This indicated the strong interfacial bonding between KH and LLDPE matrix.
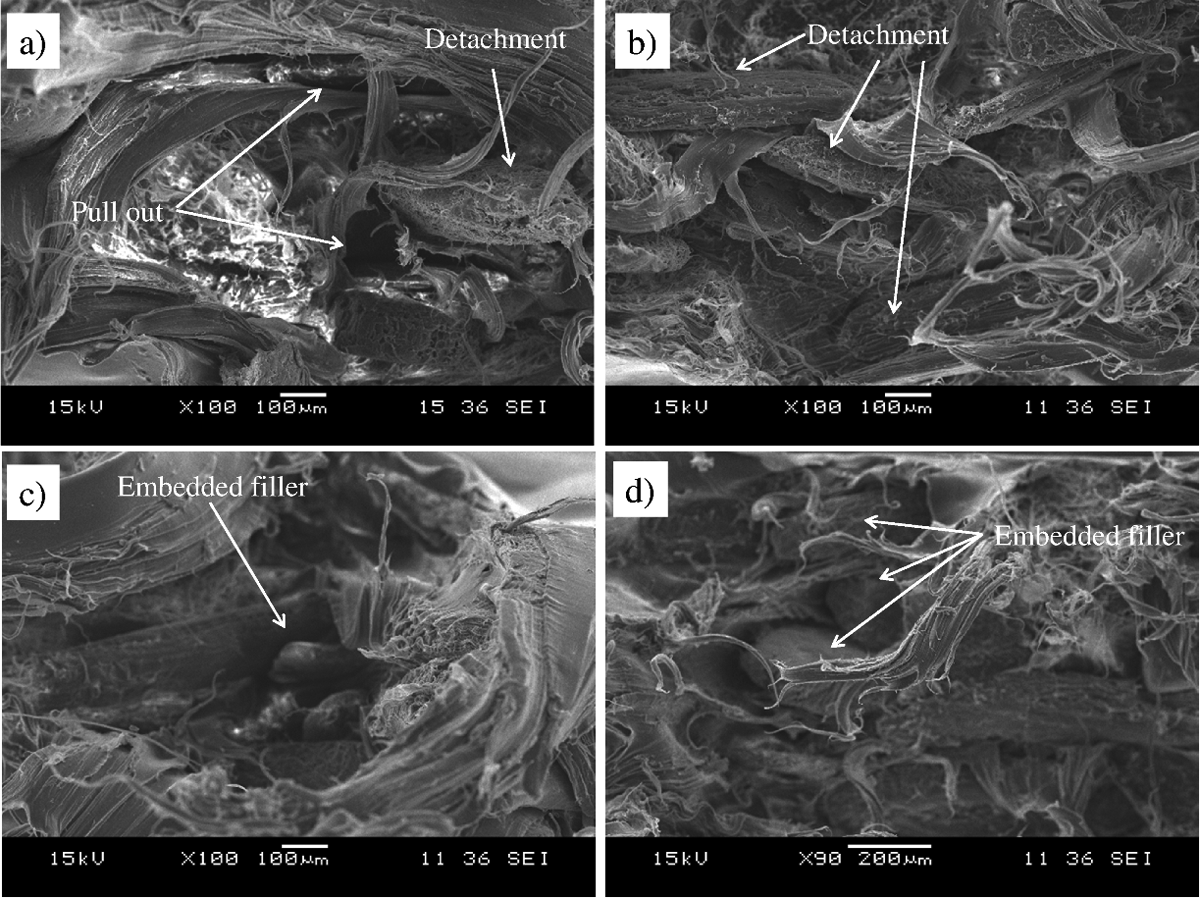
SEM micrographs of tensile fracture surface: (a) LLDPE/KH: 100/20 without PEAA; (b) LLDPE/KH: 100/40 without PEAA; (c) LLDPE/KH: 100/20 with PEAA; and (d) LLDPE/KH: 100/40 with PEAA. LLDPE: linear low-density polyethylene; KH: kapok husk; PEAA: polyethylene-grafted acrylic acid; SEM: scanning electron microscopic.
Water absorption
Figure 6 displays the water absorption of LLDPE/KH ecocomposites with and without PEAA. It can be seen that the water absorption of both LLDPE/KH ecocomposites rose with increasing KH content and immersion time. This is a common phenomenon for natural filler-based ecocomposites. 3,12,25 –27 It is because of the chemical structure of KH rich with hydroxyl groups that provides the hydrophilic behavior to ecocomposites, which is due to high water absorbability. Thus, the increase of KH content increased the water uptake ability of LLDPE/KH ecocomposites. Besides, the presence of micro-cracks between KH and LLDPE due to poor interfacial adhesion might increase water uptake by capillary action. The incorporation of PEAA reduced and altered hydrophilic properties of KH by forming covalent bond or hydrogen bond with the hydroxyl groups of KH via esterification. As a result, the reduction of hydrophilic behavior of KH by PEAA was significant, and the improvement of interfacial bonding decreased the water absorption of LLDPE/KH ecocomposites.
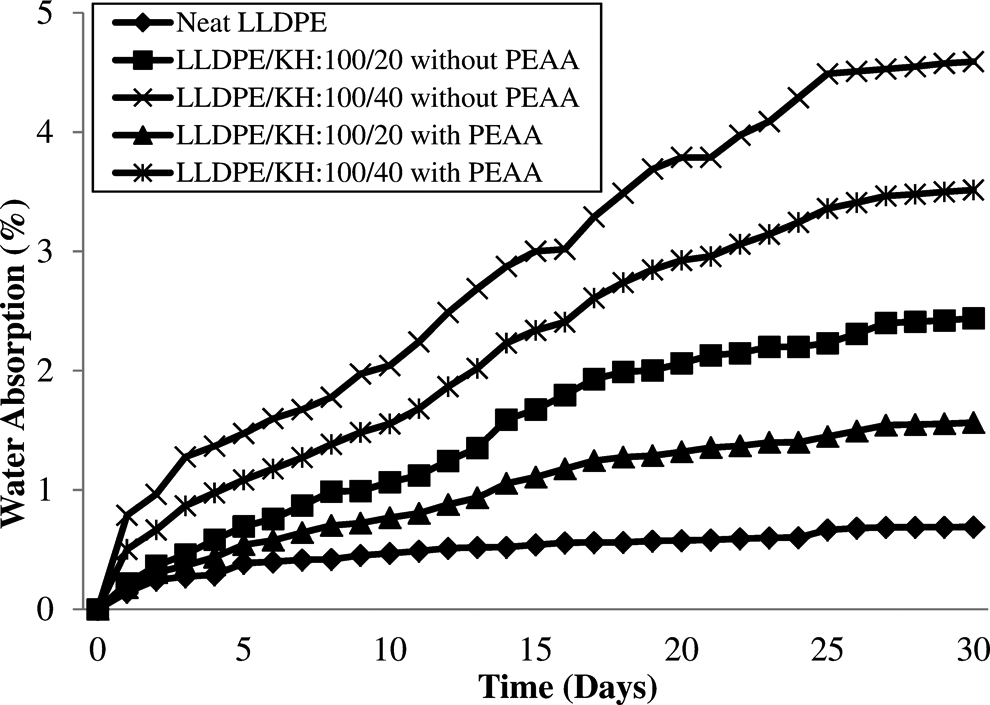
Water absorption of neat LLDPE and LLDPE/KH ecocomposites with and without PEAA. LLDPE: linear low-density polyethylene; KH: kapok husk; PEAA: polyethylene-grafted acrylic acid.
FTIR analysis
The FTIR spectra of LLDPE/KH ecocomposites with and without PEAA are shown in Figure 7. The main characteristic peak of LLDPE/KH biocomposites is listed in Table 2. The peak at 3389 cm−1 was assigned to hydroxyl groups (−OH) of hemicellulose, lignin, and cellulose in KH. This peak also indicated the hydrophilicity of LLDPE/KH ecocomposites. The intensity of absorption band at 3380 cm−1 was significantly reduced in LLDPE/KH ecocomposites with PEAA. This is because of the hydrophoxyl groups of KH, which were chemically bonded with PEAA. In addition, the peak at 1741 cm−1 was attributed to the presence of ester linkage (C–O) between KH and PEAA. This shows the evidence that the PEAA had covalently bonded with KH. The schematic reaction of PEAA and KH is illustrated in Figure 8.
Main functional group of LLDPE/KH biocomposites.

LLDPE: linear low-density polyethylene; KH: kapok husk; PEAA: polyethylene-grafted acrylic acid.
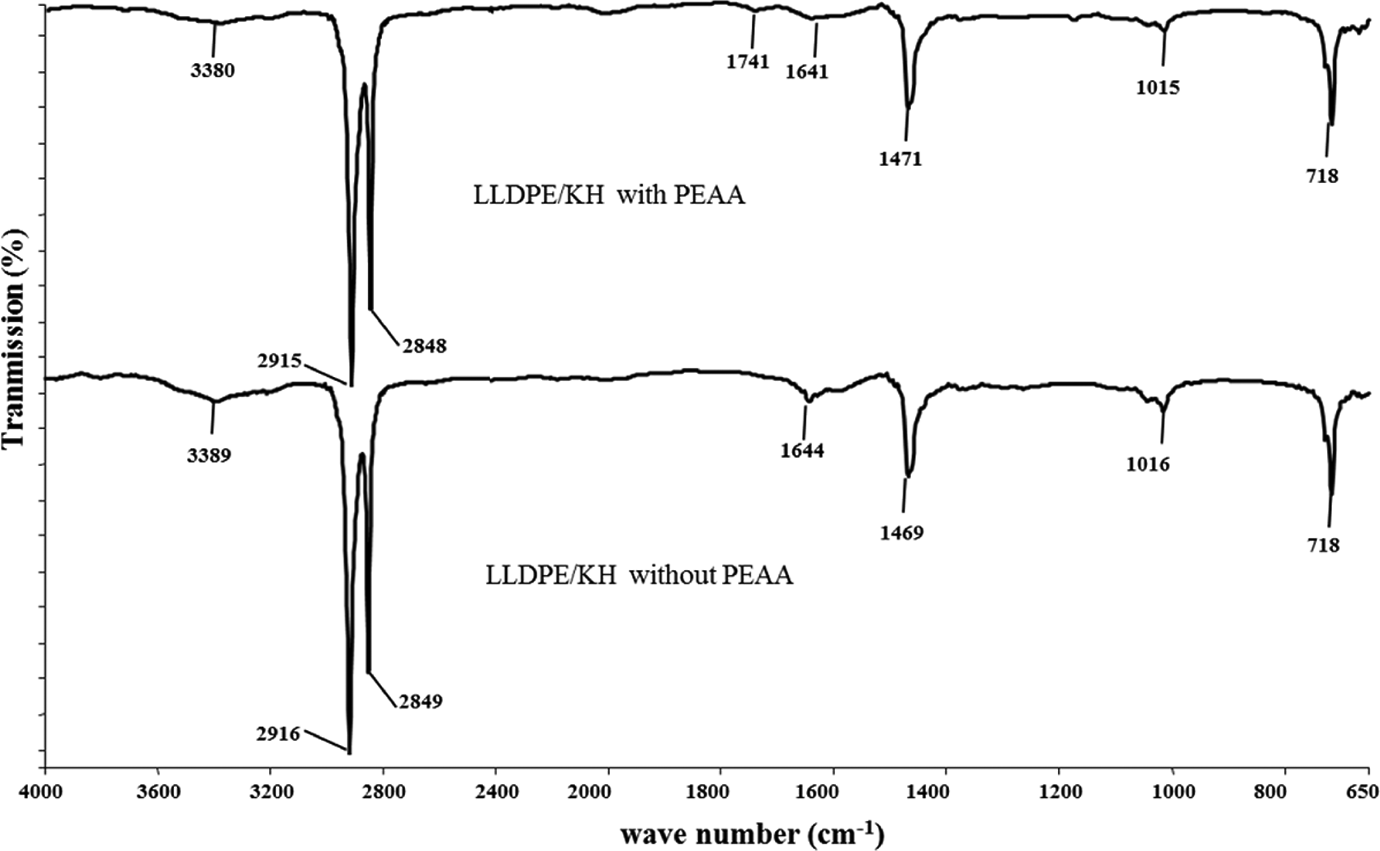
FTIR spectra of LLDPE/KH ecocomposites with and without PEAA. LLDPE: linear low-density polyethylene; KH: kapok husk; PEAA: polyethylene-grafted acrylic acid; FTIR: Fourier transmission infrared.
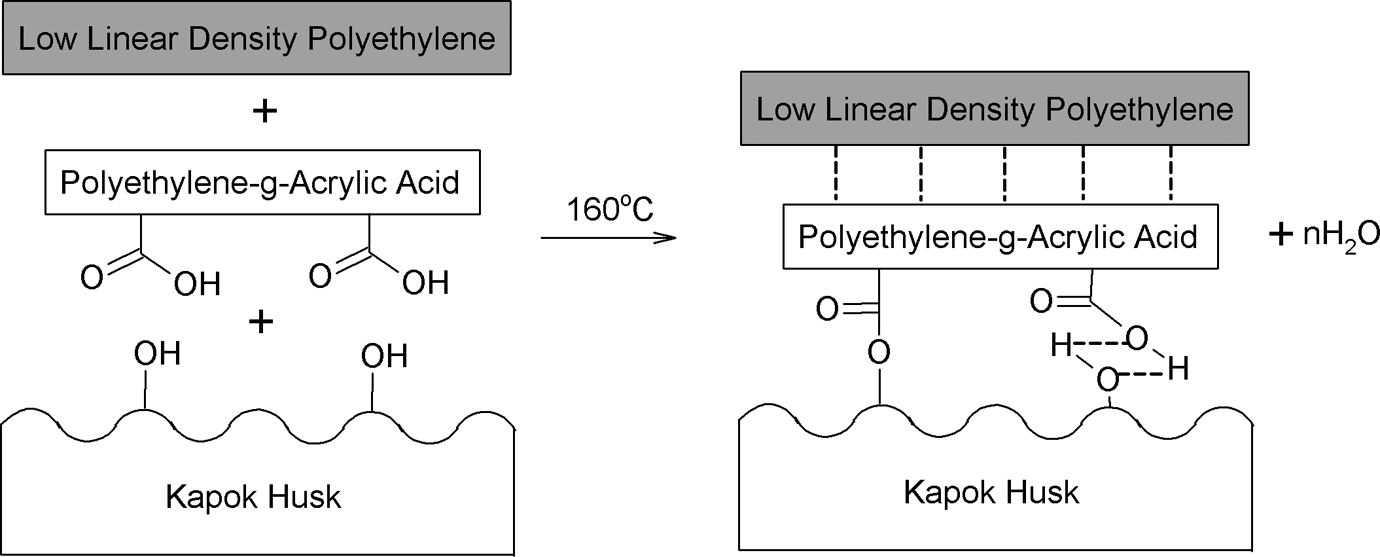
The schematic hypothetical reaction of PEAA and KH. KH: kapok husk; PEAA: polyethylene-grafted acrylic acid.
Differential scanning calorimetry
Figure 9 shows the DSC curves of LLDPE/KH ecocomposites with and without PEAA. The DSC data of both ecocomposites are summarized in Table 3. The increase of KH content reduced the crystallinity of LLDPE/KH ecocomposites. This phenomenon is due to the addition of KH that constrained the mobility of PE chains from the process of crystallization. This result was consistent with a previous study. 3 Furthermore, the LLDPE/KH ecocomposites with PEAA showed higher crystallinity compared with the LLDPE/KH ecocomposites without PEAA. This indicated that the presence of PEAA increased the crystallinity of ecocomposites by promoting the migration and diffusion of PE chain to form transcrystalline around the KH surface. Most findings have shown that for ecocomposites, the addition of copolymer-coupling agent can increase the crystallinity of biocomposites. 11,18,28 –30 However, the melting temperature of LLDPE/KH ecocomposites was not influenced by KH content and PEAA. The similar observation also found by other researchers. 11,24,31
DSC data of LLDPE/KH ecocomposites with and without PEAA.

LLDPE: linear low-density polyethylene; KH: kapok husk; PEAA: polyethylene-grafted acrylic acid; DSC: differential scanning calorimetric; T m: melting temperature; X c: degree of crystallinity of ecocomposite.
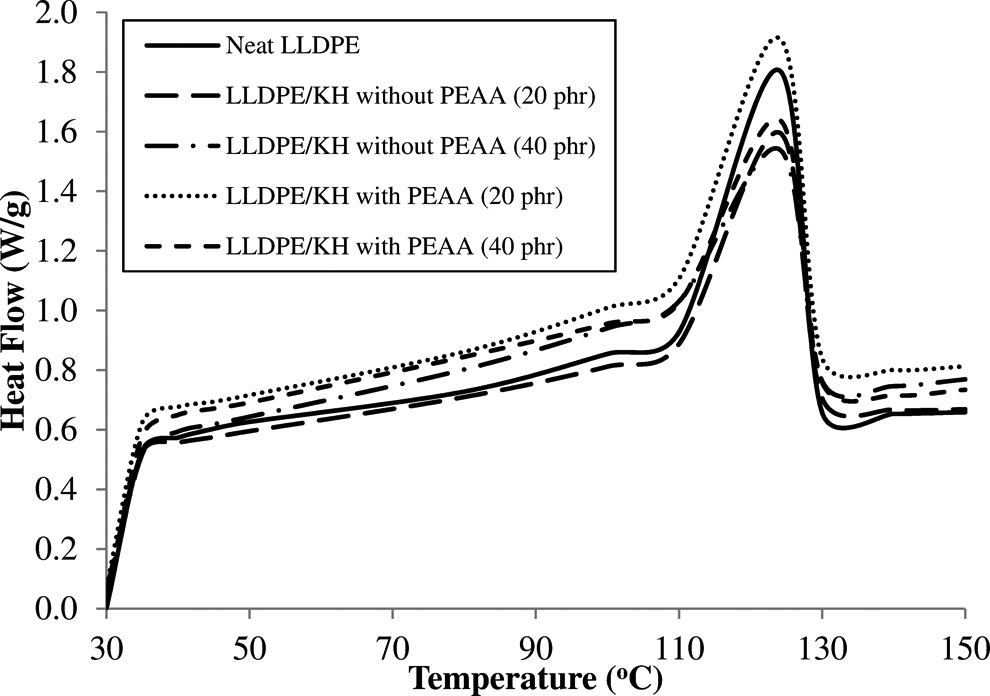
DSC curves of neat LLDPE and LLDPE/KH ecocomposites with and without PEAA. LLDPE: linear low-density polyethylene; KH: kapok husk; PEAA: polyethylene-grafted acrylic acid; DSC: differential scanning calorimetric.
Thermogravimetric analysis
Derivative TGA (DTG) and TGA curves of neat LLDPE and LLDPE/KH ecocomposites with and without PEAA are shown in Figures 10 and 11, respectively. The data from DTG and TGA curves are listed in Table 4. The DTG curve (Figure 10) shows that the neat LLDPE was decomposed in single step above temperature 330°C. The LLDPE/KH ecocomposites was decomposed in two steps, which include (i) decomposition of hemicellulose in KH and (ii) decomposition of lignin and cellulose in KH as well as LLDPE matrix. According to Table 4, LLDPE/KH ecocomposites exhibited an early thermal degradation as showed in temperature at 5% weight loss (T d5%). The increase of KH content decreased the T d5% of LLDPE/KH ecocomposites. The early thermal decomposition of LLDPE/KH ecocomposites was attributed by the weight loss of volatile compound and hemicellulose in KH. In regard to decomposition temperature at maximum rate (T dmax), the LLDPE/KH ecocomposites at 20 phr KH content showed a poor thermal stability, but the increase of KH content to 40 phr shifted the T dmax of LLDPE/KH ecocomposites to higher temperature. This is because the amount of pyrolysis materials formed from high KH content is sufficient to act as thermal protective barrier and inhibits the thermal degradation of ecocomposites. 3,7 Therefore, the thermal decomposition of LLDPE/KH ecocomposites at 40 phr of KH content is delayed. From Table 4, the residue of LLDPE/KH ecocomposites at 700°C increased with increasing KH content. As discussed, the residue was the pyrolysis materials generated from thermal decomposition of KH. Thus, change of the residue is corresponded to content of KH. Moreover, the addition of PEAA improved the thermal stability of LLDPE/KH ecocomposites, as it can be observed from the increment of T d5%, T dmax, and residue at 700°C. The PEAA was covalently bonded on KH surface and it gave better thermal stability to KH. As a result, the KH bonded with PEAA is more resistant to thermal decomposition and it improved the thermal stability of LLDPE/KH ecocomposites. According to Salmah et al., 17,18 the LDPE/palm kernel shell ecocomposites exhibited better thermal properties with PEAA.
TGA data of LLDPE/KH ecocomposites with and without PEAA.
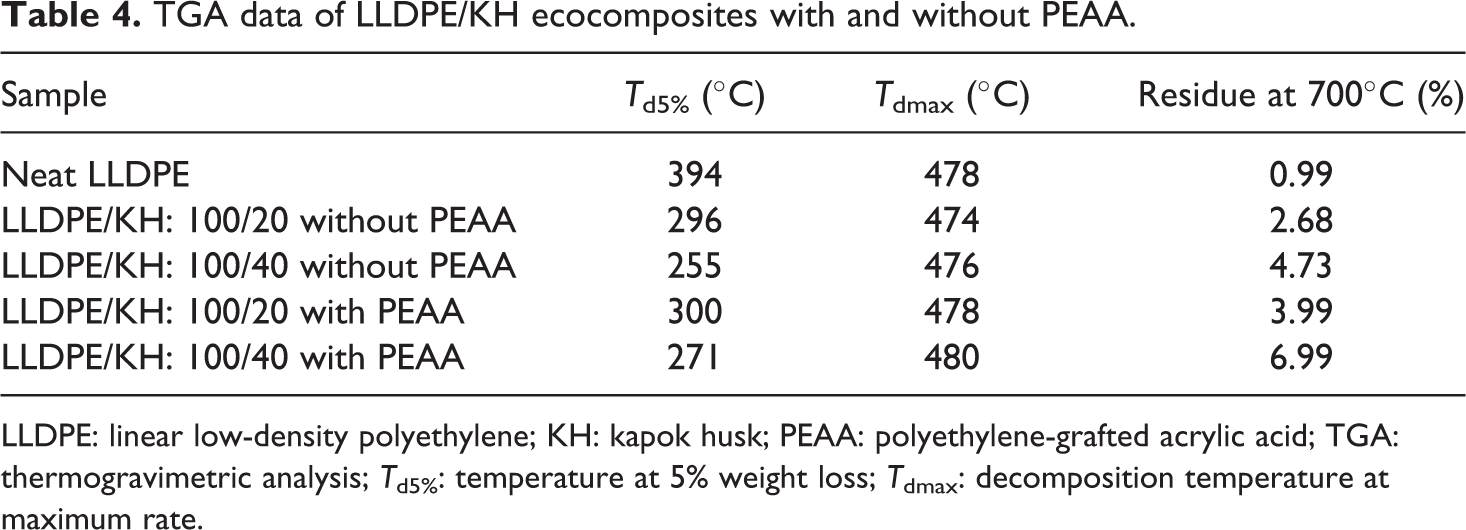
LLDPE: linear low-density polyethylene; KH: kapok husk; PEAA: polyethylene-grafted acrylic acid; TGA: thermogravimetric analysis; T d5%: temperature at 5% weight loss; T dmax: decomposition temperature at maximum rate.
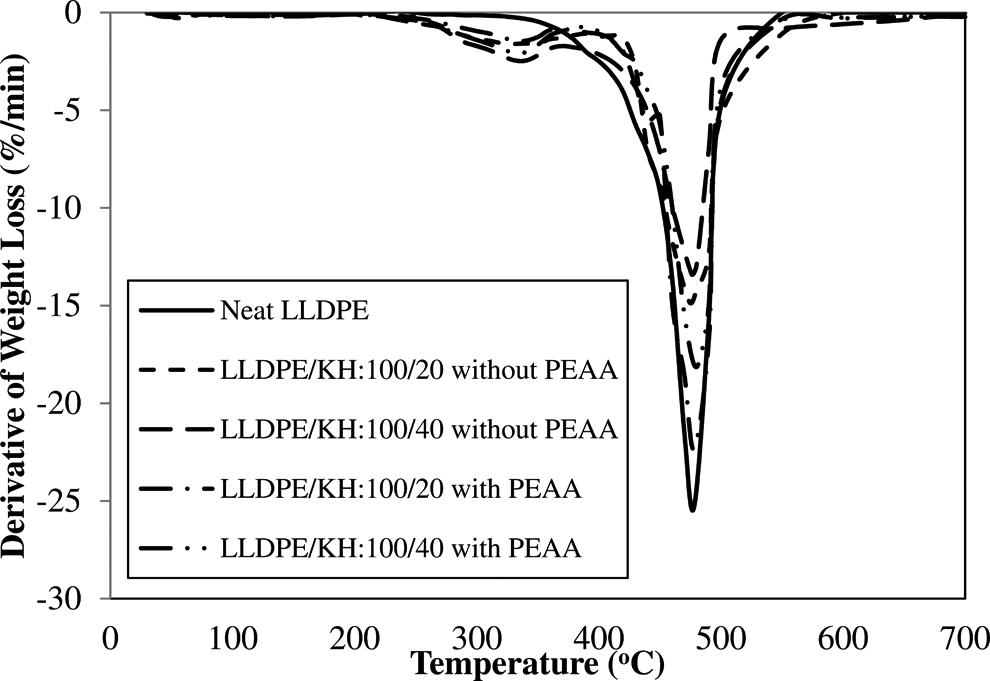
DTG curves of neat LLDPE and LLDPE/KH ecocomposites with and without PEAA. LLDPE: linear low-density polyethylene; KH: kapok husk; PEAA: polyethylene-grafted acrylic acid; DTG: derivative thermogravimetric.
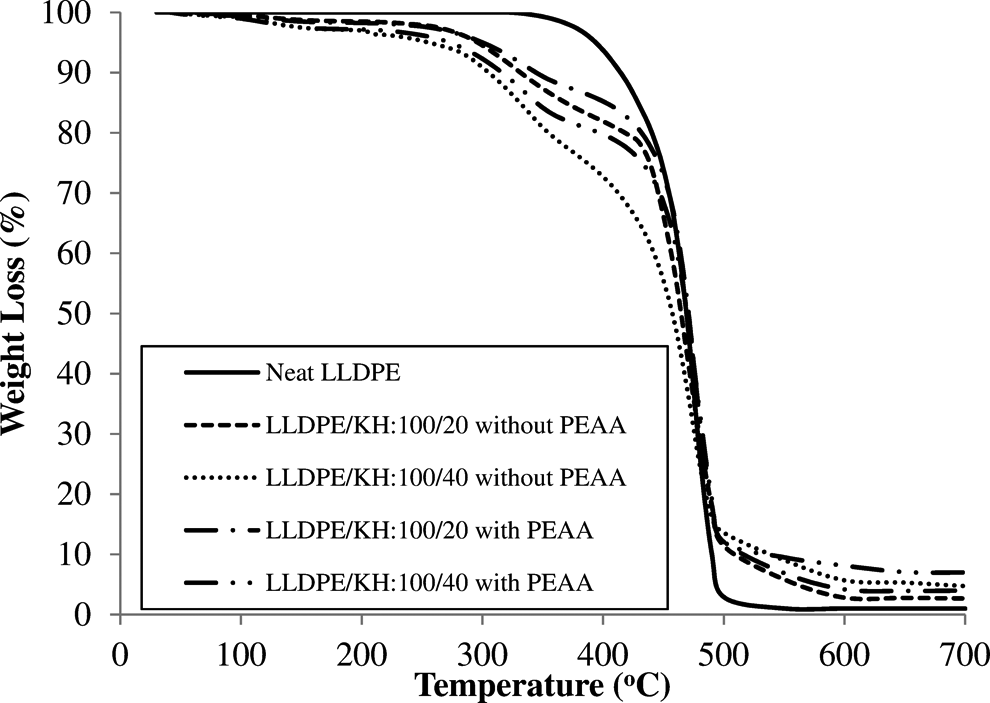
TGA curves of neat LLDPE and LLDPE/KH ecocomposites with and without PEAA. LLDPE: linear low-density polyethylene; KH: kapok husk; PEAA: polyethylene-grafted acrylic acid; TGA: thermogravimetric analysis.
Conclusion
It was found that the increase of KH content reduced the tensile strength and elongation at break, but it increased the tensile modulus of LLDPE/KH ecocomposites. The water absorption of LLDPE/KH ecocomposites increased with the change of KH content and immersion time, due to the influence of hydroxyl group from KH. The SEM micrograph also showed the poor interfacial bonding between KH particles and LLDPE matrix. The addition of KH induced poor thermal properties of LLDPE/KH ecocomposites. Whereas, the incorporation of PEAA provided a coupling effect to the LLDPE/KH ecocomposites. The presence of PEAA had significantly improved the tensile strength, tensile modulus, crystallinity, and thermal stability as well as reduced the water absorption of LLDPE/KH ecocomposites. The interfacial bonding that enhanced by the presence PEAA was proved by SEM study.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
