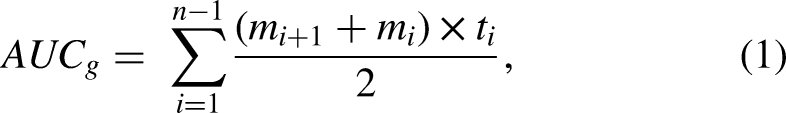Abstract
Studies examining the impact of early adversity on physiological responsivity to environmental challenges in later life yield a complex pattern of findings and ambiguity regarding the direction of effect, with some studies reporting heightened responses and others reporting dampened responses. One potential reason for these mixed findings is an oversimplified theoretical model surrounding the connection between early life stressor exposure and subsequent stress responsivity. The adaptive calibration model offersa contemporary set of assumptions aimed at providing a better understanding of the ways that early life experiences shape the stress response system to better align with current and future environments. The current study utilized a large subsample from the National Study of Daily Experiences (N = 1,605) to examine the extent to which the association between daily stressor exposure and cortisol levels varies across levels of early life adversity. Results revealed that those individuals who experienced extremely low levels of early life adversity displayed the greatest increase in cortisol levels across the day as daily stressor exposure increased. Alternatively, those individuals who experienced extremely high levels of early life adversity displayed almost no change in diurnal production of cortisol as daily stressor exposure increased. The results are discussed within the evolutionary-developmental context of the adaptive calibration model along with suggestions for future research.
Introduction
Physiological responses to environmental challenges and contexts vary tremendously between individuals, with some displaying a heightened response to a relatively minor stressor and others exhibiting more limited responses to even intense stressors. This pattern of findings has important implications as individual differences in stress responsivity have been linked to risk for negative psychosocial, neurocognitive, and health outcomes (Juster et al., 2010; McEwen & Seeman, 1999). Given these observations, it comes as little surprise that advancing our understanding of inter- and intra-individual differences in psychophysiological reactivity to stressors has garnered intensive empirical attention with several biopsychological models of human health and development being popularized (Dennis et al., 2012; Zisner & Beauchaine, 2016).
Childhood adversity is one factor that has been linked to between-individual differences in subsequent stress responses. Exposure to severe adversity, including abuse and neglect, has the potential to result in heightened stress reactivity (Conradt et al., 2014; Fearon et al., 2017; Holochwost et al., 2017). Studies focused on more specific sources of early life adversity—including maternal depression (Azar et al., 2007), low socioeconomic status (Essex et al., 2002), parental conflict (Davies et al., 2008), structural racism or discrimination (Cheadle et al., 2020), and death of a parent (Nicolson, 2004)—have reported similar patterns. These findings largely coincide with the broader literature focused on the developmental consequences of adverse childhood experiences (ACEs)—physical health problems (Danese & McEwen, 2012; Koss & Gunnar, 2018), poorer socioeconomic outcomes (Egan et al., 2015; Liu et al., 2013), and both internalizing and externalizing behavior problems (Anda et al., 2006; Duke et al., 2010; Chapman et al., 2004; Lindert et al., 2014).
Despite these observations, alternative theoretical models have been developed that may provide a more thorough explanation of the role of early life adversity on stress responsivity later in the life course. Specifically, the adaptive calibration model (ACM) is a developmental-evolutionary model aimed at connecting early life experiences and subsequent physiological, cognitive, and behavioral responses to contemporaneous stimuli including environmental stressors (Del Giudice et al., 2011; Ellis et al., 2017; Ellis & Del Giudice, 2014). Directly in line with the key arguments offered by the ACM, the current study aims to examine the potential impact of early life adversity on the association between daily stressor exposure and hypothalamic–pituitary–adrenal (HPA) axis activity assessed with diurnal salivary cortisol. To do so, we draw from a large, longitudinal sample of adults from the United States.
Childhood Adversity and Stress Responsivity
Prior research has revealed that exposures to adversity during childhood appear to exhibit a dose–response relationship with developmental delay(s) (Cprek et al., 2020) and other deleterious outcomes including physical and mental health problems (Chapman et al., 2004; Felitti et al., 1998; Merrick et al., 2017). Further, previous studies have revealed that childhood adversity clusters within families that experience other problems (Connolly & Kavish, 2019; Dong et al., 2003; Schwartz et al., 2019), as well as within racial or ethnic minority groups that are also differentially exposed to other stressors (Richards et al., 2021).
These findings have also been extended to more directly connect the accumulation of early life adverse events to individuals’ physiological dysfunction and long-term physiological “wear and tear” (Danese & McEwen, 2012; Koss & Gunnar, 2018; Krushas & Schwartz, 2022). For example, Danese and McEwen (2012) argue that adverse psychosocial experiences in childhood become “biologically embedded,” eventually culminating into dysfunction across multiple physiological systems that comprise the larger stress response system. Such patterns largely align with the allostatic load model, which posits that while physiological responses to stressors may be adaptive in the short term, the prolonged activation of the physiological systems collectively responsible for stress reactivity can accumulate, ultimately resulting in system-wide dysregulation (Juster et al., 2010; McEwen & Seeman, 1999; McEwen & Wingfield, 2003). The potential implications of early life adversity for allostatic load are relatively straightforward. Childhood adversity experiences likely necessitate reactivity, but have also been found to result in prolonged psychological stress (Frewen et al., 2019). Such stress may result in more chronic and sustained activation of the stress response system (Carbone et al., 2022) and, subsequently, increased mental and physical health problems.
Thus, a series of studies report a consistent link between early life adversity and increased reactivity within various branches of the stress response system in later life, but another set of studies have reported the opposite—increased levels of early life adversity resulting in lower physiological responses to contemporaneous stressors (Bunea et al., 2017; Koss et al., 2016; Peckins et al., 2015). Stated differently, those individuals who have experienced greater overall exposures to adversity in childhood exhibit blunted stress responses, in which stress reactivity is less pronounced compared to those who experienced lower levels of early life adversity. Importantly, these findings also directly align with previous research examining the connection between individuals who experience posttraumatic stress disorder (PTSD) and subsequent stress reactivity. More specifically, those individuals who eventually display increased PTSD symptomology are significantly more likely to also display subsequently blunted or dampened stress reactivity (Meewisse et al., 2007).
Also, in line with this observation, the results of a recent meta-analysis that included 30 datasets from k = 29 studies (Bunea et al., 2017) found that early life adversity was consistently linked with blunted cortisol reactivity with a moderate overall effect (g = −0.39), a small effect in childhood (g = −0.25), and a large effect in adulthood (g = −0.63). Previous studies have also examined the contribution of more nurturing and supportive early family environments to subsequent physiological reactivity. These studies have revealed evidence of developmental sensitivity to context where individuals who were reared in supportive and stable contexts were more likely to exhibit heightened stress reactivity when faced with an environmental challenge (Ellis et al., 2005, 2017; Shirtcliff et al., 2017). Studies have also revealed that children in more supportive contexts may be more sensitive to stressors—exhibiting more pronounced physiological responses—but also display faster recovery (Ellis et al., 2017). Collectively, these results seem to counter expectations stemming from the ACEs literature and the allostatic load perspective. Such perspectives characterize early life stressor exposure as generally maladaptive, potentially disrupting subsequent stress reactivity and culminating in a range of deleterious outcomes.
The Adaptive Calibration Model
The results presented above present a more nuanced pattern surrounding early life stressor exposure and subsequent stress responses. Namely, as evidenced by the increased stress reactivity among individuals with low early life stressor exposure, exposure to early life stressors—in low to moderate doses—may convey an adaptive response, particularly in situations in which the stressor is novel, as failure to mount such a response in the face of an unfamiliar stressor or challenge is likely maladaptive. In this way, stress responses allow individuals to gather important information from their surrounding environments, develop and individualize coping strategies, and accumulate a working knowledge base of effective responses to environmental challenges. Directly in line with this possibility, the ability to adapt to environmental challenges—or plasticity—has been previously recognized as a developmentally based trait that is the result of accumulated environmental experiences that further prime individuals to be better equipped to address future environmental challenges (Del Giudice, 2015). This conceptualization in the evolutionary-developmental literature is largely influenced by life history theory, which posits that early childhood experiences will influence reproduction, mating, and even personality and psychopathology (Del Giudice, 2016; Ellis et al., 2009; Hill & Kaplan, 1999).
These observations have been further developed into a developmental-evolutionary model referred to as the ACM (Del Giudice et al., 2011; Ellis et al., 2017; Ellis & Del Giudice, 2014). The ACM, an extension of the theory of biological sensitivity to context (BSC; Boyce & Ellis, 2005; Ellis et al., 2005; Ellis & Boyce, 2008), at least partially contributes individual differences in stress responsivity to conditional adaptation or “the evolved ability of an organism to modify its developmental trajectory (and the resulting phenotype) to match the local conditions of the social and physical environment” (Del Giudice et al., 2011, p. 1563). Stated differently, individualized stress responses can be viewed as responses adapted to prior environmental experiences, which largely direct the stress response system toward more appropriate responses for current and future challenges. Further, natural selection favored stress responsivity mechanisms that align with early environmental conditions; therefore, these early life experiences are banked and used to calibrate the stress response system across other developmental stages of the life course because early environments are assumed to predict later life environments (i.e., environmental matching). Given the importance of early life experiences, and the potential adaptive nature of exposure to early life stressors, the ACM expects a curvilinear relationship between early life adversity and stress reactivity later in the life course.
Four Adaptive Patterns to Stress Responsivity
The curvilinear association is characterized by four adaptive patterns of stress reactivity (Del Giudice et al., 2011; Ellis et al., 2017; Lin et al., 2021; Shakiba et al., 2020). The sensitive pattern is characterized by low levels of early life adversity as well as nurturing and supportive family environments during childhood. This environmental context provides increased susceptibility to positive environments and opportunities but provides little feedback regarding potential environmental challenges or stressors, effectively upregulating the stress response system in response to stress. Due to limited exposure to environmental challenges, sensitive individuals have limited information regarding adaptive strategies for overcoming environmental challenges and are, therefore, expected to exhibit increased physiological responses when faced with subsequent environmental stressors. Buffered individuals experience moderate levels of early life adversity and experience family contexts that are relatively neutral (i.e., not overly nurturing or stressful) and as a result, are expected to exhibit moderate levels of responsivity when encountering a stressor. This response is largely guided by a stress response system that is somewhat well-informed and primed to encounter and overcome environmental challenges.
The vigilant pattern is characterized by exposure to high levels of early life adversity as well as unpredictable and unstable family environments during childhood. These experiences result in a sensitive stress response system that displays increased physiological responsivity when faced with an environmental challenge later in the life course. This pattern of physiological response stems from the unpredictable and dangerous nature of the early life environment, necessitating a vigilant state to detect potential threats or dangers. Finally, unemotional individuals experience severe levels of adversity, trauma, and chronic, prolonged stressor exposure. Such experiences downregulate stress responses, resulting in blunted physiological responsivity to subsequent environmental stressors. It should be noted that the ACM specifies that the stress response system assumes domain-general responses to stressors (Del Giudice et al., 2011), that is, “all stressors are stressful.” Although much research has been conducted on the specificity of stimuli that elicit stress responses (e.g., see Dickerson & Kemeny, 2004), these responses are considered general patterns of stress reactivity in individuals regardless of stimulus.
Collectively, these four adaptive patterns characterize the complex relationship between early life adversity and subsequent physiological responsivity to environmental stressors. The curvilinear association proposed by the ACM not only provides a more detailed description of the association relative to complementary perspectives (e.g., the allostatic load model), but it also accounts for the seemingly conflicting findings surrounding the association between early life adversity and physiological stress responses. In addition, the ACM has performed well in previous tests, with studies finding evidence of the four adaptive patterns described above (Del Giudice et al., 2012; Ellis et al., 2017; Lin et al., 2021) and a more general curvilinear association between early life adversity and subsequent physiological stress response (Gunnar et al., 2009; Shakiba et al., 2020). Despite this progress, much of the research examining the ACM has focused on samples of children and has employed laboratory-based stressor tasks to assess physiological responsivity. While these approaches are certainly beneficial and have provided greater insight into the empirical validity of the primary arguments of the ACM, they are limited in their ecological validity. Research in naturalistic settings shows that unpredictable and novel (either positive or negative) stimuli elicit cortisol responses in children to respond appropriately to the stressor regardless of its valence (Flinn, 2006). In addition, the majorityof prevoius studies examine children and as such do not provide insight into longer-term patterns involving physiological responsivity during later stages of the life course.
Current Study
Directly in line with arguments offered by the ACM, the current study employs a large prospective study comprised of a nationally representative sample of adults from the United States to examine the association between early life adversity and HPA axis activity assessed with diurnal salivary cortisol. The use of a combination of retrospective self-reports, prospective longitudinal data, and daily interviews allows for a direct examination of the potential impact of early life adversity exposure on the association between daily stressor exposure and cortisol measured across multiple days.
Methods
Sample and Procedure
Data for the current study come from the first two waves of the Midlife in the United States (MIDUS) study, which is a prospective longitudinal study that is nationally representative of adults aged between 25 and 74 from the United States (Brim et al., 1996, 2004). Broadly, the MIDUS was focused on examining the impact of a broad set of psychological, social, and behavioral influences on aging and health. The first wave of the MIDUS study (MIDUS I) was collected between 1995 and 1996 and consisted of 7,108 adults. The second wave (MIDUS II) was collected between 2004 and 2006 and consisted of ∼ 70% of the participants from the MIDUS I (N = 4,963). Phone interviews were conducted during both waves of data collection and covered a broad range of topics including interpersonal relationships, health conditions, lifestyle habits, and exposure to stressful life events.
Following the completion of the MIDUS II, a subsample of 2,022 respondents were enrolled in a substudy focused on documenting daily stressor exposure and responses called the National Study of Daily Experiences (NSDE; Almeida et al., 2009). The substudy consisted of brief, semi-structured phone interviews for eight consecutive days. Starting with the second day of the study, respondents also provided saliva samples for cortisol assessment during four predetermined intervals throughout the day for four consecutive days, resulting in a total of 16 saliva samples. Of the 2,022 total participants in the NSDE, 1,735 provided at least one valid cortisol sample. After accounting for missing data on study measures, including daily stressor exposure, stressor severity, and cortisol-altering medication use, the resulting analytic sample consisted of information from 1,605 individuals, 6,117 days of cortisol collection, and a total of 23,888 cortisol data points. 1 Missing data was minimal (< 5%) across all study variables. Supplementary t-tests and binary logistic regression models did not reveal any systematic patterns of missingness. Based on the limited amount of missing data and the results of these supplemental analyses, list-wise deletion was employed.
NSDE participants were provided with a saliva collection kit prior to their first phone interview. The kit contained detailed instructions along with 16 color-coded salivette collection tubes to identify which tubes should be used for each saliva collection event. During the initial phone interview, interviewers reviewed saliva collection procedures with the participants and provided any necessary clarification. Participants were instructed to provide four saliva samples on days 2–5 of the project at each of the following intervals: (1) immediately upon waking; (2) 30 minutes after waking; (3) before lunch; and (4) before bed. Participants were asked to record the exact time each sample was provided in a log provided within the collection kit materials. As a further protocol adherence check, participants were also asked to report sample collection times during the daily phone interview. The resulting correlation between the times recorded on the log sheets and reported during the phone interviews exceeded .90 across all four daily samples (Sin et al., 2017). Participants were instructed to store daily saliva samples in their home freezers until all samples were collected. Once all saliva samples were collected, participants used materials provided in the collection kit to ship salivettes to the MIDUS Biological Core at the University of Wisconsin, Madison. Samples were stored at −60 °C and then thawed and centrifuged at 3,000 r/min for 5 min. Cortisol concentrations were assessed with luminescence immunoassay with intraassay and interassay coefficients of variation below 5% (Ryff & Almeida, 2009). Additional technical information surrounding salivary data collection and the employed laboratory protocols employed in the NSDE have been provided elsewhere (Almeida et al., 2009; Karlamangla et al., 2013; Sin et al., 2017).
Measures
Salivary Cortisol. Daily cortisol samples were employed to assess two indices of diurnal cortisol. First, the raw cortisol values were employed to examine the diurnal cortisol slope across the course of the day, from waking to bedtime. Additional information regarding the estimation of the slope is provided in the plan of analysis section below, but both linear and quadratic slope functions were estimated to allow for nonlinear deceleration across the sleep-wake cycle. Second, total cortisol output from waking to bedtime was estimated as the area under the curve with respect to ground (
Descriptive Statistics for All Study Measures (N = 1,605).

Early Life Adversity. Early life adversity was assessed following procedures outlined in previous studies (Schwartz et al., 2019; Slopen et al., 2010) and included 17 retrospective indicators from the primary MIDUS I and MIDUS II phone interviews tapping three domains. First, stressful events during childhood were assessed using nine items tapping experiences that may have caused increased stress including parental substance use problems, parental unemployment, and failing out of school. Participants indicated whether each of the nine incidents occurred (= 1) or did not occur (= 0) previously. Second, participants were asked to indicate the overall quality of their relationship with their parents when they were growing up, with separate items for maternal and paternal relationships. Provided response categories ranged between 1 (excellent) and 5 (poor). Third, abuse in childhood was assessed with six items from the conflict tactics scale tapping maternal and paternal emotional, physical, and sexual abuse. Provided response categories ranged between 1 (never) and 4 (often). Due to the differences in response categories across items, and to more effectively weight each source of early life adversity, we followed the lead of previous studies examining the MIDUS and employed confirmatory factor analysis to construct a factor score tapping the latent construct of early life adversity (Schwartz et al., 2019). A single-factor solution provided an acceptable fit to the data (comparative fit index [CFI] = .92; Tucker–Lewis index [TLI] = .91; and the root mean square error of approximation [RMSEA] = .07), and the resulting factor scores were extracted to create the early life adversity scale, with greater values indicating greater overall levels of early life adversity.
Daily Stressors. Daily stressor exposure was assessed during daily phone interviews using the Daily Inventory of Stressful Events (Almeida et al., 2002). Participants were asked to indicate whether they experienced each of the following seven events in the past 24 hr (or since the last interview): (1) argument/disagreement; (2) avoided an argument; (3) stressful event at work or school; (4) discrimination; (5) stressful event at home; (6) stressful event experienced by friend or relative; and (7) any other stressful event. Each individual event was coded dichotomously by the interviewer. For each day of the study that an individual provided one or more saliva samples, the number of stressors experienced was summed, with the resulting variety index ranging between 0 (no stressors reported) and 7 (seven stressors reported) and reflected the number of stressors reported for each day. For each daily stressor reported, participants were asked to indicate how stressful the event was for them, with response categories ranging between 0 (not at all) and 3 (very stressful). To more effectively weight stressors that were perceived as more severe, the daily stressor variety index was multiplied by the mean daily stressor severity score to create the weighted daily stressor score.
Collection Characteristics. A total of five characteristics surrounding daily saliva and phone interview data collection were utilized or included in the estimated multiple variable models as covariates. First, the self-reported saliva collection times for each collected sample were used to assess the amount of time that elapsed between saliva sample collection, allowing for the calculation of the
Participant Characteristics. To control for characteristics that may contribute to between-individual variability in cortisol changes throughout the day, a total of six individual characteristics were included in the multiple variable models as covariates. First, given previously identified associations between cortisol and mood disorders, depressive symptoms were assessed using the Screening Version of the World Health Organization Composite International Diagnostic Interview (CIDI) during MIDUS II interviews (Kessler et al., 1998). The CIDI is a validated self-administered scale asking participants to indicate whether they had experienced seven items over the past two weeks, with responses coded dichotomously (0 = no and 1 = yes). Responses were summed to reflect overall depressive symptoms, with greater values indicating increased depressive symptoms. Second, since nicotine consumption may impact cortisol (Padilla et al., 2020), a binary indicator reflecting whether the participant was a smoker during the primary MIDUS II interview was included (0 = not a smoker; 1 = smoker). Third, during the last day of saliva collection, participants were asked to report whether they had taken prescription and/or over-the-counter medications that may impact cortisol levels (e.g., steroidal inhalers or creams, allergy medication, and anti-anxiety medications). The responses were used to create a binary indicator variable identifying those who did not report medication use (0) and those who did (1). Fourth, age was calculated by subtracting the participant’s date of birth from the date of the first interview and measured continuously in years. Fifth, gender was self-reported and coded dichotomously such that 1 = male and 2 = female. Sixth, race was self-reported and coded categorically such that 1 = White, 2 = Black, and 3 = all other races.
Plan of Analysis
Linear mixed models were used to examine the extent to which the association between daily stressor exposure and cortisol varied as a function of early life adversity exposure. Given the nested structure of the data, mixed models were well-suited to address the research questions examined in the current study. More specifically, mixed models (also sometimes referred to as multilevel models or hierarchical linear models) account for the nonindependence of observations through the introduction of a series of random intercept terms, properly adjusting standard errors for multiple levels of clustering (Rabe-Hesketh & Skrondal, 2012; Raudenbush & Bryk, 2002; Singer & Willett, 2003). Further, mixed models are highly flexible, allowing for multiple levels of nestedness, permitting the estimation of interaction terms across levels of analysis, and accommodating unbalanced panels across observations. In addition to these attributes, mixed models are commonly employed when analyzing data containing salivary biomarkers including cortisol (Adam & Kumari, 2009; Hruschka et al., 2005). Similarly, given the multilevel structure of the NDSE data collection protocol—that is, saliva collection occasions nested within days, nested within participants—mixed models seem particularly well-suited for the current study. Prior to the estimation of any mixed models, the weighted daily stressor score and early life adversity measure were z-transformed to aid in interpretation.
To examine the diurnal cortisol slope from waking to bedtime, we employed a series of three-level linear mixed models to account for cortisol measures within days and days within participants. The first level analysis included measures that varied with the collection of each cortisol sample, such as the time since waking. The second level of analysis included measures that varied from day to day, including the weighted daily stressor score and collection characteristics. The third level of analysis included measures that varied between participants including early life adversity and participant characteristics. To better capture the expected decelerating diurnal pattern of cortisol over the course of the day, the number of hours since waking squared was added to three-level models as a covariate along with all other collection and participant characteristics (including hours since waking). A total of two three-level mixed models were estimated. The first model examines the main effects of daily stressors and early life adversity on the diurnal cortisol slope over the course of the day, after controlling for the included covariates. To more closely examine the extent to which the association between daily stressor exposure and the diurnal cortisol slope varied as a function of early life adversity, the second model added a multiplicative interaction term between the weighted daily stressor score and early life adversity.
To examine total cortisol output over the course of the day, we employed a series of two-level linear mixed models with
Continuous by continuous interaction terms are notoriously difficult to interpret and the resulting coefficients from estimated regression equations are typically not helpful in understanding patterns of moderation (at least in such circumstances). For these reasons, we have followed the recent suggestions aimed at more effectively presenting and accurately interpreting interaction terms in linear regression models and present the results from our models that included interaction terms graphically (McCabe et al., 2018; Mize, 2019; Preacher et al., 2007). In line with these suggestions, we present the estimated moderating effects in two ways. First, we present the marginal effects of daily stressor exposure on cortisol (either diurnal slope or
Results
The first set of models was the three-level mixed model examining diurnal cortisol slope, with results presented in Table 2. Model 1 examines the main effects of the weighted daily stressor score and early life adversity on the diurnal cortisol slope. As indicated in the first set of columns in the table, participants with greater weighted daily stressor scores had a slower decline of cortisol during the day (b = .037, 95% CI = .021; .053). An alternative interpretation is that those participants with greater overall exposure to daily stressors experienced flatter diurnal cortisol slopes throughout the course of the day. This is due to the fact that the average change is cortisol across measurement occasions is negative, which is expected since cortisol tends to decrease throughout the course of the day. In addition, since the cortisol measures were log-transformed, a simple formula can be used to reflect the percent change in cortisol for a one-unit change in daily stressor exposure
Results From Three-Level Mixed Models Examining Diurnal Cortisol Slope.
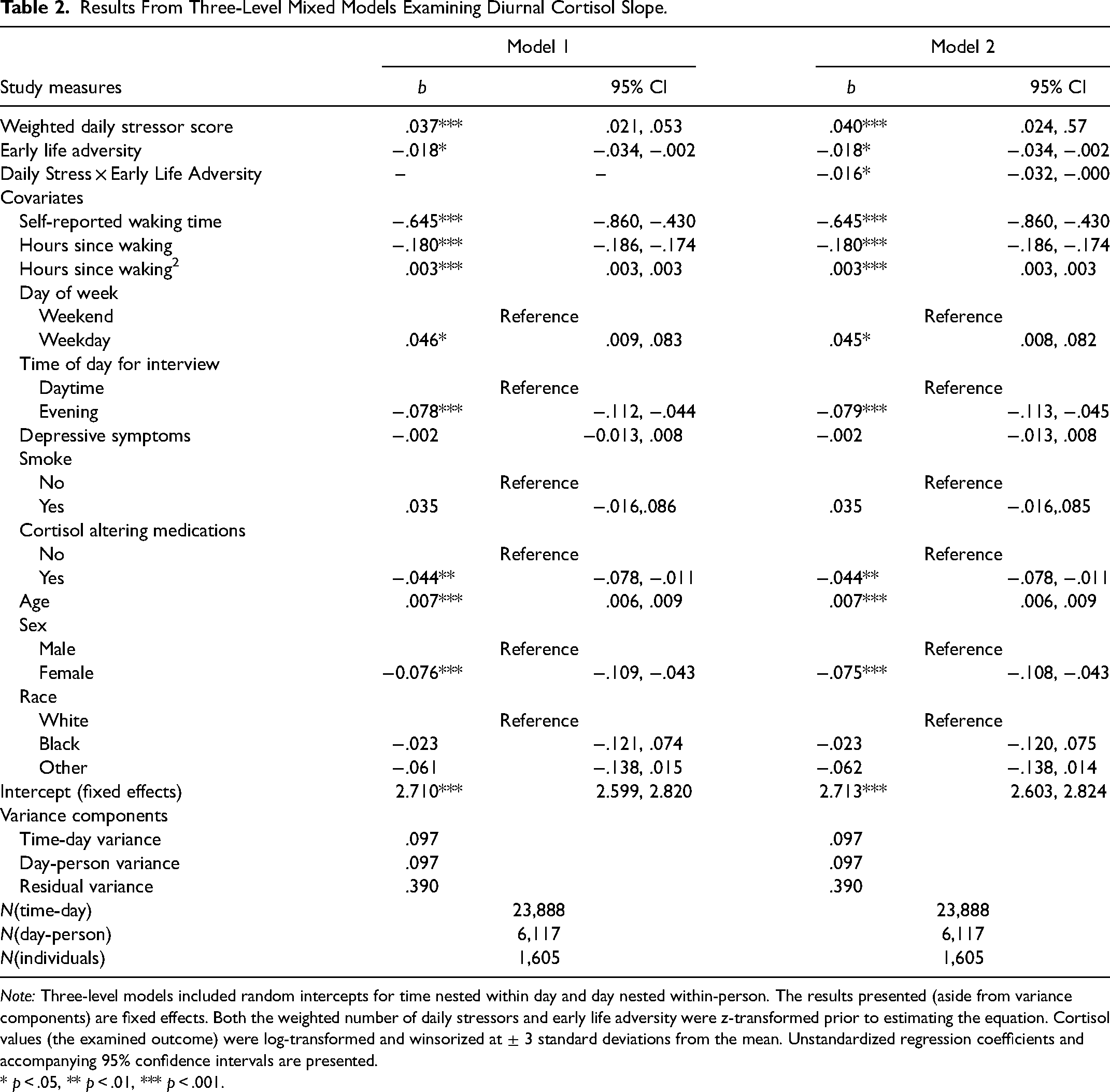
Note: Three-level models included random intercepts for time nested within day and day nested within-person. The results presented (aside from variance components) are fixed effects. Both the weighted number of daily stressors and early life adversity were z-transformed prior to estimating the equation. Cortisol values (the examined outcome) were log-transformed and winsorized at ± 3 standard deviations from the mean. Unstandardized regression coefficients and accompanying 95% confidence intervals are presented.
* p < .05, ** p < .01, *** p < .001.
Model 2 introduces a multiplicative interaction term between the weighted daily stressor score and early life adversity. The accompanying coefficient was negative and significant (b = −.016, 95% CI = −.032; −.000), but as discussed above, continuous by continuous interactions can be difficult to interpret. To better address this issue, the results are presented graphically in Figure 1. The top panel of the figure presents the marginal effects of daily stressor exposure on diurnal cortisol slope across levels of early life adversity. The marginal effects are presented with accompanying 95% confidence intervals, which are represented as error bands. Directly in line with the negative fixed effects coefficient for the product term, the association between daily stressor exposure and cortisol decreases as levels of early life adversity increase, such that the association is greatest when early life adversity is extremely low (two standard deviations below the mean) and ultimately becomes nonsignificant once early life adversity surpasses one standard deviation above the mean. Stated differently, cortisol responses to daily stressor exposure are blunted for those individuals who experienced greater levels of early life adversity.

Daily stressor influence on diurnal cortisol slope across early life adversity.
The bottom panel of Figure 1 presents the results of the interaction in a slightly different manner to further probe the results. More specifically, the figure displays the average marginal effects of daily stressor exposure on the diurnal cortisol slope for participants with extremely low, average, or extremely high levels of early life adversity. As can be seen in the figure, the predicted value of the diurnal cortisol slope for each of the three groups is similar when daily stressor exposure is extremely low. However, as exposure to daily stressors increases, the predicted values for each group begin to diverge. Notably, those individuals who experienced extremely low levels of early life adversity displayed the lowest levels of cortisol when daily stressors were low, but also displayed the highest levels of cortisol when daily stressors were high. Alternatively, those individuals who experienced extremely high levels of early life adversity displayed relatively consistent cortisol levels even as daily stressor exposure changed from extremely low to extremely high. Individuals who experienced an average level of early life adversity displayed increased levels of cortisol as daily stressor exposure increased, but not to the same magnitude as those who experienced extremely low levels of early life adversity.
The next stage of the analysis was aimed at examining total cortisol output over the course of the day, operationalized as
Results From Two-Level Mixed Model Examining Total Daily Cortisol Output.

Note: Two-level models included a random intercept term for day nested within a person. The results presented (aside from variance components) are fixed effects. The weighted number of daily stressors and early life adversity measures were z-transformed prior to estimation. Daily cortisol output was calculated as the area under the curve with respect to ground (
* p < .05, ** p < .01, *** p < .001.
The interaction results are presented graphically in Figure 2. The top panel of the figure presents the marginal effects of daily stressor exposure on

Daily stressors influence on total daily cortisol output across early life adversity.
Discussion
To a large extent, our observations can be interpreted using the assumptions of the ACM (Del Giudice et al., 2011; Ellis & Del Giudice, 2014), wherein differential levels of exposure to early life adversity systematically moderate subsequent HPA axis responsivity to daily stressor exposure. However, there were exceptions. The three-level linear mixed models examining the diurnal cortisol slope revealed that early life adversity moderated the association between the weighted daily stressor score and cortisol production across the day, such that the adrenocortical response to daily stressors was lower for those individuals who experienced greater levels of early life adversity. Further probing of this moderating effect indicated that extremely high exposure to early life adversity effectively blunted the effect of daily stressor exposure on daily cortisol production. This pattern of results directly aligns with the results from a recent meta-analysis (Bunea et al., 2017), indicating that early life adversity was associated with blunted cortisol responsivity to contemporaneous stressors, particularly within adulthood. Furthermore, this pattern of results directly aligns with the unemotional pattern specified within the ACM. More specifically, more severe and chronic exposures to trauma during childhood are expected to result in subsequently blunted physiological responses, as this pattern may be evolutionarily adaptive within environments characterized by high risk, effectively promoting fast life history strategies at the expense of social and physiological consequences (Del Giudice et al., 2011).
Further, those individuals with extremely low exposure to early life adversity experienced the greatest increase in daily cortisol production as daily stressor exposure increased. This pattern falls directly in line with the sensitive pattern outlined in the ACM, in which children reared in overwhelmingly safe and nurturing environments are more susceptible to environmental context (Del Giudice et al., 2011; Lin et al., 2021). This pattern closely resembles the “for better or worse” patterns of differential susceptibility previously described by Belsky and colleagues (Belsky et al., 1991; Belsky & Pluess, 2009), wherein some individuals are more susceptible to environmental influences regardless of whether such environments are nurturing or deleterious. In this way, sensitive individuals are expected to thrive in positive and supportive environments, but they are also expected to display heightened physiological responses to environmental challenges, as such individuals have not developed a sufficient knowledge bank comprised of effective strategies for overcoming or coping with such challenges.
Finally, those individuals with average levels of early life adversity exhibited more moderate increases in cortisol as daily stressor exposure increased, which directly aligns with the buffered phenotype outlined within the ACM. The more moderate adrenocortical response to daily stressors is attributed to a balance in the costs (i.e., long-term wear and tear or increased allostatic load) and benefits (i.e., short-term increases in physiological arousal that aid in facing and overcoming environmental challenges) of more pronounced physiological responsivity (Del Giudice et al., 2011; Shakiba et al., 2020). It is also possible that earlier exposures to adversity and other environmental stressors have also provided buffered individuals with an opportunity to gather additional pertinent information regarding overcoming environmental challenges or more effective coping strategies. This possibility directly aligns with the lifespan wisdom model, which is a developmental perspective that emphasizes the importance of exploratory risk-taking in an effort to better develop adaptive decision-making strategies when faced with similar situations in the future (Khurana et al., 2018; Romer et al., 2017; Schwartz et al., 2022). In this way, the stress response system is primed to respond to environmental stressors more effectively due to prior knowledge surrounding expected patterns of response gathered from prior experiences surrounding stressful environments.
In addition to support for the ACM, these findings also have implications for the extant literature focused on ACEs and other sources of childhood adversity and subsequent deleterious outcomes. While this literature is not centrally focused on physiological implications of adversity exposure, this connection has been previously noted (Danese & McEwen, 2012), and a large number of prior studies have identified associations between childhood adversity and subsequently upregulated physiological stress responses (Conradt et al., 2014; Fearon et al., 2017; Holochwost et al., 2017), leading to others examining ACEs within the context of evolutionary-developmental theories (e.g., Hertler et al., 2022). Furthermore, within the ACEs literature, studies have recognized that ACEs have a dose-response effect, indicating that each additional exposure to early life stressors increases the subsequent probability of a given negative outcome (Chapman et al., 2004; Felitti et al., 1998; Merrick et al., 2017). Importantly, a significant number of studies examining ACEs, including the original CDC-Kaiser Permanente ACE study (Felitti et al., 1998), have reported that more than half of the individuals in their samples have experienced at least one ACE.
These findings seem to suggest that any exposure to adversity in childhood significantly increases the probability of a range of deleterious outcomes, with the probability increasing with each additional exposure. The primary tenet of the ACM is that childhood experiences effectively calibrate the stress response system to the environment that individuals are most likely to encounter during subsequent stages of the life course (Del Giudice et al., 2011). In this way, the overly simplistic view of early life stressor exposure embraced by the ACEs literature is fixated on the costs of such experiences—which are real and indeed exist—but fail to “address the benefits of calibrating autonomic, neuroendocrine, metabolic, and immune systems to match current and future environments” (Ellis & Del Giudice, 2014, p. 2, emphasis in original). The findings from the current study reinforce this observation, as adrenocortical stressor response in adulthood significantly varied as a function of early life adversity exposure. However, the buffering effects of early life adversity were not monotonic and systematically varied depending on the level of adversity experienced. These findings have significant implications for the ACEs literature, but also for the literature focused on early life adversity more broadly, suggesting that future research in these areas should be characterized by more detailed expectations surrounding the implications of exposure to early life adversity.
One as-yet relatively unexamined area of the ACEs literature made it impossible with this current analysis to address one aspect of the ACM: Developmental switch points and/or sensitive periods. Although much is known about the cumulative effects of ACEs on myriad areas in later life, exposure timing is highly likely to affect the physiological development (Hawes et al., 2021; Walasek et al., 2022), especially given the vast physiological changes that occur throughout childhood and adolescence that are related to physical development. It follows, logically speaking, that the timing of the adverse experiences would therefore differentially affect the individual’s future stress responsivity.
In addition to the supportive findings for the ACM, the current study also yielded some conflicting findings. First, while the results provided evidence of sensitive, buffered, and unemotional patterns, we did not observe a vigilant pattern, in which high exposure to early life adversity resulted in heightened physiological responsivity to subsequent stressors. This may have been an artifact of the analytic strategies employed in the current study, as previous studies employing mixture modeling (e.g., latent class analysis or latent profile analysis) have found evidence of one or more vigilant profiles (Ellis et al., 2017; Lin et al., 2021). While such approaches are useful, particularly when examining classes or profiles of multisystem physiological responsivity, as previous studies have pointed out (Shakiba et al., 2020), these approaches are limited in that they do not allow for the examination of interactions between environmental factors. Since the current study was focused on interactions between contemporaneous exposure to daily stressors and early life adversity, mixture modeling was suboptimal. Therefore, future research would benefit from employing additional analytic methods that potentially better capture additional classes or profiles but can also accommodate environmental interactions.
Second, the estimated models examining daily cortisol output (operationalized as
Despite the contributions of the current study, the findings reported should be interpreted in light of at least six limitations. First, the current study focused on cortisol, the primary end product of the HPA axis. While cortisol has been examined within the context of the ACM in prior research (Peckins et al., 2015), the model is more directly focused on multisystem stress responsivity (Del Giudice et al., 2011; Ellis & Del Giudice, 2014); thus, future research aimed at employing a more comprehensive measure of stress responsivity would be beneficial. Second, and related, the ACM offers specific hypotheses focused on stress responsivity and recovery rather than the activity of the stress response system. Previous research has examined these hypotheses with laboratory-based stress tasks, which allow for more precise measurement of physiological responsivity and recovery. One of the contributions of the current study was a focus on everyday, organic stressor exposure, which does not permit such precision or the direct examination of stress recovery.
Third, while the overall MIDUS sample is nationally representative of U.S. adults between the ages of 25 and 74, the NSDE is only comprised of a subsample from the MIDUS study, raising questions surrounding external validity. Importantly, some demographic differences were observed between the NDSE subsample and the larger MIDUS II sample, but the examined childhood adversity measure did not significantly vary across the two samples, potentially tempering this limitation to some degree. Fourth, while the NDSE asked participants to report common daily stressors, the employed survey items were not exhaustive of all sources of possible stressors. For example, the survey asked about stressful events at home, but did not ask about family dynamics or the presence of younger children in the household. The broader focus of the employed questions may limit the generalizability of current results and future research should focus on a more specific set of daily stressors.
Fifth, and as mentioned previously, the employed analytic methods were well suited to address the nested nature of the examined data and to incorporate statistical interaction terms, but these methods did not permit the more direct identification of latent classes or profiles. Future studies employing a more flexible analytic approach that simultaneously allows for the identification of latent classes along with interactions between environmental influences would be beneficial. Sixth, measures of early life adversity relied on retrospective reports. While such data collection is consistent with previous literature (see Afifi et al., 2020; Brumley et al., 2019) and ACEs indexing (Felitti et al., 1998), future research should employ alternative measurement strategies such as hospital records, medical records, and police reports. Seventh, due to the sampling frame employed for the MIDUS study (i.e., all U.S. adults aged between 25 and 74), it is possible that the resulting analytic sample is not well populated with participants who experienced extremely high levels of early life adversity and/or daily stressor exposure in adulthood. The results of the current study may not apply to other, higher-risk populations. Future research would benefit from examining the extent to which the results of the current study replicate within additional populations that are differentially exposed to early life adversity and daily stressors in adulthood.
Despite these limitations, the current study sheds additional light on the role that early life adversity plays in calibrating and honing subsequent adrenocortical responses to current and future environmental challenges. Our findings demonstrate that early life adversity exposure is certainly impactful and meaningful, but the implications of such adversity do not appear to be monotonic and offer meaningful information that can be used to better adapt the stress response system to the current environment and to guide informed expectations about future environments. With that said, these findings in no way dismiss the consequences of early life adversity and fully acknowledge the potential harm of such experiences. Rather, our findings provide greater insight into the potential factors that explain between-individual differences in responses to early life adversity and demonstrate the importance of adopting an evolutionary-developmental framework in understanding such connections.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: In the interest of full disclosure, DAG is Scientific and Strategy Advisor at Salimetrics LLC and Salivabio LLC. These relationships are managed by the policies of the committees on conflict of interest at Johns Hopkins University School of Medicine and the University of California at Irvine.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.