Abstract
The present study aimed to extend the original focus of the dual-hormone hypothesis on testosterone and cortisol involvement in risk-taking to other sex hormones: estrogen and progesterone. The study also examined two alternative models for the proposed relationship between cortisol and sex steroids and for their joint influence on risk-taking: interaction and ratio terms. In all, 40 women and 37 men were tested for circulating sex hormones and provided self-reports on risk-taking. The findings suggest that sex hormone–cortisol ratios differentially modulate risk-taking in men and women: In men, high ratios were associated with risk-taking, whereas in women the opposite pattern was found. The findings are discussed in light of evolutionary assumptions regarding sex differences in neuroendocrine mechanisms, suggesting sex differences in neural sensitivity to sex hormones in risk-taking.
A considerable number of studies have reported sex differences in risk-taking, with men showing higher levels of risk-taking than women did in various types of risk, research designs (for a meta-analysis, see Byrnes, Miller, & Schafer, 1999), and countries (e.g., Kruger & Nesse, 2004). Given the universality of this phenomenon, an evolutionary perspective suggests that sex differences in parental investment and in intrasexual competition may underlie sex differences in risk-taking. Trivers’s (1972) elaboration of Darwin’s (1871) theory suggests that males and females differ in their parental investment strategies, which incline them to display varied degrees of intrasexual competition. Thus, males engage more frequently in male-to-male competition over mating opportunities than females do (Daly & Wilson, 1994). It has been suggested that women’s prolonged gestation and lactation periods, together with the fact that the infant’s survival depends on them, motivate them to adopt a strategy of “staying alive” that favors relative risk avoidance more so men do (Campbell, 1999).
The proposed evolutionary framework that underlies sex differences in risk-taking implies also the presence of a sex-differentiated, evolved biological infrastructure. For example, among the biological correlates, dopaminergic and serotonergic systems have been suggested as potentially important variables in risk-taking in men, with men’s greater sensation seeking deriving from a more reactive dopaminergic system (Zuckerman & Kuhlman, 2000). Hormones such as sex steroids and glucocorticoids have been also suggested as potentially important variables. Steroids have a wide range of influence, among others, on cell level, metabolism, behavior, mood, and cognition (Coates, Gurnell, & Sarnyai, 2010).
Sex steroids are chemical messengers. They include androgens, estrogens (E), and progestins, which are secreted by the gonads (ovaries in women and testes in men) and the adrenal glands in both sexes, through the hypothalamic–pituitary–gonads (HPG) axis; they affect a wide range of organs including the brain (Coates et al., 2010; Halpern, 2012). Sex steroids have been studied widely in animal and human models and are thought to have organizational as well as activational effects (Coates et al., 2010). Testosterone (T) has been linked to increased aggression (Archer, 2006), perceived social status (Eisenegger, Haushofer, & Fehr, 2011; Mazur & Booth, 1998) and sensation seeking (Roberti, 2004). Evolutionary theories suggest that T plays a significant role in modulating risky behaviors to adjust to changing social situations (Oliveira, 2004). A rise in T levels in nonhuman males, particularly in the breeding season, correlates with risky and competitive behaviors (Wingfield, Hegner, Dufty, & Ball, 1990). Empirical evidence from human studies suggests that T can play a role in individual differences in risk-taking, but findings are inconsistent. Some have documented a positive correlation between T and risk-taking, both with circulating T levels (Apicella et al., 2008; Coates & Herbert, 2008; Evans & Hampson, 2014; Ronay & von Hippel, 2010; Stanton, Liening, & Schultheiss, 2011; Vermeersch, T’sjoen, Kaufman, & Vincke, 2008b; White, Thornhill, & Hampson, 2006) and with T administration (Cueva et al., 2015; Goudriaan et al., 2010; Van Honk et al., 2004). Others did not find a significant correlation (for fluctuating T levels: Derntl, Pintzinger, Kryspin-Exner, & Schöpf, 2014; Rosenblitt, Soler, Johnson, & Quadagno, 2001; for T administration: Ortner et al., 2013; Zethraeus et al., 2009), found a negative association (Reavis & Overman, 2001), or found a nonlinear association between T and risk-taking (Sapienza, Zingales, & Maestripieri, 2009; Stanton, O’Dhaniel, et al., 2011).
Several studies have suggested also a role for E and progesterone (P) in risk-taking behaviors, but findings are again inconsistent. Some have demonstrated a positive association between E or P and risk-taking (Vermeersch, T’sjoen, Kaufman, & Vincke, 2008a; Derntl et al., 2014, respectively), whereas others failed to demonstrate such association (Reavis & Overman, 2001, for E and P; Zethraeus et al., 2009, for E), or found a negative association between E and risk-taking (Balada, Torrubia, & Arque, 1993; Derntl et al., 2014).
The inconsistent findings regarding the role of sex steroids in risk-taking behaviors can be explained based on the premise that the relationship depends on cortisol (C), the main human glucucorticoid and the end product of the hypothalamic–pituitary–adrenal (HPA) axis (Coates et al., 2010). High C level is associated with psychological stress and avoidant behaviors (Dickerson & Kemeny, 2004; Pfattheicher & Keller, 2014; Roelofs et al., 2009; Terburg, Morgan, & Van Honk, 2009; Tops & Boksem, 2011); low C level with psychological relaxation and approaching tendencies (Terburg et al., 2009). Empirical findings reveal a negative relationship between C levels and risk-taking behaviors, with higher levels of C being associated with risk-averse behaviors (Kandasamy et al., 2014; van Honk, Schutter, Hermans, & Putman, 2003).
The proposed relationship between hormones in explaining risk-taking behaviors is formulated in the dual-hormone hypothesis (DHH) for social dominance (Dabbs, Jurkovic, & Frady, 1991; Mehta & Josephs, 2010; Popma et al., 2007). DHH suggests a moderation effect of the HPA axis on the HPG axis, which regulates behavior. Reduction or excess in HPA axis activity (i.e., reduction or excess of C levels) may lead to an excess or reduction of HPG axis activity (i.e., excess or reduction of T levels). This cross talk between the HPA and the HPG axes may be understood through adaptive mechanisms that ensure the survival of organisms (Juster et al., 2016). For example, under stress conditions, the adrenal axis regulates the gonadal function by inhibiting reproductive behavior of the gonadal axis (Viau, 2002). This inhibitory mechanism is commonly found in dominance hierarchies. In many species, dominants show decreased C levels and increased T levels, whereas the opposite occurs in subordinates (e.g., Blanchard et al., 1995). The cross talk between the adrenal and the gonadal systems may affect risk-taking behavior (Mehta, Welker, Zilioli, & Carre, 2015). Empirical evidence provides support for this hypothesis, demonstrating a positive relationship between T and risk-taking behaviors only when C levels are low, across various designs (e.g., self-report, behavior; Mehta et al., 2015).
By examining the ratio between T and C, it is also possible to learn about the relationship between the HPA and HPG axes and their involvement in social behaviors such as risk-taking. It has been suggested that under conditions of threat, the T/C ratio indicates biological balance by moderating the automatic responses of approaching and avoidant behaviors, which are crucial for survival. The balance between behavioral activation and inhibition is based on the biological balance between HPA and HPG activity and on their end products, C and T (Terburg et al., 2009). The psychological balance is linked to a second balance that includes communication between cortical and subcortical areas, which is mediated by the opposite effects of C and T (Schutter & Van Honk, 2004, 2005). Therefore, the balance between these two endocrine systems may be best depicted by the ratio between their end products, aiming at capturing their opposite influences on behavior. Recently, several studies addressed this issue in their search for biological mechanisms in social aggression. They suggested that high T/C ratios predict proneness to confront threat, and therefore they predict aggressive behavior. Empirical findings provided support for this suggestion, showing that T/C ratio is a marker for aggression (Montoya, Terburg, Bos, & Van Honk, 2012; Romero-Martinez, Gonzalez-Bono, & Moya-Albiol, 2013; Terburg et al., 2009) and psychopathy (Glenn, Raine, Schug, Gao, & Granger, 2011).
The present study aims to investigate T/C ratio as a marker for risk-taking behavior. Because former studies have demonstrated the link between approaching and risk-taking behaviors (Kandasamy et al., 2014), and because these behaviors have been linked to T levels (e.g., Apicella et al., 2008; Mazur & Booth, 1998; Reavis & Overman, 2001), albeit inconsistently, there is a need to explore T/C ratio as a biomarker that combines these two hormones into one entity, to account for the interaction between the HPA and HPG axes and its connection with risk-taking behavior.
Another aim of the present study is to examine the involvement of two other sex steroids and their relations with C in risk-taking behavior. T and C interaction has been examined broadly across various social behaviors, focusing on T and C exclusively. But the theoretical infrastructure at the basis of this interaction involves a wider range of hormones that play a role in regulating social behavior, especially risk-taking. Recently, Tackett et al. (2015) have extended their focus beyond T and C and examined also the role of E in externalizing behaviors of adolescents. The authors suggested that E is an important target hormone that extends the existing evidence of the cross talk between the HPA and HPG axes. Although E is identified as a female hormone, it is produced also in males and synthesized by aromatization of the circulating T (de Ronde, Pols, Van Leeuwen, & De Jong, 2003). These findings confirmed the DHH: A positive relationship exists between E and externalizing behavior, but only when C levels are low.
Inconsistencies found in previous studies regarding the connection between the HPA and HPG axes with respect to risk-taking behaviors may be explained by sexual dimorphism in the stress response system (Del Giudice, Ellis, & Shirtcliff, 2011). Although both males and females engage in intrasexual competition, they differ in the form and degree of male–male and in female–female competition (Buss, 1988; Buss & Dedden, 1990; Walters & Crawford, 1994). Females and males compete for resources to enhance their reproductive success: Males compete for resources and status and women for enhancing offspring survival. The greater importance of personal survival in females inclusive fitness results in higher fear of physical harm and therefore inclines females toward risk aversion. In situations that entail risk-taking, such as those related to securing resources, females engage in a low-risk and indirect form of dispute resolution, unlike males (Campbell, 1999). Therefore, it is assumed that the different nature of the risks that evolution imposed on men and women, resulting in different strategies of dispute resolution, required a different supporting biological substrate for each sex. Previous studies have demonstrated different association patterns between C and T in men and women. In men, low levels of C, together with high levels of androgens, resulted in displays of antisocial behavior (e.g., Popma et al., 2007). Furthermore, low basal C levels were associated with higher increase in T levels following exposure to social stress (Bedgood, Boggiano, & Turan, 2014). Conversely, among women athletes, high basal C levels moderated reduced T reactivity in a competition (Edwards & Casto, 2015). Recently, Juster et al. (2016) have explored sex differences in C profiles, adjusted and unadjusted for basal T, E, and P. The findings revealed, among others, that T was negatively associated with reactive C among menopaused women, but not among men. P was also negatively associated with reactive C only among men. The authors suggested that differences in basal C levels may activate or deactivate the pathway linking sex hormones and behavior differentially within and between the sexes.
The accumulated empirical evidence suggests that a corresponding neuroendocrine infrastructure is needed in each sex to support adaptive stress responses, with different relationships between the HPA and HPG axes in men and women. The present study deepens our understanding of the relationship between the HPA and HPG axes by covering a wider range of sex hormones representing the HPG axis (T, E, and P), as we search for the biological basis of risk-taking behavior, based on a previously suggested mechanism involving the ratio between T and C, and expanding it to other hormones representative of the HPG axis: E and P. By examining ratio versus interaction, we expect to raise direct hypotheses regarding the relationship between sex hormones and C. Specifically, we address the potential buffering effect of C on the influence of sex hormones on risk-taking.
Based on the principles of natural selection, the interconnectivity between the HPA and HPG axes and its relation to risk-taking are expected to be moderated by sex. We hypothesize that in men, high sex hormone/C ratios predict risk-taking, whereas in women, high sex hormone/C ratios predict low risk-taking.
Method
Participants
Seventy-seven students from a college in Israel (mean age 25.78 ± 2.75 years) participated in the study. Of all, 40 participants (52%) were female (mean age 25.35 ± 3.31 years) and 37 (48%) were male (mean age 26.24 ± 2.10 years).
Participants were recruited through advertisements at the psychobiology lab of the college and received monetary compensation for their participation. All participants reported that they were in good physical health, had no psychiatric or neurologic illnesses, were not taking any psychoactive or hormone-based substances, and were heterosexual. None of the women were currently using oral contraceptives, and all women were tested between Days 10 and 14 of their menstrual cycles (follicular phase), as determined by self-reports.
Measures
The study used self-report questionnaires to measure the following.
Risk attitude
Perceptions about risk tendencies were measured using the validated general risk question of Dohmen et al. (2011): “How willing are you to take risks, in general?” Respondents rated their willingness on a scale from 1 (
Personal details
Sociodemographic information including age, gender, and income level.
Sex Hormone Measures
Endogenous hormone levels
Levels of C, E, T, and P were measured. Because of circadian variations in hormones, all participants were tested at the same time of day (08:00–08:30 a.m.). Venous blood samples (10 ml) were taken at the beginning of the test session. One blood sample was taken from each participant. Samples were batch tested in duplicates for T, E, and P levels using TSTO, eE2, and PEGE kits (Siemens Healthcare Diagnostics Inc., Tarrytown, NY) in an ADVIA CENTAUR XP system. This sandwich immunoassay technique uses direct chemiluminescent technology in a random access apparatus. For T, intraassays coefficients of variances (CVs) were 4.7% and 6.2%, and interassays CVs were 4.4% and 4.7%, for low and high T samples, respectively. For E, intraassays CVs were 11.1% and 2.7%, and interassays CVs were 2% and 2%, for low and high E samples, respectively. For P, intraassays CVs were 12.4% and 2.5%, and interassays CVs were 2.6% and 3.1%, for low and high P samples, respectively. For C, intraassays CVs were 3.69% and 3.82%, and interassays CVs were 5.45% and 1.86%, for low and high C samples, respectively.
All hormones were in the normal range for every male and female participant, except for one female participant, whose P level exceeded the mean score by 3 standard deviations (
Procedure
The study was approved by the institutional ethical review boards of the Yezreel Valley College. After giving their fully informed consent, participants completed a brief demographic questionnaire. The study began between 08:00 a.m. and 08:30 a.m. First, all participants were tested for sex hormones, after which they completed the questionnaire.
Statistical Analysis
T, E, P, C, and risk-taking were not significantly skewed. T, E, P, and C were standardized to
Results
Table 1 shows the mean hormone concentrations of C, T, E, and P for men and women. As expected, men’s T levels were higher than women’s, whereas women’s E and P levels were higher than those of men. Consistent with previous research (e.g., Mehta et al., 2015), men and women did not differ in C levels.
Means (Standard Deviations),

Tables 2 and 3 show zero-order correlations between hormones and risk-taking both for the full sample and in men and women separately.
Correlations.

***
Correlations (Males and Females Separately).

*
Interactions
A multiple regression with risk-taking scores as the dependent variable and with sex, T × C interaction, and T × C × Sex interaction revealed a significant T × C × Sex interaction, β = −2.08,
Multiple Regression Models of Sex × Hormone Interactions Predicting Risk-Taking.

Ratios
Multiple regression analyses were used to examine whether T/C, E/C, and P/C ratios and sex interact to predict risk-taking behavior (Table 5). Consistent with the hypothesis, there was a significant Sex × T/C ratio interaction for risk-taking, β = −.1.21,
Multiple Regression Models of Sex × Hormone Ratio Interactions Predicting Risk-Taking.


Sex × Testosterone/Cortisol ratio interaction for risk-taking.

Sex × Estrogen/Cortisol ratio interaction for risk-taking.
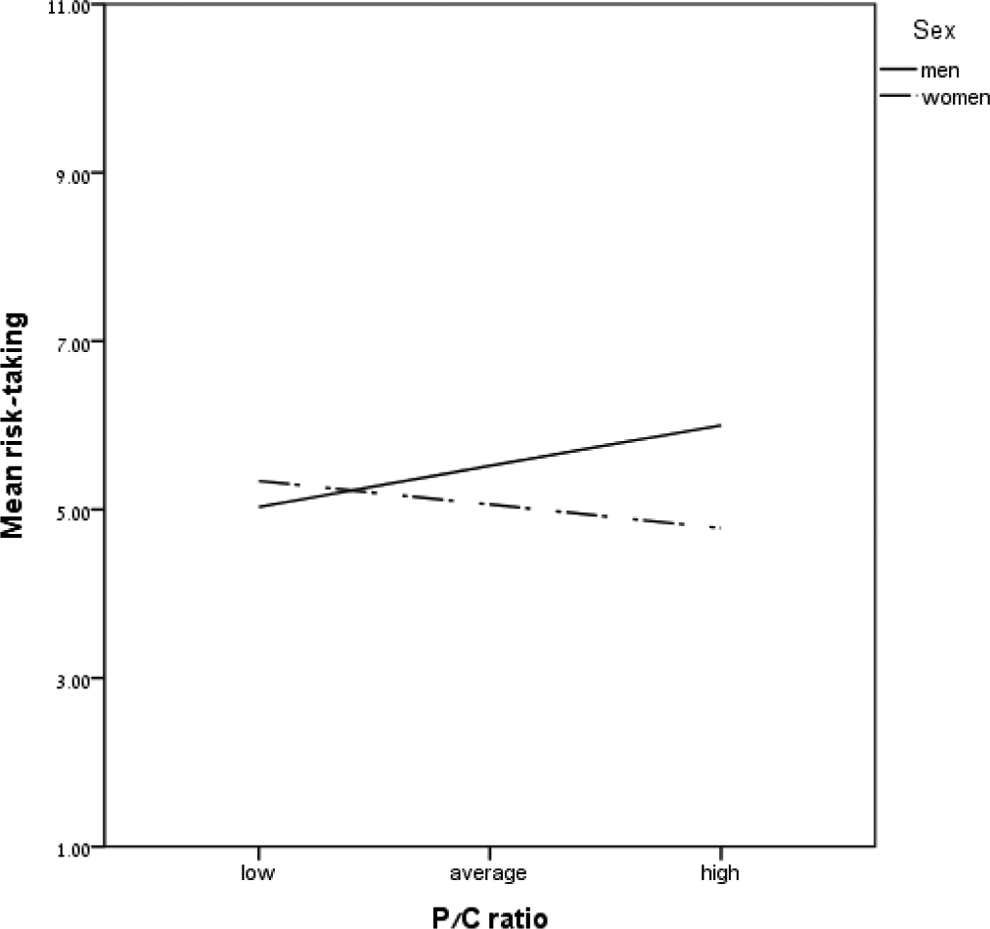
Sex × Progesterone/Cortisol ratio interaction for risk-taking.
Discussion
The current study sought to extend the original DHH concerning T and C to other sex steroids: E and P. The study explored the relationship between sex hormone/C ratios and risk-taking. Following separate analyses for the different suggested models, focusing on the interaction versus ratio term, it was found that the best model fit supports the ratio hypothesis of Terburg, Morgan, and Van Honk (2009). Considering all three sex steroids examined, our findings revealed an opposite direction of influence for men and for women. With regard to the hypothesis concerning the interaction between sex and T/C ratio predicting risk-taking, our results showed that in men, T/C was positively associated with risk-taking, that is, high levels of T and low levels of C predicted self-reported risk-taking. This finding is in accordance with previous studies based on the theoretical framework of DHH (Mehta & Josephs, 2010; Mehta et al., 2015). T/C ratio reflects the interconnections between two hormonal axes, HPA and HPG, which are critical for maintaining the balance between behavioral activation and inhibition. The direction of causality, however, is unclear. Some have proposed that T, the end product of the HPG axis, inhibits HPA axis activity (Viau, 2002); others pointed in the alternative causal direction (Mehta & Josephs, 2010). Nevertheless, the interconnectivity between the axes reflected by high T/C ratio predicts an inclination for socially aggressive behavior, specifically risk-taking, as shown in the present study.
A similar direction of influence was found with regard to the interaction between sex and E/C ratio and between sex and P/C ratio in risk-taking among men. E/C and P/C were positively associated with risk-taking, so that high levels of E or P and low levels of C predicted self-reported risk-taking. E has been identified as a female hormone, but in men it is produced and synthesized from T through the enzyme aromatase (de Ronde et al., 2003). Some have suggested that E is the main factor in the etiology of male aggression (e.g., Finkelstein et al., 1997). The present study provides support for previous findings showing an association between E and externalizing psychopathology only when C levels are low (Tacket et al., 2015). The authors stressed the importance of broadening our understanding of the HPA and HPG axes. Their study, which takes an activational approach to hormonal secretion and influence, focused on adolescence, a crucial developmental phase. Our findings in young adults support this theoretical framework, emphasizing the role of the key hormone, E, and its relationship with C in modulating risk-taking behaviors. With regard to the P/C ratio, to the best of our knowledge, there has been no previous study assessing P in men and its involvement in risk-taking. In their investigation of the involvement of gonadal hormones in sensation seeking, Daitzman and Zuckerman (1980) did not find a correlation between P levels and disinhibition (a subscale of the sensation-seeking scale) in men. Investigations of this type are more common in animal models. Studies exploring the influence of P withdrawal (PWL) on risk-taking behavior have demonstrated that following PWL male rats showed a higher incidence of open-space avoidance and more close-to-the-wall behavior, reflecting risk-avoidant behaviors, in contrast with the control group (Gulinello, Gong, & Smit, 2001; Löfgren, Johansson, Meyerson, Lundgren, & Bäckström, 2006). Recently, Juster et al. (2016) have shown that P was negatively associated with reactive C among men. In the present study, higher levels of P were associated with risk-taking, but this association was true only when C levels were low.
Our findings show that the influence patterns of sex hormones and C on risk-taking in men are aligned. In a low C condition, each of the sex hormones (T, E, and P) is associated with elevated risk-taking in men. These findings provide an important elaboration of the DHH theory, beyond the interaction between T and C, to other key gonadal hormones, E and C. The interconnectivity between the HPA and HPG axes in men reveals that high levels of sex steroids lead to risk-taking when C is low. These findings contribute to a growing body of research investigating a cluster of related tendencies and behaviors including social aggression, impulsivity, and risk-taking. Terburg et al. (2009) have suggested that motivational tendencies to confront threat, and the ensuing aggressive behaviors, originate in the emotional brain and are mediated by cognitive control. A high T/C ratio reduces the effect of cognitive control, which in turn predisposes for confrontation and social aggression. The present findings suggest that this mechanism is not limited to T, but applies also to E and P levels, interacting with C in risk-taking in men.
Examination of the results among women reveals an opposite pattern to the one exhibited by men. Both T/C and E/C were negatively associated with risk-taking, so that low levels of T or E and high levels of C predicted risk-taking. This finding contradicts the previous ones showing a positive association between T or E and risk-taking among low C individuals, including women (for T × C interaction, see Mehta et al., 2015; for E × C interaction, see Tackett et al., 2015). Previous studies have demonstrated the same result patterns for both men and women, as opposed to the present findings, in which the effects for men and women mirror each other. In women, we have found that gonadal hormones (T and E) function as a buffer for risk-taking behavior: When their levels are high and C levels are low, lower risk-taking is apparent. A few studies have documented a negative association between E levels and social behaviors relating to risk-taking in women (impulse control, thrill, and adventure seeking; Balada et al., 1993; Warren & Brook-Gunn, 1989). But the present study deepened this association through a moderating effect: The negative association between E and risk-taking is present only when C levels are low. The literature regarding the involvement of gonadal hormones in risk-taking behavior, especially in women, is inconsistent. The present study sheds new light on the combined influence of the HPA and HPG axes, which differentially modulate risk-taking behavior of men and women.
The present findings provide support for the evolutionary assumption that stress responses were selected differentially in men and women. These sex differences are supported by the neuroendocrine foundation of the stress regulatory system. In the present study, low C levels coupled with high sex hormone levels were associated with high risk-taking in men, but low risk-taking in women. These diversities may serve as an adaptive mechanism that promotes maternal and affiliative behavior for the purpose of offspring protection (McCarthy, 1995). Because offspring survival depends largely on the mother’s survival, females are motivated to adopt a strategy of staying alive that favors relative risk avoidance by comparison with men. Although both sexes engage in intrasexual competition, unlike males, females engage in a low-risk, indirect form of dispute resolution (Campbell, 1999). Therefore, it is assumed that different strategic dispute resolutions evolved in men and women, which required a different supporting biological substrate for each sex. The present study suggests that the connections between the HPA and HPG axes function differently in each sex, as a vestige of our evolutionary history. This differentiation is consistent with earlier findings concerning individual variations in risk-taking and aggression due to variability in neural sensitivity to steroid hormones (Mehta et al., 2015; Rosvall et al., 2012). Further studies must deepen our understanding of sex differences in the neuroendocrinology systems to uncover the resulting differences in other domains that interact with risk-taking such as psychopathology.
The present study has certain limitations. First, several studies have previously demonstrated racial and ethnic variability in circulating sex hormones. For example, Rohrmann et al. (2007) have found that Mexican American men had higher T levels than non-Hispanic White men, and non-Hispanic Black men had higher E levels than non-Hispanic White men. In the present study, ethnicity was not examined because of the large diversity in ethnic groups in Israel. Israeli society in the 21st century represents a mosaic of ethnic groups following massive immigration from a wide range of countries (Goldscheider, 2015). Because of the proposed linkage between ethnicity and variation in sex hormones, it is important to include ethnicity as a variable in a future study investigating the cross talk between HPA and HPG axes. Second, a word of caution is in order, given the relatively small sample size in the current study. It is possible that the present results are driven by C because of high within-subject variation in sex hormones (especially T in men, which because of the sampling schedule, at 08:00–08:30 a.m., encompasses high T variation), which in turn may override the between-subjects variation in the regression. Although the interactions between sex and hormone ratios reached significance, these findings call for further replication on larger samples and across risk-taking measurements (self-reported and behavioral). Furthermore, although we restricted the sampling schedule to 08:00–08:30 a.m., we did not control for the time since waking. Previous studies have suggested that the free C response to awakening is able to uncover subtle changes in HPA activity (Wüst et al., 2000); therefore, future studies should assess this index in investigating adrenocortical activity. Third, we measured men and women in the morning, in the follicular phase of the menstrual cycle of women not using oral contraceptives. Sex hormones and C have different circadian rhythms. To capture the complex interconnectivity between the HPA and HPG axes in risk-taking, future studies must expand the investigation and include different times of day, different phases of the menstrual cycle, and groups of women who use various contraceptives. Nevertheless, the present study expands upon the original DHH by focusing not only on T and C but also on E and P. This broader examination should apply not only to normative samples but also to psychopathological social behavior. Furthermore, previous studies exploring risk-taking, especially financial risk-taking, in experimental settings or field studies limited their investigation to male samples. In light of the findings of the present study about the differential effects of sex hormones and C on men and women, future studies on financial risk-taking should further expand the exploration by including several sex steroids and by using mixed-sex samples.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
