Abstract
Research on cooperation has contributed to a better understanding of the foundations of human social behavior. Most studies, however, have not considered fundamental social parameters such as an individual’s position in a social hierarchy. As a first step, this study investigates the modulating effects of socioeconomic status (SES) on behavior and the physiological stress response. Study participants (n = 83) played a cooperative game with computerized coplayers of four categories: similar or higher SES in combination with either high or low fairness in behavior (i.e., willingness to cooperate). All participants showed a significant increase in saliva cortisol after the game compared to a control group. Only when paired with higher SES coplayers, however, did participants show a significant subsequent decrease in cortisol concentrations. Participants’ behavior in response to a coplayer’s decisions was only affected by the degree of fairness, but not the SES, of respective coplayers. Physiologically, playing this cooperation game was a big challenge for participants as measured by salivary cortisol. Yet, the high recovery rate when playing with cooperative, higher status individuals showed the stress-protective effects of positive social interactions in the framework of social hierarchies.
The evolutionary explanation for the development of cooperative behavior, defined as costly behavior aimed to benefit other people (Nowak, 2006), is based on a direct and indirect fitness calculation between interacting individuals (Boyd & Richerson, 2009; Hamilton, 1964; Nowak & Sigmund, 2005; Trivers, 1971; West, Griffin, & Gardner, 2007). This phenomenon has been described for all levels of biological organization—including molecules, organelles, cells, and vertebrates (between individuals or groups)—which underlines its importance in the animal kingdom per se (Clutton-Brock, 2002; Dariima, Jin, Lee, Wall, & Kim, 2013; Kimura, Hase, & Ohno, 2012; Sakisaka, Ikeda, Ogita, Fujita, & Takai, 2007). During the past decades, the incorporation of different theories concerning the function of cooperation (e.g., kin selection, direct reciprocity, and network reciprocity) as well as possible mechanisms for the evolutionary stability of cooperation (e.g., punishment) has improved our understanding of this behavior in terms of evolution and sociology (Nowak, 2006). Rilling (2011) suggests a neurobiological model that regulates cooperation in primates, proposing that the emotional prerequisite for cooperation is located in the orbitofrontal cortex area of the brain. This area, together with the brain reward system, enforces cooperation and is affected in response to unreciprocated cooperation (Rilling et al., 2008). Correspondingly, the reward system seems to be sensitive to psychosocial stressors as corroborated by increased cortisol concentrations (Oei, Both, van Heemst, & van der Grond, 2014). Psychosocial stressors activate the hypothalamic–pituitary–adrenal (HPA) axis, thereby triggering a cascade of hormone secretions from the brain. This ultimately promotes a peripheral increase in the cortisol release from the adrenal gland. Secreted cortisol molecules are transported to the brain and inter alia negatively affect the brain reward system (Chrousos, 1998). While high cortisol hormone levels from chronic stress are disastrous to health across a variety of species, short-term cortisol elevations followed by efficient recovery have positive effects in terms of physiological arousal and increased attention and memory (e.g., Schwabe, Bohringer, Chatterjee, & Schachinger, 2008; Smeets, Giesbrecht, Jelicic, & Merckelbach, 2007).
In humans and nonhuman primates, social hierarchies are thought to represent key psychosocial stressors (Abbott et al., 2003). Hierarchy systems have evolved in all nonhuman primates (e.g., dominance hierarchy) and are associated with asymmetric access to resources, also in human societies (e.g., socioeconomic status [SES]; Wallner, Machatschke, & Fieder, 2008). In nonhuman primates, hypercortisolism—a chronic and pathological elevation of cortisol exposure over the day—is generally associated with subordination, in which affected individuals receive less social support (Sapolsky, 2005). Studies on humans showed that individuals holding a—subjectively—low SES tend to have chronically increased basal cortisol levels (Adler, Epel, Castellazzo, & Ickovics, 2000; Kunz-Ebrecht, Kirschbaum, & Steptoe, 2004) and especially flattened slopes of—declining—diurnial cortisol secretion (Agbedia et al., 2011). Low subjective SES is usually accompanied by feelings of a lack of control in day-to-day life (Adler et al., 2000). Correspondingly, a lack of sufficient social coping outlets accompanies the exposure to higher frequencies of potentially severe stressors in subjects with a low SES (Uchino, 2006). This is regarded as one major reason why such persons develop hypercortisolism (Gersten, 2008; Sapolsky, 2005) and could result in low-SES subjects seeking additional nonsocial coping outlets such as gambling or drug abuse (Pampel, Krueger, & Denney, 2010; Wood & Griffiths, 2007). Interestingly, besides well-investigated coping outlets such as social support in laboratory experiments (Heinrichs, Baumgartner, Kirschbaum, & Ehlert, 2003), establishing cooperation itself might represent a mediating factor in coping with psychosocial stress by influencing HPA activity. A recent study documented low stress reactivity and decreasing saliva cortisol concentrations in response to successful cooperation in a Lego-house building task (Moya-Albiol et al., 2013). The observed rates of recovery to the basal state were comparable to those in studies providing people with different coping outlets after exposing them to acute psychosocial stress (Heinrichs et al., 2003) such as low subjective social status (Von Dawans, Fischbacher, Kirschbaum, Fehr, & Heinrichs, 2012).
While research has been done on the relationship between social status or cooperative effort and physiological patterns of cortisol reactivity and recovery (Moya-Albiol et al., 2013; von Dawans et al., 2012), little is known about the combined influence of both factors on endocrine stress and behavior. Coping mechanisms and reward system activation might favor seeking cooperative outcomes in interactions with high-status individuals in order to benefit from otherwise restricted resources (both material and nonmaterial ones such as information). This could potentially boost one’s own status because of the proximity to a high-status individual and would be in line with Michael Chance’s (1967) observations of primate behavior: Attention is preferentially directed toward high-in-status conspecifics (for a recent review on attentional bias in humans and nonhuman primates, see Koski, Xie, & Olson, 2015), and humans preferentially learn from others who have high status (e.g., Atkinson, Mesoudi, & O’Brien, 2012). From a proximate, mechanistic perspective, this means that if cortisol is a modulator of social interaction, and the cortisol response is sensitive to relative SES differences, then the cortisol responses of individuals interacting in a potentially cooperative situation could differ along the gradient of subjective SES. Therefore, this study was designed to investigate how SES differences and the degree of cooperation modulate cortisol reactivity and rates of recovery as well as future cooperative behavior. For this, we programmed a cooperative game. Participants were randomly assigned to computer-simulated higher or similar SES partners who reciprocated cooperative behavior largely or little. We predicted higher cortisol reactivity and lower rates of recovery in subjects holding a similar SES during an interaction. Furthermore, lower cortisol reactivity and higher recovery rates were expected for subjects facing cooperative partners. The investigation of the combined effects of SES and (un)successful cooperation on cortisol responses and behavior was of exploratory nature.
Materials and Method
Participants and Study Design
For the study, 106 (n = 91 experimental group; n = 15 control group) healthy male students (mean age: 21.4 years ± 2.8 SE) were recruited from the University of Vienna. During 3 weeks, the study was carried out in a computer lab at the university (beginning either at 08:30 a.m. or 09:45 a.m.). The lab had no windows, and light and temperature (21°C) conditions were constant. The study design was programmed using z-Tree experimental software 3.3.10 (Fischbacher, 2007). It consisted of a game period followed by two relaxation periods and lasted for a total of 60 min. The game period was designed to last for about 20 min.
In response to a stressor, increases in saliva cortisol are usually used as a measure of cortisol reactivity. Time of recovery to the basal state is another important index for the severity of a psychosocial stressor (Sapolsky, Romero, & Munck, 2000; Seeman & Robbins, 1994) and coping ability. To evaluate the cortisol rate of recovery to the basal state, a subsequent relaxation period lasted 40 min in total. During the first 20 min of relaxation, subjects watched photographs of restorative landscapes followed by educational cartoons for another 20 min. To evaluate the effects of the laboratory condition on participants’ physiological response, we also conducted a control group testing. In this group, an additional educational cartoon replaced the game stage.
At the beginning of the study, subjects were randomly assigned to a computer. Opaque partitions separated the workplaces and ensured anonymity of decision-making. Participants received a written game instruction, which was orally repeated by the instructor. They then had to fill out a short questionnaire on personal, SES, and health data and were informed that their personal and SES data are forwarded to the coplayer and that they receive the same information about the coplayer. However, participants actually played against computer-simulated coplayers, a fact that participants were not instructed about until debriefing. We defined SES by age, monthly net income, degree of education, and supervising responsibility in companies of either self-employed or employed persons (Featherman & Hauser, 1977; Fieder et al., 2005). A typical higher SES coplayer was above 35 years of age, held at least a master’s degree, supervised more than five people in his job, and received a monthly net income of above €3,000 (approximately double the mean monthly net income of an Austrian male employee in the year 2012—data: Statistics Austria). In contrast, a similar SES (sim-SES) coplayer was less than 25 years old, held at most a bachelor’s degree, was a supervised employee, and received a monthly net income of below €750. Our participants’ characteristics matched well with those of sim-SES coplayers. Ninety-seven percent of the participants were undergraduates, only 3 of the 25 employed subjects had supervising responsibility, and 11 had a monthly net income of slightly more than €750.
Game Design
Situations in which people lack control over a stressor are known to elicit clear and pronounced endocrine stress responses (Dickerson & Kemeny, 2004). Therefore, we designed the game in such a way that players do have only limited control over the game outcome, that is, participants were always first movers and were instructed that their coplayers could observe their choices before responding. Coplayer responses were labeled “fair” when they cooperated (i.e., the coplayer bore the cost of a participant receiving a positive share) and “unfair” when they refused to cooperate (i.e., the coplayer did not bear the costs of a participant receiving a positive share). The payoff structure and coplayer programming were designed to make a coplayer’s behavior more reasonable and the game less controllable: 100 experimental currency unit (ECU) if both players choose the fair option, 0 ECU if only the participant chooses fair (110 ECU for coplayer), 90 ECU if only the coplayer chooses to play fair (0 ECU for coplayer), and −10 ECU if both players refused to choose the fair option. Coplayers were programmed to always choose the unfair option in response to unfair choices by participants and to reciprocate fair choices in 45% (high fairness) or 20% (low fairness) of the cases, respectively. These categories yield four different types of coplayers: sim-SES/low fairness, sim-SES/high fairness, higher SES/low fairness, and higher SES/high fairness. Scenario participants played two subsequent repeated cooperation games (eight rounds each). The game was designed to last about 20 min in order to elicit a pronounced stress response. Coplayers in both games were of the same category, with only descriptive details differing. This way, the effects of individual associations with a specific coplayer’s profile are reduced, which outweighs the benefits of intraindividual comparisons with regard to SES and cooperation in a time-constrained setting (e.g., directed attention fatigue). SES profiles of players were displayed at the beginning and after Round 4 of each game. The game was designed to last about 20 min in order to elicit a pronounced stress response.
A participant’s financial compensation (in euro) corresponded to his earnings (in ECU) at the end of the experiment. ECU 100 equated to €1. All research protocols and the study design were reviewed and approved by the Ethic Commission of the University of Vienna # 00020.
Saliva Cortisol Collection and Analysis
The sampling regime included three resting samples on nonexperimental working days. For these samples, all subjects received written instructions and were urged to collect morning samples on an empty stomach (approximately at 8 a.m.). Throughout an experimental session, three saliva samples were collected: Sample 1 (test) immediately after the game, recovery Sample 1 (Recovery 1) 20 min after the game, and recovery Sample 2 (Recovery 2) 40 min after the game. For the latter samples, a collection request was displayed on the computer screen. All samples (3–5 ml) were collected in plastic tubes and stored at −20°C until analysis. Saliva cortisol levels were measured using an enzyme-linked immunoassay as described in Machatschke and colleagues (2008). Cross reactions with relevant steroids were 4-pregnene-11β, 21-diol-3, 20-dione 6.2%; 4-pregnene-11β, 17α, 21-triol-3, 20, dione 100%; 5α-pregnane-11β, 17α, 21-triol-3, 20, dione 4.6%; 5α-pregnane 3α, 11β, 17α, 21-tetrol-20-one 0.8%; and 5β-pregnane-3α, 11β, 17α, 21-tetrol-20-one 0.1%. All other steroids cross reacted with 0.01%. The intra- and interassay coefficients of variance were 8.9% and 11%, respectively. The saliva concentrations did not systematically differ between those groups of participants beginning the study at 08:30 a.m. and those beginning at 09:45 a.m. Hence, these were pooled in the following analyses or, as in the case of mixed model calculus, a time component was included as random factor.
Medication as Intervening Variable
Eight of the ninety-one players who participated in the experiments reported to suffer from allergic asthma that is treated with inhaled corticosteroids. In such individuals, cortisol responses are often delayed and markedly attenuated (Heijsman, de Vries, Wolthuis, & Kamps, 2011; Wlodarczyk, Gibson, & Caeser, 2008) also in response to acute psychosocial stress (Buske-Kirschbaum et al., 2003). Acutely administered corticosteroids reduce asthma symptoms significantly but do also have—maladaptive—side effects on decision-making (Montoya, Terburg, Bos, & Van Honk, 2012; Putman, Antypa, Crysovergi, & van der Does, 2010), as they reduce fear (Buchanan, Brechtel, Sollers, & Lovallo, 2001; Montoya et al., 2012; Oei, Tollenaar, Spinhoven, & Elzinga, 2009) and increase risk seeking (Putman, Hermans, & van Honk, 2010) or even aggressiveness (Wallner & Machatschke, 2009). Therefore, the authors decided to exclude these participants from the statistical analyses presented in the Results section. Full mixed model calculus—including those eight participants receiving asthma medication—are provided in the Appendix section (see Appendix Tables A1–A2).
Statistics
To test for increased cortisol reactivity in response to the experiment, saliva cortisol after-test concentrations (test) between the test and control group were compared using an independent sample t-test. Significance tests and bias-corrected 95% confidence intervals (BC-CI) are based on 9,999 bootstrap replicates.
Cortisol ratios between after-test values (test) and recovery values (20 min and 40 min after the test) were calculated by applying the following formula: recovery value/(recovery value + test). Both values were used to assess poststressor recovery to the basal state in participants. Poststressor recovery as well as absolute differences in cortisol concentrations throughout the testing were analyzed using mixed effect models with a player’s repeated measurements and a time component as random factors. Here, we regressed absolute concentrations of repeated samples (resting, testing, and relaxing values) and cortisol ratios of players to respective grouping variables (SES and fairness of a coplayer). In both models, response variables were grand mean centered to help interpret fixed effects.
Individual differences in fairness frequencies were analyzed with generalized mixed effect models with a logit link function and binomial variance (lme4 package, version 1.0-4, Bates, Maechler, Bolker, & Walker, 2013) with a player’s repeated choices and a time component as random factors. Due to the repeated measurements design, both games were analyzed separately and compared thereafter. Decisions to either play fair or unfair were regressed to respective grouping variables (SES and fairness of a coplayer) as well as the round of decision. This procedure enabled not only examining group differences concerning mean probabilities but also assessing behavior changes over time. Significance was set at p ≤ .05. All statistical calculations were carried out using the SPSS software package version 19 and R statistical software (R Core Team, 2013; version 2.8.1, http://www.r-project.org).
Results
Effects on Cortisol Levels and Recovery
A comparison of saliva cortisol concentrations between test, mean ng/ml 27.59 (±1.64 SE), and control group mean ng/ml 20.59 (±2.98 SE) showed a significant increase in cortisol secretion in response to the experimental game for test group participants (t = 1.66, p = .049, 95% BC-CI [0.30, 14.21]). The comparisons between test and control groups after 20 min and 40 min of recovery were not statistically significant (t = 1.30, p = .218, 95% BC-CI [−3.04, 11.02]; t = .31, p > .761, 95% BC-CI [−8.52, 9.32]; see Figure 1).

Comparison of cortisol concentrations between experimental group and controls. Whiskers represent mean ± 1 SE, p ≤ .05.
To evaluate influencing factors on cortisol reactivity and the ratio of recovery to the basal state, we applied mixed model regressions. Based on absolute values of cortisol concentrations, no group differences with regard to coplayer SES or coplayer fairness (Table 1) could be detected. However, regressions on the ratio of recovery (after game/resting cortisol) showed a significant effect of a coplayer’s SES (Table 1). For subjects interacting with coplayers of higher SES, the ratio of recovery to the basal state was significantly increased in comparison to those subjects facing coplayers of similar SES (see also Figure 2). Neither experiencing fairness in coplayer’s behavior nor a combined effect of a coplayer’s fairness and SES significantly influenced the ratio of recovery in response to the experiment.
(Generalized Linear) Mixed-Model Regressions on Physiological Postgame Recovery (Ratio of Recovery) and Absolute Cortisol Concentrations.

Note. n = 83. Numbers are regression estimates with SEs in parentheses and absolute p values. Numbers in brackets are 95% confidence intervals (based on 9,999 bootstrapped replicates). SES = socioeconomic status of a coplayer; Fairness = probability for coplayer cooperation.

Intragroup courses of cortisol concentrations in players interacting with coplayers of either similar (a) or higher (b) socioeconomic status (SES; cortisol concentrations: 1 = resting, 2 = after test, 3 = relaxing after 20 min, 4 = relaxing after 40 min). Different line colors code for the reactivity (red) in response to the cooperation game as well as the recovery to the basal state (green) thereafter. Error bars represent mean ± 1 SE. The ratio of after game/resting values was significantly higher when playing with a higher SES coplayer.
Effects on Cooperative Behavior
In general, participants decreased their cooperative effort, as more rounds were played throughout both games. Mixed model regressions on decisions in Game 1 and Game 2 (see Table 2) further revealed significant differences in behavior in relation to the fairness of the interaction partner. Participants adopted a coplayer’s behavior during each game (see also Figure 3a and b). SES however did not affect a participant’s behavior neither alone nor in interaction with a coplayer’s fairness.
Generalized Mixed Effect Models With a Player’s Repeated Choices in Game 1 and a Time Component as Random Factors.
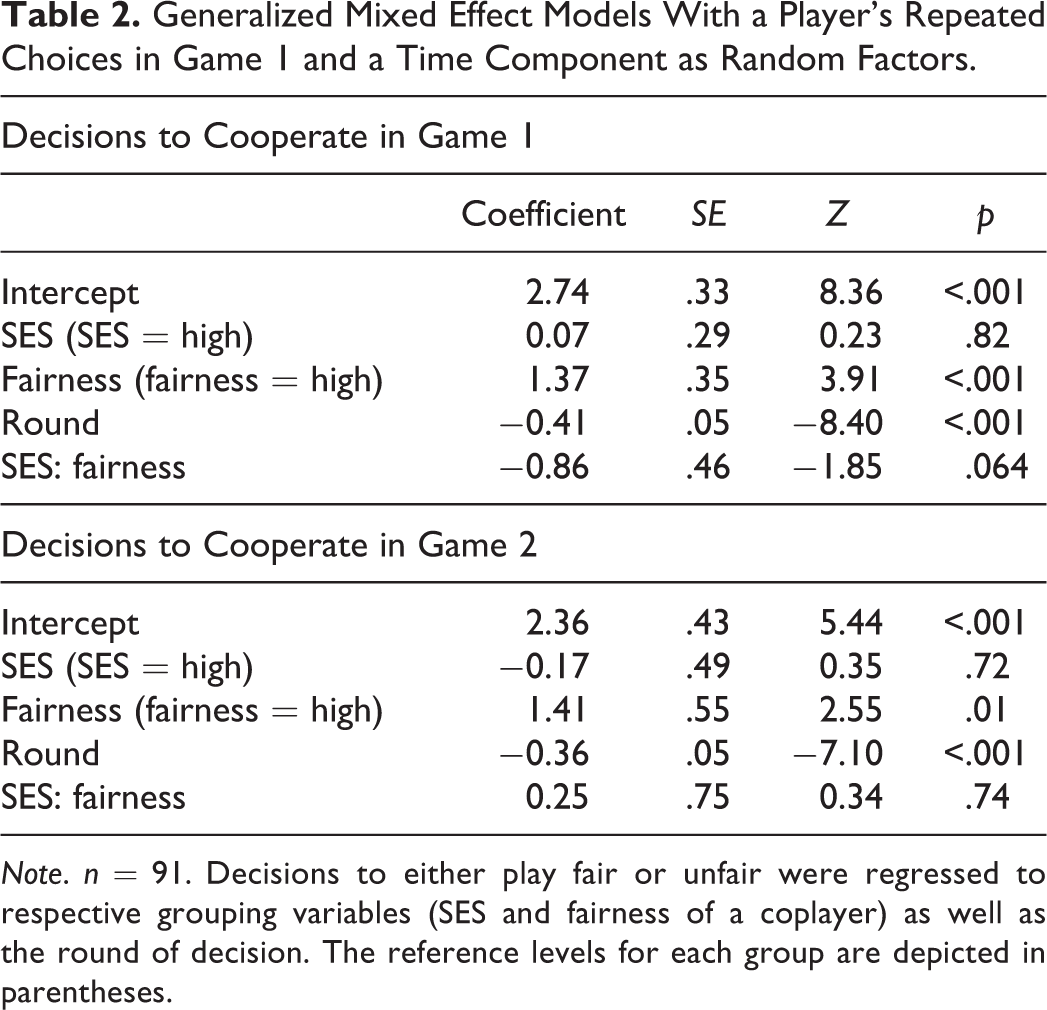
Note. n = 91. Decisions to either play fair or unfair were regressed to respective grouping variables (SES and fairness of a coplayer) as well as the round of decision. The reference levels for each group are depicted in parentheses.

Proportions of participants’ choices to cooperate throughout eight rounds of playing in Game 1 (a) and Game 2 (b). The different lines code for the four possible types of coplayers: sim-SES/low fairness (blue line), sim-SES/high fairness (green line), higher SES/low fairness (red line), and higher SES/high-fairness coplayers (black line). High fairness of the coplayer leads to more cooperation. However, there is a general pattern of decreasing cooperation over time.
Discussion
This study investigated the effects of SES difference and fairness on cortisol responses and cooperative choices in a male student sample. Our results show that playing a cooperation game itself (i.e., acting as first mover in a sequential cooperation game) triggered significantly increased cortisol concentrations in all players. Moreover, we found distinctive saliva cortisol responses under different socioenvironmental game conditions. Participants paired with a player of similar demographic status showed no significant decrease in saliva cortisol concentrations over time. In contrast, participants paired with coplayers with higher SES attributes showed significantly increased ratios of recovery to the basal state.
Hence, besides traditional frameworks such as the trier social stress test (Dickerson & Kemeny, 2004; Kirschbaum, Pirke, & Hellhammer, 1993), experiments involving sequential cooperation as modeled in this study can induce significant HPA activity. The observed patterns of increased stress reactivity for all participants throughout the testing correspond to those of the subjects who failed to cooperate in a Lego-house building task (Moya-Albiol et al., 2013). Possibly, the behavior programming limiting the maximal probability of reciprocated fair choices by a coplayer to only 45% was regarded as being too low to count as truly fair or cooperative by participants in general and might have triggered feelings of failed overall cooperation in response to our testing. Also, humans might typically behave more variably than programmed for coplayers in this study. More research is needed to verify this and also to clarify in how far other interactions involving sequential decision processes can constitute a psychosocial stressor that can sufficiently elevate stress response.
The time of recovery to the basal state is an important index for the severity of a psychosocial stressor (Sapolsky, 2005) and for the long-term effects of repeated exposure to such stressors. The rates of poststressor cortisol concentrations significantly increased in all participants. With respect to cortisol decrease during relaxation, however, we observed significant drops only for the group of subjects interacting with higher SES coplayers—irrespective of their behavior programming. Faster cortisol recovery can be related to increased sensitization processes in the brain reward system of participants during the testing (Oei et al., 2014). In this respect, an interaction with higher SES subjects may have represented a stress buffering option in itself. Higher status involves better access to limited resources and heightens the value of actual and future cooperation for lower-in-status conspecifics (Campbell, Marsden, & Hurlbert, 1986; Von Rueden, Gurven, & Kaplan, 2008). Thus, from an evolutionary point of view, it could be physiologically rewarding to interact with a high-in-status conspecific even if cooperative interactions occurred rather rarely (e.g., 20% of fair choices). This is because spatial and temporal proximity and the amount of past positive interactions might facilitate receiving privileges. Studies on other primate species put this in an evolutionary comparative perspective and hint toward an evolutionary stable strategy. Henazi and Barrett (1999), for example, suggested that grooming represents an interchange currency in nonhuman primates, which is used to increase the level of tolerance between dominant and subordinate individuals. This in turn either reduces aggression levels or increases access to limiting resources for the subordinates.
Our results, however, show that—at least in a low-authoritarian social setting like ours—a rewarding fast relaxation is achieved through various behavioral strategies that need to be investigated in follow-up studies. We could not detect clear profiles of stress recovery that are rooted in combined effects of status and behavior of coplayers. Therefore, our findings do not support findings by Dawans, Fischbacher, Kirschbaum, Fehr, and Heinrichs (2012) who suggest that the initiation of potentially successful cooperation is a strategy to cope with acute psychosocial stress. The authors showed that, in response to the trier social stress test (Kirschbaum et al., 1993), participants significantly increased their cooperative effort toward strangers in a cascade of social dilemma situations including fairness (von Dawans et al., 2012). One nonexclusive interpretation for this lack of agreement can be that a diversification of coping strategies could be rooted in the adoption of behavior observed in high-status individuals. Since in numerous primate species high-in-status conspecifics receive more attention than others (Koski et al., 2015), and learning from others is biased toward learning from those with higher status (Atkinson et al., 2012), an adoption of behavior might be an effective mechanism to provide an evolutionary advantage through culture transmission and coordinated group activity (Barkow, 2014).
Lahti and Weinstein (2005) propose that much of the observed variation in commitment to a group can be explained by “a rule of stability-dependent cooperation, where the adaptive level of individual commitment varies inversely with the stability of the social group” and that “moral norms serve a group stability insurance function” (p. 47). It reflects the trade-off between competition with fellow group members and the stability of the group relative to other groups. Power disparity, however, is a mediating variable insofar, as it might overwhelm the effects of group stability. In that case, the possibility to modify the level of service to the group is very limited for most individuals. Thus, status differences might play an important role in stress-induced cooperation, a concept that should be addressed in future studies.
In those subjects facing sim-SES coplayers, we observed neither an alternation in cooperative choices during testing nor noticeable physiological stress recovery. Here, recovery after exposure to our setting was markedly delayed and did not reach the basal state even 40 min after testing. This suggests exposure to a significant psychosocial stressor. We therefore conclude that similar subjective SES during an interaction indeed made a difference in our experiment. While in subjects interacting with higher SES coplayers, established coping mechanisms were automatically applied during the testing, subjects facing sim-SES coplayers recovered much slower. This resulted in cortisol responses similar to those in losers of contest experiments in humans or even rodents (Aguilar, Jiménez, & Alvero-Cruz, 2013; Jiménez, Aguilar, & Alvero-Cruz, 2012; Wallner & Dittami, 2003) and might support predictions derived from the biosocial model of status and dominance (Mazur, 1985). Future studies should therefore explicitly focus on patterns of cooperation involving status differentials together with different coping mechanism models. Also, as more recent studies show (Mehta & Josephs, 2010; Pfattheicher, Landhäuser, & Keller, 2014), investigating the interplay between status, cortisol and testosterone could help to clarify possible findings and provide deeper insights into status-induced differences in male behavior and stress physiology.
In evolutionary psychology, cooperation and social hierarchies are two important concepts to understand current and past behavior. Often these two are investigated separately, yet in any natural interaction among humans, one can barely be observed without the other. Our study experimentally manipulated both of them and documented male students’ cortisol response.
In conclusion, we find the SES of interacting partners to have modulatory effects on saliva cortisol secretion rates in participants while not impinging on the degree of cooperative effort toward (un)cooperatively playing coplayers. Thus, status differentials made a difference in this study. Physiologically, acting as first mover with coplayers of similar status seemed to be a big challenge that markedly prolonged stress recovery. In contrast, participants interacting with coplayers holding a higher SES were better able to cope with this type of stressor. Although more work has to be done to better understand the modulating effects of SES and behavior on HPA functioning, this study highlights potential mechanisms of this relationship and opens directions for future research.
Footnotes
Appendix
Generalized Mixed Effect Models With a Player’s Repeated Choices in Game 1 and a Time Component as Random Factors.

| Decisions to Coperate in Game 1 | ||||
|---|---|---|---|---|
| Coefficient | SE | Z | p | |
| Intercept | 2.830 | .302 | 9.347 | <.001 |
| SES (SES = high) | −0.136 | .21 | −0.647 | .517 |
| Fairness (fairness = high) | 0.76 | .214 | 3.546 | <.001 |
| Round | −0.401 | .046 | −8.735 | <.001 |
| Medication (medication = yes) | −0.365 | .357 | −1.022 | .306 |
| Decisions to cooperate in Game 2 | ||||
| Intercept | 1.978 | .361 | 5.485 | <.001 |
| SES (SES = high) | 0.20 | .332 | 0.062 | .951 |
| Fairness (fairness = high) | 1.570 | .343 | 4.578 | <.001 |
| Round | −0.313 | .046 | −6.80 | <.001 |
| Medication (medication = yes) | −0.979 | .569 | −1.721 | .085 |
Note. n = 91. Decisions to either play fair or unfair were regressed to respective grouping variables (SES and Fairness of a Coplayer) including medication as well as the round of decision. The reference levels for each group is depicted in parentheses. The results show that we did not observe clear patterns in behavior in response to our experimental setting that can be interpreted in line with recent findings on impacts on behavior through asthma medication.
Acknowledgments
We would like to thank Pia Stephan and Markus Koppensteiner for their helpful comments as well as Markus Riedler for his assistance in preparing the figures. S.W. was supported by the Emerging Field “Comparative Human Life History: A Multilevel Approach,” University of Vienna, Faculty of Life Sciences.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
