Abstract
Individual differences in morningness/eveningness are relatively stable over time and, in part, genetically based. The night-owl pattern is more prevalent in men than in women, particularly after puberty and before women reach menopause. It has been suggested that eveningness evolved relatively recently in human evolutionary history and that this trait may be advantageous to individuals pursuing short-term mating strategies. Consistent with this hypothesis, eveningness is associated with extraversion, novelty-seeking, and in males, with a higher number of sexual partners. In this study, I investigated whether eveningness is associated with short-term relationship orientation, higher risk-taking, and higher testosterone or cortisol. Both female and male night-owls were more likely to be single than in long-term relationships than early morning individuals. Eveningness was associated with higher risk-taking in women but not in men; this association was not testosterone-dependent but mediated by cortisol. Female night-owls had average cortisol profiles and risk-taking tendencies more similar to those of males than to those of early-morning females. Taken together, these findings provide some support to the hypothesis that eveningness is associated with psychological and behavioral traits that are instrumental in short-term mating strategies, with the evidence being stronger for women than for men.
Introduction
There is substantial individual variation in people's propensities to go to sleep early or late in the evening and wake up early or late in the morning. At one extreme of the distribution, early-morning (EM) people typically go to bed early in the evening and get up early in the morning; they usually exhibit optimal cognitive performance early in the morning, and do not function effectively in the evening. In contrast, night-owls (NO) go to sleep late in the evening and wake up late in the morning; they perform better in the evening or at night and have trouble functioning in the morning (e.g., Horne, Brass, and Pettitt, 1980; Horne and Ostberg, 1977; see Adan et al., 2012, and Tankova, Adan, and Buela-Casal, 1994, for reviews). Although many people easily self-identify as having one sleep pattern or the other, others fall in between the EM and the NO extremes and do not appear to have a clear sleep pattern or preference. Individual differences in morningness/eveningness (also known as chronotype) are generally stable over time and show a moderate degree of heritability (h2 = 0.45; Hur, 2007; Hur, Bouchard, and Lykken, 1998; Klei et al., 2005). Environmental factors (e.g., geographic and seasonal variation, work schedule), however, also make important contributions to variation in sleep pattern (e.g., Leonhard and Randler, 2009; Natale, Adan, & Fabbri, 2009).
One of the most consistently reported findings concerning individual variation in sleep pattern involves sex differences. Men sleep, on average, for fewer hours than women at any age, and at around the time of puberty (children tend to have the early-morning sleep pattern) men also begin to show a greater tendency to be night-owls than women (Adan and Natale, 2002; Tonetti, Fabbri, and Natale, 2008; Tsai and Li, 2004). This sex difference in sleep pattern persists into adulthood, but becomes weaker or disappears after women reach menopause (Randler, 2007; Randler and Bausback, 2010). The sex difference in sleep pattern is accompanied by differences in some physiological variables. For example, melatonin levels in women peak earlier at night than in men (Gibertini, Graham, and Cook, 1999; Kerkhof and Van Dogen, 1996). Furthermore, in both women and men, skin conductance peaks earlier in the day in EM than in NO types (Wilson, 1990), whereas cortisol levels peak about 1 hour earlier in EM than in NO types, and the amplitude of the cortisol rhythm is smaller and more flattened in NO than in EM types (Bailey and Heitkemper, 2001; see also Rosenberg, Maximov, Reske, Grinberg, and Shah, 2014, for differences in brain function).
The sex difference in sleep pattern (but not in sleep amount) and its relation to puberty and menopause led Piffer (2010) to propose a hypothetical scenario for the evolution of the night-owl pattern. Specifically, he speculated that since humans evolved from diurnal primate ancestors, morningness was probably the ancestral evolutionary condition for our species (see also Kanazawa and Perina, 2009), whereas eveningness may represent a relatively new trait that evolved due to its mating-associated fitness benefits. Increased safety from predation and other ecological dangers during early human evolution may have increased opportunities to engage in social and mating activities in the late evening hours, when adults are less burdened by work or child-rearing. Individuals - both male and female - with a new genetic predisposition for eveningness presumably benefited more from these new social and mating opportunities, particularly if these individuals were not permanently pair-bonded, or if they were pair-bonded but engaged in extra-pair mating. In this scenario, NO males had a higher reproductive success than EM males, thus a genetic predisposition for eveningness gradually became more and more prevalent in the male population (EM males, however, continued to reproduce successfully, e.g., in the context of long-term pair-bonds with faithful EM females). Finally, since eveningness increased the fitness of men more than the fitness of women, eveningness became a sexually dimorphic trait, being more prevalent in men than in women. In a series of studies inspired by this hypothetical evolutionary scenario, Piffer and collaborators reported that in men, eveningness (but not average sleep duration) was positively associated with number of sexual partners (Gunawardane, Piffer, and Custance, 2011; Piffer, 2010; Randler et al., 2012a).
Regardless of whether or not individual differences in sleep patterns are associated with variation in mating success in contemporary human societies, a question may arise as to whether eveningness is associated with behavioral or psychological traits that favor short-term mating strategies. This is because different traits promoting short-term mating strategies are likely to evolve in clusters rather than independently from one another. One obvious candidate is personality. Extraversion is a personality dimension that is generally linked with higher short-term mating success (Nettle, 2005; Randler et al., 2012a), and several studies have reported that NO people are generally more extraverted than EM people (Díaz-Morales, 2007; Matthews, 1988; Randler et al., 2012a; but see Tonetti, Fabbri, and Natale, 2009). Moreover, personality studies have reported that eveningness is also associated with higher impulsivity and novelty-seeking (e.g., Caci, Robert, and Boyer, 2004; Digdon and Howell, 2008), as well as with narcissism, psychopathy, and Machiavellism (Jonason, Jones, and Lyons, 2013), which is also consistent with the hypothesized functional significance of this trait for short-term mating strategies.
Besides personality, two additional traits that favor short-term mating, and should therefore be associated with eveningness, are risk-taking and short-term relationship-orientation (i.e. the propensity to be single or to engage in short-term romantic relationships) (e.g., Baker and Maner, 2008; Kelly and Dunbar, 2001). There is no previous research on chronotype and relationship orientation, and so far only one study has investigated the relationship between sleep pattern and risk-taking. In this study, Killgore (2007) examined different measures of risk-taking (Brief Sensation-Seeking Scale, Evaluation of Risks Scale, and the Balloon Analog Risk Task) in a sample of 54 young adults (29 male, 25 female) who were subdivided by a median split into an Evening (n = 28) and a Morning (n = 26) group based on self-reported sleep patterns and preferences. He reported that Evening types scored higher than Morning types on the BSS scale and in Total Risk-Taking Propensity (a cumulative Index obtained by combining all the items in the ER Scale), but there were no differences in the behavioral Balloon test. Sex differences in risk-taking and their possible interaction with chronotype were not considered in this study. Moreover, it was not clear how men and women were distributed into the Evening and Morning groups after the median split of the subject population. This is problematic because men generally score higher than in women in all measures of risk-taking (e.g., Byrnes, Miller, and Schafer, 1999; Figner and Weber, 2011). Therefore, if men were over-represented in the Evening group and under-represented in the Morning group, then differences in risk-taking in relation to sleep pattern were confounded by sex.
The sex variable was not taken into consideration also by the three studies by Piffer and colleagues (Gunawardane et al., 2011; Piffer, 2010; Randler et al., 2012a), which were all conducted with male subjects. Sex, however, is an important aspect of the sexual selection hypothesis of eveningness evolution. Clearly, both men and women engage in short-term mating and the night-owl sleep pattern occurs in both sexes, though it is more prevalent in men than in women. Therefore, when investigating whether eveningness is associated with traits that favor short-term mating strategies, both men and women should be considered.
In this study, I tested the hypothesis that eveningness is associated with greater propensities for risk-taking and for short-term relationship orientation. Specifically, I tested the prediction that NO females should be more similar to NO males (or to males in general) than to EM females in risk-taking and short-term relationship orientation. Since sexually dimorphic hormonal variables (testosterone and cortisol levels are higher in men than in women; Maestripieri, Baran, Sapienza, and Zingales, 2010) may be potential physiological mechanisms underlying the link between a predisposition for eveningness and traits such as risk-taking and short-term relationship orientation, I tested the prediction that NO females should be more similar to NO males (or to males in general) than to EM females in their salivary concentrations of testosterone and cortisol. Furthermore, if testosterone or cortisol differed significantly in relation to sleep patterns, I tested its possible role as a physiological mechanism mediating the relationship between social and physiological variables. Finally, I compared the number of sexual partners between NO and EM males to assess whether I could replicate the previously reported association between male eveningness and higher number of sexual partners.
Materials and Methods
Participants
Study participants were 501 master's students (348 males, 153 females) in the Booth Business School at the University of Chicago. Participation in the study was one of the requirements for a course and therefore it was mandatory for the entire 2008 cohort of MBA students. Students, however, were paid $20 or more for their participation. The use of human subjects was approved by the IRB and all students were asked for informed written consent for their participation in the study.
Procedures
For all study participants (n = 501), information on the following variables was collected: age, sex, relationship status, financial risk aversion, self-reported risk-taking tendencies, and salivary cortisol and testosterone concentrations measured before and after the experimental risk aversion test. For a subset of study participants (n = 201; 110 males, 91 females), additional information was collected about self-reported baseline stress level, sleep pattern, sleep amount, and number of previous sexual partners (only for male participants) (see below for details).
Assessment of financial risk aversion and salivary Cortisol and testosterone
Study participants were asked to take a 90 minute computerized test in which they played a game that assessed their tendencies for financial risk aversion, along with other economic games (Sapienza, Zingales, and Maestripieri, 2009). All students were tested on two days. Tests were conducted in the afternoon, between 1:30 and 5:00 p.m. Taking the test was a stressful experience (see Maestripieri et al., 2010). To assess changes in cortisol and testosterone in relation to the test, two saliva samples were collected from each study participant, one at the beginning of the test session and the other 2 hours later, after the participants completed their tests. Hormone changes from the pre-test to the post-test were calculated using the regressor variable method, namely as the unstandardized residuals of a regression analysis with pre-test hormone as the predictor and post-test hormone as the dependent variable. This method ensures that the measure of hormone change is independent of the pre-test scores (see Maestripieri et al., 2010). Approximately 2–3 ml of saliva was collected by passive drool into plastic vials. Although salivary concentrations of testosterone and cortisol are lower in the afternoon than in the morning, afternoon hormone levels are more stable and therefore better suited for psychoneuroendocrine studies. All samples were immediately placed into dry ice and transported to Dr. Robert Chatterton's Endocrinology Laboratory at Northwestern University, where they were frozen at −80 °C until assayed. Before assay, samples were thawed and centrifuged to reduce viscosity. Salivary concentrations of testosterone and cortisol were measured by radioimmunoassay, using antisera prepared within the laboratory (Chatterton, Vogelsong, Lu, and Hudgens, 1997). Cross-reactivity of the cortisol antiserum with cortisone was nonexistent; cross-reactivities of the testosterone antiserum with dihydrotestosterone and androstenedione were 13 and 0.2%; those for androsterone, etiocholanone, estradiol, and dehydroepiandrosterone were all less than 0.1%. The lower sensitivity of the assays was 0.07 ng/ml for cortisol and 7.5 pg/ml for testosterone. Intra-assay coefficients of variation (CVs) were all < 10% and inter-assay CVs were < 15%, consistent with published data for other assays from this laboratory (Chatterton et al., 1997). All samples were assayed in duplicate, and the average of duplicates was used in all analyses.
Self-report measures
Self-reported risk-taking. Risk-taking tendencies were assessed by asking participants to answer the question “Are you generally fully prepared to take risks?” on a 0 to 10 scale, with 0 = unwilling/unprepared and 10 = willing/fully prepared.
Sleep pattern. Study participants were asked the question “Are you a night-owl or an early morning person?” If participants could not identify themselves as being one pattern or the other, they were given the option of answering “I don't know.” This single-item measure for the assessment of chronotype represents a simplification of the 5-item “reduced Morningness-Eveningness Questionnaire” (rMEQ; Adan and Almirall, 1991), which in turn is a validated simplification of the 19-item Morningness-Eveningness Questionnaire (MEQ) developed by Horne and Ostberg (1976). In the rMEQ, individuals are asked four questions about the time at which they prefer to get up or go to sleep and the time at which they perform best or feel tired. Item 5 in the rMEQ requires that the individuals self-identify as “definitely a morning type,” “rather more a morning than an evening type,” “rather more an evening than a morning type,” or “definitely an evening type.” Individuals are then classified into three groups (Morning Type, Neither Type, Evening Type) or two groups (Evening Type, Morning Type). Investigations of the psychometric properties of the rMEQ have shown that this scale has high inter-item correlations and high coefficient alpha (e.g., Caci Deschaux, Adan, and Natale, 2009; Chelminski, Petros, Plaud, and Ferraro, 2000). This means that people's self-assessment of chronotype (item 5) accurately reflects their self-assessment of circadian behavior and preferences (items 1–4). The single-item assessment of chronotype used in this study requires a self-identification of the subjects as a morning or an evening-type, similar to item 5 of the rMEQ. The psychometric properties of the rMEQ suggest that this 5-item scale and the single-item measure should result in a similar classification of chronotypes. The single-item measure, however, provides a more conservative assessment of chronotype than the rMEQ (particularly when this scale is used to assign study participants to two groups with a median split) because participants who answer “I don't know” are excluded from data analyses.
Sleep amount. Participants were asked to estimate how many hours per night they usually slept.
Relationship status. Subjects were asked whether they were single or in a long-term relationship. The long-term relationship category was defined as being with a romantic partner for at least 6 months up to the time of testing, including being married. By definition, the single category included both single individuals and individuals who were in a short-term (less than 6 months) relationship at the time of testing.
Number of previous sexual partners. Male participants were asked to indicate the number of different individuals with whom they had sexual intercourse in their life.
Information was also collected on background variables such as the participants' age, height, weight, general health, smoking and drug use, handedness, sexual orientation, and use of contraceptives. Women were asked whether they used hormonal contraceptives at the time of testing, the first and the last day of their most recent menstrual flow period, the average length of their menstrual cycles, and whether cycles were regular or irregular. None of the above background variables had any significant effects on the variables of interest in this study.
Results
Sex differences in variables and correlation between risk-taking measures
With one exception (cortisol responses to stress), there were significant sex differences in all the variables considered in this study. Males (n = 326) were significantly older than females (n = 147), t(471) = 5.45, p < 0.0001, but the average difference in age was only about 1 year (males M ± SD = 28.72 ± 2.39; females = 27.41 ± 2.48). Males (n = 110) reported sleeping a lower number of hours than females (n = 91) (males M ± SD = 7.0 ± 0.85; females = 7.41 ± 0.88), t(199) = −3.33, p < 0.001. Males (n = 324) scored significantly higher than females (n = 145) in the self-reported measure of risk-taking, t(467) = 4.1, p < 0.0001 (see Figure 1a), and significantly lower than females in the experimental measure of financial risk-aversion (males: M ± SD = 3.61 ± 4.02; females = 5.57 ± 4.58), t(472) = −4.65, p < 0.0001. Self-reported risk-taking was significantly negatively correlated with financial risk aversion, r(444) = −0.25, p < 0.0001 (a similar significant correlation was also found when male and female subjects were analyzed separately). Given this correlation, and to avoid redundancy, only the self-reported measure of risk-taking was used in subsequent analyses. Males (n = 319) were significantly more likely to be single than in a relationship when compared to females (n = 144), χ2 (1) = 4.92, p = 0.02 (see Figure 2a).

(a) Risk-taking scores (mean ± SEM) in males and females; (b) Risk-taking scores (mean ± SEM) in NO and EM males and females
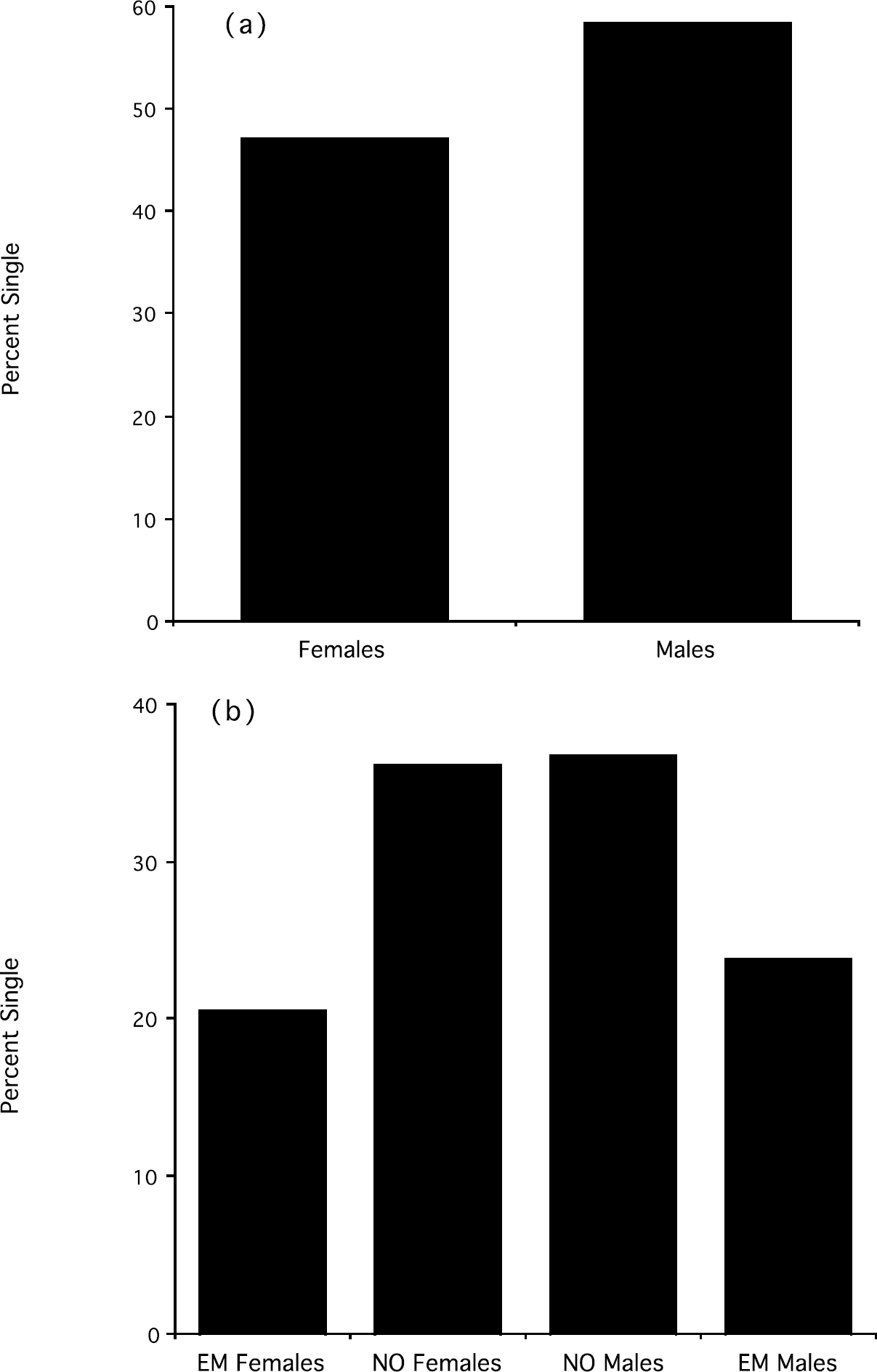
(a) Percentage of single individuals in males and females; (b) Percentage of single individuals in NO and EM males and females
Males (n = 349) had significantly higher average cortisol levels than females (n = 152), t(499) = 3.62, p < 0.0001 (see Figure 3a). Males (n = 349) also had significantly higher average testosterone levels than females (n = 152), t(499) = 17.49, p < 0.00001 (see Figure 4a). There was no significant sex difference in the cortisol response to stress (males: M ± SD = 0.01 ± 4.18; females = −0.007 ± 3.29; t(499) = 0.06, ns), but males had significantly higher T residuals (males: M ± SD = 6.21 ± 1.93; females = −14.12 ± 2.34; t(499) = 6.14, p < 0.0001), indicating that their testosterone dropped in response to stress significantly more than in females.

(a) Average cortisol concentrations (mean ± SEM) in males and females; (b) Average cortisol concentrations (mean ± SEM) in NO and EM males and females
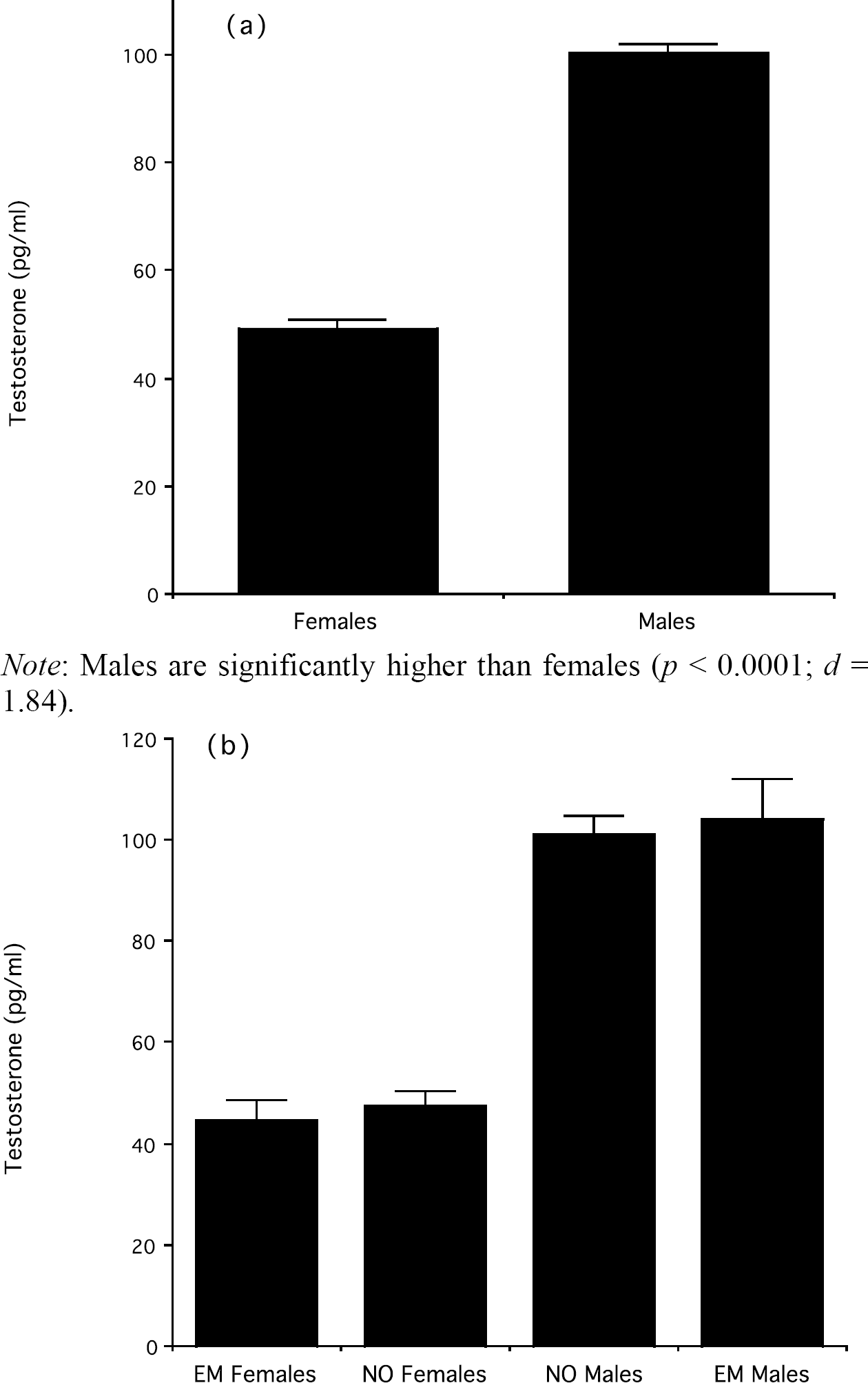
(a) Average testosterone concentrations (mean ± SEM) in males and females; (b) Average testosterone concentrations (mean ± SEM) in NO and EM males and females
Sleep pattern, risk-taking, and relationship status
Of the 201 study participants who were asked the question “are you a night-owl or an early morning person?” 170 of them self-identified as being either a NO or an EM type (68 NO males, 21 EM males, 48 NO females, and 33 EM females), whereas 31 other people answered “I do not know.” Overall, night-owls were approximately twice as frequent as early-morning types (116 NOs vs 54 EMs). The distribution of the two sleep patterns was significantly different in males and females, χ2 (1) = 5.75, p = 0.01: Among males, NOs were more than three times as common as EMs (68 vs 21) whereas among females, NOs were approximately 1.5 times as common as the EMs (48 vs 33) (see Figure 5). There was no significant effect of sleep pattern on the amount of sleep (ANOVA; F(1, 166) = 0.10; ns) or a significant interaction between sleep pattern, sex, and sleep amount, F(1, 166) = 2.70, ns (NO males: M ± SD = 7.08 ± 0.89; EM males = 6.86 ± 0.73; NO females = 7.27 ± 0.90; EM females = 7.53 ± 0.86).
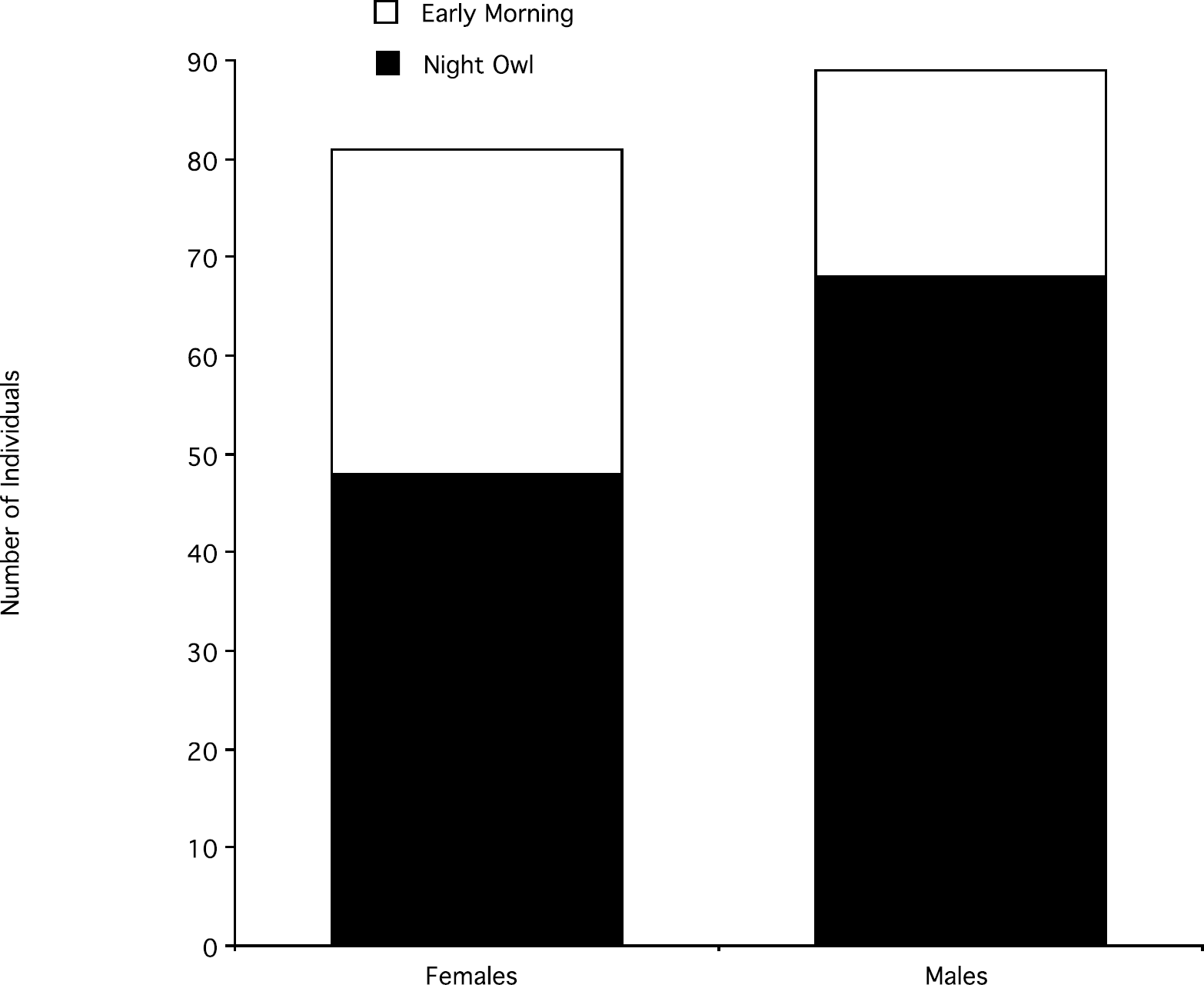
Relative number of males and females who reported being EM or NO
An ANOVA examining the effects of sleep pattern and sex on risk-taking revealed no main effect of sleep pattern, F(1, 166) = 0.45, ns, but a significant interaction between sleep pattern and sex, F(1, 166) = 5.83, p = 0.01. Bonferroni-Dunn post-hoc tests revealed that EM females scored significantly lower on risk-taking than individuals in the other three groups (all p values < 0.05). NO males, EM males, and NO females were not significantly different from each other (see Figure 1b).
The percentage of individuals who were single was similar in NO males and NO females and significantly higher than the percentage of individuals who were single in EM males and EM females, χ2 (1) = 3.42, p < 0.05 (see Figure 2b). Therefore, Night-Owls, regardless of sex, were more likely to be single than Early-Morning individuals. Although NO males reported having had, on average, almost twice as many sexual partners as EM males (M ± SD = 11.00 ± 14.99 vs 6.36 ± 5.29), the difference between groups failed to reach statistical significance, t(54) = 1.30, ns.
Sleep pattern, hormones, and risk-taking
ANOVA revealed no significant main effect of sleep pattern on average cortisol levels, F(1, 166) = 2.40, ns, but a significant interaction between sleep pattern and sex, F(1, 166) = 3.96, p < 0.05. Similar to the effects of sleep pattern and sex on risk-taking, post-hoc tests revealed that EM females had significantly lower average cortisol levels than individuals in the other three groups (all p values < 0.05; see Figure 3b), whereas NO males, EM males, and NO females were not significantly different from each other. Unlike cortisol, there was no significant main effect of sleep pattern on average testosterone, F(1, 166) = 0.01, ns, and no significant interaction between sleep pattern and sex, F(1, 166) = 0.4, ns (see Figure 4b).
Measures of cortisol or testosterone change in response to stress were not affected by sleep patterns. Thus, there was no significant main effect of sleep pattern on the cortisol stress response, F(1, 166) = 0.49, ns, and no significant interaction between sleep pattern and sex, F(1, 166) = 1.19, ns (NO males: M ± SD = −0.45 ± 3.21; EM males = −0.26 ± 4.47; NO females = 0.10 ± 2.85; EM females = −0.89 ± 2.29). Similarly, there was no significant main effect of sleep pattern on testosterone residuals, F(1, 166) = 0.63, ns, and no significant interaction between sleep pattern and sex, F(1, 166) = 1.86, ns; NO males: M ± SD = 7.16 ± 28.25; EM males = 18.34 ± 39.87; NO females = −14.91 ± 22.18; EM females = −17.83 ± 26.02).
Variation in average cortisol or in risk-taking was not correlated with variation in sleep amount regardless of sex (average cortisol and sleep amount: r(168) = 0.05, ns; risk-taking and sleep amount: r(168) = 0.09, ns). Furthermore, there was no significant difference in sleep amount in relation to relationship status, t(168) = −0.15, ns. Thus, discrete variation in sleep patterns was a more powerful predictor of variation in cortisol, risk-taking, and relationship status than continuous variation in sleep amount.
The hypothesis that cortisol mediated the influence of sleep pattern and sex on risk-taking was tested with a four-step multiple regression approach (Baron and Kenny, 1986). In Model 1, sleep pattern and sex were a significant predictor of risk-taking (β = −0.17, t = −2.31, p = 0.02). Sleep pattern and sex were also a significant predictor of average cortisol (Model 2: β = −0.24, t = −3.22, p = 0.002). Finally, cortisol significantly predicted risk-taking (Model 3: β = 0.21, t = 2.78, p = 0.006; see Figure 6). In a multiple regression model (Model 4), the effect of sleep pattern and sex on risk-taking was no longer significant after controlling for cortisol (β = −0.13, t = −1.72, ns), whereas the effect of cortisol on risk-taking remained significant after controlling for sleep pattern and sex (β = 0.18, t = 2.30, p = 0.02). These results support the cortisol mediation hypothesis. Finally, there were no significant differences in risk-taking or average cortisol between individuals who were single or in a relationship (cortisol M ± SD: single = 7.42 ± 2.87; relationship = 7.68 ± 3.41; t(168) = −0.25, ns; risk-taking, single = 6.57 ± 1.81; relationship = 6.35 ± 2.02; t(168) = 0.68, ns), suggesting that relationship status played no significant role in accounting for the association between sleep pattern, cortisol, and risk-taking.

Correlation between average cortisol levels (across sex and sleep pattern groups) and risk-taking scores
Discussion
The hypothesis that eveningness should be associated with higher risk-taking was supported by results obtained in women but not in men. Night-owl females were indistinguishable from NO and EM males in risk-taking, and all three groups scored higher than the EM females. The prediction that NO males should score higher in risk-taking than EM males was not supported. In a previous study, however, eveningness was found to be associated with higher risk-taking in both men and women (Killgore, 2007; but see the Introduction for a criticism of this study).
The hypothesis that eveningness should be associated with greater propensity for short-term relationship orientation was supported in both men and women, as NO males and females were more likely than EM males and females to be single than in a relationship. NO males reported, on average, a higher number of sexual partners than EM males (11.00 vs 6.36); this difference was not statistically significant but it might have reached significance with a larger sample size. Although demonstrating that NO individuals have a higher number of sexual partners is not a crucial test of the adaptiveness of eveningness, if the finding by Piffer and collaborators could be replicated in both men and women, it would contribute to a better understanding of the functional significance of individual differences in chronotype and their possible evolutionary origin.
In this study, the association between eveningness and risk-taking in women was testosterone-independent, as NO females were similar to EM females in testosterone levels. Average cortisol, instead, was a significant predictor of risk-taking across sexes, and a regression-based mediation analysis indicated that cortisol likely mediated the effect of sleep pattern on risk-taking in women. Men had significantly higher average cortisol than women but, as with risk-taking, NO females were indistinguishable from NO and EM males in average cortisol levels, and all three groups had higher cortisol than EM females.
The finding that cortisol was more strongly associated with chronotype and risk-taking than testosterone was somewhat surprising, since Randler et al. (2012b) presented evidence that eveningness is associated with higher testosterone in men. However, cortisol and testosterone were positively correlated in this subject population and variation in average testosterone levels accounted for variation in financial risk aversion among women (Maestripieri et al., 2010; Sapienza et al., 2009).
Unlike variation in average cortisol levels, which may potentially be the result of variation in sleep patterns and/or reflect stable individual differences in arousability or reactivity to the environment (see also Adam, Hawkley, Kudielka, and Cacioppo, 2006; Decker and Aggott, 2013, for evidence that high-functioning adults have high cortisol levels, as elevated cortisol may provide an “adaptive boost” to meet daily demands), variation in cortisol responses to stress was not associated with sleep patterns or with risk-taking. Similarly, variation in average sleep amount (i.e., self-reported number of hours of sleep per night) was not a significant predictor of average cortisol or of risk-taking. Therefore, it appears that variation in average cortisol and in risk-taking is specifically associated with individual differences in morningness/eveningness, especially in women, rather than with sleep amount or stress reactivity.
The origin of individual differences in sleep patterns is probably, in part, genetic and, in part, environmental; regardless of relative contributions of genes and environment to this trait, different sleep patterns could result in different cortisol profiles, and cortisol, in turn, could affect variation in people's propensities to take risks. It is also possible, however, that correlations between sleep pattern, cortisol, and risk-taking are the product of independent associations with a fourth variable not considered in this study, such as personality (for a discussion of personality and risk-taking, see Zuckerman and Kuhlman, 2000). Establishing causal relationships between variables is difficult without an experimental manipulation of these variables, which was beyond the scope of this study. The possibility that sleep patterns, cortisol profiles, and risk-taking propensities (and possibly also relationship orientation) are all linked together through personality is still consistent with the hypothesis that this is a cluster of traits functionally linked to short-term mating strategies in both sexes.
The lack of significant differences between NO and EM males in cortisol or risk-taking may be the result of the low number of EM males in the subject population (n = 21) and does not necessarily argue against the hypothesis that eveningness and risk-taking are associated with short-term mating strategies. Although eveningness may be an important component of short-term mating strategies, there may be other aspects of short-term mating strategies that are unrelated to sleep pattern, especially in men.
Although a great deal of research has already been conducted on human morningness/eveningness, with a few exceptions (e.g., Jonason et al., 2013; Kanazawa and Perina, 2009; Piffer, 2010), most of this research has not been conducted from an evolutionary perspective and therefore has not investigated the possible adaptive significance of variation in this trait. The relationship between chronotype, sex, relationship orientation, and risk-taking highlighted in this study, however, suggests that further tests of adaptive hypotheses are warranted.
The present study has some methodological limitations, including the way chronotype was assessed, the lack of personality measures, the small sample size, particularly for the EM males, and the use of a subject population consisting of MBA students with particular demographic and chronotype characteristics. Future studies are needed to replicate these results and assess the extent to which they can be generalized to other populations. The hypotheses and the data presented here, however, can stimulate and guide future research on the evolutionary significance of chronotype and contribute to the elucidation of this poorly understood aspect of human biology and behavior.
Footnotes
Acknowledgements
This study was funded by intramural grants from the University of Chicago. I thank Paola Sapienza and Luigi Zingales for their contributions to the research project from which these data were obtained. I also thank Daniel Traficonte and Claire Wilson for assistance in preparing this manuscript and Davide Piffer and Samuele Zilioli for helpful comments and discussion.
