Abstract
Introduction
Heart failure (HF) patients are prone to postoperative hemodynamic instability after cardiac surgery, possibly due to upregulated renin-angiotensin-aldosterone system (RAAS) activity. We evaluated perioperative RAAS activity in control and HF patients and its impact on acute kidney injury (AKI) incidence.
Materials and Methods
Pre-specified analysis of a prospective observational cohort study (VASOR). Plasma renin, angiotensinogen and aldosterone were measured perioperatively in 18 control and 18 HF patients.
Results
RAAS blocker use was identical in both groups. HF patients displayed a diminished preoperative renal function. AKI occurred in 1 control and 8 HF patients. Preoperatively, HF patients exhibited 5-fold higher renin and aldosterone levels, identical aldosterone/renin ratios, and modestly lower angiotensinogen levels than controls. During surgery, renin rose 10-fold higher in HF patients. Aldosterone increased proportionally, but was blunted when normalized for renin (aldosterone/renin ratio). This was not due to angiotensinogen depletion. Postoperatively, angiotensinogen increased 50% in both groups, allowing aldosterone to increase, despite a modest postoperative renin decline. Preoperative renin levels and the surgery-induced renin rise predicted postoperative AKI.
Conclusions
HF patients exhibit dysregulated RAAS responses to surgery may reflect impaired angiotensin I-angiotensin II conversion. Elevated renin signals RAAS dysregulation, identifying patients who might benefit from angiotensin II infusion.
The Netherlands Trial Register (NTR5647); http://www.trialregister.nl/trialreg/admin/rctview.asp?TC=5647.
Introduction
Vasoplegia, a significant complication after cardiac surgery, is characterized by hypotension resulting from profound, vasoconstrictor-resistant vasodilation in the presence of normal cardiac function, which, if not treated vigorously, leads to an increase in morbidity and mortality.1,2 While the underlying mechanisms are not fully understood, the renin-angiotensin-aldosterone system (RAAS) has emerged as an important player. Several studies proposed disturbed RAAS activity in septic or cardiac surgery patients, while exogenous angiotensin II (Ang II) administration turned out to be beneficial.3–5
RAAS stimulation, required to maintain vascular tone at the time of cardiopulmonary bypass (CPB) during cardiac surgery, involves the release of renin. 6 This may occur acutely, depending on the amount of renin stored in the kidney. Chronic RAAS stimulation, resulting from the compensatory, neurohumoral activation seen in heart failure (HF), involves the additional upregulation of renin-synthesizing cells, further increasing renin levels. Blood pressure is inversely related to renin release, implying that high renin levels are an indication of hypotension. 7 In agreement with this concept, elevated plasma renin levels during CPB and in sepsis patients predict postoperative hemodynamic instability and the incidence of acute kidney injury (AKI), as well as mortality.4,58–11
A renin increase normally results in parallel rises in Ang I and II. Ang II subsequently suppresses renin release via a negative feedback loop, to prevent excessive RAAS upregulation. Angiotensin-converting enzyme inhibitors (ACEi) or angiotensin II receptor blockers (ARB) block this feedback loop, thus upregulating renin.12,13 Depending on the extent and duration of RAAS blockade, renin may rise several 100-fold.13,14 This is particularly true in HF patients 15 . These patients often already display increased RAAS activity inherent to their disease. ACEi and ARBs will increase renin even further, 16 while concomitant application of diuretics (resulting in sodium loss, a well-known stimulator of renin release) will also stimulate RAAS activity. As a consequence, HF patients typically have very high renin levels. This may eventually lead to angiotensinogen depletion, so that a further renin rise (as seen during CPB), may no longer increase Ang I in parallel. A further complication is that patients undergoing cardiac surgery on CPB display ACE downregulation, either as a consequence of endothelial dysfunction or because the pro-inflammatory factors that are released during CPB suppress ACE.4,17 This would result in a diminished Ang II upregulation for a given rise in Ang I. Finally, the Ang II responsiveness of these patients might be diminished due to the Ang II type 1 (AT1) receptor downregulation that is expected to occur when the body is continuously exposed to high Ang II levels. 18
To better understand the association of the RAAS with postoperative hemodynamic instability and thus, with vasoplegia and the incidence of AKI, the present study compared RAAS activity perioperatively in patients with and without HF undergoing cardiac surgery. Such knowledge may help to design new treatment strategies resulting in better patient outcomes, including Ang II infusion.
Methods
Study design
VASOR is a prospective, observational study, conducted at a University Medical Center with the aim to examine the vascular responsiveness in patients with HF undergoing cardiac surgery. The protocol has been published. 19 Patients with and without HF undergoing cardiac surgery on CPB were compared. The study is registered at The Netherlands Trial Register (NTR5647) and was performed in line with the principles of the Declaration of Helsinki. The institutional medical ethical committee approved the protocol (22 May 2015, P14.298). The study was reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplement). 20
Study population
Patients scheduled for cardiac surgery with the use of CPB between 2016-2018 were screened for eligibility. To form comparable study groups, patients were selected according to the expected CPB and aortic cross-clamp duration. No individual matching based on baseline characteristics was performed. Patients were assigned to either the HF or the control (non-HF) group. HF was defined according to the European Society of Cardiology guidelines. 16 Inclusion criteria for the HF group were: 1) diagnosed with HF and 2) left ventricular ejection fraction (LVEF) ≤ 35%. Patients that were included in the control group were 1) not diagnosed with HF and 2) had a LVEF > 50%. Exclusion criteria were: 1) age <18 years; 2) incapacitated adults; 3) emergency operation; 4) preoperative need of moderate or high dosages of intravenous inotropic support (dobutamine or dopamine), vasopressin and/or mechanical support; 5) aortic valve insufficiency > grade 1; 6) the use of nitrates, α-adrenergic blockers or phosphodiesterase type 5 inhibitors; 7) endocarditis; and 8) acute heart failure. All patients gave written informed consent for study participation ( Figure S1 ). Diuretics, ACEi and ARB were discontinued the morning of the procedure and were reinitiated postoperatively when hemodynamically appropriate
Vasoplegia was defined as the continuous need of vasopressors (norepinephrine ≥ 0.2 µg/kg/min for at least 12 consecutive hours or terlipressin) in combination with a cardiac index ≥ 2,2 L/min/m2 for at least 12 consecutive hours, starting within the first 3 days postoperatively. Intraoperatively, a mean arterial pressure (MAP) ≤ 65 mmHg was corrected using norepinephrine or phenylephrine. Norepinephrine was started postoperatively if the MAP was ≤ 65 mmHg and the cardiac index was normal (after adequate resuscitation with fluids, if necessary), aiming for a MAP > 65 mmHg. When a norepinephrine dosage of more than 1 μg·kg–1·min–1 was required, terlipressin was started, initiated with a 0.8 mg bolus, followed by a continuous infusion of terlipressin.
Study outcomes
This pre-specified analysis of the VASOR study aims to determine the preoperative levels and the perioperative response of RAAS components in patients with and without HF. We measured renin and angiotensinogen as indicators of angiotensin-generating capacity, and aldosterone as an indicator of Ang II-responsiveness. The primary end-point was the change in plasma renin between the preoperative and the after cessation of CPB levels (Δ renin). Secondary endpoints included: 1) change in plasma angiotensinogen levels after cessation of CPB (Δ angiotensinogen); 2) change in plasma aldosterone levels after cessation of CPB (Δ aldosterone); 3) correlation between preoperative plasma renin levels and the amount and duration of norepinephrine use postoperatively; 4) correlation of CPB duration with Δ renin; 5) incidence of AKI according to the KDIGO guidelines within 72 h from cardiac surgery (using the serum creatinine criterion as well as the urine output criterion) and its correlation with the preoperative renin levels. 21 According to the KDIGO guidelines, AKI is defined as the presence of any of the following: a) increase in serum creatinine by 0.3 mg/dL or more (26.5 μmol/L or more) within 48 h, b) increase in serum creatinine to 1.5 times or more than the baseline of the prior 7 days, c) urine volume less than 0.5 mL/kg/h for at least 6 h. The latest preoperative serum creatinine levels were considered as baseline reference.
Blood samples
EDTA plasma samples were obtained through an arterial line at 4 different time points: 1. before induction [T1], 2. after induction [T2], 3. 15 min after cessation of CPB [T3], and 4. on day one postoperative (24 h after surgery) [T4]. The samples were centrifuged (1550 g, 10 min, 4°C) and plasma was stored at −80°C until analysis was performed.
Measurement of RAAS components
Plasma renin was measured with a commercial immunoradiometric kit (Beckman Coulter, Immunotech, Prague, Czech Republic). The detection limit of this assay is 2 pg/mL, with an inter-assay variability of 7%. Angiotensinogen in plasma was measured as the maximum quantity of Ang I that was generated during incubation with excess recombinant renin. This assay has a detection limit of 0.5 pmol/mL, and an inter-assay variability of 10%. Aldosterone was measured by solid-phase radioimmunoassay (Beckman Coulter, Immunotech, Prague, Czech Republic), with a detection limit of 14 pg/mL, and an inter-assay variability of 8%. The aldosterone/renin ratio was calculated as an indicator of renin-dependent synthesis of aldosterone.
Statistical analysis
This study is an exploratory, pre-specified analysis of the VASOR study. 19 The sample size calculation was therefore based on the primary outcome of VASOR, i.e.,change in systemic vascular resistance after phenylephrine administration at baseline. Baseline patient characteristics were described using summary statistics. Continuous variables were reported as mean ± SD or as median with interquartile range. Differences between groups (HF vs. control) were compared using an unpaired Student's t-test, or a Median test with Yates's correction for continuity when necessary. The Wilcoxon signed ranked test was used to compare differences within groups. Categorical data were reported as percentages and differences were compared based on Chi-square or Fisher's exact test, when appropriate. Missing values for two patients at timepoint 4 were imputed using linear regression. These samples were not collected because the patients did not have an arterial line in situ. Subgroup analyses were performed to test differences of angiotensinogen levels between patients receiving transfusions and steroids perioperatively. Similarly, aldosterone levels were also tested among patients receiving intraoperative cardioplegia or not.
Spearman correlation analysis was used to determine the correlation between the preoperative renin concentration and postoperative use of noradrenaline (maximum and total dosage in μg/kg/min and duration in minutes). A similar analysis was used to assess the correlation between the CPB duration and Δ renin. The area under the receiver-operating characteristics curve (AUC) and respective 95% confidence intervals (CI) were calculated to measure the discrimination of patients at risk for developing postoperative AKI based on the preoperative renin concentration and Δ renin. To assess the magnitude of the association of the preoperative renin levels with the postoperative AKI a binary logistic regression was performed. The VASOR study was powered for vascular responsiveness; no separate a priori power calculation was performed for AKI or vasopressor dependence. 19 Accordingly, analyses of AKI and norepinephrine usage are exploratory and hypothesis-generating.
The significance level was set at p < 0.05. Statistical analysis was performed using SPSS (version 25.0, IBM, Armonk, NY) and R software (Version 4.1.0, 2022.0201, R Foundation for Statistical Computing, Vienna, Austria).
Results
In total 36 patients (18 HF, 18 control) were enrolled in the VASOR study. At T4, 2 plasma samples were not available for analysis. Therefore, a total of 142 plasma samples were analyzed. Baseline characteristics can be found in Table 1. Overall, HF patients exhibited a trend towards worse preoperative renal function and had a lower systolic blood pressure and higher white blood cell count compared to control patients (p = 0.090, 0.034 and 0.016, respectively). Although the use of diuretics was more common in the HF group (89% vs 28%, p < 0.001), no difference was found in the use of ACEi or ARB between the 2 groups (61% vs 50% in HF and controls respectively, p = 0.738). There was a trend for higher usage of mineralocorticoid receptor antagonists in the HF group (44% versus 11%, p = 0.06). The patients’ intraoperative characteristics were similar, while HF patients required more inotropic and vasopressor support during the postoperative course (Table 2). AKI occurred in 8 HF patients, compared to 1 control patient (44% vs 6%, p = 0.018, Table S1 ). Postoperative vasoplegia occurred in 1 control patient.
Baseline characteristics

Data are presented as mean ± SD, median (IQR), or %.
Intraoperative characteristics

Data are presented as mean ± SD, median (IQR), or %.
RAAS components
HF patients were characterized by significantly higher preoperative (T1) renin (24.8 [9.6-49.8] vs. 4.2 [2.2–9.8] pg/mL, p = 0.005) and aldosterone (130 [69.5–268] vs. 68.1 [29.8–95.4] pg/mL, p = 0.005) levels than control patients (Figure 1). Their preoperative angiotensinogen levels tended to be lower, although this did not reach statistical significance (1202 [870–1641] vs. 1350 [1150–1605] pmol/mL, p = 0.134).
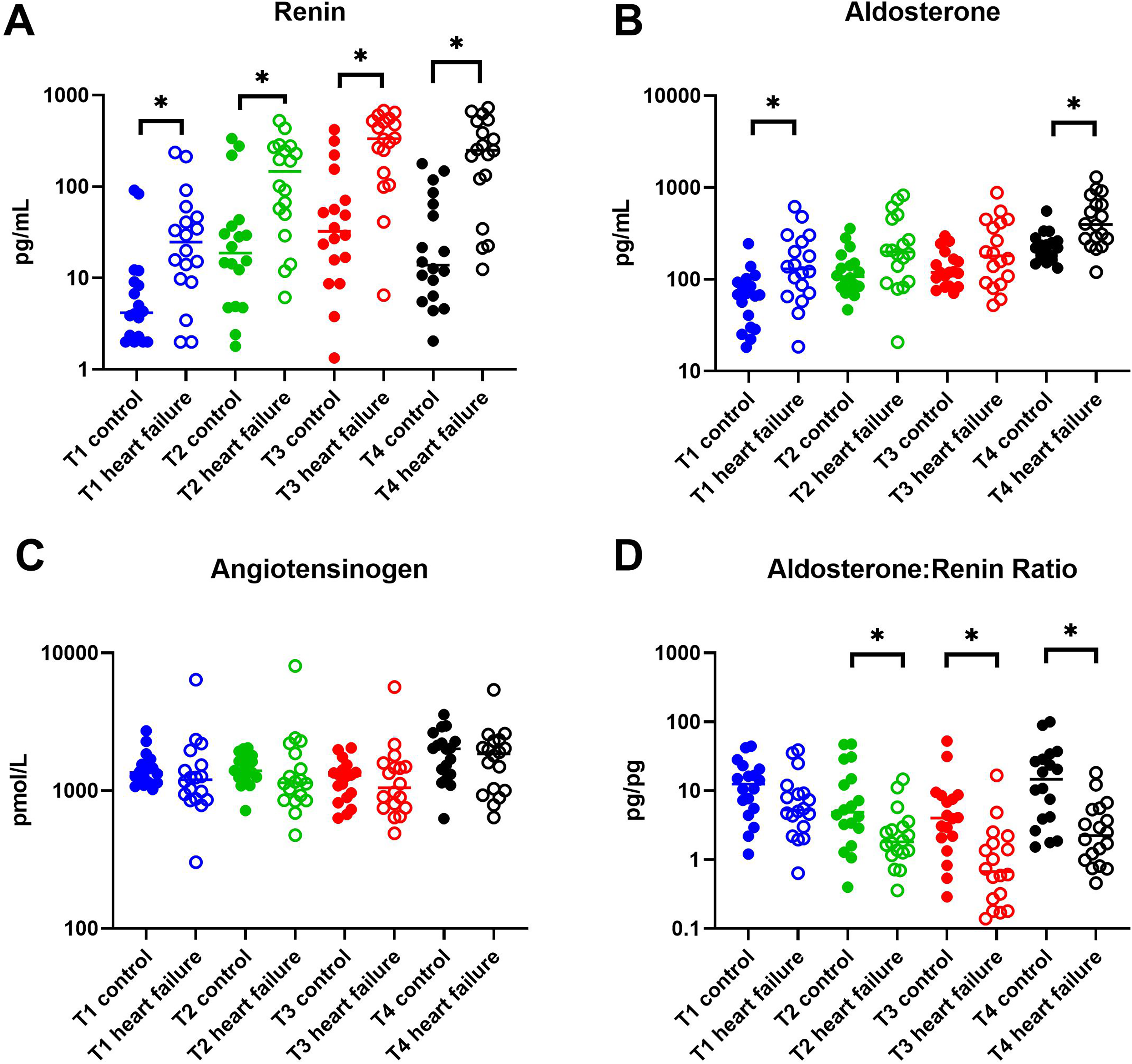
Perioperative concentrations of A, renin; B, aldosterone; C, angiotensinogen; D, aldosterone/renin ratio. T1: before induction of anesthesia, T2: after induction of anesthesia, T3: after cessation of cardiopulmonary bypass, T4: postoperative day 1. Squares: control patients; triangles: heart failure patients. Individual data points and the median are depicted. *P < 0.05.
Renin levels rose approximately 10-fold during cardiac surgery in both groups, reaching the highest levels after CPB cessation (Figure 1A; p < 0.001). Interestingly, the net increase after CPB (T3) in the HF group was 10-fold larger than in the control group (Δ renin: 296.9 [119.5–458.97] vs. 27.9 [11.8–81.7] pg/mL, p < 0.001). Aldosterone increased in parallel with renin (Figure 1B), but when normalized for renin (aldosterone/renin ratio) (Figure 1D), the aldosterone rises in the HF group were greatly diminished (p = 0.008, p < 0.001 and p = 0.008 at after anesthesia induction [T2], after CPB cessation [T3] and at postoperative day 1 [T4] respectively). This suggests that the body's responsiveness to rises in renin (and thus angiotensin) is blunted. Importantly, at baseline, the aldosterone/renin ratio was similar in control and HF patients (p = 0.182), implying that the diminished aldosterone response in HF patients occurred only at the time of surgery and the day thereafter. Postoperatively, renin decreased, although its levels on day 1 postoperative were still greatly elevated versus the preoperative situation (p < 0.001 in both groups). Despite the drop in renin, aldosterone levels doubled postoperatively vs. after CPB (T3) in both groups (p < 0.001 and p = 0.011 for HF and controls respectively).
Angiotensinogen dropped by 10-20% during the operation (Figure 1C), potentially reflecting the renin rises, but returned to levels above baseline at postoperative day 1 (T4) (p = 0.022 and p = 0.018 vs. preoperative [T1], in HF and controls respectively). At all timepoints, although not significant, the angiotensinogen levels in HF patients were below those in control patients. Angiotensinogen correlated negatively with renin, and this correlation was identical in control and HF patients at all time points (Figure 2). CPB duration did not correlate with Δ renin (rs(34) = −0.048, p = 0.779).

Association between log renin and log angiotensinogen. T1: before induction of anesthesia, T2: after induction of anesthesia, T3: after cessation of cardiopulmonary bypass, T4: postoperative day 1.
Results were unaltered when examined within RAAS blocker users (data not shown).
Hematocrit, blood transfusion, steroid use, and cardioplegia
The above-mentioned changes in RAAS components might relate to changes in blood volume and composition due to obligatory hemodilution, blood transfusion, steroid application and potassium exposure (due to potassium-rich blood cardioplegia). The preoperative hematocrit and its change (≈25%) during the operation were identical in HF and control patients (Figure 3A). This implies that the drop in angiotensinogen during the operation may largely be attributed to hemodilution. Twenty-four patients (16 HF, 8 control) received blood transfusion (during the intraoperative period and/or the postoperative day 1), and their changes in angiotensinogen were identical to those in the 12 patients that did not receive a blood transfusion (Figure 3B). This indicates that the postoperative rise in angiotensinogen cannot be attributed to blood transfusion. Twelve patients (6 HF, 6 control) received steroids (hydrocortisone n = 5, dexamethasone n = 5, prednisolone n = 1, prednisolone + hydrocortisone n = 1). Their postoperative rises in angiotensinogen were not different from those who did not receive steroids (Figure 3C), implying that this rise could also not be attributed to exogenous steroid administration. It was also unrelated to changes in C-reactive protein (Table S1). Finally, 32 patients received cardioplegia intraoperatively, and their aldosterone rises, if anything, were more modest (p = NS) compared those in patients who did not receive cardioplegia (Figure 3D). Thus, the rise in aldosterone cannot be attributed to exogenous potassium administration.

Perioperative hematocrit and hormone levels in patients by condition and treatment type. A, perioperative hematocrit values in control and heart failure patients; B, perioperative concentrations of angiotensinogen in patients receiving or not receiving a blood transfusion; C, perioperative concentrations of angiotensinogen in patients receiving or not receiving steroids; D, perioperative concentrations of aldosterone in patients receiving or not receiving cardioplegia. Individual data points and the median are depicted. T1: before induction of anesthesia, T2: after induction of anesthesia, T3: after cessation of cardiopulmonary bypass, T4: postoperative day 1. Squares: control patients; triangles: heart failure patients. Individual data points and the median are depicted. *P < 0.05.
Prediction of AKI based on renin levels
The preoperative renin levels showed fair predictive performance for postoperative AKI only in HF patients (Figure 4A, AUC 0.681, 95% CI 0.414–0.948). The optimal cutoff value for the preoperative renin levels in HF patients was ≥ 15 pg/mL, with a sensitivity of 75% and a specificity of 50%. According to the binary logistic regression analysis, one unit increase in the preoperative renin levels is associated with a 3.5% increase in the odds of developing postoperative AKI (beta coefficient 0.035, 95% CI 1.004–1.067).

ROC curves for A, the preoperative and B, the Δ renin on AKI prediction. Δ renin = change in renin concentration between before the operation and after the cessation of cardiopulmonary bypass. AKI = acute kidney injury; ROC = receiver operating characteristic.
The Δ renin exhibited an unsatisfactory predictive performance with an AUC of 0.513 (95% CI 0.229-0.796) (Figure 4B). The optimal cutoff value for Δ renin was 261 pg/mL, with also a sensitivity of 75% and a specificity of 50%.
Postoperative hemodynamic instability
A Spearman's correlation analysis was performed to assess the relationship between the preoperative renin levels and the postoperative hemodynamic instability as assessed by the use of noradrenaline. Preoperative renin levels correlated positively with both the postoperative maximum and total dosage of noradrenaline (rs(34) = 0.447, p = 0.006 and rs (34) = 0.433, p = 0.008, respectively), as well as the duration of noradrenaline administration (rs(34) = 0.361, p = 0.031).
Discussion
This study provides detailed insight into RAAS changes during cardiac surgery on CPB, highlighting the distinct response in HF patients. First, it provides evidence that in HF patients, despite a marked increase in renin levels intraoperatively, there is no corresponding increase in RAAS activity. Secondly, it confirms that high preoperative renin levels are predictive of postoperative AKI in HF patients and the use of noradrenaline required to manage postoperative hemodynamic instability. These findings are suggestive for RAAS disruption beyond renin, and would explain why Ang II application might be useful in such patients.
High renin levels are indicative of significant renin upregulation, implying that the number of renin-synthesizing cells in the kidney has increased considerably. 22 Consequently, any renin stimulus will cause even larger increase in renin concentrations. Both ACEi/ARBs and diuretics contribute to higher preoperative renin levels, via loss of Ang II–mediated feedback and sodium/volume depletion, respectively. When given together, this likely results in synergy.23,24 In our cohort ACEi/ARB use was similar in HF and controls (61% vs 50%) while also the perioperative RAAS response patterns were similar in ACEi/ARB users and non-users. Thus, ACEi/ARB exposure is unlikely to account for the between-group differences we observed. Yet, diuretics were more often used by HF patients, implying that these drugs may, on top of the inherent effects of HF, have contributed to the larger renin rise.
The renin levels in our HF patients at baseline were, on average, five times higher than those in controls. The preoperative renin levels in this study were associated with the incidence of postoperative AKI in HF patients and the use of noradrenaline. When placed in a broader context, our findings are consistent with observations from ICU cohorts in which renin levels have been associated with mortality.10,25 We believe that this is an indication of a disrupted RAAS activity in HF patients that ultimately leads to a dampened RAAS response to surgery. The net result of this is insufficient vasoconstriction which causes hypotension necessitating increased concentrations of noradrenaline to maintain hemodynamic stability. The combination of hypotension, hypoperfusion and the use of high noradrenaline dosages can have detrimental effects on kidney perfusion. This further supports the interpretation of renin as a possible biomarker of circulatory stress across surgical and critical-illness settings. Of note, our study included HF patients with reduced LVEF (≤ 35%), thereby representing an advanced HF cohort. Disease progression in HF is known to be associated with greater neurohormonal activation, leading to higher renin release and possibly to impaired renal perfusion.26,27
This sequalae may therefore amplify vulnerability to postoperative AKI.
Renin correlated negatively with angiotensinogen, and this correlation was comparable at all timepoints in both control and HF patients (Figure 2). This suggests that the renin-angiotensinogen interaction (i.e., the generation of Ang I) was identical under all conditions. Angiotensinogen levels were lower in HF and decreased modestly in both groups during surgery, potentially reflecting the rise in renin at that time point. The relative renin rise in both groups was identical (10-fold), but in absolute terms the renin rise in the HF patients was 10-fold higher. Here, another important player appeared to be blood dilution, indicated by a hematocrit drop of around 25% in both groups of patients. This implies that in reality the renin increase was even higher, while the surgery-induced drop in angiotensinogen might be attributed largely to blood dilution. Other factors that should be considered here are capillary leak and fluid extravasation, keeping in mind that angiotensinogen is likely distributed equally over the entire extracellular fluid. 28 From these data it can be concluded that the angiotensinogen levels were sufficiently high (i.e., in the Km range), even during surgery, to allow a parallel rise in Ang I synthesis corresponding to the rise in renin.
Given the fact that Ang II acts as a stimulator of aldosterone release, high renin levels should immediately result in a parallel rise in Ang II and aldosterone levels (Figure 5). Indeed, this was the case at baseline, where the aldosterone/renin ratio was identical in controls and HF patients. However, during surgery, the aldosterone/renin ratio dropped 4-5-fold in the HF patients versus the control group. Consequently, the aldosterone levels became identical in both groups after CPB (T3), despite >10-fold higher renin levels. This difference persisted on the day after the operation (T4). Hence, renin-mediated RAAS activation was ineffective in HF patients during surgery. Since this did not concern Ang I generation, it should reflect Ang I-II conversion by ACE or diminished AT1 receptor responsiveness. Given the diminished Ang II/I ratios reported by others in critically ill patients, the former explanation seems the most likely.4–5,29 An important site of Ang I-II conversion are the capillary endothelial cells of the lungs. 30 Since the lungs are excluded from the circulation during CPB, a drop in the Ang II/I ratio might be expected. 6 It could even underlie, at least partially, the rise in renin. However, since CPB occurred in both patient groups, there must be an additional player in the HF patients. The aldosterone/renin ratio remained suppressed on the day after surgery, when the pulmonary circulation had been restored. A likely candidate is endothelial dysfunction, related to the surgery-induced inflammatory response. 31 Potentially, such dysfunction occurs in both patient groups, but is worse in HF patients. Nevertheless, alternative mechanisms such as adrenal hyporesponsiveness or AT1 receptor downregulation/desensitization in chronic HF could also contribute. Importantly, considering that similar RAAS dysregulation has been observed in patients with sepsis, COVID-19, and vasoplegia, i.e., critically ill patient populations with high morbidity and vasopressor needs, the presence of the same pattern in HF patients is notable. Routine cardiac surgery patients typically recover well with low postoperative risk, but this resemblance to severely ill cohorts suggests that HF patients represent a more fragile population and require closer perioperative monitoring and more thorough preoperative planning.
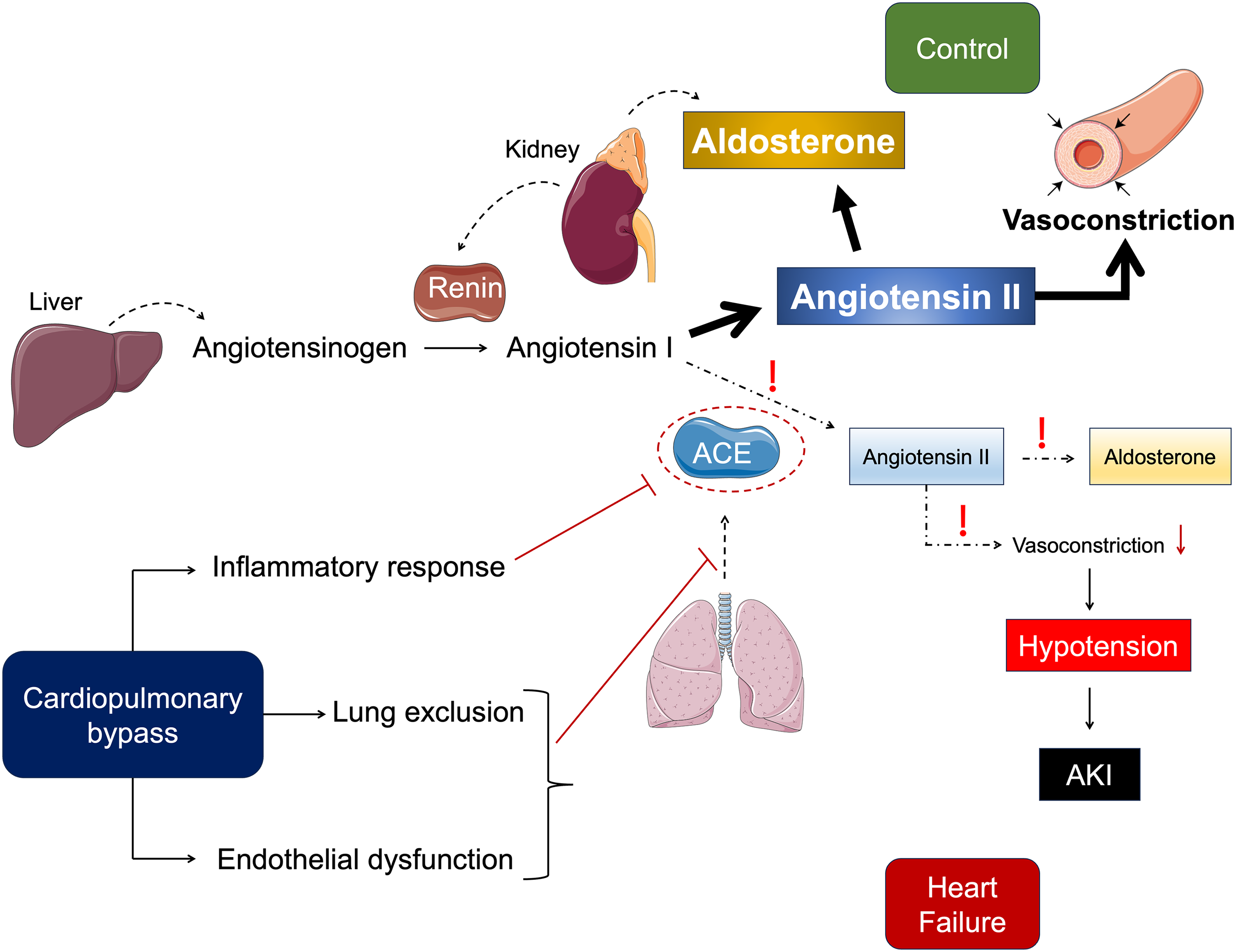
Renin-angiotensin-aldosterone system dysregulation in heart failure patients undergoing cardiac surgery on cardiopulmonary bypass. ACE: angiotensin-converting enzyme, AKI: acute kidney injury.
The inflammatory response to surgery might also underlie the significant upregulation of angiotensinogen on the day after the operation. For instance, cortisol is a well-known inducer of angiotensinogen. 32 Other vital players in the systemic inflammatory response to cardiac surgery, like interleukin-6 and tumor necrosis factor alpha, have also been reported to stimulate hepatic angiotensinogen expression and production. 33 The use of CPB is known to cause a systemic release of both interleukin-6 and tumor necrosis factor alpha.34,35 Our data exclude exogenous steroids as significant contributors, given that the angiotensinogen rises were similar with and without steroid application. Although renin started to decline modestly post-operation, when combined with an almost 50% rise in angiotensinogen, it is still possible to induce a rise in angiotensin generation at T4. Consequently, post-operation a further rise in aldosterone was observed, despite a drop in renin. Our data do not support that this rise related to the use of potassium-containing cardioplegia, although the number of patients that did not receive cardioplegia was small.
Our study has several limitations. First, it is a small, single-center study in which patients with and without HF were compared. Therefore, the findings of this study might not be applicable in other patient populations and should, thus, be confirmed in larger patient cohorts. Second, this is a pre-specified analysis of another study which was powered for vascular responsiveness and not for clinical outcomes. With our sample size, only relatively large differences in RAAS activation and clinical outcomes would be expected to reach statistical significance; small or moderate associations cannot be reliably detected. Therefore, analyses of AKI and vasopressor dependence are exploratory and underpowered and require confirmation in larger, prospectively powered cohorts. In addition, we included patients who were treated with RAAS blockers. Although RAAS blockers were withheld on the morning of surgery, ACEi/ARB-induced renin elevation is known to persist for 24–48 h after discontinuation, and therefore a proportion of the preoperative and early intraoperative renin levels might still partly reflect residual RAAS blockade. 36 Importantly, while residual ACEi/ARB effects may partly contribute to elevated baseline renin, they cannot explain the disproportionate intraoperative renin rise or the markedly blunted aldosterone response observed in HF patients, suggesting that HF-related RAAS dysregulation remained the dominant driver of these findings. Another limitation is that we did not measure Ang I, Ang II or ACE. Angiotensin measurement would have required sampling conditions that prevent angiotensin generation and metabolism ex vivo, while for ACE we should have obtained samples without the ACE inhibitor EDTA. Nevertheless, our data are in full agreement with normal RAAS physiology. In this patient cohort, only a few patients developed vasoplegia making it difficult to study any relevant associations. However, the correlation between preoperative renin levels and postoperative use of noradrenaline suggests that elevated renin may also predict the occurrence of postoperative vasoplegia, given its association with hemodynamic instability and thus, the need for postoperative noradrenaline.
Perspectives
HF patients, despite exhibiting very significant renin increases intraoperatively, do not appear to exhibit a proportional increase in Ang II, as reflected by the aldosterone levels, indicating a disruption of RAAS activation. This disruption is probably due to endothelial damage and/or the absence of pulmonary ACE at the time of CPB. Patients not appropriately responding to a rise in renin could potentially benefit from targeted Ang II therapy to treat postoperative vasoplegia. Preoperative renin levels may help to select such patients, like they also predict postoperative AKI and noradrenaline use. This hypothesis should be further examined in future prospective studies. Given the huge difference in renin levels between different patient populations (control and HF patients with or without ACEi or ARB treatment), it is likely that the cut-off point will differ. To determine cut-off points, larger studies are required.
Supplemental Material
sj-pdf-1-jra-10.1177_14703203251414310 - Supplemental material for Disturbed RAAS response to cardiac surgery in heart failure patients
Supplemental material, sj-pdf-1-jra-10.1177_14703203251414310 for Disturbed RAAS response to cardiac surgery in heart failure patients by Olga Papazisi, Marieke E. van Vessem, Saskia L.M.A. Beeres, Remco R. Berendsen, Bart J.A. Mertens, Ingrid M. Garrelds, Sesmu M. Arbous, Robert J.M. Klautz, Meindert Palmen and A.H. Jan Danser in Journal of the Renin-Angiotensin-Aldosterone System
Supplemental Material
sj-docx-2-jra-10.1177_14703203251414310 - Supplemental material for Disturbed RAAS response to cardiac surgery in heart failure patients
Supplemental material, sj-docx-2-jra-10.1177_14703203251414310 for Disturbed RAAS response to cardiac surgery in heart failure patients by Olga Papazisi, Marieke E. van Vessem, Saskia L.M.A. Beeres, Remco R. Berendsen, Bart J.A. Mertens, Ingrid M. Garrelds, Sesmu M. Arbous, Robert J.M. Klautz, Meindert Palmen and A.H. Jan Danser in Journal of the Renin-Angiotensin-Aldosterone System
Footnotes
Abbreviations
Acknowledgements
We would like to thank Madhavi Harhangi for her assistance in preparing the figures.
Ethical considerations
The study is registered at The Netherlands Trial Register (NTR5647) and was performed in line with the principles of the Declaration of Helsinki. The institutional medical ethical committee approved the protocol (22 May 2015, P14.298).
Consent to participate
All patients gave written informed consent for study participation as part of the VASOR study.
Consent for publication
Not applicable.
Author contributions
Conception and study design: OP, MV, SB, MP, AD; data acquisition, analysis and/or interpretation: all authors; statistical analysis: OP, MV, BM; manuscript preparation: OP, MP, AD; manuscript editing: all authors; manuscript revision and final approval: all authors. OP, MP and AD had full access to all the data and take responsibility for the integrity of the data and accuracy of data analysis.
Funding
This work was supported by Stichting Zabawas. Stichting Zabawas had no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Declaration of conflicting interest
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data supporting the results presented in this paper can be made available upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
