Abstract
Introduction
The cardiorenal syndrome is a term loosely defined as the development of renal dysfunction in the presence of heart failure. The interaction between the kidney and the heart is complex, as disorder of one organ can perpetuate the dysfunction of the other. 1 Renal insufficiency continues to be the most powerful predictor of mortality in patients hospitalized with acute heart failure.2,3 It is estimated that for every 0.5 mg/dL increase in serum creatinine, there is a 15% increase in all-cause mortality. 3 Many factors have been linked to the development of acute renal insufficiency in patients with heart failure, including the drugs used in acute treatment. In patients with moderate to severe congestion and systemic vasoconstriction, the addition of nesiritide, nitroglycerin, or nitroprusside, is often used to provide rapid improvement in hemodynamics. Reduction of preload and afterload with any of these intravenous (IV) vasodilators rapidly decreases ventricular filling pressures, which may enhance cardiac output and increase renal perfusion.4–6 Nesiritide, recombinant b-type natriuretic peptide, may also antagonize the renin–angiotensin–aldosterone system (RAAS) and enhance sodium excretion. 7 While this additional physiologic effect of nesiritide has the potential to influence renal function in patients with heart failure, studies evaluating nesiritide’s effects on glomerular filtration rate (GFR), diuresis, and renal function have not demonstrated a clear beneficial effect.8,9 No studies have compared the effect of the various IV vasodilators on renal function in patients with acute heart failure already being diuresed. In comparative studies of nesiritide to nitroglycerin and placebo, nesiritide improved hemodynamics without an indication for a greater risk of worsening renal function.10,11
Previous studies have used absolute or relative changes in serum creatinine to define acute kidney injury (AKI), but there has been no consensus in the definitions used. The Acute Dialysis Quality Initiative (ADQI) Group recently proposed a standard method of evaluating AKI, known as the risk, injury, failure, loss, and end stage (RIFLE) classification criteria, which has been validated in critically ill patients. 12 The criteria detect and classify AKI into categories that correlate with the clinical outcomes such as intensive care unit (ICU) length of stay, hospital length of stay, and in-hospital mortality. The development of AKI defined by the RIFLE criteria has been associated with worse outcomes in patients with heart failure; however, experience with this classification system in this population remains limited. 13
The purpose of this study was to compare the safety and efficacy of the IV vasodilators, nitroglycerin and nesiritide, in the treatment of acute heart failure as assessed by indices of renal function and injury, including the RIFLE criteria. This data support consideration of renal effects as an important factor in clinical decision making when selecting an IV vasodilator for the management of acute heart failure.
Methods
A retrospective cohort study was conducted in patients with acute heart failure on aggressive diuretic regimens requiring the use of nitroglycerin or nesiritide for the relief of congestion. The study was approved by the Los Angeles County + University of Southern California (LAC + USC) Medical Center Institutional Review Board.
Patient Population
Adult patients admitted to the cardiac intensive care unit (CICU) at LAC + USC Medical Center from January 2008 to July 2009 were screened for inclusion. Patients were included if they were admitted with acute heart failure and received IV nitroglycerin or nesiritide for at least 6 hours. Patients were excluded if they required continuous renal replacement therapy, hemodialysis, or were not prescribed an IV diuretic regimen concomitantly.
Data Collection
Data were extracted using the CICU Eclipsys computer system and verified with paper charts when necessary. Eclipsys is a computerized system used in the CICU for daily documentation for each patient. Records in this system include medication orders and administration times, laboratory tests, hourly clinical monitoring (vital statistics, hemodynamics, fluid balance, etc), and health care provider notes. Data collected related to the study objectives included baseline demographics (age, gender, height, weight, ethnicity, past medical, and social history), heart failure characteristics (New York Heart Association functional classification prior to need for hospitalization, left ventricular ejection fraction, and etiology), hourly vital statistics and urine output, serum chemistries, and in-hospital medication regimens.
Statistical Analysis
The primary safety endpoint was the incidence of AKI defined by the RIFLE classification system (creatinine criteria) 13 or a traditional renal endpoint defined as an acute rise in creatinine of 0.3 mg/dL or 25% from the baseline. The urine output criteria were not applied to this patient population as diuretics were required in all the participants included. Secondary safety endpoints included incidence and degree of hypotension (systolic blood pressure, SBP <90 mm Hg, <80 mm Hg or mean arterial pressure, MAP <50 mm Hg). The secondary safety endpoints were assessed by chi-square test. Additional outcomes included absolute and relative changes in GFR as assessed by the Modified Diet in Renal Disease (MDRD) formula, estimated creatinine clearance (eClcr) by Cockcroft-Gault equation, blood urea nitrogen (BUN) and serum creatinine (Scr), which were assessed by the paired t test. Baseline demographics were compared between groups by 1-way analysis of variance or chi-square test for continuous and nonparametric data, respectively.
The primary efficacy endpoint was the mean hourly urine output after initiation of IV vasoactive therapy assessed at 6 hours, 24 hours, and at the end of the treatment. Comparison at each time point was assessed by independent samples t test. Significance was set at P < .05.
Results
Patient Population
A total of 131 patients received nitroglycerin (N = 94) or nesiritide (N = 37) and met the inclusion criteria. The mean age of the study population was 57 ± 12 (mean ± SD) years, and 67.2% of the study population was male. The cause of heart failure was unknown in a large proportion of patients (ischemic heart disease 30.1% and dilated cardiomyopathy 24.3%). The mean left ventricular ejection fraction (LVEF) was 38 ± 17%. The majority of patients had a baseline New York Heart Association Functional Classification of II (50.6%) or III (32.2%) prior to admission. Baseline characteristics (Table 1 ) were similar between study groups, with the exception of a significant difference in history of alcohol use (nesiritide 48.1% vs nitroglycerin 62.8%, P = .02). A total of ninety-four percent of patients were on furosemide, of which 69.5% were on a continuous infusion. The mean maximum dose for the continuous infusion was 7.1 ± 3.2 mg/h for the nesiritide group and 6.4 ± 2.5 mg/h for the nitroglycerin group (P = .24). While not statistically significant, patients in the nesiritide treatment group had a lower LVEF (nesiritide 34 ± 15% vs nitroglycerin 40 ±17%, P = .05), higher mean baseline serum creatinine (nesiritide 1.44 ± 0.59 mg/dL vs nitroglycerin 1.31 ± 0.80 mg/dL, P = .37), and lower mean baseline SBPs (nesiritide 114 ± 24 mm Hg vs nitroglycerin 123 ± 23 mm Hg, P = .06) than the nitroglycerin group. The mean duration of study drug exposure was 38.6 ± 35.7 hours for nesiritide, with a mean maximum dose of 0.01 ± 0.002 mcg/kg per minute. Mean duration for nitroglycerin was 30.7 ± 22.6 hours, with a mean maximum dose of 128 ± 120 mcg/min. The mean time from admission to initiation of IV vasodilatory therapy was 29 ± 42 hours.
Patient Demographics

NOTE: P value for nesiritide vs nitroglycerin. Abbreviations: ACE/ARB, angiotensin converting enzyme inhibitor or angiotensin receptor blocker; CCB, calcium channel blocker; CI, continuous infusion; HTN, hypertension; IHD, ischemic heart disease; LVEF, left ventricular ejection fraction; NTG, nitroglycerin.
Comparative Safety Endpoints
There was no difference in the incidence of acute renal injury between groups (Table 2 ) by RIFLE creatinine criteria (nesiritide 18.9% vs nitroglycerin 22.2%, P = .88) or by the traditional acute renal insufficiency definition (nesiritide 21.6% vs nitroglycerin 34.4%, P = .16). The absolute decrease in GFR calculated by the MDRD formula 24 hours after therapy initiation was significantly greater for nitroglycerin than for nesiritide (nesiritide −0.7 ± 18.2 mL/min per 1.73 m2 vs nitroglycerin −9.1 ± 20.5 mL/min per 1.73 m2, P = .03). Mean absolute change in eClcr decreased to a greater degree in nitroglycerin-treated patients; however, the difference did not meet statistical significance compared to nesiritide. Mean change in BUN at 24 hours also favored nesiritide (nesiritide −0.2 ± 9.8 mg/dL vs nitroglycerin 4.2 ± 9.1 mg/dL, P = .02).
Comparative Effect on Renal Function and Blood Pressure

NOTE: P value for nesiritide vs nitroglycerin. Abbreviations: BUN, blood urea nitrogen; eClcr, estimated creatinine clearance (using Cockcroft-Gault equation); GFR, glomerular filtration rate (estimated by Modification of Diet in Renal Disease equation); MAP, mean arterial pressure; MDRD, modified diet in renal disease; Scr, serum creatinine; SBP, systolic blood pressure.
The incidence of hypotension was higher in the nesiritide group with 25.7% of patients experiencing a SBP of less than 80 mm Hg vs 8.0% of nitroglycerin patients (P = .01). Only 2 patients (1 nitroglycerin and 1 nesiritide) had their vasodilatory therapy stopped for the low SBP. A total of 17.1% of nesiritide-treated patients experienced a MAP less than 50 mm Hg compared to only 4.5% of nitroglycerin-treated patients (P = .03). Over the 24-hour period after drug initiation, SBP was significantly lower at 1, 2, 6, 12, and 24 hours in the nesiritide group (Figure 1 ). In-hospital mortality was 7%, with no difference between groups (Table 2).
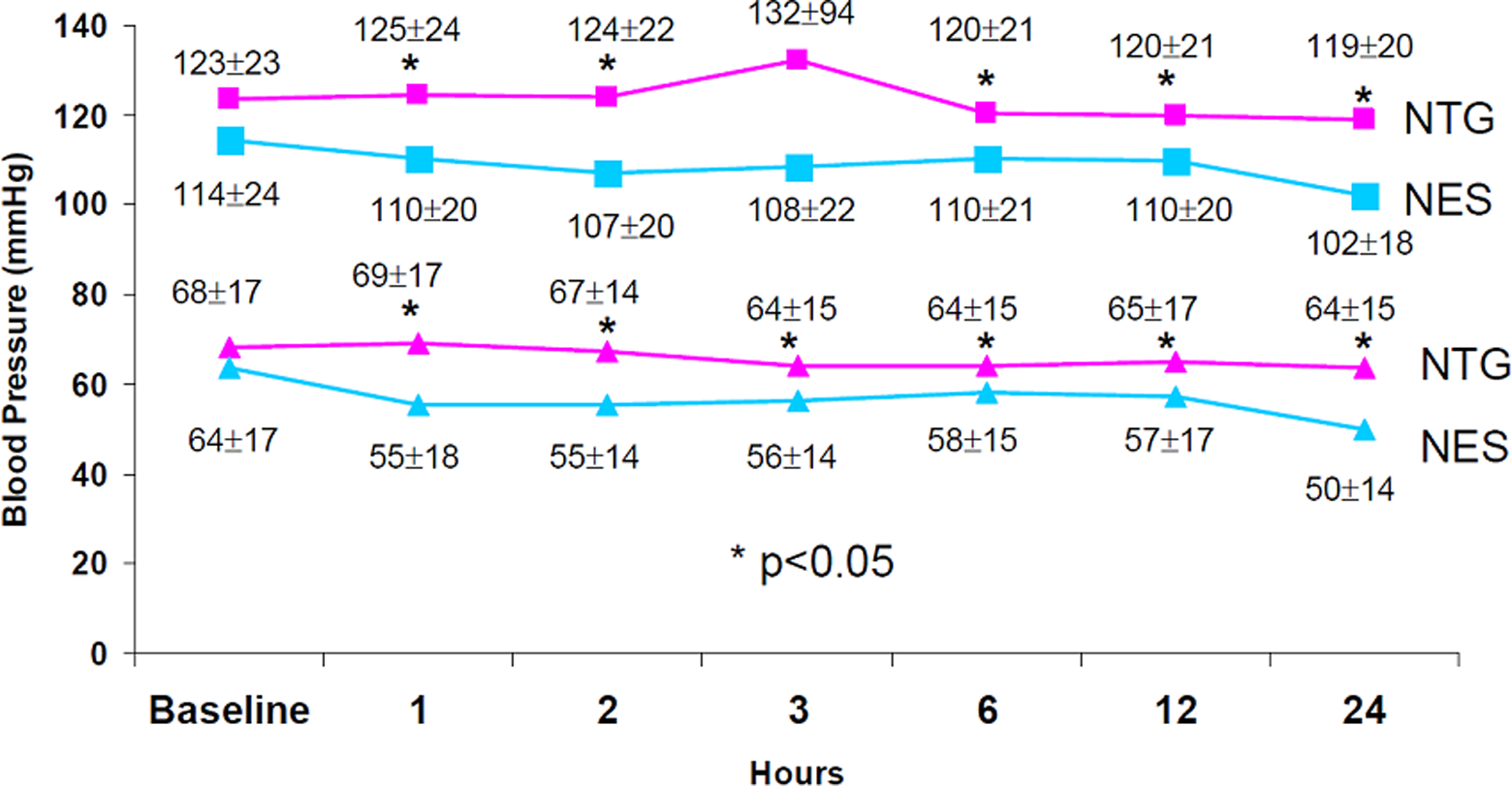
Blood pressure trends. NTG indicates nitroglycerin; NES, nesiritide.
Comparative Efficacy Endpoints
There was no significant difference in mean hourly urine output at 6 hours, 24 hours, or at end of treatment between nesiritide and nitroglycerin (Table 3 ). Total urine output at the end of treatment was 6419 ± 6562 mL and 4249 ± 3445 mL for nesiritide and nitroglycerin, respectively (P = .06).
Comparative Effect on Urine Output

Abbreviation: UO, urine output in milliliters.
Discussion
Patients with acutely decompensated heart failure often require the addition of nesiritide or nitroglycerin to their diuretic regimen to facilitate relief of congestion which may also enhance diuresis. Several studies have linked the development of renal insufficiency with negative clinical outcomes, 3 although there is limited data with most drugs used in the treatment of acute heart failure. In this study, there was no difference in the incidence of acute renal injury between nitroglycerin- and nesiritide-treated patients as assessed by the RIFLE creatinine/GFR criteria or the traditional definition of acute renal insufficiency. Interestingly, the traditional definition detected a greater frequency of renal injury compared to the RIFLE definition. Only the creatinine/GFR criteria for the RIFLE classification system was used because the mandated use of diuretics in acute heart failure negates the utility of the urine output criteria. Only one other study has applied the RIFLE criteria in the acute heart failure setting. 13 Hata et al found that the patients with acute heart failure that developed AKI stayed in the ICU about 4 days longer and in the hospital about 23 days longer than those patients that did not develop AKI. The distribution of AKI in the Hata study was 40.1%, 18.6%, and 0.2%, corresponding to risk, injury, and failure categories, respectively. 13 Our study found 16.2% and 2.7% of patients in the nesiritide group fell into the Risk and Injury category, respectively, compared to 20% and 2.2% in the nitroglycerin group. No patients in our study developed AKI classified as Failure.
Although we did not detect a difference between nesiritide and nitroglycerin in the development of acute renal injury, our results do support a potential differential effect on renal function. There was a significant difference in the change in GFR at 24 hours after initiation between therapies. The results suggest preservation of renal function by nesiritide and a slight decline in nitroglycerin-treated patients. The effect on GFR was mirrored by the changes in BUN, with no change in nesiritide-treated patients and an increase in nitroglycerin-treated patients. Although nesiritide has not been shown to improve the renal function in heart failure, our results are consistent with its potential to preserve renal function in conditions that would normally challenge the kidney including concomitant diuretics, heart failure, and cardiac surgery.8,14–17 The magnitude of the observed differences in GFR and BUN in our analysis may be clinically important. An acute decrease in GFR of 10 mL/min in patients with acute heart failure has been associated with a 7% increase in mortality. 3 In addition, elevated BUN on admission and discharge has been associated with survival independent of serum creatinine in patients hospitalized with heart failure. 18 In a retrospective analysis of the Outcomes of a Prospective Trial of Intravenous Milrinone for Exacerbations of Chronic Heart Failure study, every 5 mg/dL increase in BUN was associated with an 8% increase in risk of death at 60 days. 19
Few studies have reported the effect of IV vasodilators on renal function. Cogan et al assessed the effect of the vasodilator nitroprusside on renal clearance in patients with acute heart failure and found that nitroprusside increased the cardiac output and renal blood flow; however, it failed to increase GFR. 4 Leier examined the regional blood flow response to IV nitroglycerin in patients with acute heart failure, and found renal blood flow actually decreased despite increases in cardiac output. In the same study, nitroprusside was found to increase renal blood flow and cardiac output. 5 No information on GFR or urine output was reported. In a renal hemodynamic evaluation of nesiritide in patients with acute heart failure, despite an increase in cardiac output, no effect on renal blood flow or renal vascular resistance was detected. 6
Our study demonstrated a significant increase in mean hourly urine output after initiation of either nesiritide or nitroglycerin compared to baseline, with no difference between therapies. It appears both nesiritide and nitroglycerin may have had the greatest effect on urine output within the first 6 hours of treatment. There was a statistically significant difference in total urine output at the end of the treatment with nesiritide compared to nitroglycerin. However, this may have been influenced by fluid intake and the extent to which the patients were fluid overloaded, which were not assessed in this study. Although not statistically significant, the average duration of treatment for the nesiritide-treated patients was also approximately 8 hours longer than for nitroglycerin, which may also account for this difference.
A proposed mechanism for the development of renal insufficiency in patients with acute heart failure is renal hypoperfusion from hypotension or excessively reduced ventricular filling pressures.20,21 A decreased blood supply may lead to a reduction in oxygen delivery, cellular apoptosis and the RAAS, sympathetic nervous system, and inflammatory pathway activation. There was a significant difference in the incidence of hypotension between groups. Patients in the nesiritide group had a lower SBP at baseline which may account for the greater incidence in SBPs of less than 80 mm Hg, MAPs lower than 50 mm Hg, and lower blood pressures over the 24-hour period after treatment. Although the nesiritide was associated with lower blood pressures, it did not appear to have an adverse effect on renal function. Whether this is a reflection of nesiritide exerting a renal protective effect is unknown.7,17 It may also be possible that the magnitude of reduction in blood pressure seen in the nesiritide-treated patients was not substantial enough to lead to decreased renal perfusion.
There was no significant difference in the incidence of death between patient groups. Baseline characteristics suggest that the patients treated with nesiritide were sicker prior to initiation of therapy as they had a significantly lower LVEF and baseline SBP. Low SBP on admission has also been a predictor for worse outcomes. 2 However, this study was not powered to detect subtle differences in patient outcomes.
Our study has limitations. Inherently, retrospective analyses only allow for detection of associations, and cause and effect cannot be determined. Our findings must also be interpreted under the context of these IV vasodilators being used in patients receiving IV loop diuretics. Whether nesiritide or nitroglycerin exert independent effects on GFR or BUN remains a question to be answered.
Conclusion
The incidence of renal injury was not different between nesiritide- and nitroglycerin-treated patients with acute heart failure; however, nitroglycerin was associated with a decline in GFR and increase in BUN despite higher baseline and on-treatment blood pressures. Prospective studies to clarify the renal effects of IV vasodilators are warranted.
Footnotes
Acknowledgment
The authors thank Tesfa Ghebreyesus for his assistance in extracting data from the ICU computer system to conduct this study.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Tien Ng has received compensation as a consultant for Otsuka America Pharmaceutical, Inc.
The author(s) received no financial support for the research and/or authorship of this article.
