Abstract
Background:
Renin-angiotensin-aldosterone system (RAAS) activation in heart failure with reduced ejection fraction (HFREF) is detrimental through promotion of ventricular remodeling and salt and water retention.
Aims:
The aims of this article are to describe RAAS activity in distinct HFREF populations and to assess its prognostic impact.
Methods:
Venous blood samples were prospectively obtained in 76 healthy volunteers, 72 patients hospitalized for acute decompensated HFREF, and 78 ambulatory chronic HFREF patients without clinical signs of congestion. Sequential measurements were performed in patients with acute decompensated HFREF.
Results:
Plasma renin activity (PRA) was significantly higher in ambulatory chronic HFREF (7.6 ng/ml/h (2.2; 18.1)) compared to patients with acute decompensated HFREF (1.5 ng/ml/h (0.8; 5.7)) or healthy volunteers (1.4 ng/ml/h (0.6; 2.3)) (all
Conclusion:
PRA is significantly elevated in ambulatory chronic HFREF patients but is not associated with worse outcome. In contrast, in acute HFREF patients, PRA is associated with cardiovascular mortality or HF readmissions.
Introduction
Renin-angiotensin-aldosterone system (RAAS) activation in heart failure with reduced ejection fraction (HFREF) has detrimental long-term effects such as water and salt retention as well as promoting adverse ventricular remodeling. Outcomes in HFREF patients have drastically improved during the past two decades through strategies that have targeted RAAS activation.1–4 Plasma renin activity (PRA) and plasma aldosterone levels are biomarkers that quantitatively reflect RAAS activation and might be used for risk stratification in HFREF. Indeed, previous studies have linked higher levels of RAAS activation to more advanced disease stages and worse outcomes both in acute and chronic HFREF.5–9 Most of these studies have focused on PRA as renin is the rate-limiting step of the RAAS, and a more reliable reflection of RAAS activation compared to serum aldosterone.10,11 However, these studies largely predate the current era of HFREF treatment in which angiotensin-converting enzyme inhibitors (ACE-i), angiotensin receptor blockers (ARB), beta-blockers and mineralocorticoid receptor antagonists (MRA) are guideline-recommended therapies. Indeed, nowadays cardiologist uptitrate ACE-i/ARB and beta-blockers to the highest achievable dose without intolerable side effects (e.g. systolic blood pressure <90 mmHg, symptomatic orthostatic hypotension, decline in renal function, hyperkalemia). 12 Moreover, whether RAAS activation during decongestive therapy has prognostic significance remains unclear. 13 Therefore, the objective of this study is to describe the extent of RAAS activation, and its prognostic impact in well-characterized HFREF populations on optimal medical therapy.
Methods
Study design
This prospective cohort study was carried out in a single tertiary care center (Ziekenhuis Oost-Limburg, Genk, Belgium) between September 2011 and October 2015. The study complies with the Declaration of Helsinki and the institutional review board approved the study protocol. All participants provided written informed consent before any study-specific intervention was performed.
Study population
Patients were eligible for study inclusion if ⩾18 years of age and able to give informed consent.
Study endpoint
Cardiovascular mortality and HF readmissions (defined as hospitalizations because of signs or symptoms of congestion or low cardiac output that warranted treatment with parenteral drugs) were prospectively registered in all study patients from inclusion up till three years after which they were censored.
Laboratory measurements
Venous blood samples were obtained at the moment of study inclusion with the patient in the supine position after an adaptation period of 30 minutes. Plasma NT-proBNP levels were measured by the Roche Diagnostics Assay (Roche, Rotkreuz, Switzerland). PRA was determined using the Gamma-coat*radio immunoassay (DiaSorin, Sallugia, Italy). Plasma aldosterone levels were assessed by the Aldosterone Maia radioimmunoassay (Adaltis, Rome, Italy).
Within the subpopulation of acute decompensated HFREF sequential venous blood samples were obtained before the start of intravenous therapy (baseline), after three days of decongestive therapy, and during ambulatory follow-up approximately six weeks after discharge. Treating physicians were blinded to test results and treatment during hospitalization was at their own discretion.
Statistical analysis
Continuous variables are expressed as mean±standard deviation, if normally distributed, or otherwise by median (interquartile range). Normality was assessed by the Shapiro-Wilk statistic. Categorical data are expressed as percentages and compared with the Pearson χ²-test. One-way analysis of variance (ANOVA) testing or the Kruskal-Wallis
Results
Study population
Seventy-six healthy volunteers, 72 patients with acute decompensated HFREF and 78 ambulatory chronic HFREF patients were included. Table 1 summarizes their baseline characteristics. Compared to healthy controls, acute and chronic HFREF patients were older and had a severely impaired LVEF (25±10 vs 33±7, respectively). Neurohormonal blocker use was high in both cohorts of HFREF patients. However, compared to chronic ambulatory HFREF patients, fewer patients with acute decompensated HFREF were on maintenance therapy with an ACE-i or ARB (50% vs 87%). Instead, 26% of acute decompensated patients were taking oral vasodilators (hydralazine and/or nitrates). Loop diuretic use was highest in the cohort of acute HFREF patients.
Baseline characteristics of the study population.

ACE-i: angiotensin-converting enzyme inhibitor; ARB: angiotensin receptor blocker; bpm: beats per minute; HFREF: heart failure with reduced ejection fraction; MRA: mineralocorticoid receptor antagonist; NT-proBNP: N-terminal of the prohormone of B-type natriuretic peptide; NYHA: New York Heart Association; PRA: plasma renin activity.
RAAS activation in distinct populations of HFREF
PRA was significantly higher in ambulatory chronic HFREF patients (7.6 ng/ml/h (2.2; 18.1)) compared to acute decompensated HFREF patients (1.5 ng/ml/h (0.8; 5.7)) or healthy volunteers (1.4 ng/ml/h (0.6; 2.3)) (both

Plasma renin activity (PRA) in healthy volunteers, patients with acute decompensated heart failure with reduced ejection fraction (HFREF) and ambulatory chronic HFREF.
Uni- and multivariable regression analysis for significant determinants of plasma renin activity in the total study population.

To correct for non-normal distribution, PRA was logarithmically transformed. Beta and standard error (S.E.) for continuous variables reported per standard deviation change. ACE-i: angiotensin-converting enzyme inhibitor; ARB: angiotensin receptor blocker; bpm: beats per minute; MRA: mineralocorticoid receptor antagonist; NT-proBNP: N-terminal of the prohormone of B-type natriuretic peptide.
PRA during decongestive therapy and uptitration of neurohormonal blockers in patients with acute decompensated HF
During decongestive treatment (from baseline to day 3) in patients with acute decompensated HFREF, the absolute and relative increase in PRA was +2.4 ng/ml/h (−0.2; 6.9) and +107% (−12%; 553%), respectively (both

Plasma renin activity (PRA) from admission until six weeks after discharge in patients with acute heart failure and reduced ejection fraction. PRA levels rise during decongestive therapy and neurohumoral uptitration.
Use of neurohormonal blockers and loop diuretics in patients presenting with acute decompensated heart failure and reduced ejection fraction at baseline (BL), after three days (D3) of decongestive treatment, and at six weeks (6W) after discharge.

ACE-i: angiotensin-converting enzyme inhibitor; ARB: angiotensin receptor blocker; MRA: mineralocorticoid receptor antagonist.
PRA and clinical outcome
During the entire follow-up period, 36 events occurred in patients with acute decompensated HFREF (22 patients died from cardiovascular causes and 14 patients were readmitted for worsening HF). PRA levels at admission in the highest tertile were associated with a significantly increased event rate (log rank=0.035) (Figure 3). There was no difference in outcome between patients with a PRA rise vs decline during recompensation (from baseline until day 3) (log rank=0.96) (Figure 4, Table 3 Supplementary Appendix). In the subgroup with ambulatory chronic HFREF patients, one death and six HF readmissions occurred. PRA levels were not associated with clinical outcome (log rank = 0.99) (Figure 3). Baseline characteristics of acute decompensated and chronic stable HFREF patients per tertile are presented in the supplementary appendices.

Kaplan–Meier curves for the combined endpoint of heart failure-associated hospitalization and cardiovascular mortality in patients with acute decompensated heart failure with reduced ejection fraction (HFREF) (upper panel) and ambulatory chronic HFREF (lower panel) according to tertiles of plasma renin activity (PRA).
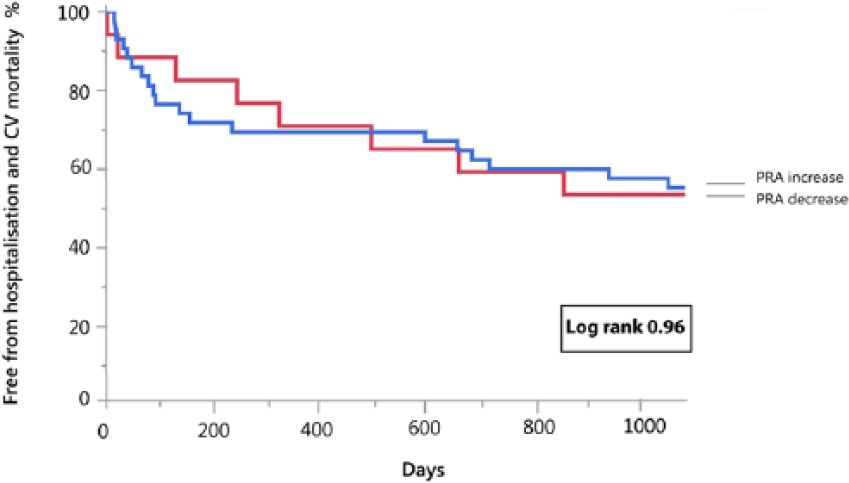
Kaplan–Meier curve for the combined endpoint of heart failure-associated hospitalization and cardiovascular mortality in patients hospitalized for acute heart failure with plasma renin activity (PRA) increase vs PRA decrease during decongestive therapy.
Discussion
Longitudinal data from distinct well-characterized HFREF populations in the current era of treatment with neurohormonal blockers provide a unique opportunity to examine RAAS activation. The primary findings of this study are (1) significant PRA is present in ambulatory chronic HFREF patients without signs and symptoms of congestion, while PRA seems depressed during episodes of acute decompensated HFREF with clear signs of volume overload; (2) PRA levels correlate significantly to worse outcomes only in patients with acute decompensated HFREF before initiation of decongestive treatment; and (3) treatment with neurohormonal blockers significantly influence neurohormonal levels.
The RAAS
To better appreciate the study results, a brief review of the RAAS is useful. Renin, an enzyme released by juxtaglomerular cells of the renal afferent arteriole, starts a cascade in which angiotensinogen is cleaved first into angiotensin I, which is further metabolized to angiotensin II (Ang II) by ACE. Ang II causes systemic and renal arteriolar vasoconstriction, promotes renal tubular sodium and water reabsorption, and is a potent stimulator of aldosterone release from the adrenal glands. Upon an acute drop of the cardiac output, RAAS activation helps to preserve organ perfusion in general and the glomerular filtration rate in particular.16–18 Renin is released from the afferent arteriole in response to three main stimuli: (1) decreased arterial blood pressure sensed by baroreceptor cells in the afferent arteriolar vessel wall, (2) decreased chloride concentrations in macula densa cells lining the renal tubules at the end of Henle’s loop, and (3) sympathetic nerve system activation.19,20 As a result, renin release is physiologically inhibited by normal or elevated systemic blood pressure and a diet high in salt.21–23 Persistent and excessive RAAS activation causes adverse ventricular remodeling and contributes to fluid retention with signs and symptoms of congestion.24–30
High PRA in ambulatory chronic HFREF
More than two decades ago, before the standard use of neurohormonal blockers in HFREF, Francis et al. compared neurohormonal activation—including PRA—in healthy volunteers vs asymptomatic HFREF patients vs HFREF patients with signs and symptoms of congestion. The authors concluded that neurohormonal activation already occurred in patients with left ventricular dysfunction before the onset of symptoms, which was further exaggerated as overt HF ensued and diuretics were added to therapy. 7 Remarkably, important increases in neurohormonal activation were mainly seen in the patients with symptomatic HFREF, while the PRA increases in asymptomatic patients were modest. This is in contrast to our findings in a contemporary cohort of HFREF patients, where the most pronounced PRA rise was observed in ambulatory chronic HFREF patients without signs and symptoms of congestion but well treated with ACE-i, ARB, beta-blockers and MRA.
Indeed, ACE-i and ARB tend to lower aldosterone concentrations, but increase PRA, while beta-blockers might lower both, and MRA increase both.31–37 Although none of the patients included in this study were taking an angiotensin receptor blocker-neprilysin inhibitor (ARNI), in light of the new HF guidelines it may be interesting to point out that previous studies have demonstrated that the association of sacubitril, a neprilysin inhibitor, does not affect PRA or serum aldosterone concentration.38,39 Hypertensive patients treated with neurohormonal blockers demonstrate increased PRA and plasma aldosterone levels.40–42 Yet, the individual response to medication varies greatly because of genetic polymorphisms.6,43–47 Our data corroborate this as we observed a wide spread both in PRA and plasma aldosterone levels among patients with ambulatory chronic HFREF. Overall, patients with ambulatory chronic HF had significantly higher levels of PRA compared to acute decompensated HF patients while exhibiting a much lower risk. Thus, the association between PRA and outcome seems to apply only to the higher-risk groups of HF patients. Therefore, most probably the PRA and serum aldosterone levels do not reflect disease-related (harmful) RAAS activation and are not a reliable surrogate for downstream receptor activation.48,49 Interestingly, the RAAS consists of two main axes: the classical ACE/Ang II and the counteracting ACE2/angiotensin 1 to 7 (Ang 1–7) axis. 50 ACE2, a homolog of ACE, degrades Ang II into Ang 1–7. Ang 1–7 exerts a wide array of actions, many of which are opposite to those attributed to Ang II (vasodilation, decrease in fibrosis and cardiomyocyte hypertrophy, inhibition of aldosterone secretion etc.).50,51 In our study we measured PRA, the rate-limiting step of the RAAS system and common to both axes and serum aldosterone. However, we did not measure Ang II, Ang 1–7 or other products of intermediate steps of the complex RAAS. It has been shown that in response to ACE-i, PRA rises but plasma levels of Ang 1–7 also increase. A high percentage of ambulatory chronic HF patients, but also acute decompensated patients were on ACE-i as well as MRA. Therefore, it may be a logical hypothesis that the increased renin activity due to efficient blockade of the classical ACE/Ang II axis leads to stimulation of the counteracting and beneficial axis of the RAAS (ACE2/Ang 1–7), which may be partly responsible for the beneficial effects of this therapy in HF patients. This could be an interesting topic for further research for the development of new targets for HF therapies.
PRA in acute HFREF
As neurohormonal activation is often perceived as the key driver in HFREF disease progression, it may seem odd that PRA and serum aldosterone levels are significantly lower in patients with signs and symptoms of congestion. Furthermore, this contradicts former observations in medication-naive HFREF patients. 7 Yet, most chronic HFREF patients in the current study were on maximally tolerated dosages of neurohormonal blockers and also have rather low blood pressure. Both are powerful predictors of PRA levels in our overall population (Table 2). In contrast, most patients with acute decompensated HFREF present with elevated rather than low arterial blood pressure, a finding also present in the current study. 52 Intriguingly, this might indicate that the RAAS in advanced HFREF treated with neurohormonal blockers remains appropriately responsive to hemodynamic changes including blood pressure and volume overload. Also, the most important increase in RAAS activation during the treatment of acute HFREF is seen in the first days of hospitalization, which seem to be linked to decongestive therapy (reduction in plasma volume as well as intensified diuretic therapy) and introduction and/or uptitration of neurohormonal blockers. Therefore, one might speculate that PRA could be a potential surrogate for effective circulatory volume assessment.
Prognostic value of PRA
PRA levels correlate significantly to worse outcomes only in patients with acute decompensated HFREF before decongestive treatment. High levels of neurohormones in stable HFREF patients are not predictive for rehospitalization due to water and salt retention or death. Also, the relation of high PRA levels and negative outcome in acute decompensated HFREF might not be driven by higher neurohormonal activation but rather reflect more advanced disease in this subgroup, reflecting volume overload and low pressure, both known to be related to worse outcome.50,51 In conclusion, it seems that the association between an RAAS biomarker and adverse outcomes applies only to the setting and not to its absolute value.
Study limitations
First, we recruited and compared two groups of HFREF patients. Although we were able to characterize these groups in detail, it is uncertain to what extent observed differences in RAAS activation were due to heterogeneity between groups. The fact that all patients were recruited from a single institution, the limited sample size and event rates makes findings hypothesis-generating and ask for separate confirmation. Second, patients with urgent need for administration of loop diuretics, inotropic/vasodilator or mechanical support were not included in this study. Thus, our data do not apply to severely decompensated HF patients or patients in cardiogenic shock. Third, all patients were put in a semi-supine position for an adaptation period before a venous blood sample was drawn. However, besides physical activity, PRA also depends on many other variables such as circadian rhythm, sodium intake, and presence/absence of disease states that could not be accounted for in this cohort study. 53
Conclusion
PRA is decreased in a state of acute decompensation compared to ambulatory chronic HFREF. An increase in PRA activity is observed in the majority of patients during decongestive treatment and neurohormonal blocker uptitration. However, increased PRA is associated with adverse outcomes only in the setting of acute decompensated HFREF before initiation of decongestive treatment.
Footnotes
Declaration of conflicting interests
None declared.
Funding
P. N., F.H.V., P.M. and W.M. are researchers for the Limburg Clinical Research Program (LCRP) UHasselt–ZOL–Jessa, supported by the foundation Limburg Sterk Merk (LSM), Hasselt University, Ziekenhuis Oost-Limburg and Jessa Hospital. F.H.V. and P.B.B. are supported by a PhD fellowship of the Research Foundation–Flanders (FWO). P.N. and M.D. are supported by a research grant provided by Vision4Life-Sciences.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
