Abstract
Introduction
The prognostic role of the AGTR1 A1166C genetic polymorphism in patients with acute myocardial infarction (AMI) is controversial and inconsistent across various study populations.
Materials and Methods
We conducted a cross-sectional analysis with 6 months of follow-up. All participants were identified for AGTR1 A1166C polymorphism using the polymerase chain reaction method, with follow-up on survival status at 6 months from the date of admission. The Cox regression model and survival curves were estimated using the Kaplan–Meier curve and corrected with confounding factors.
Results
The proportions of AA, AC and CC genotypes were 88.1%, 10.9%, and 1%, respectively. Mortality after 6 months occurred in 31 cases (30.7%). 6-month survival analysis through Kaplan-Meier curves showed a trend with lower cumulative survival of the AC and CC genotype group with p = 0.02, and the outcomes were still significant with p = 0.05 when corrected for confounding factors. Although Kaplan-Meier curves and Cox regression models indicated a marginally elevated incidence of heart failure within the AC and CC genotype populations following a six-month follow-up period, the statistical analysis revealed no significance.
Conclusion
AGTR1 A1166C gene polymorphisms could be an important factor in increasing all-cause mortality in patients after AMI.
Introduction
Acute myocardial infarction (AMI) remains a public health problem with high incidence rates, high mortality complications, and a significant reduction in the patient's quality of life. AMI affects more than 7 million patients worldwide every year, consuming high medical costs and causing a huge socio-economic burden. 1 The global burden of cardiovascular disease (CVD) and myocardial infarction (MI) has shifted to low- and middle-income countries, which account for more than 80% of cardiovascular deaths.1,2 In clinical practice, AMI is a common cardiovascular emergency with high mortality and morbidity. This is the most severe manifestation of coronary artery disease, accounting for more than one-third of deaths in developed countries each year.3,4 AMI leads to in-hospital mortality with high proportion, and two-thirds fold higher when non-adherence to guidelines of treatment. 5 Increased use of treatment and lifestyle changes have significantly reduced deaths due to MI in recent decades. However, the mortality rate due to AMI is still high.6,7
AMI is a multifactorial disease with a complex mechanism including interactions between genetic factors and environmental factors. Genetic factors can contribute about 50–60% to the pathogenesis of MI. 8 In addition to demographic factors, comorbidities, clinical and paraclinical characteristics, and treatment methods, the prognosis of death in patients with AMI is also influenced by genetic factors. Many genetic markers related to AMI have been identified, including genes encoding components of the renin–angiotensin–aldosterone (RAA) system. The RAA system plays an important role in blood pressure regulation, hemodynamic stability and homeostasis. Dysregulation of the RAA system is related to the pathophysiology of many different cardio-metabolic diseases, including AMI. 9 Excessive activation of the RAA system through the adverse effects of angiotensin II includes vasoconstriction, secretion of aldosterone, direct effects on cardiomyocytes, stimulating myocardial hypertrophy, growth of vascular smooth muscle cells and fibroblasts causing remodeling, cardiac dysfunction, heart failure, arrhythmia and progression of atherosclerosis, leading to severe cardiovascular events and death in patients with AMI. 10
Genes of the RAA system have been studied a lot in MI pathology, including the AGTR1 gene. The AGTR1 gene encodes the type 1 angotenin II receptor located on the long arm of chromosome 3 (3q21-25) and has the most studied variant A1166C (rs5186).8,11 The AGTR1 CC genotype is least common in both Asian and other populations around the world.12–15 Over the past three decades, the AGTR1 A1166C gene variant has been studied and found to be related to the onset or severe of hypertension diabetic nephropathy, and cancer, especially CVD such as CAD, AMI, heart failure (HF), and arterial stiffness.16–22 Still, results vary across different countries, geographic regions, and races. Data on genotype prevalence and association of this gene variant with characteristics and prognosis of major adverse cardiovascular events (MACE) including death and heart failure after acute MI have not been fully detected in Asian countries. Furthermore, the study of the AGTR1 A1166C gene variant may provide useful information for individualizing and optimizing treatment and prevention of AMI in today's era of precision medicine. Therefore, this study aim to find out the prevalence of the AGTR1 A1166C gene variant as well as determining whether these variants are associated with all-cause morbidity and heart failure in Vietnamese population, thereby contributing to the management strategy of AMI patient. Our study's outcomes could contribute to the management strategy of AMI patients.
Methods
Study design, population, and ethical approval
We conducted a cross-sectional analysis with 6 months of follow-up study at Can Tho University of Medicine and Pharmacy Hospital, Can Tho City, Vietnam between June 1, 2022 and June 1, 2024. The Ethics Committee in Biomedical Research of the University of Medicine and Pharmacy at Can Tho City approved this research with the approval number: 22.270.HV/PCT-HDDD. Enrolled patients had to sign a consent form to participate in the study, and all of the information of patients was kept confidential.
Inclusion criteria: Eligible patients were ≥18 years old with a confirmed diagnosis of AMI according to The Fourth Universal Definition of Myocardial Infarction (2018) 23 including ischaemic symptoms and abnormality of electrocardiogram. 24
Exclusion criteria: Medical history of AMI; percutaneous coronary intervention; coronary bypass surger; Loss of contact during follow-up of all-cause mortality; Refused to provide the informed consent or did not want to take part in this study.
Sample size
The sample size was calculated according to the one proportion estimated formula. 25 As for identifying the prevalence of AGTR1 A1166C, according to the study by Franco et al. (2007), 26 the prevalence of the AGTR1 A1166C gene variant was 8%. The association of mortality and gene polymorphism assessment is based on the mortality prevalence of the Duy Cong Tran et al. (2024) study, 19 reported as 10.9% (p = 0.109). We calculated n ≥ 79 (with p = 0.08, α = 5%, d = 0.06). We conducted on 101 AMI patients to minimize bias due to sample loss.
Study variables and data collection methods
Enrolled patients were examined for clinical characteristics, risk factors, and comorbidities. Baseline and clinical characteristics included: Age; Sex; Classification of Killip (Classified into 4 grades from I to IV indicating the severe of admission condition);27 New York Heart Association (NYHA) (4 classes of the clinical symptoms severe); 28 and Global Registry of Acute Coronary Events (GRACE) score (Assess base on patient's age, Killip grade, systolic blood pressure (SBP), change ST in the electrocardiogram, circulatory arrest, creatinine level, cardiac biomarkers increase, heart rate, and risk stratification into 3 degrees mild, moderate, and severe) 29 on admission. Risk factors and comorbidities also examined by clinical physicians included: Family history of premature CAD; Overweight and obesity (World Health Organization (WHO) classification for Asian body mass index (BMI), overweight when BMI ≥ 23 Kg/m2); 30 Smoking (Still smoking or quit less than 5 years); 31 Hypertension (European Society of Cardiology and European Society of Hypertension (ESC/ESH) 2018, hypertension when SBP ≥ 140 mmHg and/or diastolic blood pressure (DBP) ≥ 90 mmHg), 32 Type 2 diabetes mellitus (T2DM) (American Diabetes Association (ADA) recommendations 2020); 33 Dyslipidemia (The National Cholesterol Education Program Adult Treatment Panel III Guidelines (NCEP – ATP III)). 34
Laboratory tests were performed by experienced cardiologists to confirm the diagnosis of AMI. The data collected included: electrocardiogram (NIHON KODEN 9620-12000); 24 first hour (1st) and second hour (2nd) of troponin T high sensitive (hs) concentration (Cobass e411); echocardiography assessed left ventricular ejection fraction (LVEF) (Philips Affiniti 50); order serum biochemistry for patient's optimal treatment (urea, creatinine, glucose, aspartate transaminase (AST) and alanine transaminase, aka alanine aminotransferase (ALT)) (Boehringer Mannheim). Diagnosis confirms the type of MI when it meets all of the criteria following The Fourth Universal Definition of Myocardial Infarction (2018). 23
Patients were followed up for all-cause mortality in the hospital, first month, third month, and sixth month from admission. Re-examination patients received clinical symptoms assessment, serum biochemistry, and echocardiography for assessed appearance of MACE. Survival or mortality was recorded through regular rechecks or telephone calls.
Genotyping of AGTR1 A1166C
Polymerase chain reaction (PCR) was used to assess the patient's genotype: (1) 2 mL of venous blood was collected in EDTA-anticoagulant tubes (Store at 2–8 °C for a maximum of 72 h); (2) Genomic DNA was extracted by silica column-based protocol (TopPURE® BLOOD DNA EXTRACTION KIT, ABT, Vietnam) according to manufacture; (3) 5 uL of DNA samples were added to 15 uL of PCR tube including 4 µL HOT FIREPOL® MULTIPLEX MIX READY TO LOAD, 3 µL 4 primers (Appendix 1), 8 ml double distilled water for tetra primer-amplification refractory mutation system-PCR (T-ARMS-PCR). Thermal cycling for PCR protocol includes 3 stages (C1000 Thermal Cycler), denaturation at 95 °C for 3 min, followed by annealing at 60 °C, and elongation at 72 °C (Each reaction was accompanied by a DNA-free negative control to control for contamination); (4) Select a purified and representative genotype from an electrophoresed PCR product sample, and perform a gene sequencing reaction using the Sanger method. Including 3 types of AGTR1 A1166C genetic polymorphism: AA, AC, and CC.
Study outcome
The primary outcome of finding out the prevalence of the AGTR1 A1166C gene variant as well as determining whether these variants are associated with all-cause mortality and heart failure in the Vietnamese population. The secondary outcome for assessing the relationship of AGTR1 A1166C gene polymorphisms with risk factors and severe hospitalized admission. Thereby contributing to the novelty perspective and clinical management strategy of AMI patients with AGTR1 A1166C gene variants.
Statistical analysis
SPSS 22.0 software for Windows (IBM, Armonk, USA) was used to process the study data. The normal distribution of continuous variables was determined based on the Kolmogorov-Smirnov test. Continuous variables were presented as mean ± standard deviation if normally distributed or median (interquartile range) if not normally distributed. Categorical variables were expressed as frequencies (percentages). Hardy-Weinberg equilibrium was evaluated by the Chi-squared test. The baseline characteristics of AA, AC, and CC genotype groups were compared using the Chi- squared or Fisher exact tests for categorical variables and the ANOVA or the Kruskal–Wallis rank tests for continuous variables depending on data distribution. The differences between the factors in the deceased group and the survivor group were assessed using the Chi-squared or Fisher exact tests for categorical variables and the Student t test or Mann–Whitney U test for continuous variables depending on the variables’ standard distribution. The association between AGTR1 A1166C genetic polymorphism and 6-month all-cause mortality, heart failure was analyzed using the Cox regression model in the dominant (AA versus AC and CC). The Cox regression model and survival curves were estimated using the Kaplan–Meier method and compared using a log-rank test and hazard ratio (HR) of confounding factors. Confounding factors added to the Cox regression model were age ≥ 60, smoking, T2DM, hypertension, dyslipidemia, GRACE > median (152 scores), LVEF ≤ 40%, Killip grade 4, and NYHA grade IV. The results of tests were considered statistically significant when the p value ≤ 0.05.
Results
Baseline characteristics
There were 101 patients diagnosed with AMI recruited in the study (Figure 1). The most common genotype was AA with 88.1%, AC at 10.9%, and CC at 1%. Males accounted for 54.5%, and the mean age was 65.7 ± 12.4 years. The most common risk factor was hypertension (81.2%), dyslipidemia was 56.4%, and the least common was a family history of premature coronary artery disease (5%). The clinical form of non-ST-elevation acute MI has almost the same rate as that of acute ST-elevation MI (51.5% vs 48.5%). Most patients with acute MI have grade I according to the Killip classification (63.4%), and Killip 4 accounts for 18.8%. 90 patients (89.1%) had increased high sensitive Troponin T concentration at the time of admission ≥ 0.053 ng/mL. 12 cases (11.9%) with LVEF decreased ≤ 40% and 67 cases (66.3%) had preserved LVEF ≥ 50%. All of the baseline characteristics and tests were not different between groups of genotype (Table 1).
Baseline characteristics and AGTR1 A1166C genotypes distribution of the population.

CAD: coronary artery disease; T2DM: Type 2 diabetes mellitus; GRACE: Global Registry of Acute Coronary Events; NYHA: New York Heart Association; LVEF: left ventricular ejection fraction; hs: high sensitive; SD: standard deviation; IQR: Interquartile range; STEMI: ST-segment elevation myocardial infarction.
= Chi-squared test.
= Fisher's exact test.
= Independent Samples T test.
= Mann-Whitney U test.
Relationship assessment of AGTR1 A1166C gene polymorphisms with risk factors and severe hospitalized admission
The hospital admission GRACE score with 167.98 ± 60.81 (Table 1). Figure 2 shows the changes in GRACE score during the hospitalization period between groups of genotypes indicating the relation of severe conditions. There was no difference in GRACE score at admission, discharge, and between groups of genotypes.

Study population flowchart. DNA = Deoxyribonucleic acid, AMI = Acute Myocardial Infarction.

Stratification of mortality risk according to GRACE score within genotype population. GRACE = Global Registry of Acute Coronary Events, PCI: Percutaneous coronary intervention.
Coronary artery disease risk factors were analyzed with a univariable logistic analysis model as shown in Figure 3(a), majority trend of OR > 1 but the data was found no significance (p > 0.05). In addition, the severe hospitalized admissions were also assessed through the proportion of severe GRACE, in-hospital death, Killip score ≥ 2, STEMI, and combining factors (for patients who meet all of the conditions) analyzed according to the dominant genetic model AA versus AC, and CC. Overall, OR > 1 with all of the factors indicated the trend in AC and CC gene polymorphisms (Figure 3(b)). However, there was no statistically significant.
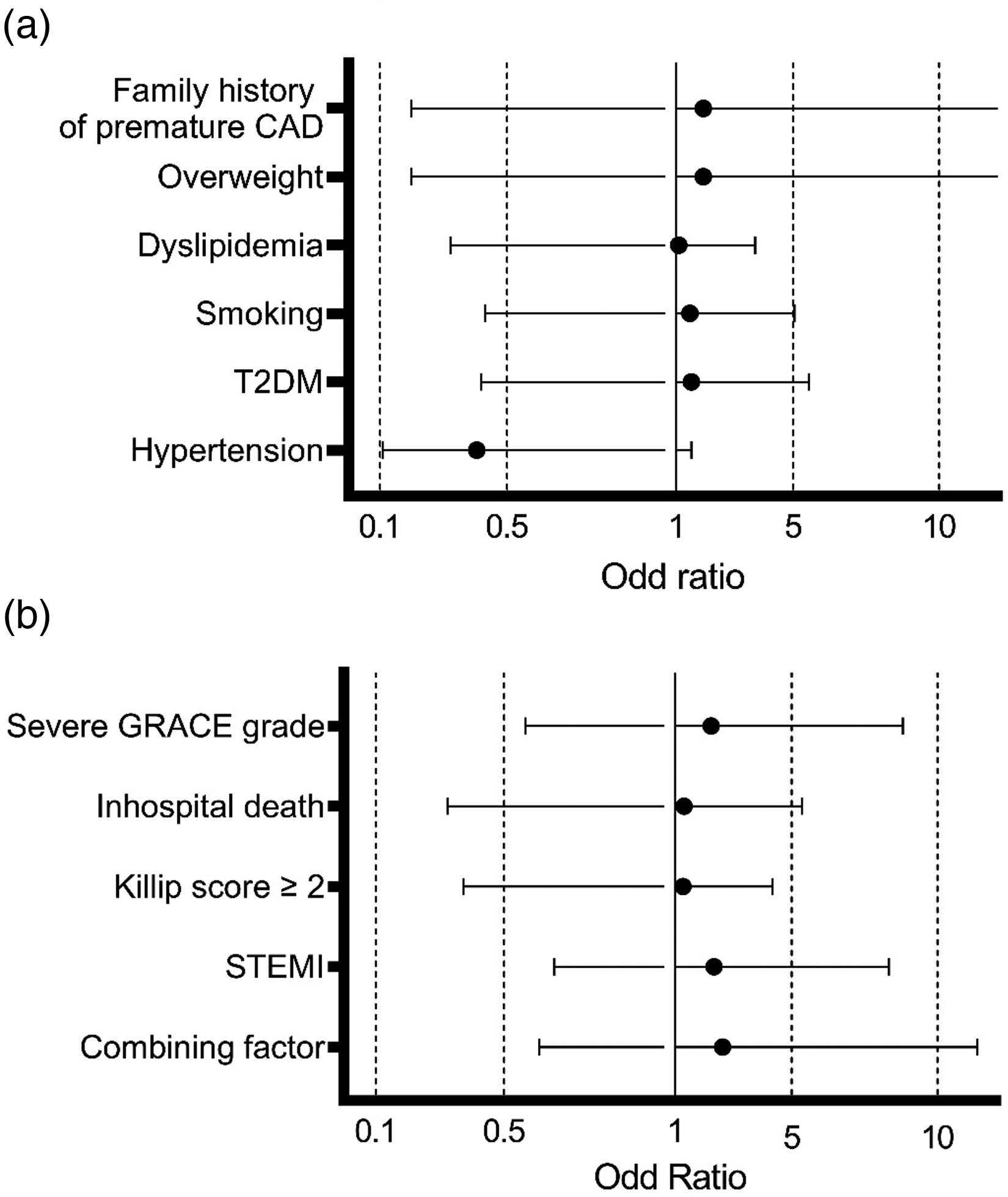
Association between AGTR1 A1166C with (a) coronary artery disease risk factors and (b) severe hospitalized admission in the AA versus AC and CC dominant genetic model. CAD = Coronary artery disease, T2DM = Type 2 diabetes mellitus, GRACE = Global Registry of Acute Coronary Events, STEMI = ST-segment elevation myocardial infarction, Combining factors including = GRACE severe, in-hospital death, Killip ≥ 2, and STEMI.
Survival analysis of 6-month all-cause mortality and heart failure of AGTR1 A1166C gene polymorphisms in acute myocardial infarction patients
Through univariate Cox regression analysis, the AGTR1 A1166C gene variant was statistically significantly associated with all-cause mortality within 6 months in patients after AMI in the dominant genetic model AC and CC compared to AA (Figure 4). 6-month survival analysis through Kaplan-Meier curves showed a trend with lower cumulative survival of the AC and CC genotype group with p = 0.02 (Figure 4(a)). Cox regression multivariate correction for confounding factors also showed the increasing trend of mortality in AGTR1 A1166C gene polymorphism (Figure 4(b)). After 6 months of follow-up, Kaplan-Meier curves (Figure 4(c)) and Cox regression model (Figure 4D) showed a slightly higher proportion of HF in the AC and CC genotype populations. However, the data showed no significance (Figure 4(c) and (d)).

Kaplan-Meier curves in the genotype patient group AC and CC compared to AA with (a, c) Non-correction for confounding factors of (a) mortality and (c) heart failure, (b, d) Multivariate correction for confounding factors with Cox regression of (b) mortality and (d) Heart failure. HF = Heart failure.
Multivariable analysis of the association between these two genetic groups and 6 months of all-cause mortality and heart failure through the Cox regression model. Figure 5 showed NYHA grade IV, LVEF ≤ 40, and AGTR1 A1166C gene polymorphism related to mortality proportion after 6 months of AMI, with hazard ratio was 27.40 (95% CI = 2.82–265.60, p = 0.004), 4.05 (95% CI = 1.37–6.63, p = 0.012), 2.59 (95% CI = 1.00–6.74, p = 0.05), respectively. In addition, heart failure did not show statistically significant (p > 0.05) (Figure 5).
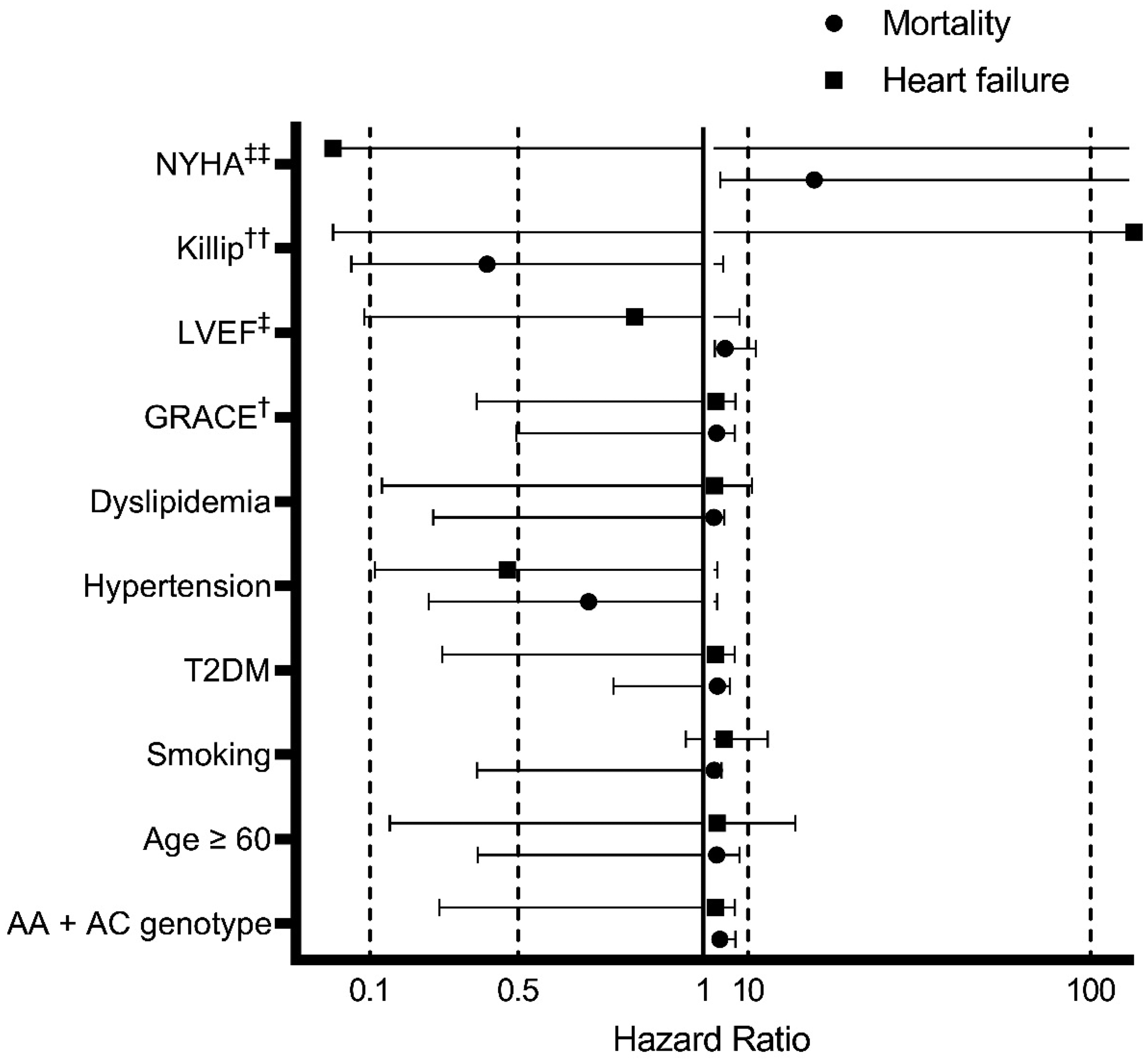
The hazard ratio of risk factors and comorbidities in mortality and heart failure were assessed through Cox regression in the AA versus AC and CC dominant genetic model. GRACE = Global Registry of Acute Coronary Events, NYHA = New York Heart Association, LVEF = left ventricular ejection fraction, T2DM = Type 2 diabetes mellitus, † = GRACE score ≥ median 152 scores, ‡ = LVEF ≤ 40%, †† = Killip grade 4, ‡‡: NYHA grade IV.
Discussion
Principal findings
Our study was conducted on 101 AMI patients in the Vietnam population (Figure 1). The prevalence of AGTR1 A1166C gene polymorphisms was 10.9% for the AC genotype and 1% for the CC genotype (Table 1). AGTR1 A1166C gene polymorphisms showed a trend in severe hospitalized admissions but were not statistically significant (Figures 2 and 3). 6-month survival analysis through Kaplan-Meier curves showed a trend with lower cumulative survival of the AC and CC genotype group with p = 0.02 (Figure 4(a)) and the outcomes were still significant with p = 0.05 when corrected for confounding factors (Figure 4(b)). Therefore, AGTR1 A1166C gene polymorphisms could be an important factor in increasing all-cause mortality in patients after AMI. The Cox regression model showed NYHA grade IV, LVEF ≤ 40, and AGTR1 A1166C gene polymorphism increased the mortality risk after 6-month follow-up of AMI patients with hazard ratio was 27.40 (95% CI = 2.82–265.60, p = 0.004), 4.05 (95% CI = 1.37–6.63, p = 0.012), 2.59 (95% CI = 1.00–6.74, p = 0.05), respectively.
Possible explanations and comparision with other studies
Our study outcomes showed the AC and CC genotypes of the AGTR1 A1166C gene variant were 10.9% and 1%, respectively (Table 1). The prevalence of the genotype in other studies was similar when compared to our outcome. Studies in Asian populations such as China, India, and Japan reported the similar prevalence: Masahiko Hara et al. (2013) 35 in the Japanese population AC was 14.99%, CC was 0.6%; Rupinder Kaur et al. (2012) 36 in the Indian population AC was 20.6%, CC was 0.6%; Shaoyong Su et al. (2014) 37 in the Chinese population AC was 20.6%, CC was 0.24%; Duy Cong Tran et al. (2024) 19 in the Vietnamese population AC was 9.2%, CC was 1.1%. Other populations show differences: Tiret L et al. (1994) in France 12 in France, the AC and CC genotype prevalence were higher with 46.65% and 7.17%, respectively of the population; Kee F et al. (2000) 14 in Ireland with 42.2% and 7.8%; Messias Antônio Araújo et al. (2005) 38 in Brazil with 35.5% and 10%; Peter Kruzliak et al. (2013) 39 in Slovakia with 43.92% and 23.89%; Sounira Mehri et al. (2011) 13 in Tunisia with 44.91%, and 18.64%. Therefore, Asian races and populations had found a significantly lower proportion of AGTR1 A1166C gene polymorphism when compared with the Caucasian race.
Stratification of mortality risk according to GRACE score also found no difference in GRACE score at admission, discharge, and between groups of genotypes (Figure 2). Our outcome did not find an association between the AGTR1 A1166C gene polymorphism risk factors and severe hospitalized admission in patients with AMI through univariable logistic analysis of the dominant genetic model AA versus AC and CC (Figure 3(a) and (b)). Sounira Mehri et al. (2011) in Tunisia, assessed 118 AMI patients and found that the genotypes were not related to factors such as age, gender, health care, diabetes, hypertension, and smoking. 13 Similarly, the study by Duy Cong Tran et al. (2024) also found the majority OR > 1 but no significant association between the gene and risk factors for CAD in 548 patients with AMI. 19 Yang Y et al. (2017) performed a meta-analysis of 41 case-control studies including 11,837 hypertensive patients and 11,020 controls to evaluate the association between AGTR1 A1166C polymorphism score and hypertension (OR = 1.21; 95% CI: 1.05–1.40; p = 0.009). 22 Messias Antônio Araújo et al. (2005) in Brazil conducted the cross study with 110 AMI patients and also found no association with OR = 1.35 (p > 0.05). 38 In general, the association between the AGTR1 A1166C gene variant and CAD risk factors is conflicting between studies. This association may be positive in subjects who have not had AMI, while studies in patients with AMI have mostly not detected this association. This could be explained by differences in genotype prevalence in the study populations, the high prevalence of CAD risk factors made the difference in distribution genotype. Therefore, AGTR1 A1166C gene polymorphism indicated the trend in the association with CAD risk factors and severe hospitalized admission. However, similar to other studies the association was not significant.
Our study found the AGTR1 A1166C gene polymorphism was statistically significantly associated with all-cause mortality within 6 months in patients after AMI in the dominant genetic model AC and CC compared to AA with p = 0.02 (Figure 4(a)). In addition, Cox regression multivariate correction for confounding factors also showed the increasing trend of mortality in AGTR1 A1166C gene polymorphism with p = 0.05 (Figure 4(b)). Although Kaplan-Meier curves and Cox regression models indicated a marginally elevated incidence of heart failure within the AC and CC genotype populations following a six-month follow-up period, the statistical analysis revealed no significance (Figure 4(c) and (d)). NYHA grade IV, LVEF ≤ 40, and AGTR1 A1166C gene polymorphism related to mortality proportion after 6 months of AMI (Figure 5). Peter Kruzliak et al. (2013) in Slovakia, have also shown the genotype to be a prognostic factor for mortality in patients with AMI with OR = 6.484, 95% CI = 1.036–40.598, p = 0.036. 39 In a large prospective study by Emmanuelle Filippi-Codaccioni et al. (2005), 970 AMI patients in France reported AGTR1 CC genotype was an independent predictor of cardiovascular death after AMI (RR = 2.8; 95% CI: 1.2–6.5; p < 0.021) others association factors such as age (RR = 2.8; 95% CI: 1.6–5.0; p < 0.001), hypercholesterolemia (RR = 2.1; 95% CI: 1, 2–3.7; p < 0.014) or reduced left ventricular ejection fraction (RR = 2.7; 95% CI: 1.5–4.8; p < 0.002). 40 In contrast with our study, Masahiko Hara et al. (2014) study of 8957 Japanese AMI patients found there was no statistical significance in the cumulative survival through Kaplan-Meier estimated (log-rank p = 0.484). The difference could be due to the time when the study was conducted from 1998 to 2008, the limitation of health management of AMI in the period could lead to bias. In addition, the study by Franco E et al. (2007) concluded that the AC genotype was associated with mortality (through Kaplan Meier curves), MI, and coronary revascularization in young Italian patients with AMI ≤ 45 years old during a follow-up period of 9 ± 4 years. However, the study did not correct for confounding factors in survival analysis. 26 Therefore, the AGTR1 A1166C gene polymorphism could associated with all-cause mortality within 6 months in patients after AMI.
Strengths and weaknesses of the study
Our study had a sample collection process designed, including, and excluding criteria, and the methods were clearly described and reproducible. The novelty of our study is that the outcome has given the reference of AGTR1 A1166C gene polymorphisms prevalence. In addition, the variant of the genotype of AC and CC plays an important factor in increasing all-cause mortality in patients after AMI, and the outcomes were still significant when corrected for confounding factors. Therefore, our study data and outcome could be strong evidence for further study in the field.
However, our study was conducted with marginal sample size and evaluated only in 1 hospital, which might lead to bias in national and area baseline characteristics. A multicenter study with the same or larger sample size is required to accurately embody the outcomes. In our research, with a follow-up period of 6 months, patient mortality between groups could change in a longer period of follow-up. Additionally, the combination of CYP2C19 gene polymorphism in the Vietnamese population could significantly affect the treatment outcomes which were not assessed by our study. 41 Therefore, a study with a more extended evaluation period is needed to give more substantial evidence for the conclusion of the association of AGTR1 A1166C gene polymorphisms with all-cause mortality.
Conclusion
The prevalence of AGTR1 A1166C genetic polymorphism in the population is small. The AGTR1 A1166C genotype of AC and CC plays an important factor in increasing all causes of 6-month mortality in patients after AMI, and the outcomes were still significant when corrected for confounding factors. In addition, the genotype is not associated with 6-month HF. However, further studies with longer follow-up periods and larger sample sizes are required to consider the prognostic role of the AGTR1 A1166C variant in patients with AMI.
Supplemental Material
sj-docx-1-jra-10.1177_14703203241312627 - Supplemental material for The prognostic value of AGTR1 A1166C gene polymorphism in all-cause mortality and heart failure in patients with acute myocardial infarction
Supplemental material, sj-docx-1-jra-10.1177_14703203241312627 for The prognostic value of AGTR1 A1166C gene polymorphism in all-cause mortality and heart failure in patients with acute myocardial infarction by An Viet Tran, Anh Phuong Nguyen, Phat Tan Tran, Khue Duy Nguyen, Thuan Tuan Nguyen, Bao Lam Thai Tran and Cao Thi Tai Nguyen in Journal of the Renin-Angiotensin-Aldosterone System
Supplemental Material
sj-xlsx-2-jra-10.1177_14703203241312627 - Supplemental material for The prognostic value of AGTR1 A1166C gene polymorphism in all-cause mortality and heart failure in patients with acute myocardial infarction
Supplemental material, sj-xlsx-2-jra-10.1177_14703203241312627 for The prognostic value of AGTR1 A1166C gene polymorphism in all-cause mortality and heart failure in patients with acute myocardial infarction by An Viet Tran, Anh Phuong Nguyen, Phat Tan Tran, Khue Duy Nguyen, Thuan Tuan Nguyen, Bao Lam Thai Tran and Cao Thi Tai Nguyen in Journal of the Renin-Angiotensin-Aldosterone System
Supplemental Material
sj-docx-3-jra-10.1177_14703203241312627 - Supplemental material for The prognostic value of AGTR1 A1166C gene polymorphism in all-cause mortality and heart failure in patients with acute myocardial infarction
Supplemental material, sj-docx-3-jra-10.1177_14703203241312627 for The prognostic value of AGTR1 A1166C gene polymorphism in all-cause mortality and heart failure in patients with acute myocardial infarction by An Viet Tran, Anh Phuong Nguyen, Phat Tan Tran, Khue Duy Nguyen, Thuan Tuan Nguyen, Bao Lam Thai Tran and Cao Thi Tai Nguyen in Journal of the Renin-Angiotensin-Aldosterone System
Footnotes
Acknowledgments
The authors are grateful to the physicians, and patients who participated in the study.
Data accessibility statement
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and informed consent statements
The Ethics Committee in Biomedical Research of the University of Medicine and Pharmacy at Can Tho City approved this research with the approval number: 22.270.HV/PCT-HDDD. Enrolled patients had to sign a consent form to participate in the study, and all of the information of patients was kept confidential.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
