Abstract
Objective:
The previous studies on angiotensin converting enzyme (ACE) insertion/deletion (I/D) genetic polymorphism and glioma risk were inconsistent. Therefore, we performed a meta-analysis to assess the association between ACE I/D polymorphisms and glioma risk.
Methods and Results:
In total, four populations (1110 cases and 1335 controls) on ACE I/D polymorphism were included. Overall, the meta-analysis demonstrated no significant association between ACE I/D polymorphism and glioma risk. In addition, the analysis of the association of ACE I/D polymorphism and clinical grade also showed no significant association.
Conclusion:
Our meta-analysis didn’t find a significant association between ACE I/D polymorphism glioma risk. However, further studies with larger sample size and more ethnic groups are required to confirm the results.
Introduction
Glioma is the common malignant disease of the central nervous system with a very poor prognosis. Over the past 40 years, the median survival has only slightly improved and <50% of patients survived 5 years post diagnosis of glioma. 1 Of note, having a first-degree family member’s history of brain tumor has been found to increase risk approximately two-fold. 2 However, the etiology and pathogenesis of glioma is not fully understood. Although several occupations, environmental carcinogens and diet have been reported to elevate glioma risk, studies in humans showed no definitive association with occupational or environmental exposure and the risk of glioma. Genetic factors such as single nucleotide polymorphisms (SNPs) were recently found to be linked to glioma risk. 3
Angiotensin converting enzyme (ACE) is a member of the renin-angiotensin system (RAS), with the main function of converting angiotensin I (Ang I) into angiotensin II (Ang II). Studies have shown that ACE expression is significantly increased in many tumors, involved in tumor angiogenesis, cell growth and metastasis. 4 There is also substantial evidence indicating that Ang II plays a critical role in the metastasis of various cancers. Furthermore, a number of basic and meta-analytic studies have shown that ACE inhibitors reduce the metastatic potential of tumors. 5 Previous studies have revealed that the ACE serum level and ACE activity are governed by gene polymorphisms of ACE insertion/deletion (I/D). 6 In the recent years, ACE I/D genetic polymorphisms has been concerned in the susceptibility of glioma.7–10 However, the previous reports were inconsistent. Therefore, our present study aimed to perform a meta-analysis to assess the association between ACE I/D polymorphisms and glioma susceptibility.
Materials and methods
Search strategy
To search the literatures for all reported observational studies of associations between ACE I/D polymorphisms and glioma risk, we used the following keyword: (glioma, or glioblastoma, or brain tumor), (polymorphism, or single-nucleotide polymorphism, or SNP, or variant) and (angiotensin converting enzyme or ACE). First, two investigators (M.L Sun and Y.P Sun) independently search the MEDLINE, Pubmed, Web of Science, EMBASE, CENTRAL Library, Google scholar database for articles published before Mar 2020. CNKI and VIP database were also searched for researches published in Chinese. Furthermore, references of original articles and review articles are also screened.
Inclusion criteria
The studies included in the meta-analysis must meet all the following criteria: (1) evaluating the association between ACE I/D polymorphism and glioma risk; (2) using case-control design; (3) sufficient data for genotype frequency of II, ID and DD in cases and controls.
Data extraction
All data from eligible reports were independently abstracted by two independent investigators (M.L Sun and Y.P Sun) with the standard protocol. The following information was extracted from each study: (1) first author’s name; (2) year of publication; (3) study design; (4) country origin; (5) frequency of genotype numbers in cases and controls, (6) baseline characteristics, and (7) tumor type and stage were determined according to the world health organization (WHO) criteria. 11
Statistical analyses
All the analyses were performed using the Review Manager 5.3 package (The Cochrane Collaboration, Oxford, UK). We evaluated the association between ACE I/D polymorphism and glioma risk by calculating pooled ORs and 95% confidence intervals (CIs). Z-test was used to determine the significance of pooled ORs and p < 0.05 was considered statistically significant. Heterogeneity between studies was evaluated by Cochran’s Q with a significance level of 0.10. If heterogeneity existed (p < 0.10), a random-effect model was used. Otherwise, a fixed effect model was used. Publication bias was not assessed in this study because the number of included studies was less than 10 studies (www.cochranehandbook.org).
Results
Characteristics of the included studies
The literature search and selection approach were showed in Figure 1. Initially, a total of 78 studies were screened. 25 studies were excluded because of duplications. After reading titles and abstracts, 32 studies were excluded. After reading the full-text, 17 studies, including 2 reviews, 11 without complete context, 2 used repeated population and 2 about basic research, were excluded.

Flow chart for selection of studies.
Thus, four studies7–10 including 1110 cases and 1335 controls on ACE I/D polymorphism and glioma risk were included in this meta-analysis. All the studies were case-control studies and the distributions of the genotypes in the control populations were consistent with HWE in all the four studies (Table 1).
The characteristics of eligible studies considered in the meta-analysis.

Quantitative analysis results
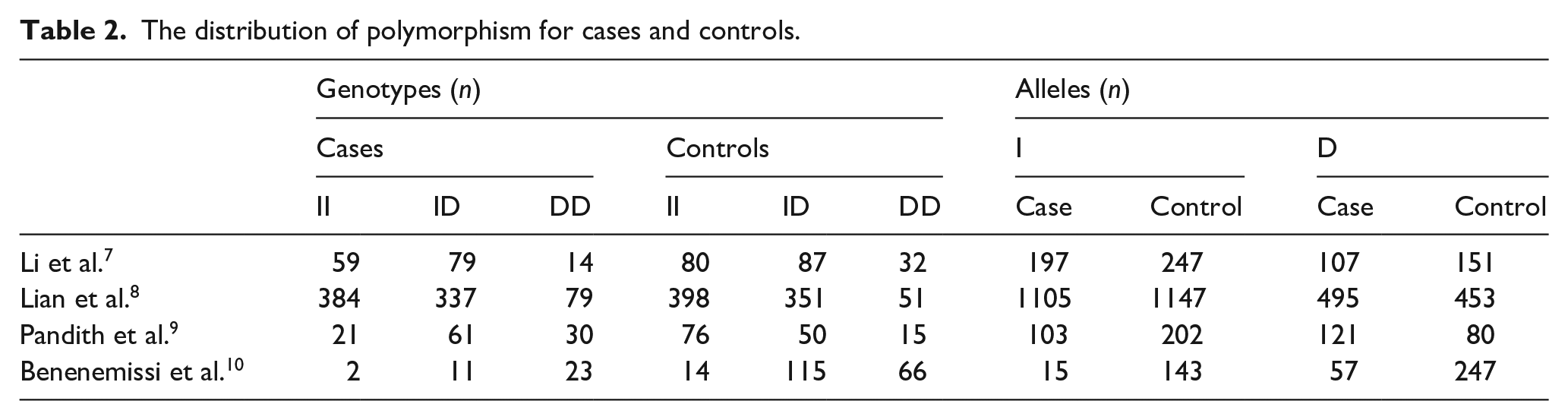
The summary of ACE I/D polymorphism in glioma and control groups was described in Table 2. For the association between ACE I/D polymorphism and glioma, four studies including 1110 cases and 2335 controls were included for the analysis. Overall, the meta-analyses demonstrated no significant association between ACE I/D polymorphism and glioma risk. As shown in Figure 2(a), compared with ACE ID/II genotype, subjects with DD genotype showed a non-significant risk for glioma (OR 1.96; 95%CI 0.73 to 5.27; p = 0.18). Compared with II genotype, ACE ID/II genotype carriers also did not show a significantly increased risk for glioma (OR 1.66; 95%CI 0.81 to 3.38; p = 0.16) (Figure 2(b)). In addition, we analyzed the risk for glioma between DD genotype and I allele carriers (ID/II), and we also did not find statistical association (OR 1.71; 95%CI 0.82 to 3.56; p = 0.15) (Figure 2(c)).
The distribution of polymorphism for cases and controls.

Forest plot of studies evaluating the relationship between ACE I/D polymorphism and glioma risk: (a) DD vs II; (b) DD/ID vs II; (c) DD vs ID/II.
The summary of ACE I/D polymorphism in different glioma grade subgroups (III-IV VS. I-II) was described in Table 3. For the association between ACE I /D polymorphism and glioma grade, 3 studies consisted of 630 cases of grade III-IV and 482 cases of grade I-II were analyzed. We assessed the risk for high glioma grade (III-IV VS. I-II) between D allele carriers and DD homozygotes, which also showed no significant association (OR 0.81; 95%CI 0.41 to 1.59; p = 0.54) (Figure 3).
The distribution of polymorphism for clinical grade of glioma.


Forest plot of studies evaluating the relationship between ACE I/D polymorphism and high grade (III-IV) glioma risk.
Discussion
Glioma accounts for approximately 70% to 80% of all brain tumors and more than 50% of patients suffering from glioma survive for less than 5 years after diagnosis. 12 However, the etiology and risk factors were not thoroughly clear. Studies in humans showed no definitive association with occupational or environmental exposure and the risk of glioma. Therefore, some researchers have focused on the association between genetic polymorphisms and glioma risk and found a series of susceptible genes. 13 To our best knowledge, our present study is the first meta-analysis to evaluate the association between ACE I/D polymorphism and glioma risk. The results suggested that ACE I/D polymorphism might not be involved in the development of glioma.
The RAS is an important system for the regulation of cardiovascular function and plays an important role in maintaining blood pressure and stabilizing the internal environment of electrolytes. 14 Ang II, an important member of RAS, which is produced by angiotensin I under the action of ACE, not only regulates the stability of the internal environment, but also participates in regulating the growth, growth and migration of tumor cells. Recently, Ang II has been found to promote tumor cell proliferation, tumor blood vessel formation, and inhibit tumor cell differentiation. 4 This suggests that inhibiting the production of Ang II or blocking its effect is expected to be a new measure for the treatment of malignant tumors. ACE catalyzes the conversion of Ang I to Ang II and is therefore thought to be involved in tumor formation. Animal experiments have confirmed that the use of ACE inhibitors can slow tumor growth and tumor blood vessel formation. 15 In view of this, ACE inhibitors are expected to become new antitumor drugs for the prevention and treatment of tumors.
Differential levels of ACE in the plasma of various tumor patients affects the biological activity of the tumor. Studies found that the ACE I/D polymorphism affected the plasma ACE concentrations. In patients with D-allele carriers, the plasma ACE activity levels and concentrations were higher than those in I-allele carriers. 16 Recently, Pandith et al. 9 reported that ACE DD genotypes were highly presented in glioma cases 26.8% versus 10.6% in controls and conferred 5-fold risk for predisposition in glioma cases. Lian et al. 8 also found that glioma cases had a significantly higher frequency of DD genotype than controls. Benenemissi et al. 10 reported that ACE DD genotype was highly presented in glioma cases than controls and conferred 3.64-fold risk for predisposition in glioma cases compared with ID genotype. However, Li et al. 7 reported that the ID genotype was associated with an increased risk of glioma compared with DD genotype. All the above results demonstrated that ACE I/E polymorphism may play an important role in tumor diagnosis and treatment. This prompted us to try to clarify the relationship between ACE I/D polymorphism and glioma risk. In our current analysis, we first analyzed the DD genotype and glioma risk, and no significant association was found. Furthermore, we compared the II homozygotes with ID/DD genotype carriers and we also did not find significant association. To obtain more information, we assessed the risk for high glioma grade (III-IV vs I-II) between D allele carriers and DD homozygotes, which also showed no significant association. All these results suggested that ACE I/D polymorphisms might not be involved in glioma progression.
Several limitations should be mentioned. First, because few studies are available in this field, the populations were homogeneous and publication bias was not assessed in this study; Second, most studies included in this meta-analysis were from Asia and only 1 study was from Algerian. Thus, we did not investigate the ethnic difference. Third, a single gene mutation may have a limited effect on cancer. Whether other known gene polymorphisms of the renin-angiotensin system also affect gliomas, and the interaction between different genes, should also be considered.
Overall, although the current meta-analysis didn’t find a significant association between ACE I/D polymorphism and glioma risk, further large-scale, multi center, controlled, prospective investigations are needed to confirm our results.
Footnotes
Acknowledgements
We thank Hongyan Yang and Guohong Zu for their help in revising the paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants of Science and Technology Plan of Jinan Health and Family Planning Commission (No. 2018-1-01) and Jinan City’s 2019 Science and Technology Development Plan (No. 201907031).
