Abstract
Objective:
Previous case-control studies on the relation between angiotensin-converting enzyme (ACE) gene insertion/deletion (I/D) polymorphism and breast cancer did not reach the same conclusion. In the present study, we aimed to further evaluate the relationship between the ACE gene I/D polymorphism and breast cancer.
Methods:
We selected 13 case-control studies related to ACE gene I/D polymorphism and breast cancer by searching PubMed, EMBase, Chinese Biomedical Literature Database, Chinese CNKI, and Wanfang database. To test the heterogeneity between each study, we utilized the Q-test and I2 test. To merge the odds ratio (OR) and 95% confidence interval (CI), we utilized the random effects model during the analyses.
Results:
The present study included 1997 patients with breast cancer and 8404 cancer-free control subjects. By meta-analysis, we did not find any association of ACE gene I/D polymorphism with breast cancer in different model (DD vs (ID+II): OR=1.28, 95% CI (0.90–1.81), p=0.16; II vs (ID+DD): OR=0.99, 95% CI (0.81–1.21), p=0.93; D allele vs I allele: OR=1.15, 95% CI (0.94–1.41), p=0.18).
Conclusion:
We concluded that ACE gene I/D polymorphism was not associated with breast cancer.
Introduction
Breast cancer is one of the most common malignant tumors in women. 1 During the past decade there has been increasing scientific interest in understanding the complex relationship between breast cancer and renin–angiotensin system (RAS).2–4 The RAS plays an important role in blood pressure and cardiovascular homeostasis.5,6 However, recently published data indicated that angiotensin II, the main biologically active peptide of RAS, contributed to breast cancer development and progression.7,8 The production of angiotensin II is regulated by angiotensin-converting enzyme (ACE) which the serum levels is governed by genetic variation at the ACE locus. Recently, breast cancer is considered as a complex disease resulting from interaction between genetic background and environmental factors.9–12 Previous studies indicated that some certain genetic polymorphisms were recognized to be associated with the risk of breast cancer.9–15 Among these genes, ACE gene polymorphism was considered as a risk factor of breast cancer in several published papers,16–23 however, the results of these studies were inconsistent. Namazi et al. and Alves Corrêa et al. found ACE genetic polymorphism was associated with breast cancer risk.16,17 And Yaren et al. and González-Zuloeta et al. also confirmed the previous studies.19,20 However, Pei et al. and Zhang et al. did not find any association between ACE gene I/D polymorphism and breast risk.24,25 Although there have been published meta-analyses which suggested that ACE I/D polymorphism may not be a genetic risk factor for breast cancer,24–26 there are many further studies27,28 which have been published. Therefore, we collected all published case-control studies to perform an update meta-analysis to further explore the relation between ACE gene polymorphism and breast cancer.
Materials and methods
Literature collection and screening
To identify all the articles that explored the association of ACE polymorphisms with breast cancer risk, we conducted a computerized literature search of PubMed, EMBase, Chinese Biomedical Literature Database, Chinese China National Knowledge Infrastructure (CNKI), and Wanfang database using the terms “breast cancer (Mesh),” “angiotensin-converting enzyme” or “ACE,” “polymorphism”, or Single nucleotide polymorphism “SNP”, or “genotype”, or “insertion/deletion”, or “I/D”, or “D/I”, or “D/D” or “I/I”, without any restriction on language or publication year. By means of online retrieval and literature review, references obtained using the above-mentioned databases were reviewed again to ensure that no relevant studies are missed.
The inclusion criteria for the present study are as follows: (a) independently published case-control or cohort studies on the relation between ACE polymorphism and breast cancer; (b) similar themes and methods; and (c) sufficient information upon genotype counts. The studies were excluded if relevant data were not available or there was heterogeneity of gene polymorphism in the control population.
Quality assessment and data extraction
Two reviewers (Xiao-Lei Li and Hai-Ou Qu) independently evaluated the studies and extracted the data using a standard approach according to the above-mentioned inclusion criteria. Discrepancies were resolved through discussion. We utilized the Cochrane Handbook 5.2 quality evaluation criteria to assess the methodological quality of included studies. Those studies that have been reported repeatedly or were poor-quality studies were excluded from included literatures.
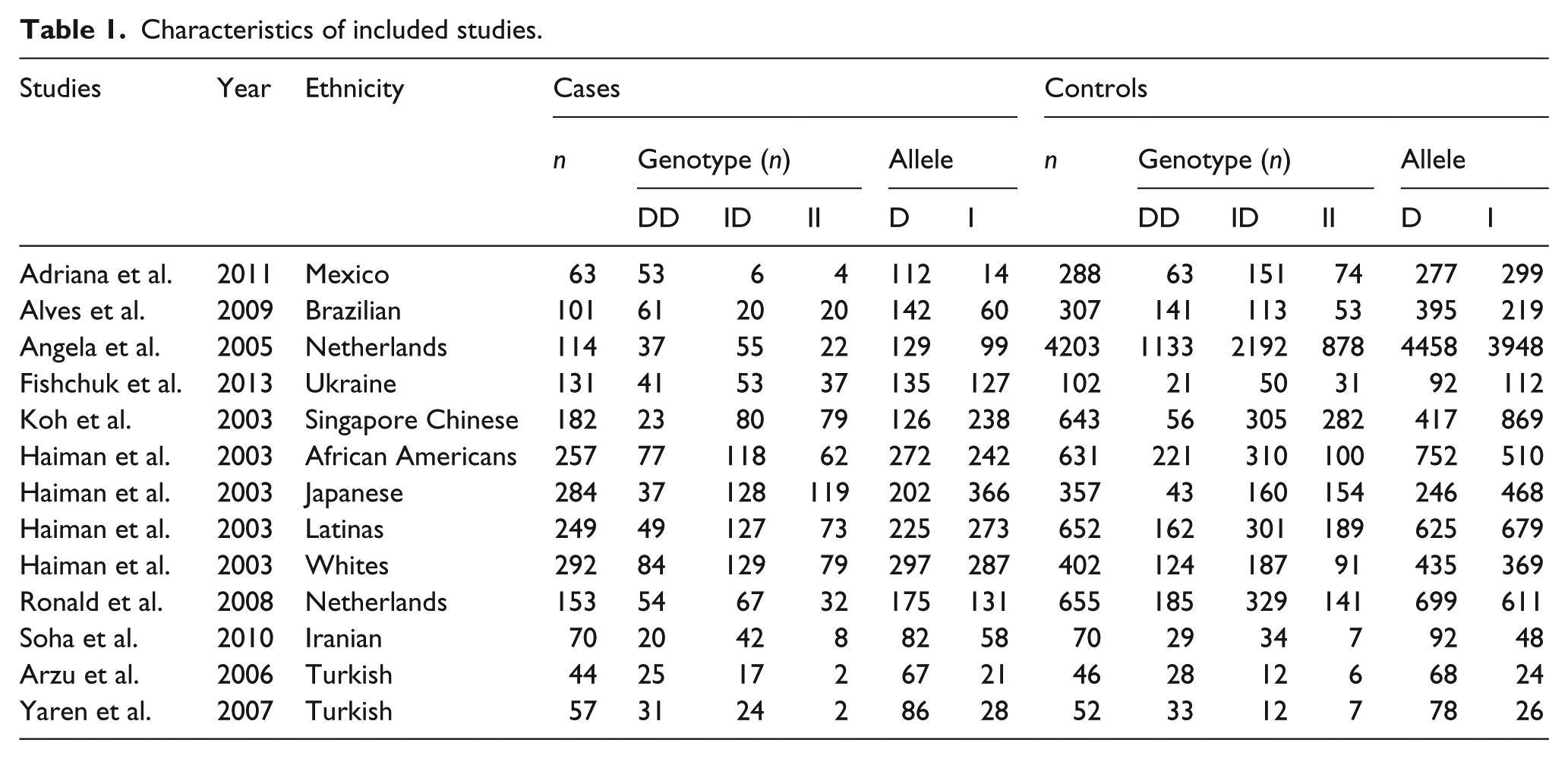
For each study we abstracted the first author’s last name, year of publication, ethnicity of participants, numbers of cases and controls, frequency of insertion or deletion genotypes. Hardy–Weinberg equilibrium was assessed using the χ2 test.
Statistical analysis
We performed the present meta-analysis utilizing RevMan 5.2 software which provided by the Cochrane Collaboration. We utilized Q-test and I2 test to examine the heterogeneity between each study. We utilized the odds ratio (OR) for efficacy analysis statistics. In the present study, we selected the random effects model to merge the OR. Analysis of sensitivity includes the difference of point estimation and confidence intervals (CIs) of the combined effects value of different models to observe whether it changes the result. To test the publication bias, we utilized RevMan 5.2 statistical software to make the funnel plot. A value of p<0.05 was considered as a significant difference.
Results
Study identification
As shown in Figure 1, 116 studies were preliminarily detected; 91 studies were excluded because of having no RAS gene polymorphisms, only gene expression analysis, and not being relevant to breast cancer. Fifteen studies were further excluded because of only being a review paper, no ACE I/D genotype, and duplicated publications. Therefore, a total of 10 studies16–23,27,28 including 13 independent case-control studies, with a total of 1997 patients with breast cancer and 8408 cancer-free control subjects, were included in this research, see Table 1.

Flow diagram of study identification. ACE: angiotensin-converting enzyme; I/D: insertion/deletion; RAS: renin–angiotensin system.
Characteristics of included studies.
Quantitative synthesis
The result of meta-analysis of the association between breast cancer and ACE gene polymorphism in 13 case-control studies is shown in Figures 2, 3, and 4. The heterogeneity test of the various studies revealed heterogeneous results (DD vs (ID+II): I2=86%, p<0.0001; D allele vs I allele: I2=83%, p<0.0001; II vs (ID+DD): I2=52%, p<0.01); therefore, we used the random effects model in the analysis. No significant association between ACE I/D polymorphism and susceptibility to breast cancer was identified in any of the genetic models (DD vs (ID+II): OR=1.28, 95% CI 0.90–1.81, p=0.16; II vs (ID+DD): OR=0.99, 95% CI 0.81–1.21, p=0.93; D allele vs I allele: OR=1.15, 95% CI 0.94–1.41, p=0.18).
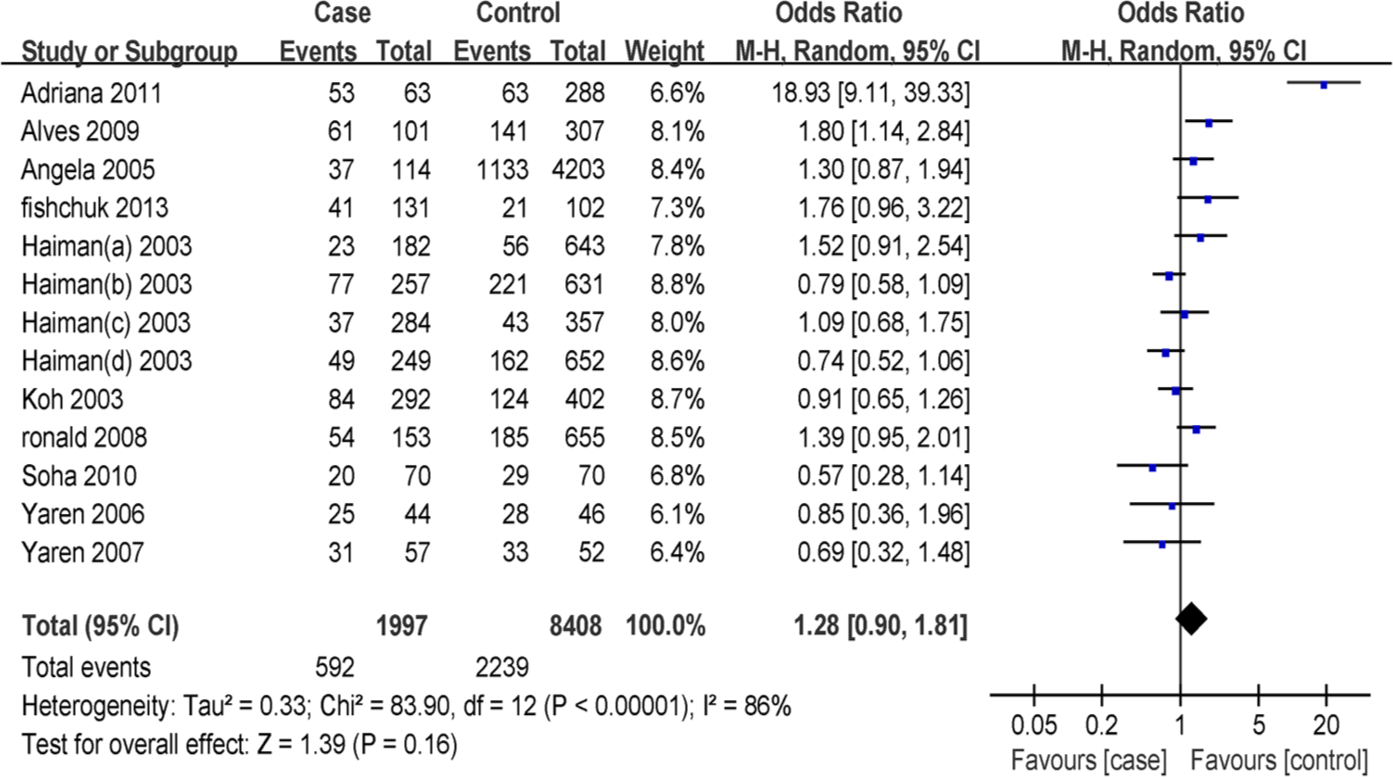
Forest plot of breast cancer risk associated with angiotensin-converting enzyme (ACE) insertion/deletion (I/D) polymorphism (DD vs ID+II). The squares and horizontal lines correspond to the study-specific odds ratio (OR) and 95% confidence interval (CI), respectively. The area of the squares reflects the study-specific weight. The diamond represents the pooled results of OR and 95% CI. In this analysis, the random-effects model was used.

Forest plot of breast cancer risk associated with angiotensin-converting enzyme (ACE) insertion/deletion (I/D) polymorphism (D allele vs I allele). The squares and horizontal lines correspond to the study-specific odds ratio (OR) and 95% confidence interval (CI), respectively. The area of the squares reflects the study-specific weight. The diamond represents the pooled results of OR and 95% CI. In this analysis, the random-effects model was used.

Forest plot of breast cancer risk associated with angiotensin-converting enzyme (ACE) insertion/deletion (I/D) polymorphism (II vs ID+DD).The squares and horizontal lines correspond to the study-specific odds ratio (OR) and 95% confidence interval (CI), respectively. The area of the squares reflects the study-specific weight. The diamond represents the pooled results of OR and 95% CI. In this analysis, the random-effects model was used.
Publication bias analysis
We utilized RevMan 5.0 software to analyze the publication bias; the funnel plot (Figure 5) shows that the points are evenly distributed and symmetrical, and most of the points are within the 95% CI. And the shape of funnel plots showed no obvious asymmetry and the result of Egger’s test did not show statistical evidence for bias. This indicates that there is no publication bias, and the result of the study is credible.
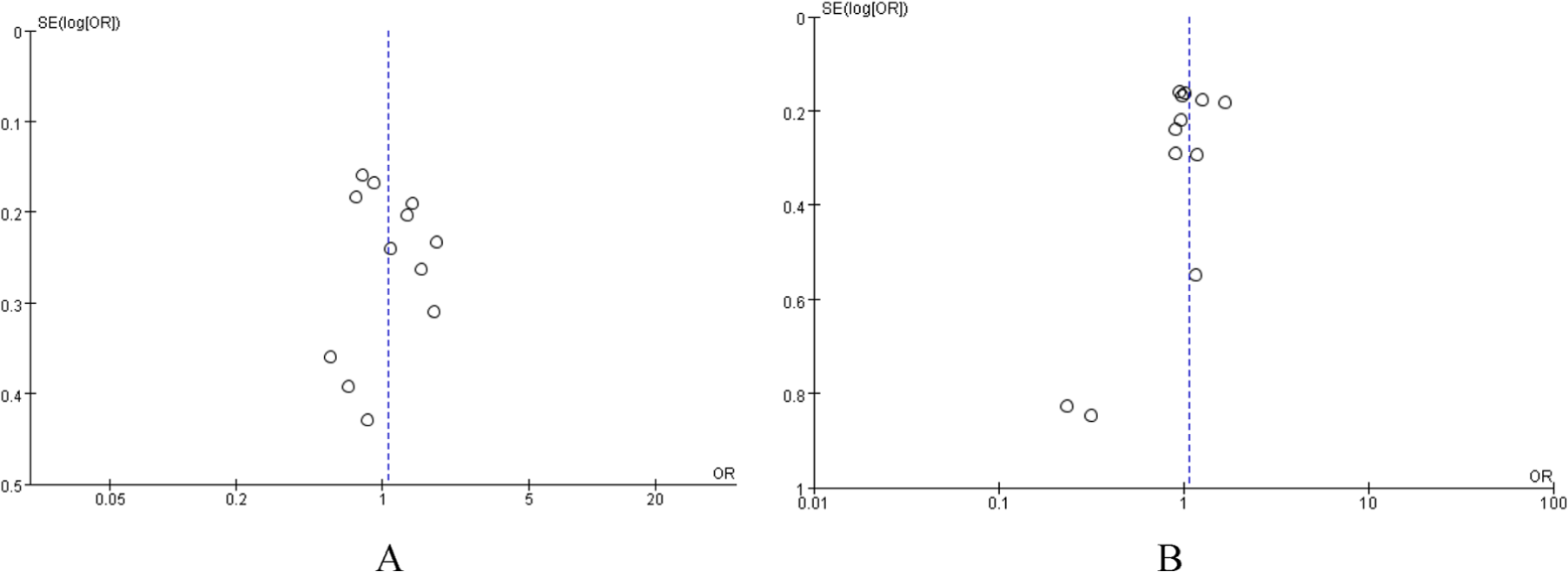
Begg’s funnel plot for publication bias test. Each circle denotes an independent study for the indicated association. Log[odds ratio (OR)], natural logarithm of OR. Horizontal line stands for mean effect size. (A) DD vs ID+ II, (B) II vs ID+ DD.
Sensitivity analysis
We deleted one single study from the overall pooled analysis each time to check the influence of the removed data set to the overall ORs. The pooled ORs and 95% CIs were not significantly altered when any part of the study was omitted, which indicated that any single study had little impact on the overall ORs.
Discussions
In the present study, we performed a meta-analysis to evaluate the association of ACE gene I/D polymorphism with breast cancer. We did not find any association between ACE gene polymorphism and breast cancer.
ACE not only has an important role in the regulation of blood pressure and cardiovascular homeostasis,5,6 but also influences tumor cell proliferation, migration, angiogenesis and inflammation.7,8 Previous studies indicated that ACE inhibitor could lower the breast cancer risk. 29 In recent years, more and more publications suggested that the ACE I/D polymorphism is related to breast cancer risk.16–23 Koh et al. 23 were the first to report the association of ACE I/D polymorphism with breast cancer. From then on, there have been many further studies published. However, these studies used a small number of samples. The results may reflect chance observations rather than true associations. One published meta-analysis including 10 case-control studies did not show any significant association between ACE I/D polymorphism and breast cancer risk. 24 However, there were several studies which have not been included in this meta-analysis.
The present study which included 13 independent case-control studies has shown that ACE gene I/D polymorphism was not associated with the susceptibility of breast cancer. However, there is still a need for further research and screening of etiological relations between the functional polymorphism loci of the ACE gene and the susceptibility of breast cancer.
Several limitations should be considered when interpreting these results. First, all studies involved in our study are of different ethnicity; second, the different research methods may increase the heterogeneity of these studies; third, a small sample of some included case-control studies may reduce the test power. To some extent, this reduced the reliability and comprehensiveness of the results of this systemic evaluation.
In conclusion, the present study suggested that there is no association between ACE gene I/D polymorphism and breast cancer.
Footnotes
Conflict of interest
The author declares that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
