Abstract
Objective:
This study aimed to investigate the effect of anti-osteoporosis therapy on plasma aldosterone concentration (PAC), plasma renin concentration (PRC) and the aldosterone/renin ratio (ARR) in patients with postmenopausal osteoporosis.
Methods:
In 60 patients with postmenopausal osteoporosis, bone mineral density (BMD), PAC and PRC were measured before and after treatment with alendronate (70 mg/week, n=22) or recombinant human parathyroid hormone (20 μg/day, n=35) for 48 weeks.
Results:
PAC was negatively correlated with the T-score of lumbar spine BMD and femoral neck BMD (lumbar r=−0.386, p<0.01; femoral neck r=−0.262, p<0.05). With the improvement in lumbar BMD after anti-osteoporosis treatment (T-score −3.4±0.5 vs. –3.1 ±0.4, p<0.0001), PAC decreased from 182.8±53.2 to 143.7±68.6 pg/mL (p<0.0001), PRC increased from 7.8±11.6 to 39.2±50.0 μIU/mL (p<0.0001) and the ARR decreased from 74.8±75.2 to 13.1±17.1 pg/μIU (p<0.0001). At baseline, 58% (35/60) of the patients had an ARR >37 pg/μIU, and the proportion decreased to 8% (5/57) after treatment.
Conclusion:
Treatment with alendronate or parathyroid hormone causes decreased PAC and increased PRC, resulting in a decreased ARR in postmenopausal women with osteoporosis.
Keywords
Introduction
Osteoporosis is a disease characterised by low bone mass, destruction of bone microstructure and increased risk of fracture. Osteoporosis is a form of degenerative disease that increases in prevalence with age, especially in postmenopausal women, of whom 44% suffer from postmenopausal osteoporosis.1,2
Previous studies have shown a significant increase in blood pressure in postmenopausal women, with 38% of postmenopausal women suffering from hypertension. 3 The renin–angiotensin–aldosterone system (RAAS) is a crucial factor in the regulation of blood pressure and water–sodium balance. Recent studies have shown that 5–10% of hypertension cases are caused by primary aldosteronism (PA).4,5 The plasma aldosterone–renin ratio (ARR) is a widely accepted method for screening PA. In a cross-sectional study of 324 subjects, plasma aldosterone concentration (PAC) and the ARR increased with decreasing bone mineral density (BMD) in postmenopausal women. 6 The false-positive rate of the ARR in screening tests in postmenopausal women with osteoporosis was significantly higher than that in postmenopausal women with osteopenia and normal bone mass, suggesting that the decrease in BMD was closely related to the increase in plasma aldosterone. 6 However, the effect of anti-osteoporosis therapy on PAC, plasma renin concentration (PRC) and the ARR has not been reported.
Alendronate is an extensively used anti-osteoporotic drug. In addition, intermittent low-dose parathyroid hormone (PTH) promotes bone formation, reversing bone loss and increasing BMD, and alendronate is also considered an effective treatment for osteoporosis.
This study aimed to evaluate the changes in PAC, PRC and the ARR prospectively in postmenopausal women with osteoporosis treated with alendronate or PTH for 48 weeks.
Methods
Patients and study design
The subjects were enrolled from the Department of Endocrinology, the First Affiliated Hospital of Chongqing Medical University, from July to October 2014. The inclusion criteria were: women aged 45–80 years with natural menopause for more than three years, a diagnosis of osteoporosis (i.e. a T-score of ⩽–2.5 for BMD at the lumbar spine or total hip or a T-score between −2.5 and −1.0 for BMD at the lumbar spine or total hip and at least one postmenopausal fragility fracture and three or more lumbar vertebrae in the L1–L4 region could be measured by dual-energy X-ray absorptiometry (DXA)) and a body mass index (BMI) between 18 and 30 kg/m2. Those who met any of the following exclusion criteria were excluded: patients with diseases known to affect calcium or bone metabolism; patients with secondary osteoporosis; patients who had received anti-osteoporosis treatment, including PTH, diphosphonate for injection, calcitonin, androgen, oestrogen, active vitamin D, oral bisphosphonates or selective oestrogen receptor regulators, six months before inclusion; patients who received oestrogen or progesterone supplements two months before inclusion; patients with paroxysmal sleep apnoea syndrome, severe kidney dysfunction, ischaemic heart disease, malignant tumour or other serious diseases; abnormal laboratory examination, including bone alkaline phosphatase over the normal upper limit; glutamic oxaloacetic transaminase or glutamic pyruvic transaminase more than twice the normal upper limit; abnormal thyrotropin or parathyroid hormone; and serum calcium over the normal upper limit. During the trial, it was recommended that participants consume a liberal-sodium diet.
After a one-week placebo washout period, all subjects were randomly divided into two groups – the alendronate group and the PTH group – and underwent anti-osteoporosis treatment for 48 weeks. Patients in the PTH group were injected with recombinant human PTH (rhPTH) 1–34 (Suzhou Jinmeng Biotechnology Co., Ltd, Wujiang, PR China) at a dose of 20 μg/day; patients in the alendronate group were treated with alendronate (Fushanmei; Hangzhou Mershadong Pharmaceutical Co., Ltd, Hangzhou, PR China) at a dose of 70 mg/week, while patients in both groups were treated with calcium carbonate (Casilid; General Electric Pharmaceutical Co., Ltd, Shanghai, PR China) at a dose of 600 mg/day. All subjects received basic therapy, including 500 mg calcium and 200 IU vitamin D3 per day. During the treatment, the subjects were examined every four weeks to assess compliance and adverse reactions. Blood pressure, height, weight, waist circumference and blood samples were collected, and BMD was measured before and 48 weeks after treatment.
Laboratory examination and BMD measurement
Blood samples for PAC, PRC, serum calcium, phosphorus, triglycerides, total cholesterol, blood glucose and PTH were drawn from the cubital vein in the morning (8:00–10:00am) after 8–10 hours of fasting and after the subjects had been out of bed for at least two hours and had rested in a seated position for 5–15 minutes.
The ARR was calculated as the ratio of PAC to PRC. When the ARR was ⩾37.0 pg/μIU, it was deemed to be positive for the screening test. 7 Patients with positive screening results proceeded to the captopril challenge test (CCT) to exclude or confirm the diagnosis of PA. Patients received 50 mg captopril orally at 8:00–9:00am after sitting or standing for at least one hour. Blood samples were collected for the measurement of PRC and PAC at baseline and two hours after the challenge, with the patient remaining seated during this period. The diagnosis of PA was established if post-CCT PAC was >110 pg/mL. 8
PRC and PAC were measured by automated chemiluminescence immunoassays (LIAISON; DiaSorin, Saluggia, Italy). Serum calcium, phosphorus, total cholesterol and triglycerides were measured by an automatic biochemical analyser; blood glucose was measured by the glucose oxidase method; and the level of PTH was detected by chemiluminescence immunoassay.
BMD was measured by DXA (QDR 4500A Discovery; Hologic, Mississauga, Canada), which was performed on the hip (total hip and femoral neck regions) and the lumbar spine (L1–L4). 9 An anthropomorphic prosthesis was used to control the quality of the BMD measurement every day. The intraday repetitive standard deviations of the lumbar spine, total hip and femoral neck were 0.005, 0.006 and 0.007 g/cm2, respectively. The bone mass of the subjects was evaluated according to the BMD results, which were expressed by T-score. The criteria for the diagnosis of osteoporosis were as follows: BMD of the lumbar L1–L4, femoral neck or total hip >2.5 standard deviations (T-score ⩽–2.5) lower than that of healthy young women. 10
Statistical analysis
IBM SPSS Statistics for Windows v21.0 (IBM Corp., Armonk, NY) was used for statistical analysis. The Kolmogorov–Smirnov test was used to test the normality of measurement data, and non-normally distributed data were analysed after logarithmic transformation (log10). Measurement data are expressed as the mean±standard deviation. The paired-samples t-test was used to assess the difference between before and after treatment in the group, and the independent-samples t-test was used in the comparison between the groups.
Results
A total of 60 postmenopausal women with osteoporosis were included in this study. During the treatment, three patients were lost to follow-up, and a total of 57 subjects completed the study, including 22 patients in the alendronate group and 35 patients in the rhPTH group. The three patients lost to follow-up were excluded from the analysis. The average age was 62.6±6.0 years, and the average duration of menopause was 14.7±7.6 years. There was no significant difference in age, duration of menopause, BMI, waist circumference, blood pressure, blood glucose, serum calcium, phosphorus, triglycerides, total cholesterol or PTH between the two groups (Table 1).
Baseline characteristics of postmenopausal osteoporosis patients.

Values are presented as means±SD.
BMI: body mass index; WC: waist circumference; SBP: systolic blood pressure; DBP: diastolic blood pressure; FPG: fasting plasma glucose; PTH: parathyroid hormone; NS: not significant; SD: standard deviation.
At baseline, the BMD T-scores of the lumbar spine, total hip and femoral neck were −3.4±0.5, −1.9±0.6 and −2.5±0.5, respectively. After 48 weeks of anti-osteoporosis treatment (alendronate or PTH), the lumbar spine BMD T-score was significantly improved compared to baseline (–3.4±0.5 vs. –3.1±0.4, p<0.0001; Figure 1(a)). There was no significant change in total hip or femoral neck BMD T-scores. After 48 weeks of treatment, the BMD T-scores of the lumbar spine, total hip and femoral neck in the alendronate group were significantly better than those before treatment. After 48 weeks of treatment, the lumbar spine BMD T-score increased significantly, the total hip BMD T-score decreased and the femoral neck BMD T-score did not change significantly in the rhPTH treatment group (Table 2).

Lumbar spine T-score (a) and plasma concentrations of aldosterone (b) in postmenopausal women with osteoporosis at baseline and 48 weeks after treatment. Group data are presented as means with error bars.
Comparison before and after treatment.
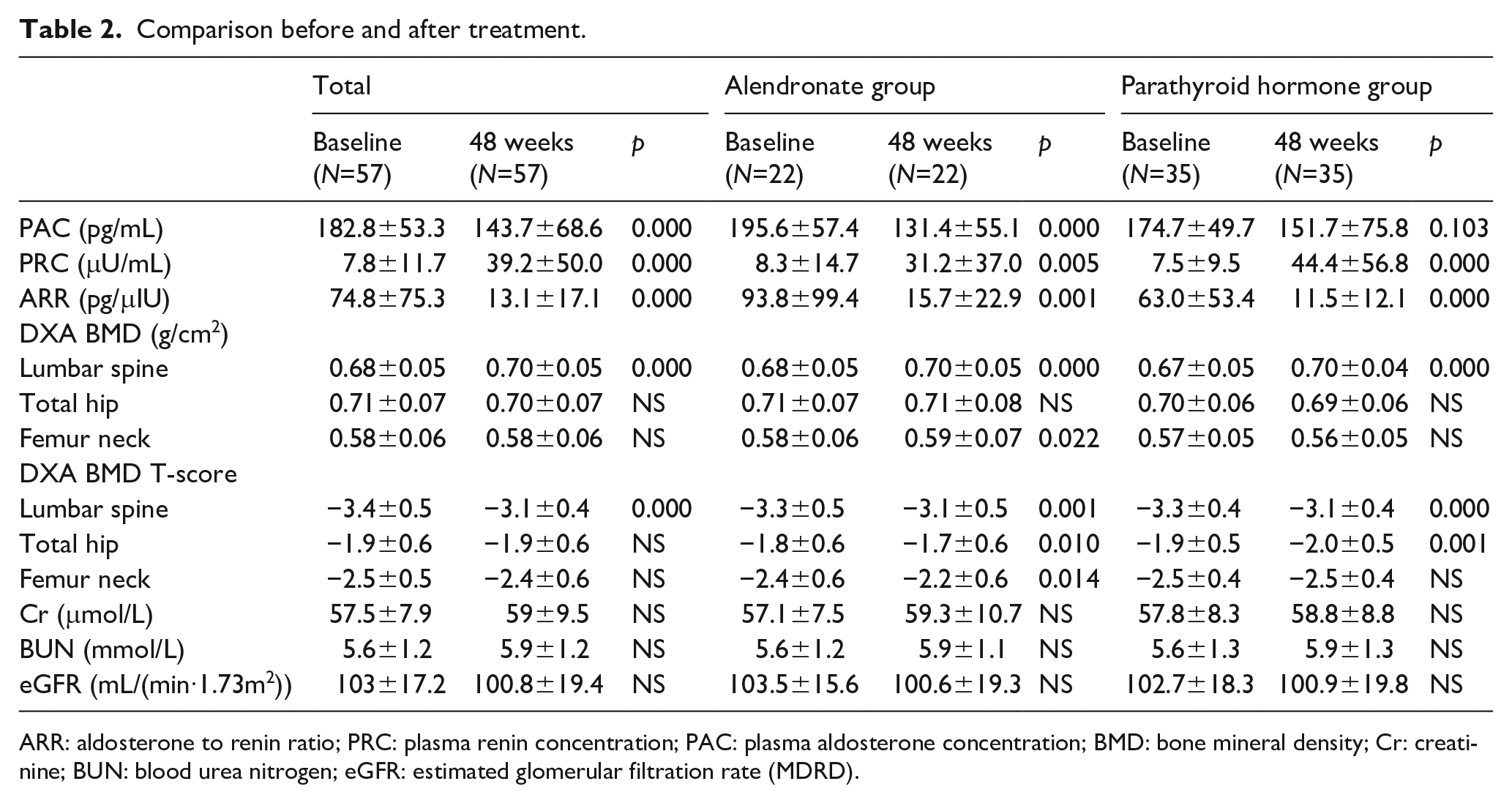
ARR: aldosterone to renin ratio; PRC: plasma renin concentration; PAC: plasma aldosterone concentration; BMD: bone mineral density; Cr: creatinine; BUN: blood urea nitrogen; eGFR: estimated glomerular filtration rate (MDRD).
At baseline, the PAC, PRC and ARR of all subjects were 182.8±53.2 pg/mL, 7.8±11.6 μIU/mL and 74.8±75.2 pg/μIU, respectively. After 48 weeks of anti-osteoporosis treatment, PAC decreased significantly (182.8±53.2 vs. 143.7±68.6, p<0.0001; Figure 1(b)), and PRC increased significantly (7.8±11.6 vs. 39.2±50.0, p<0.0001), resulting in a significant reduction in the ARR (74.8±75.2 vs. 13.1±17.1, p<0.0001). Both alendronate and rhPTH caused obvious changes in PRC and ARR. However, alendronate caused a significant decrease in PAC, while the decrease in PAC in the rhPTH group was not significant (Table 2).
In all subjects, PAC was negatively correlated with lumbar spine BMD T-score and femoral neck BMD T-score at baseline (lumbar spine r=−0.386, p<0.01; femoral neck r=−0.262, p<0.05; Figure 2).

Associations between the aldosterone concentration and lumbar spine T-score (a), and femur neck T-score (b). Individual values (dots) with association line.
Using an ARR of ⩾37.0 pg/μIU as the positive screening cut-off, at baseline, 35 subjects were ARR positive and 25 were ARR negative, resulting in an ARR-positive rate of 61% (35/57). After 48 weeks of treatment, only five subjects were ARR positive, resulting in an ARR-positive rate of 8% (5/57). Subjects who were ARR negative were excluded from having PA, and subjects who were ARR positive underwent CCT. A diagnosis of PA was ruled out in all subjects who were ARR positive by CCT. Thus, the ARR results in these subjects were false-positives, and the false-positive rate of ARR decreased from 61% to 8% after 48 weeks of anti-osteoporosis treatment (Figure 3). In addition, there was no significant difference in the proportion of false-positive ARR between hypertensive patients (9/15) and non-hypertensive patients (26/42; 60.0% vs. 61.9%).

Percentage of women with false-positive ARR at baseline and 48 weeks after treatment, using ARR ⩾37.0 pg/μIU as the cut-off for positive screening.
Discussion
This study suggested that the BMD T-score was negatively correlated with PAC in postmenopausal women with osteoporosis. We found that with the improvement in BMD in postmenopausal women with osteoporosis who received anti-osteoporosis treatment, PRC increased and PAC decreased, resulting in an increase in the ARR and the false-positive rate of the ARR.
Our previous studies showed that PAC and ARR in postmenopausal women with osteoporosis were significantly higher than those in women with normal bone mass, 6 suggesting that BMD is closely related to the RAAS. This population study of anti-osteoporosis treatment further confirmed the relationship between BMD and the RASS; that is, low BMD may lead to an increase in PAC, and an increase in PAC may inhibit the production of PRC, resulting in an increase in the ARR and the likelihood of ARR false-positives.
There are few studies on the effect of anti-osteoporosis treatment on the RAAS. In an Italian study, 40 postmenopausal women with osteopenia were treated with 60 mg/day raloxifene for six months, and the levels of plasma aldosterone and renin were lower than those before treatment. However, there was no significant difference. 11 A similar study in Japan found that plasma aldosterone and renin levels did not change significantly in postmenopausal women with osteoporosis or osteopenia (41 cases) after six months of raloxifene (60 mg/day) treatment. 12 The above results are different from those of this study. On the one hand, raloxifene is an oestrogen receptor regulator, and its anti-osteoporosis mechanism is different from that of alendronic acid and PTH. On the other hand, the treatment period of raloxifene is relatively short. In addition, the changes in BMD after treatment were not shown in those studies.
The mechanism underlying the effect of BMD on plasma aldosterone is not clear, and it is speculated that it may be related to bone metabolism or aldosterone metabolism related hormones. A genomic study of the Framingham cohort found that aldosterone interacted with bone metabolism, and some key molecules of the aldosterone synthesis signalling pathway (NR3C2, MR, PIK3R1, PRKCH and SCNN1B) were also involved in bone metabolism. 13 In addition, type 1 PTH receptor14,15 was found in the adrenal gland. In vitro, it was found that PTH stimulated the secretion of aldosterone 16 via adrenal glomerulosa zone cells. Infusion of PTH could increase the level of aldosterone metabolites in urine. 17 However, the relationship between PTH and aldosterone does not explain the findings of this study because our study and previous studies have shown that long-term use of diphosphate does not significantly affect plasma PTH levels.18,19 Therefore, the specific mechanism of the relationship between BMD and aldosterone metabolism needs to be further elucidated.
There are some limitations to this study. First, the sample size was relatively small. Although there were only 60 subjects, the changes in PAC and ARR were significant after anti-osteoporosis treatment. In addition, this study lacked a control group and did not observe changes in PAC or ARR in patients with natural disease development. As urinary sodium and urinary calcium excretion were unavailable in this study, whether the changes in aldosterone and renin after anti-osteoporosis treatment are related to calcium or sodium metabolism is unclear.
Conclusions
PAC, ARR and false-positive rates of the ARR decreased significantly with the improvement in BMD in postmenopausal women with osteoporosis treated with alendronate or PTH. It is suggested that we should be more careful in interpreting the results of the ARR for screening tests in postmenopausal women with osteoporosis.
Supplemental Material
Authorship_change_request_form – Supplemental material for Effects of anti-osteoporosis therapy on plasma aldosterone and renin
Supplemental material, Authorship_change_request_form for Effects of anti-osteoporosis therapy on plasma aldosterone and renin by Qingfen Hu, Kangla Liao, Longwei Zhang, Xiaoyu Shu, Zhixin Xu, Yuyang Qiu, Qifu Li and Shumin Yang in Journal of the Renin-Angiotensin-Aldosterone System
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Key Research and Development Plan, Major Project of Precision Medicine Research (2017YFC0909600, sub-project: 2017YFC0909602, 2017YFC0909603), the National Natural Science Foundation of China (81670785, 81870567, 81800731, 81800701, 81970720) and the Joint Research Project of Chongqing Science and Technology Commission and Traditional Chinese Medicine (MSXM20192015).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
