Abstract
Objective:
The diagnosis of primary aldosteronism (PA) among the older-aged population has posed a crucial challenge. Among patients over 50 years old, this trial assessed comparability of the performance of two PA diagnostic tests: losartan and captopril suppression tests.
Methods:
A post-hoc subgroup analysis from a prospective cohort was conducted by the TAIPAI (Taiwan Primary Aldosteronism Investigation) group between July 2003 and July 2006. Of the 160 patients in the cohort, 60 patients over 50 years old received captopril and losartan tests to confirm PA.
Results:
Among the 60 patients over 50 years old, 31 patients had PA confirmed by standardized protocol. The area under the receiver-operating characteristic (ROC) curve for post-captopril aldosterone was significantly less than that for post-losartan plasma aldosterone concentration (PAC) (0.87 vs 0.94, p=0.02). Using the aldosterone-renin ratio (ARR)>35 with PAC>10 ng/dl, the specificity was 82.76% vs 93.1% and the sensitivity was 77.42% vs 87.10% for the captopril and losartan tests, respectively. The equivalence between the two tests were confirmed by the exact McNemar’s test (p=1.0).
Conclusion:
The losartan test showed comparable accuracy to confirm PA. Verification of this “elderly-friendly” confirmatory test will be the first step to prepare a specific diagnostic model of PA for the older-aged population.
Keywords
Introduction
As populations are aging worldwide, the global burden of hypertension has dramatically increased as the elderly persons have the highest prevalence of this disease.1–3 The unique characteristic of hypertension in the near-elderly and elderly is that secondary causes are more prevalent and different compared to the younger population.4,5 Primary aldosteronism (PA) has long been considered a rare cause of hypertension in near-elderly and elderly. 6 An increasing body of literature has demonstrated a greater prevalence of PA, about 10% in patients with increasing hypertension severity, presenting a growing concern. 7 Long-standing aldosterone excess has been proved to induce arterial stiffness and endothelial injury. 8 PA is also related to kidney damage, 9 metabolic syndrome, 10 and left ventricular hypertrophy. 11 Therefore, an accurate and safe diagnostic test for these populations is an unaddressed clinical and epidemiological concern.
As the capacity to adjust excess fluid level is gradually declining in the near-elderly and elderly, oral sodium loading, saline infusion and fludrocortisone suppression tests may not be the first choice especially if patients also have been diagnosed with heart failure, advanced liver disease, and renal insufficiency. Consequently, the captopril challenge test may be the only suitable diagnostic test. Compared to the captopril test, the losartan test has demonstrated potentially better diagnostic accuracy in general population while losartan also provides a better safety profile than captopril regarding the development of angioedema, a potentially life-threatening event.12–14 In this post-hoc analysis, we aimed to verify the performance of the losartan test for PA in patients older than 50 years old. As PA becomes increasingly prevalent in this specific age range, the safety issues and applicability of diagnostic tests would become a critical concern in this population.
Materials and methods
Patients
From July 2003 to January 2006, 64 patients older than 50 years old were enrolled into the Taiwan Primary Aldosteronism Investigation (TAIPAI) database after follow-up screening at, or referral to, the National Taiwan University Hospital and its affiliated hospitals. Since this was a subgroup analysis from our previous published prospective cohort study investigating the diagnostic accuracy of losartan test, 12 we carefully examined the study power according to the subgroup sample size. Based on the study sample size of 30 in each group with a 0.70 proportion of positive ratings, and 0.60 kappa, we have 90% power for a two-sided test. 15 The initial evaluation included: (a) age at onset of hypertension or hypokalemia of <35 years; (b) difficult-to-control hypertension after therapy initiation; (c) the clinical occurrence of a hypertensive crisis; (d) the presence of hypokalemia or metabolic alkalosis, or a random aldosterone-renin ratio (ARR) >35 ng dl−1 per ng ml−1h−1; and (e) evidence of adrenal incidentaloma with hypertension or hypokalemia. Difficult-to-control hypertension was defined as patients taking more than three antihypertensive medications while their blood pressure (BP) still did not reach their therapeutic target. 16 Hypertensive crises are characterized by severely elevated BP, usually higher than 180/110 mm Hg, along with progressive or impending target organ damage. 17 The ethnic composition of our cohort reflected a typical Taiwanese population. 18
All antihypertensive medications were discontinued for at least 21 days before the study. Diltiazem and/or doxazosin were administered for control of marked high BP when required. 19 Medications that might interfere with the renin-aldosterone axis such as steroids, sex hormones, licorice, or non-steroidal anti-inflammatory drugs were also withheld for at least six weeks. All patients consumed a low-salt diet with 6 g of NaCL daily and were supplied with potassium during the testing period if hypokalemia was identified. The database was constructed for quality assurance in one medical center (National Taiwan University Hospital, Taipei, Taiwan) and its three branch hospitals in different cities (National Taiwan University Hospital Yun-Lin branch, Yun-Lin, southern Taiwan; En Chu Kong Hospital, Taipei County; and, Tao-Yuan General Hospital, Tao-Yuan, middle Taiwan). All patients requiring confirmation of primary aldosteronism through a suppression test or adrenal venous sampling (AVS) were recruited and the data were prospectively collected. This post-hoc subgroup analysis from the previous prospective cohort study was approved by the Institutional Review Board of National Taiwan University Hospital(NTUH No. 9461700402).
Confirmation of primary aldosteronism
In this subcohort, a diagnosis of pheochromocytoma was made in one patient, renal artery stenosis was diagnosed in one patient, and polycystic kidney disease was diagnosed in one patient. Only one patient who did not give his informed consent was excluded. The remaining 60 patients received both captopril and losartan tests (Figure 1). The two tests were performed on two consecutive days. As previously reported, the time-to-peak plasma concentration of losartan was proportional to the dose and was ~1.5 h with the 50 mg dose. 20 With oral captopril, the time-to-peak was >0.5 h. 21 In our previous head-to-head comparative study, 12 we have set the sampling period as at least one half-life of plasma renin activity (PRA) or plasma aldosterone concentration (PAC) because the biological half-life of aldosterone is ~30 min 22 and the half-life of PRA is ~15 min. 23 The patients were asked to sit for at least 10 min for the baseline blood samples at 0900 , and allowed to ambulate moderately until the second sampling. The second blood samples were obtained either 1 h after the administration of 50 mg of captopril, or 2 h after the administration of 50 mg of losartan. 12
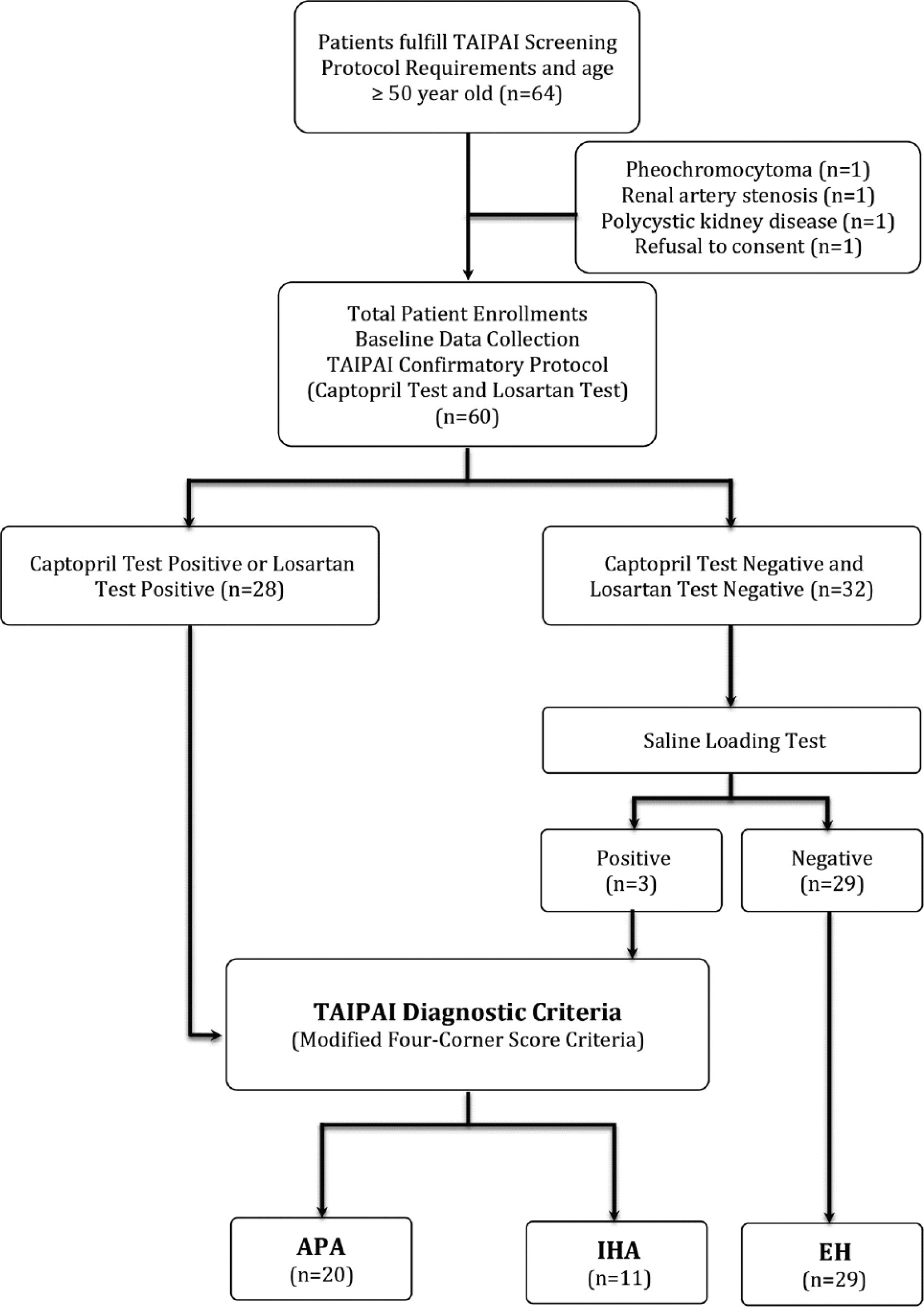
Study flow diagram.
An ARR >35 with a PAC >10 ng/dl (>277 pmol/l) after the administration of captopril or losartan was defined as a positive test for PA. We constructed an ARR >35 (ng dl−1 per ng ml−1h−1) because this value had the best sensitivity and specificity to differentiate PA from essential hypertension (EH) in the TAIPAI database. Patients with both negative captopril and losartan suppression tests underwent a saline infusion test on a separate day to evaluate the autonomous secretion of aldosterone. After at least 1 h in the supine position, 0.9% NaCl solution (2 l) was administered intravenously from 0800–1200, and blood samples for PRA and PAC were drawn before and at the end of the saline infusion. Patients with PAC >10 ng/dl after saline infusion were diagnosed with PA in the TAIPAI database. 24
Differential imaging studies
Computerized tomography (CT) of the adrenal glands with a non-ionic iodinated contrast agent was done on all enrolled patients, with at least 3-mm contiguous slices in a normal surrounding. Although there were no strict measurements of normal adrenal size, CT imaging was considered abnormal when any volume greater than 10 mm3 was detected. Those with inconclusive CT findings underwent dexamethasone suppression adrenocortical scintigraphy with CT (NP-59, I-131-6-beta-iodomethyl-19- norcholesterol & NP59-SPECT/CT). 25 Bilateral adrenal venous sampling (AVS) was required if image studies were ambiguous. Successful venous cannulation was defined as the ratio of the cortisol level of the adrenal vein to that of the inferior vena cava >3. Lateralization of aldosterone secretion was defined by a greater than four-fold difference in the aldosterone/cortisol ratio between the two adrenal glands. 24
Histopathological studies
All of the surgically removed adenomas were re-evaluated by a histopathologist in the TAIPAI study group who was blinded to the clinical data. A histological diagnosis of aldosterone-producing adenoma (APA) was based on well-defined, encapsulated tumors predominantly consisting of foamy clear cells. 26 Adenoma appeared as nodules of clear cells in sheets or nests that were sharply demarcated by a pseudo-capsule and compressed the non-neoplastic, uninvolved adrenal gland. 27 Adenomas were differentiated from nodular adrenal hyperplasia by their solitary and well-circumscribed nature.27,28 Adrenal glands from the idiopathic hyperaldosteronism (IHA) patients were marked by diffuse hyperplasia of cells resembling those of normal zona glomerulosa with or without macro- or micro-nodules. 29
Measure of aldosterone and renin
The concentration of aldosterone was measured by radioimmunoassay (RIA) using commercial kits (Aldosterone MAIA Kit, Biochem ImmunoSystems, Bologna, Italy) as previously described.30,31 The detection limit was 10.0 pg ml−1 with a 90% confidence interval, with the normal range of 70–350 pg ml−1 in an upright position. PRA was measured as the generation of angiotensin I in vitro using a commercially available RIA kit (Incstar Corporation, Stillwater, Minnesota, US). Its normal range was 2.63±1.32 ng ml−1h−1 with the patient in an upright position. In our three centers over a period of 13 years, the same aldosterone and renin assays were used.
Diagnostic criteria
Identification of APA in hypertensive patients required all of the following “modified four-corner score” criteria:12,24,32 (a) evidence of autonomous excess aldosterone production based on an ARR >35 ng dl−1 per ng ml−1h−1 and a PAC > 10 ng dl−1after any confirmatory test; (b) lateralization of aldosterone secretion at adrenal vein sampling or during dexamethasone suppression NP-59 SPECT/CT; 25 (c) evidence of adenoma on a CT; and (d) pathologically proven adenoma after an adrenalectomy, and cure of hypertension without antihypertensive agents or improved hypertension, potassium, PAC, and PRA as described. 32
IHA was established based on the following criteria: 33 (a) evidence of autonomous excess aldosterone production based on an ARR >35 ng dl−1 per ng ml−1h−1 and a PAC > 10 ng dl−1after any confirmatory test; (b) non-lateralization of aldosterone secretion at adrenal vein sampling, or after undergoing dexamethasone suppression adreno-cortical scintigraphy; 25 (c) evidence of bilateral diffuse enlargement on a CT; and (d) evidence of diffuse cell hyperplasia in the pathology studies. In patients with negative captopril and losartan tests, the pre-specified ARR <35 ng dl−1 per ng ml−1h−1 and PAC < 25 ng dl−1, and negative salt-loading results were considered to be diagnostic of EH.
Statistical analysis
The primary objective of the study was to compare the diagnostic accuracy of the losartan test vs the captopril test for PA in patients older than 50 years old. The data were provided as the mean values±standard deviation (SD)). As the data of PAC, PRA and calculated ARR were not normally distributed, median level with interquartile range were provided. 34
Statistical analyses were performed using STATA version 12.0 statistical software (StataCorp LP, College Station, Texas, USA). A normal distribution was attained by appropriate transformations of skewed variables such as PAC and ARR. Comparisons of variables between PA and EH were based on t-test statistics. The κ-test was used to evaluate the agreement of defining PA between captopril and losartan suppression tests. The results were expressed as a kappa coefficients and were classified according to the scale of Landis and Koch. 35 The exact McNemar’s test was used to check equality among captopril and losartan tests and the reference standard – the “modified four-corner score” criteria. The sensitivity, specificity, accuracy, positive percent agreement and negative percent agreement for both losartan and captopril tests were calculated and compared by using receiver-operating characteristic (ROC) curves. In addition, the age- and potassium-adjusted probabilities of having PA according to the results of both suppression tests were also computed. The p-value equating significance was <0.05.
Results
The demographics of PA patients and EH patients are shown in Table 1. EH patients tended to be older. There were no statistically significant differences in sex, body mass index (BMI), systolic and diastolic blood pressures between the PA and EH groups. The basal levels of serum potassium (SK), PRA, PAC, and ARR were statistically significantly different between the PA and EH patients (all p<0.05).
Main demographic and biochemical characteristics of the study patients.
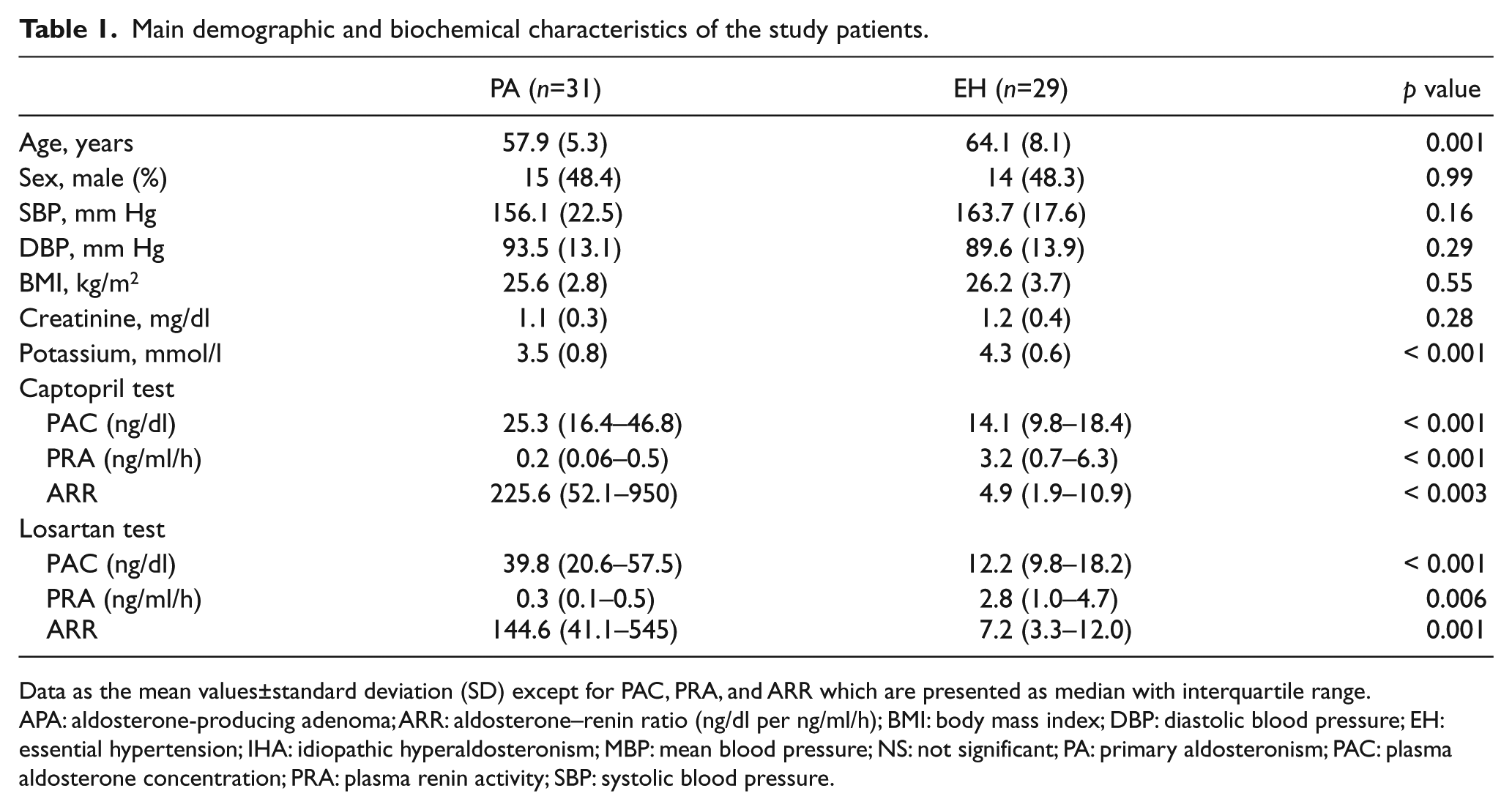
Data as the mean values±standard deviation (SD) except for PAC, PRA, and ARR which are presented as median with interquartile range.
APA: aldosterone-producing adenoma; ARR: aldosterone–renin ratio (ng/dl per ng/ml/h); BMI: body mass index; DBP: diastolic blood pressure; EH: essential hypertension; IHA: idiopathic hyperaldosteronism; MBP: mean blood pressure; NS: not significant; PA: primary aldosteronism; PAC: plasma aldosterone concentration; PRA: plasma renin activity; SBP: systolic blood pressure.

Receiver-operating characteristic (ROC) curves for the detection of all primary aldosteronism by (a) post-test plasma aldosterone concentration (PAC), and (b) post-test aldosterone-renin ratio (ARR).
Comparison of diagnostic performances between losartan and captopril tests.

The adjusted probability of having primary aldosteronism (PA) by using post-captopril and post-losartan aldosterone-renin ratio (ARR) at the cut-off value of 35 ng/dl per ng/ml/h.
Discussion
This study verified the diagnostic value of losartan challenge test in near-elderly and elderly patients, a subpopulation of major interest in current global health care.36,37 The losartan test was initially thought to be less diagnostically powerful but has been recently validated as a comparable test to the captopril test in the general population by our previously published head-to-head study. 12 The current nested case-cohort investigated the diagnostic accuracy of the losartan test in the population over 50 years old. Since the pharmacodynamics/pharmacokinetic profiles are different among older-aged patients for ACEI (angiotensin-coverting-enzyme inhibitor), analyzing this subgroup provides additional clinical insight to previously published work.12,38,39 In addition, our study has demonstrated a good agreement between captopril and losartan tests with kappa value of 0.67 (p<0.01).
In this study, we found that the frequency of APA is higher than IHA in patients over 50 years old in contrast to the current concept of PA regarding the prevalence of subtype. 40 This “reversing” phenomenon reflects the multiple and intertwined problems of approaching PA in the near-elderly and elderly. First, the prevalence of PA in elder persons has rarely been the focus of contemporary literature, perhaps the most significant grey area in the management of hypertension on older-aged patients. Second, the presence of clinical comorbidities poses noteworthy limitations to the current PA diagnostic system such as adherence and tolerance to multi-step diagnostic tests. Finally, the cut-off value in many morbid subgroups remains undetermined e.g. patients with end stage renal disease (ESRD) and congestive heart failure (CHF). All of the above-mentioned perspectives may lower the awareness of primary clinicians toward PA in daily practice with a blunt and insensitive approach.
Although previously thought to be an extremely rare condition, PA is now considered as one of the most common causes of secondary hypertension.41,42 Also, a family history of early hypertension (<40 years), 40 indicating an increased pretest probability, has long been considered a triggering factor of the PA screening test.40,43 However, the prevalence of PA may be stable across the entire age spectrum. 4 Meanwhile, the average age at which PA was diagnosed in recent published studies from five continents is 52.1 years old (Table 3), nearly the US Census Bureau’s definition of the “older” population. 44 The average hypertension duration is 10.1 years which echoes the tendency of delayed diagnosis and the wastage of medical resources under current clinical practices (Table 3). Furthermore, the hypothetical PA onset age is approximately 41.8 years old (Table 3 and Figure 4). By updating the epidemiological profile, public awareness of PA in cases over 50 years old has the potential to increase.
Recent studies (study population more than 10 patients) of primary aldosteronism (PA) provide both mean age at the diagnosis of PAand the patients’ mean duration of hypertension including primary care and referral settings published between 1 January 2000 and 4 July 2012 from two electronic databases, MEDLINE and EMBASE.The key words “primary aldosteronism” or “hyperaldosteronism” were used and two reviewers (CC Kuo and VC Wu) independently screened the titles, abstracts, and contents to identify potentially eligible studies and to avoid the duplication by collate authors’ name and affiliation to minimize the possibility of extracting repeat data from the same study group.

HTN: hypertension; PA: primary aldosteronism.
Hypothetical mean age of PA onset=the average of the five continents’ (mean age of PA at diagnosis – mean duration of hypertension)=41.8 years.
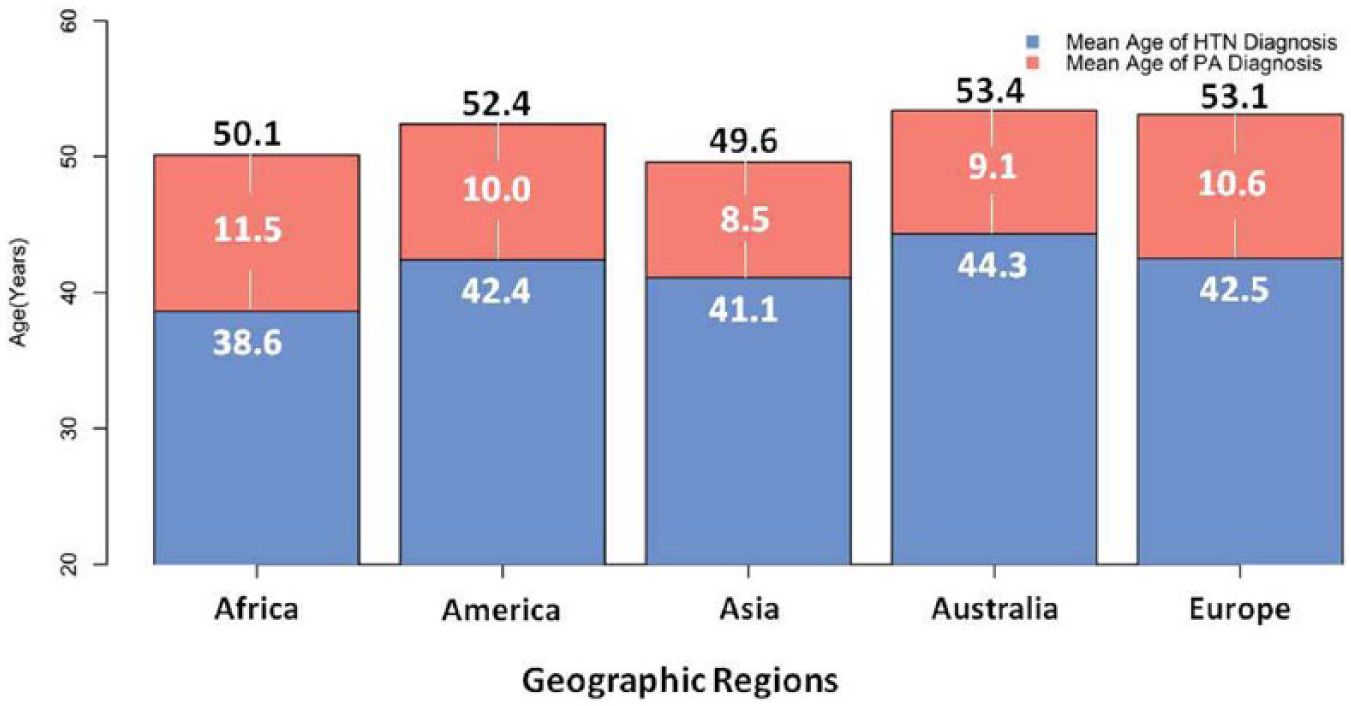
The mean primary aldosteronism (PA) age at diagnosis and hypertension (HTN) duration summarized from recent PA studies between January 2000 and July 2012 in five continents. The number in each red bar indicates the mean duration of hypertension.
The call for adequate diagnostic testing of PA in the near-elderly and elderly is still largely unaddressed as are the clinical practice guidelines for other more common diseases. 75 The current three-step diagnostic system of PA greatly compromises the adherence among the elderly. Furthermore, multiple comorbid conditions often limit the selection of diagnostic tests. In patients with hypertensive pulmonary edema, congestive heart failure or kidney diseases that are vulnerable to fluid-overload, salt-loading and fludrocortisone tests are obviously inappropriate. Furthermore, to avoid possible angioedema from captopril in such a susceptible population, 14 the losartan test turns out to be a feasible choice. From the standpoints of safety and test adherence, conducting a large prospective study to verify the losartan test in an ethnically diverse population would be fundamental to formulate a specific diagnostic model for patients over 50 years old.
In this post-hoc analysis restricting the sample to patients over 50 years old, the proportion of APA is significantly higher than IHA. Although this prevalence profile might be affected by the selection criteria in our TAIPAI screening protocol, 24 the curability of APA offers a cost-benefit opportunity to avoid unnecessary long-term anti-hypertensive medication. The accurate diagnosis of IHA also facilitates the use of effective target therapies such as spironolactone and eplerenon. 76 And timely diagnosis of PA in elder population may considerably lower the risk of long-term complications ranging from cardiovascular diseases,77,78 kidney damage, 9 to metabolic syndrome 79 related to aldosterone excess and consequently interrupt the vicious cycles of PA and coexisting organ damage.
The strength of our study is its prospective head-to-head design and that all patients underwent both captopril and losartan suppression tests. Moreover, the rigorous protocol based on a three-step investigation minimizes the possibilities of misclassification and ascertainment bias.24,40
There are some limitations to the study. First, as a study investigating a confirmatory test, larger numbers would offer a more accurate unbiased estimation on the sensitivity and specificity of the losartan test. 80 The relatively small sample size may also mask the true difference due to insufficient statistical power. Yet the results seem promising since, even with limited sample size, the losartan test is comparable to the captopril test as a useful confirmatory tool in hypertensive patients over 50 years old. Second, a selection bias may be inherent in the selection criteria. Regarding the proportion of subtypes of PA, the TAIPAI protocol employed hypokalemia as a selection criteria which may have resulted in selective inclusion of a more severe form of PA, APA. 81 Third, spectrum bias is an inevitable issue for studies conducted in tertiary medical centers, which may overestimate the performance of the diagnostic tests. This bias may be even more prominent for confirmatory tests since the study population would have been highly selected after the screening protocol. However, due to the head-to-head design, it is fair to compare both captopril and losartan tests and verify the “rule in” and “rule out” performances of the losartan test. 82
Also, we are aware that confirmatory tests based on perturbation of the renin-angiotensin system will lead to exclude patients with angiotensin II-responsive APA. 83 Therefore, our finding may not be generalized to non-angiotensin II autonomous APA. Finally, the target population was restricted to Asian population in our study; therefore, the statistical inference toward other race/ethnicities needs to be further validated.
In summary, we found that the losartan test is comparable to the captopril test in patients older than 50 years old. With comparable diagnostic accuracy, the losartan test has a better safety profile compared to the captopril test which is an advantage in the case management of patients over 50 years old. We believe the verification of this “elderly-friendly” confirmatory test will be the first step to prepare a specific diagnostic model of PA for the near-elderly and elderly in contrast to current ‘one-size-fits-all’ practice.
Research highlights
Adequate diagnostic testing of PA in older-aged persons is unaddressed in current guidelines.
The losartan test showed comparable diagnostic performance to the captopril test in older-aged persons.
The diagnosis of PA was delayed 10.1 years on average resulting in a huge waste of medical sources.
Footnotes
Acknowledgements
The authors wish to acknowledge the members of the Taiwan Primary Aldosteronism Investigation (TAIPAI) Study Group: Vin-Cent Wu, Yen-Hung Lin, Yi-Luwn Ho, Hung-Wei Chang, Lian-Yu Lin, Fu-Chang Hu, Kao-Lang Liu, Shuo-Meng Wang, Kuo-How Huang, Yung-Ming Chen, Chin-Chi Kuo, Chin-Chen Chang, Shih-Chieh Chueh, Ching-Chu Lu, Shih-Cheng Liao, Ruoh-Fang Yen, Wei-Chou Lin, Bor-Sen Hsieh and Kwan-Dun Wu.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported in part by a National Heart, Lung, and Blood Institute training grant T32 HL7024-38, Ta-Tung Kidney Foundation, and the Taiwan National Science Council (grant NSC 96-2314-B-002-164, grant NSC 96-2314-B-002-033-MY3, and grant NSC 97-2314-B-002-155-MY2).
