Abstract
Introduction:
Performing measurements of plasma renin activity (PRA) or direct renin concentration (DRC) and aldosterone concentration, we should be well informed about requirements concerning blood sample processing.
Material and methods:
Forty-seven patients had blood collected in the supine and upright positions. Blood was withdrawn into two EDTA2K tubes and one with clot activator. One EDTA2K tube was cooled at +4°C and centrifuged at +4°C whereas the other was prepared at room temperature. PRA and DRC were measured by radioimmunoassay (RIA) and radioimmunometry (IRMA), respectively, in both cooled and not cooled plasma samples, and aldosterone was measured by RIA in not cooled plasma and in serum.
Results:
In all the groups, with low, medium, and high values of PRA and direct renin, the temperature of sample processing within 30 minutes had no marked influence on the final result (correlation coefficient for renin was 0.9994, and for PRA, 0.8297). The measured concentrations of aldosterone also showed high correlation (r = 0.9790) but were markedly higher in plasma.
Conclusion:
The measurements of DRC, and to a lesser extent PRA, were similar regardless of temperature condition during the 20–30 minutes necessary for blood sample processing. Aldosterone concentrations in plasma vs serum samples appeared to be markedly higher.
Introduction
Hypertension is one of the most common cardiovascular diseases, and the search for treatable causes and effective diagnostic methods is a challenge for doctors. Primary aldosteronism (PA) is one of the causes of hypertension. 1 Currently, it is assumed that it may affect 5%-12% of patients with hypertension, so there is a need to improve diagnostic methods, especially for effective screening.2,3
Measurements of plasma renin activity (PRA) or direct renin concentration (DRC) and aldosterone concentration are used for investigation in cases of suspected PA. There are many factors that can have an influence on these biochemical results, such as: drugs, body position, time of day, diet, method of blood collection (avoiding stasis and hemolysis), temperature and time of blood processing and laboratory method.4–6 Performing these measurements, we should be well informed about all precautions and requirements concerning proper blood sample processing.
Aims of the study
To investigate whether cooling samples after their collection and during centrifugation influences the results of PRA and DRC measurements.
To investigate whether the results of aldosterone concentration measured in serum and EDTA2K plasma are similar and can be referenced to the same reference values.
Material and methods
Forty-seven patients with an indication for testing the renin-angiotensin-aldosterone system had blood collection after a night’s rest in the supine position (0hr) and again after walking for two hours (2hr). At each collection, blood was withdrawn into two EDTA2K tubes (plasma) and one with a clot activator (serum). After blood collection, one tube was transported to the laboratory in a cool state (within 10 minutes) and the other tube at ambient (room) temperature (also within 10 minutes). The cooled at +4°C blood sample was centrifuged at +4°C (10 minutes, 3500 rpm) whereas the not cooled sample was centrifuged at ambient (room) temperature (10 minutes, 3500 rpm). The biological material (serum, EDTA2K-plasma) was frozen at −30°C and stored until assayed.
Aldosterone was measured by radioimmunoassay (RIA) (ZenTech, RIAZENco, Belgium). Analytical sensitivity was 1.4 ng/l, functional sensitivity was 25 ng/l. The reference value at rest was 10–160 ng/l, and in the upright position was 35–300 ng/l. Intra-assay coefficients of variation (CV) were: 13.7% for 42 ng/l, 3.8% for 177 ng/l and 5.3% for 278 ng/l, and inter-assay CV values were: 18.6% for 38 ng/l, 7.5% for 169 ng/l and 6.2% for 284 ng/l.
PRA was measured by radioimmunoassay kit (RIA), (CISbio International, France). The standard 90-minute incubation time at 37°C was applied for generation of angiotensin I. Analytical sensitivity was 0.1 ng/ml per hour. The reference value at rest in the supine position was 0.2–2.8 ng/ml per hour, and in the upright position was 1.5–5.7 ng/ml per hour. Intra-assay CV values were: 4.3% for 1.4 ng/ml per hour, 6.5% for 3.8 ng/ml per hour and 7.2% for 16.0 ng/ml per hour. Inter-assay CV values were: 9.9% for 1.4 ng/ml per hour, 4.9% for 3.9 ng/ml per hour and 8.5% for 15.8 ng/ml per hour.
Renin was measured by radioimmunometric kit (IRMA) manufactured by CISbio International (France). According to the kit producer, analytical sensitivity was 1 ng/l. The reference value at rest in the recumbent position in patients between 20 and 40 years of age was 3.6–20.1 ng/l, and between 41 and 60 years, 1.1–20.2 ng/l. In the upright position in patients between 20 and 40 years, the reference value was 5.1–38.7 ng/l, and between 41 and 60 years, 1.8–59.4 ng/l. The intra-assay CV values were: 3.6% for 3.8 ng/l, 1.5% for 65.4 ng/l and 0.9% for 262 ng/l. The inter-assay CV values were: 5.0% for 4 ng/l, 3.6% for 72 ng/l, and 4.0% for 263 ng/l.
The study was approved by the bioethics committee at The Medical Centre of Postgraduate Education.
All results are expressed as median and mean ± SD. Differences between the groups are presented as percentages. The relationships between the compared results were expressed using correlation analysis.
Results
Among 47 investigated patients, PA was diagnosed in four patients (adrenal hyperplasia in three and aldosteronoma in one).
In the group of results with PRA < 1 ng/ml per hour, the median values at ambient (room) temperature vs +4°C were as follow: PRA 0.3 vs 0.3 and renin 3.3 vs 3.3. In the group of results with PRA between 1 and 10 ng/ml per hour, the median values were: PRA 2.6 vs 2.5 and renin 13.6 vs 14.3, and in the group of results above 10 ng/ml per hour, the median was 16 vs 14 for PRA and 86 vs 75 for renin (Table 1). Correlation between measurements at ambient (room) temperature and +4°C was r = 0.8297 for PRA and r = 0.9994 for renin (Figures 1 and 2).
Comparison of plasma renin activity (PRA) and direct renin concentration (DRC) results at ambient (room) temperature (18–25°C) and cool temperature (+4°C).

PRA: plasma renin activity, temp: temperature, hr: hour, n: number of patients.

Correlation of PRA results at ambient temperature (18–25°C) vs cool temperature (+4°C).

Correlation of DRA results at ambient temperature (18–25°C) vs cool temperature (+4°C).
Individual results of PRA and renin (DRC) at ambient (room) temperature vs +4°C are presented in Table 2.
Individual results of PRA and renin (DRC) at ambient temperature (18–25°C) vs cool temperature (+4°C).

PRA: plasma renin activity. DRC: direct renin concentration; hr: hour; PRA lower at +4°C in position: 1, 2, 15, 17 and 25. PRA lower at 18–25°C (ambient temperature) in position: 2, 5, 23, 34, 39 and 41.
The measured concentrations of aldosterone in plasma (EDTA2K) and serum samples showed high correlation (r = 0.9790) (Figure 3), but appeared markedly higher in plasma than in serum. The median serum aldosterone concentration was 258 ng/l (range 68–2100 ng/l), and the median concentration in plasma (EDTA2K) was 444 ng/l (range 150–2226 ng/l). The differences between pairs of plasma and serum samples ranged from 37% to 144% (median 75%) (Table 3).
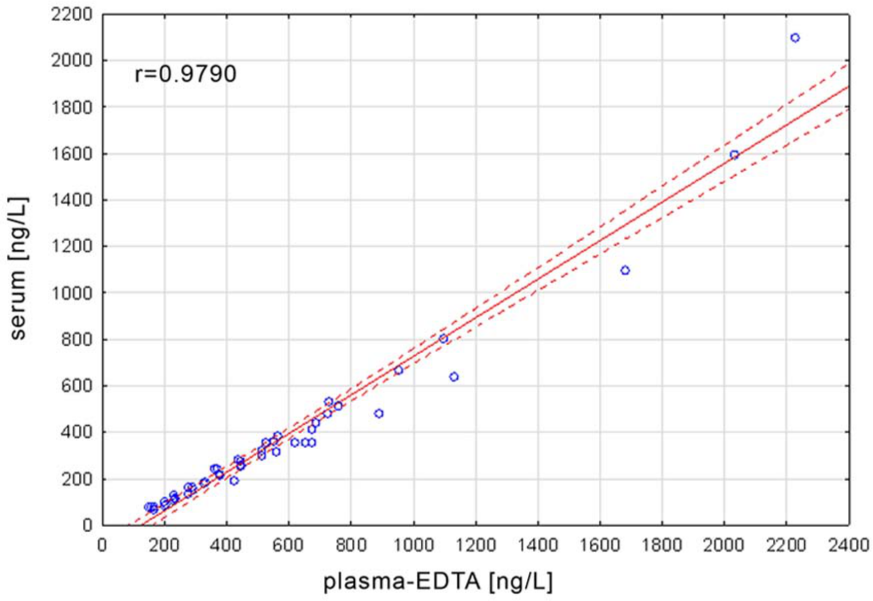
Correlation between aldosterone concentrations measured in serum and plasma-EDTA2K.
Comparison of aldosterone results in plasma (EDTA2K) and serum samples.

r= 0.9790.
Discussion
In the past, blood samples for PRA have been collected and transported to laboratories on ice and centrifuged at 2–4°C to avoid enzyme activation and uncontrolled initiation of angiotensin I production.
Reports of possible renin cryoactivation at low temperature led to initial confusion and the suggestion that blood samples should be processed at room temperature. This controversial question was therefore further investigated.
Brossaud and Corcuff showed that PRA decreased significantly when blood samples were stored for five hours at room temperature as compared to +4°C storage. 7 They stated that within that period enzyme degradation at room temperature was more important than potential cryoactivation, and decided to maintain sample processing at +4°C.
Similarly, Locsei et al. showed that PRA and direct renin decreased significantly when blood samples were kept at room temperature for two hours before processing, but they changed only marginally if blood samples were placed at 0–5°C for the same period of time. 8
From our study it appears that when processing blood samples, cool or room temperature during the initial 20–30 minutes has no significant effect on the results of direct renin, and to a lesser extent, on PRA. Therefore, a refrigerated centrifuge is not necessary at the site of blood drawing, if the blood samples can be further processed either in ambient or cool conditions within 20–30 minutes in the local laboratory. Otherwise, according to suggestions by Locsei et al., they could be placed at 0–5°C and transported to a laboratory in less than two hours 8 or possibly quickly centrifuged and transported frozen on dry ice.
A limitation of this part of our study is that we have tested only blood samples processed within 20–30 minutes and that in the PRA assay we applied only the standard 90-minute incubation time for generation of angiotensin I.
A comprehensive and detailed review concerning methodology of PRA and DRC and precautions necessary to ensure accuracy of these assays was presented by Campbell et al. 9
Determining aldosterone seemed simpler and less problematic. Nevertheless, there are reports of differences in aldosterone results with the use of different aldosterone assays. 5
Currently used assays allow determination of aldosterone both in serum and in plasma, but do not differentiate the range of reference values for them. Therefore we were surprised by the results of our study comparing the aldosterone concentrations in serum and in EDTA-plasma samples obtained from the same subjects at the same time. We observed that using the RIA-CT assay the measured aldosterone concentrations in plasma samples were markedly higher than in serum samples, and these differences ranged from 37% to 144%.
Confirmation of our observation can be found in the study by Taylor et al., who measured aldosterone in plasma and serum using high-performance chromatography and tandem mass spectrometry. In their study aldosterone values were similar for EDTA and heparin plasma samples, but were much lower in the plain clot samples (51.3%) and the serum separator samples (42.2%). 10
Hartman et al., comparing aldosterone results in EDTA-plasma and heparin-plasma samples, obtained concordant results too; however, they did not investigate serum samples from the same patients. 11
Our findings seem to have practical value for laboratory practice because reference values for plasma and serum samples should not be the same.
At this moment, however, we cannot explain the different results in plasma and serum. We can add only that such differences were also reported for some other substances and were considered to be due to the so-called “matrix effect.”
Conclusions
Aldosterone concentrations in plasma (EDTA2K) vs serum samples appeared to be markedly higher and therefore reference values should be calculated separately for plasma and serum samples.
The results of DRC and, to a lesser extent, of PRA, were similar regardless of temperature condition during the approximately 20- to 30-minute period necessary for blood collection, centrifugation and preservation at -30°C.
Footnotes
Conflict of interest
None declared.
Funding
This work was supported by The Medical Centre of Postgraduate Education grant n° 501-1-08-11/12.
