Abstract
Introduction:
This study aimed to investigate whether mononucleotide polymorphisms of the angiotensinogen gene at promoter were associated with the blood-pressure-lowering response to telmisartan treatment.
Materials and methods:
After a two-week single-blind placebo run-in period, 148 patients with mild-to-moderate primary hypertension received monotherapy with 80 mg/day of telmisartan and then were followed up for eight weeks. The -6A/G and -20A/C polymorphisms of the angiotensinogen gene at promoter were determined through polymerase chain reaction and restriction fragment length polymorphsim analysis. The relationship between these polymorphisms and changes in blood pressure was observed and evaluated after eight weeks of treatment.
Results:
There were no significant differences between -6A/G, -20A/C polymorphisms of the angiotensinogen gene and blood pressure reductions after treatment, p>0.05.
Conclusion:
It is suggested that angiotensinogen-6 A/G and angiotensinogen-20 A/C polymorphisms were not associated with the antihypertensive response to telmisartan treatment in Chinese patients with hypertension.
Keywords
Introduction
Essential hypertension (EH) is a complex disorder considered to be the consequence of interactions between genetic and environmental factors. 1 It is established that around 40% of blood pressure (BP) changes are due to genetic heritability and 50% due to environmental factors. 2 Antihypertensive therapy is an important method to treat hypertension. However, the responses of patients to antihypertensive treatment are diverse. 3 Past efforts to identify factors affecting responses to therapy included phenotypic (age, race) 4 and biochemical (renin profile, insulin sensitivity)5,6 methods, but none of these have found extensive clinical utility. The advent of the Human Genome Project has generated rejuvenated interest in the pursuit of pharmacological therapy targeted to individuals genetically identified as most likely to benefit from a specific treatment. Studies have suggested that BP response and outcomes associated with antihypertensive drugs are influenced by genetic variations.7,8
The renin–angiotensin–aldosterone system (RAAS) plays a central role in the modulation of BP and frequently contributes to high BP. 9 Genetic variation in the genes encoding products of the RAAS has been studied extensively as candidate genes for EH.10–13 The major active peptide of RAAS is angiotensin II, the production of hydrolysis of angiotensin I under the action of angiotensin-converting enzyme, the vast majority of which is mediated via the angiotensin II type 1 receptor (AT1R). 14 Angiotensin receptors are found extensively in the body: vascular smooth muscles, adrenal cortical globular cells, and cells in some parts of the brain, heart and kidney organs. Angiotensin II, with the angiotensin receptor, exerts its physiological functions by increasing BP through vasoconstriction, stimulating secretion of aldosterone, exciting the sympathetic nervous system, and so on. 15 Angiotensin receptor blockers (ARBs) are widely used antihypertensive drugs. ARBs act directly on the AT1R to block its vasoconstrictive response to angiotensin II and to inhibit angiotensin II-stimulated release of aldosterone. Variations in genes of the RAAS have been investigated with regard to the therapeutic responsiveness to antihypertensive drugs: however, the results of these studies are controversial.16–19 The aim of this study was to investigate the association between the angiotensinogen (AGT) gene -6A/TG and -20A/C polymorphisms at promoter and the BP response to AT1R blocker telmisartan in Chinese hypertensive patients.
Materials and methods
Study subjects
In this study both male and female Chinese Han patients who met the following criteria were included: age 18–80 years; history of essential hypertension; diastolic blood pressure (DBP) greater than or equal to 95 mmHg and lower than or equal to 109 mmHg; systolic blood pressure (SBP) lower than 180 mmHg. The exclusion criteria were as follows: secondary hypertension, congestive heart failure, cerebrovascular accident, myocardial infarction within the past three months; a documented history of unstable angina pectoris within the past three months; any clinically important abnormal laboratory findings, such as alanine aminotransferase (ALT) or creatinine level twice the upper limit of normal; pregnancy or lactation in women. This study was approved by the appropriate ethics committees. Informed consent was given to all of the participating patients before any study procedures.
Study design
BP was measured by a well-trained doctor or nurse using a mercury sphygmomanometer after the patient had rested for at least 10 min in a seated position, and was determined as the mean of three measurements taken two minutes apart. A total of 164 patients were recruited with EH in our study. All antihypertensive agents were withdrawn before the start of a two-week placebo period. At the end of the placebo period, 11 patients were excluded and a total of 153 qualified patients were given telmisartan, 80 mg orally, once daily for eight weeks. Five patients withdrew from the study because of failing to follow up. Thus, 148 patients completed the eight-week trial, and their data were used for the present study. Blood was collected from all patients for genotype analysis.
Definition of study variables
Baseline information on the following variables was included in the analysis: gender, age, body mass index (BMI; kg/m2), BP, heart rate (HR), laboratory variables included the serum levels of ALT, creatinine, glucose, uric acid, total cholesterol (TC), triglyceride (TG), high-density lipoprotein (HDL) and low-density lipoprotein (LDL), sodium, potassium, and angiotensin II.
Detection of AGT gene polymorphisms
Genomic DNA was isolated from peripheral leukocytes separated from red blood cells according to the procedure of Tilzer et al. 20 The AGT gene polymorphisms were determined by polymerase chain reaction (PCR) and restriction fragment length polymorphsim (RFLP). A 20-µL PCR reaction mixture contained 10× PCR buffer 10 µL, 1.5 mM MgCl2 2 µL, 0.1 mM each dNTP 2 µL, 0.5 µM primer 2 µL, 1 U Taq enzyme (Takara) 2 µL and 100 ng genomic DNA 2 µL. The primers for the two polymorphic sites are shown in Table 1. The annealing temperatures of the two variant sites PCR were set up differently as -6 at 59°C and -20 at 62°C. After PCR amplification, restriction enzymes DsiE I and Eco0109 I (New England Biochemicals) were respectively applied to digest the PCR products for the -6 and -20 sites. The digested PCR products were then loaded on a 3% ethidium bromide-stained agarose gel to identify the product sizes.
Primer sequences used for amplification of angiotensinogen (AGT) single nucleotide polymorphisms (SNPs).

Statistical analysis
Data are reported as means±standard deviation (SD). The χ2 test was used to evaluate categorical variables and test for Hardy–Weinberg equilibrium (HWE) of polymorphisms. Differences in biochemical parameters and BP were assessed using a paired t-test. Linear regression modeling was used to determine the correlation of BP response with genotypes, following normalization with pretreatment BP, age, gender, BMI, blood glucose, TC, TG, HDL, and LDL. SPSS 20 software was applied for statistical analysis. A two-tailed p value less than 0.05 was considered to be significant.
Results
Characteristics of the study subject
Data from a total of 148 patients was analyzed in this study. All patients had mild-to-moderate hypertension. The basic characteristics of patients are presented in Table 2. After eight-week treatment with telmisartan, both SBP and DBP were remarkably decreased (p<0.001). The serum angiotensin II level was obviously increased at the end of the eight-week treatment (p<0.001). No significant differences were observed for the other parameters.
Changes of patient characteristics and blood pressure (BP) response to the treatment.
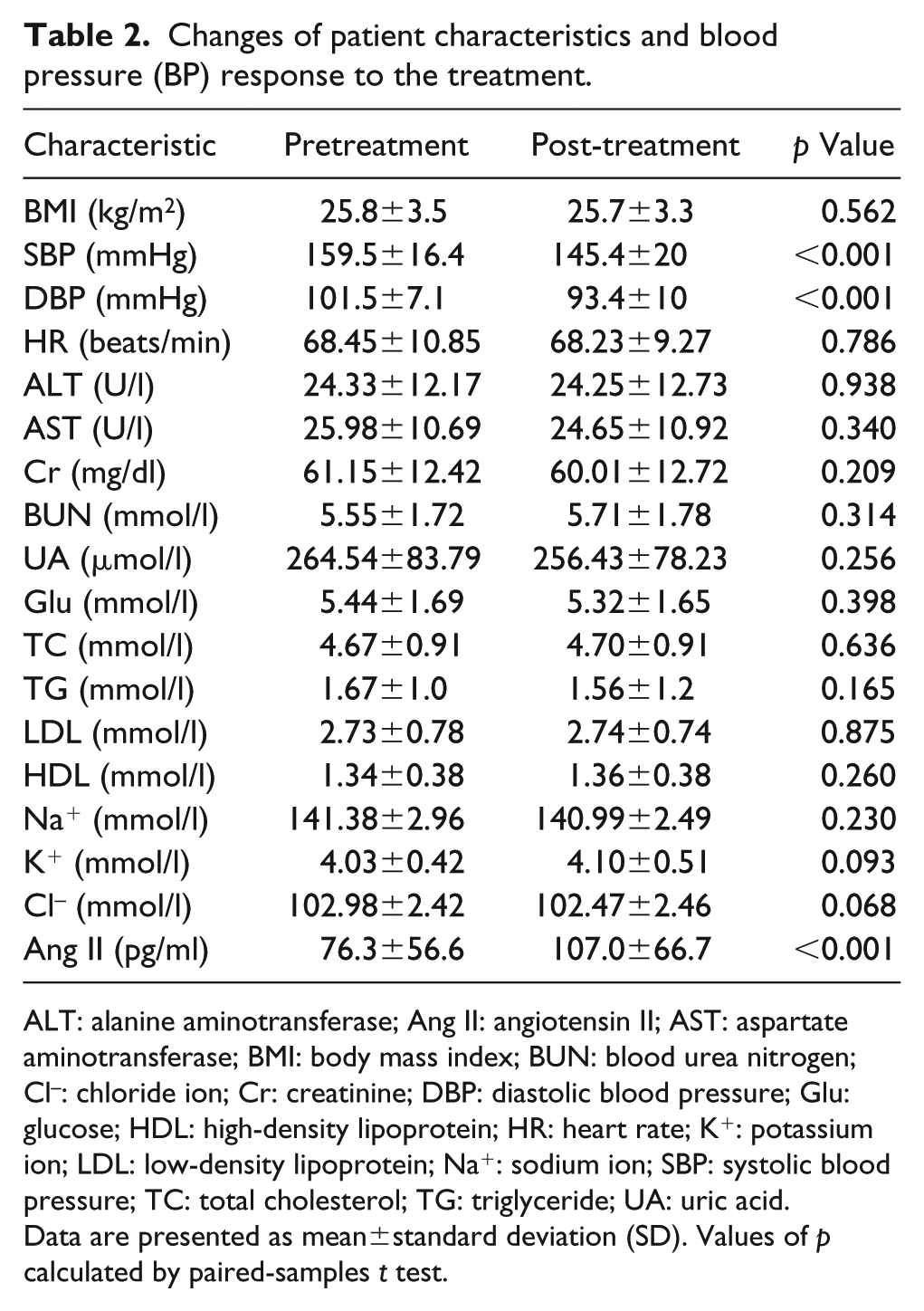
ALT: alanine aminotransferase; Ang II: angiotensin II; AST: aspartate aminotransferase; BMI: body mass index; BUN: blood urea nitrogen; Cl–: chloride ion; Cr: creatinine; DBP: diastolic blood pressure; Glu: glucose; HDL: high-density lipoprotein; HR: heart rate; K+: potassium ion; LDL: low-density lipoprotein; Na+: sodium ion; SBP: systolic blood pressure; TC: total cholesterol; TG: triglyceride; UA: uric acid.
Data are presented as mean±standard deviation (SD). Values of p calculated by paired-samples t test.
Genotype and antihypertensive response to telmisartan
Genotype and allele distributions of the study population are summarized in Table 3. Genotype and allele frequencies of each polymorphism in the study population were in Hardy–Weinberg equilibrium (p>0.05). The changes in BP response to antihypertensive treatment in relation to genotypes of AGT gene polymorphisms are shown in Table 4. Four subjects carrying homozygous AGT-6G allele were found, so GG and AG genotypes were used in combination for analyses. Homozygous AGT-20C allele was also found in four subjects, so CC and AC genotypes were used in combination for analyses. We did not find any association of AGT-6A/G and -20 A/C polymorphisms with SBP or DBP responses to telmisartan.
Genotype and allele frequencies of the -6A/G and -20A/C polymorphisms of the angiotensinogen (AGT) gene in the study population.

Single nucleotide polymorphisms (SNPs) of angiotensinogen (AGT) gene in linear regression and blood pressure (BP) response to antihypertensive drugs.

Two SNPS were first respectively considered by themselves (alone) as a predictor of the BP response, then after adjustment for the concomitant variables including pretreatment BP, age, gender, body mass index, glucose, triglycerides, total cholesterol, high-density lipoprotein, and low-density lipoprotein (after covariates). Standardized β is the standardized regression coefficient; R2 is the percentage of interindividual variation in BP response explained by predictors in the model.
Discussion
AGT is a protein that interacts with renin to produce angiotensin I, the prohormone of angiotensin II. The level of AGT in plasma directly affects production of angiotensin II. Jeunemaitre et al. first identified the role of AGT gene polymorphism in human EH. 21 They found a 17% excess of AGT gene in hypertensive sibling pairs in linkage studies and estimated that mutations at the AGT gene might be a predisposing factor in at least 3–6% of individuals younger than 60 years of age with hypertension. The AGT gene is located on chromosome 1q42-q43, with a total length of about 12 KB and is composed of five exons and four introns. The core promoter region is located in the TATA box and transcription start site, AGT gene core promoter region upstream of a cis element hAG core promoter element 1 combined with core promoter transcription factors AGT factor 1, with the sequence upstream and downstream of the 3 ‘end enhancer element interaction, influence the basis of AGT gene transcription rate. 22
AGT-6 A/G and -20 A/C are located in the transcriptional regulatory region of AGT, affecting the transcriptional activity of AGT genes. 23 Liu et al. reported that there may be a synergistic effect between AGT-6 A/G and -20 A/C polymorphisms and exon 2 235M/T polymorphism, which may be related to the occurrence of primary hypertension in Chinese Han people. 24 Gu et al. performed a meta-analysis (5932 patients) to assess the association of the A-6G and A-20C polymorphisms in the promoter region of AGT gene and hypertension and found that these two polymorphisms were associated with EH in the Chinese population. 25 Markovic et al. confirmed that AGT gene -6 A/G and -20 A/C polymorphisms are associated with primary hypertension in African-Americans and Caucasian populations. 26 However, some studies have not found a correlation between AGT gene -6 A/G and -20 A/C polymorphisms and primary hypertension.27,28
Several studies evaluated the impact of the AGT gene on the response to antihypertensive drugs. Jiang et al. have examined the relationship between the RAAS genotypes including -6A/G polymorphisms of the AGT gene and the BP response to diuretic treatment in a Chinese Han ethnic population. 29 They found that BP dropped more after diuretics in patients with AGT-6 G allele than in patients with AGT AA genotype (p=0.025). Filigheddu et al. studied the relationship between the polymorphism of RAAS gene and the effect of fosinopril on hypertension, and found no correlation between AGT gene -6 A/G polymorphism and the effect of fosinopril on hypertension. 30 At present, there are few reports on the correlation between the AGT gene -20 A/C and the therapeutic effect of antihypertensive drugs. In this study, we investigated the relationship between the AGT gene promoter region -6 A/G and -20 A/C polymorphisms and telmisartan antihypertensive effect after telmisartan was given to patients with mild and moderate hypertension of the Han nationality for eight weeks. No association was found between AGT-6 A/G and -20 A/C genotypes and the telmisartan antihypertensive effect.
Hypertension is a polygenic disease and antihypertensive drug treatment is the most important way to control hypertension clinically. “Personalized medicine” as an effective method can improve outcomes and reduce costs. How to select the most suitable antihypertensive drugs for each patient and conduct individualized treatment has become a new challenge. Specific genotypes might predict the response to specific antihypertensive treatment. A limitation of this study is due to the relatively small size, and more prospective studies should be carried out to confirm the results.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
