Abstract
Objective:
The objective of this article is to investigate the renoprotecive effects of exendin-4 in a mouse model of unilateral ureteral obstruction (UUO) and explore the putative mechanisms.
Methods:
Male Balbc mice underwent sham operation or UUO surgery, and then received intraperitoneal injection of vehicle or exendin-4, respectively. After 14 days, mice were sacrificed and the left kidneys were collected and analyzed by histology, immunohistochemistry, Western blot, quantitative real-time reverse transcription polymerase chain reaction, radioimmunoassay and enzyme-linked immunosorbent assay.
Results:
As compared to the sham group, mice that underwent UUO surgery developed more severe tubular injury and interstitial fibrosis, as well as higher expression of fibronectin (FN), collagen-1 (Col-1) and α-smooth muscle actin (α-SMA). Also, we observed higher expression of angiotensin-converting enzyme (ACE) while lower expression of angiotensin-converting enzyme 2 (ACE2), higher levels of intrarenal angiotensin II (Ang II) while lower levels of intrarenal angiotensin-(1–7), and higher expression of transforming growth factor β1 (TGF-β1) and phosphorylation of Smad3 (p-Smad3) in the obstructed kidneys. Impressively, these pathologic changes were significantly attenuated in the mice group of UUO treated with exendin-4.
Conclusion:
Our present study indicates for the first time that exendin-4 exerts renoprotective effects in an experimental model of UUO, partly through regulating the balance of the intrarenal renin-angiotensin system and then inhibiting the Ang II-mediated TGF-β1/Smad3 signaling pathway.
Keywords
Introduction
Chronic kidney disease (CKD) affects about 13% of the adult population and has already become a health care problem worldwide. Further, numerous studies show that a considerable proportion of CKD patients will eventually develop end-stage renal disease or cardiovascular death, highlighting the need for better prevention and management for CKD.1,2 Interstitial fibrosis, characterized by excessive deposition of extracellular matrix (ECM) proteins in the tubulointerstitial compartment, is thought to be the common final pathway of all forms of CKD, whose leading cause is diabetic nephropathy.3,4 Therefore, it is mandatory to develop rational strategies aiming at interstitial fibrosis to combat the progression of CKD.
Although the exact mechanism of CKD is not yet fully elucidated, accumulating evidence has pointed to the critical role of excessive activation of the intrarenal renin-angiotensin system (RAS) in the pathogenesis of CKD.5-7 Among this fascinating system, angiotensin II (Ang II) is the most powerful biologically active component and considered to be a central mediator of interstitial fibrosis.5,8,9 However, our recognition of the RAS has become more convoluted since the discovery of a novel enzyme called angiotensin-converting enzyme 2 (ACE2) in 2000. It shares 42% homology with angiotensin-converting enzyme (ACE) and mediates the degradation of Ang II to angiotensin-(1–7) (Ang-(1–7)), acting in a counterregulatory manner to ACE.6,7 Therefore, the balance between the two enzymes affects the production and accumulation of Ang II. Considerable research has observed that the high expression of ACE as well as low expression of ACE2 results in excessive accumulation of intrarenal Ang II, which plays a detrimental role in kidney diseases.10-12 Overall, the above findings support the idea that restoring the balance between ACE and ACE2 and hence decreasing the level of intrarenal Ang II may serve as a target for CKD treatment.
Exendin-4, a glucagon-like protein-1 (GLP-1) analog, was originally isolated from the venom of the Gila monster lizard (Heloderma suspectum). 13 Nowadays, synthetic exendin-4 has been widely used in the treatment of patients with type 2 diabetes. Moreover, many studies have indicated that exendin-4 exerts renoprotective effects through various mechanisms in addition to the control of glucose homeostasis.14-18 However, until now, it has been unclear whether exendin-4 modified the activity of the intrarenal RAS as part of the underlying mechanisms for renoprotection.
Therefore, the present study was designed to explore the possibility that exendin-4 attenuates interstitial fibrosis by regulating the balance of the intrarenal RAS and then inhibiting the Ang II-mediated transforming growth factor β1 (TGF-β1)/Smad3 signaling pathway. We examined the effect of exendin-4 on a mouse model of unilateral ureteral obstruction (UUO), an excellent model to study the pathogenesis of CKD without underlying hypertensive and diabetic conditions.
Material and methods
Animals
All animal procedures were performed in accordance with the guidelines of the Laboratory Animal Committee at the Southern Medical University. Eight-week-old BALB/c mice (weighing 20 to 25 g each) were randomly divided into four groups, which consisted of a sham group (n = 6), a sham treated with exendin-4 group (n = 6), a UUO group (n = 8) and a UUO treated with exendin-4 group (n = 6). The UUO model was established under intraperitoneal pentobarbital-induced anesthesia. Then the left ureter was visualized via a flank incision and ligated with 4-0 silk. The sham operation was performed identically but without ureter ligation. Mice received an intraperitoneal injection of either exendin-4 (Sigma-Aldrich, USA) at a dose of 36 ug/kg per day or vehicle, respectively. After 14 days, mice were sacrificed and the left kidneys were collected for further analysis.
Quantitative real-time polymerase chain reaction (PCR)
Total RNA was obtained from renal cortex tissues using TRIzol Reagent (Takara, Otsu, Japan) and then was used to synthesize the complementary DNA (cDNA) using M-MLV Reverse Transcriptase (Invitrogen, Carlsbad, USA). Quantitative real-time PCR was performed on an ABI 7500 Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). The primers used are listed in Table 1.
Sequences of quantitative real-time PCR primers used in the present study.

PCR: polymerase chain reaction; FN: fibronectin; Col-1: collagen-1; α-SMA: α-smooth muscle actin; TGF-β1: transforming growth factor β1; ACE: angiotensin-converting enzyme; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Western blot
Western blot analysis was performed as described previously. 19 Briefly, equal amounts of proteins were electrophoresed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and electrotransferred to polyvinylidene difluoride (PVDF) membranes (Merck Millipore, MA, USA). After blocking with 5% non-fat dry milk or 5% bovine serum albumin (BSA) dissolved in Tris-buffered saline with Tween 20 (TBST) for one hour, the membranes were incubated at 4°C with gentle shaking overnight with each of the antibodies listed in Table 2. Then the membranes were washed three times for 10 minutes with TBST and incubated with horseradish peroxidase-conjugated secondary antibodies (LI-COR, NE, USA) for one hour. The bands were visualized with Odyssey infrared scanning (LI-COR) and densitometry was performed using Gel-Pro32 software.
Characteristic of antibodies used in the present study.

FN: fibronectin; Col-1: collagen-1; p-Smad3: phosphorylation of Smad3; α-SMA: α-smooth muscle actin; TGF-β1: transforming growth factor β1; ACE: angiotensin-converting enzyme; GAPDH: glyceraldehyde 3-phosphate dehydrogenase; WB: Western blot; IHC: immunohistochemistry.
Histological and immunohistochemical analysis
The kidney tissues were fixed in 4% paraformaldehyde. Samples were embedded in paraffin and then cut into slices with a thickness of 4 um. Hematoxylin and eosin (H&E) (Leagene, Beijing, China) and Masson’s trichrome (Maixin, Fuzhou, China) stain kit were used to assess morphological changes and degree of interstitial fibrosis, respectively. Immunohistochemical staining was performed to assess the renal tissue protein expression of fibronectin (FN), collagen-1 (Col-1), α-smooth muscle actin (α-SMA), ACE and ACE2 as previously described. 19 All sections were observed under an Olympus B×40 photomicroscope (Olympus, Tokyo, Japan). Five randomly selected cortical fields per mouse were assessed using Image Pro-plus 6.0 software.
Radioimmunoassay (RIA)
Kidney amounts of Ang II were measured with an Iodine [125I] Angiotensin II Radioimmunoassay kit (Beijing North Institute of Biological Technology, Beijing, China) according to the manufacturer’s instructions.
Enzyme-linked immunosorbent assay (ELISA)
The concentration of Ang-(1–7) was determined using a mouse Ang-(1–7) ELISA kit (CSB-E13763m; Cusabio Biotech). Each sample contained the same amount of protein and the absorbance was read at 450 nm using an EL×800 microplate reader (BioTek, VT, USA).
Statistical analysis
All results were presented as the mean ± standard deviation (SD). Statistical analysis was performed using SPSS software version 19.0 (SPSS Inc, Chicago, IL, USA). The comparison of values between groups was measured by one-way analysis of variance (ANOVA), followed by a least-significant difference (LSD) or a Dunnett’s T3 post hoc test. A p < 0.05 was considered statistically significant.
Results
Effect of exendin-4 on pathological changes of the obstructed kidneys
As seen in Figure 1(a), H&E staining revealed normal architecture of tubules in the sham group and severe tubular injury in the UUO group. In contrast, exendin-4 treatment significantly reduced tubular injury of the obstructed kidneys as compared to the UUO mice. Masson’s trichrome staining also showed that UUO mice developed more interstitial fibrosis compared with the sham group. However, interstitial fibrosis was significantly attenuated by exendin-4 treatment compared with UUO mice (Figure 1(b)).

Effect of exendin-4 on pathological changes of the obstructed kidneys. sham+Ex-4: sham + exendin-4; UUO: unilateral ureteral obstruction; UUO + Ex-4: unilateral ureteral obstruction + exendin-4.
Effect of exendin-4 on messenger RNA (mRNA) expression of FN, Col-1 and α-SMA
As shown in Figure 2, the obstructed kidneys in the UUO group exhibited increased mRNA levels of FN, Col-1 and α-SMA as compared to the sham mice with or without exendin-4 treatment (p < 0.01). However, exendin-4 treatment significantly reduced the mRNA expression of FN and α-SMA (p < 0.01 and p < 0.05, respectively). But no significant difference in renal cortical mRNA expression of Col-1 was observed between the UUO and UUO treated with exendin-4 groups (p > 0.05).
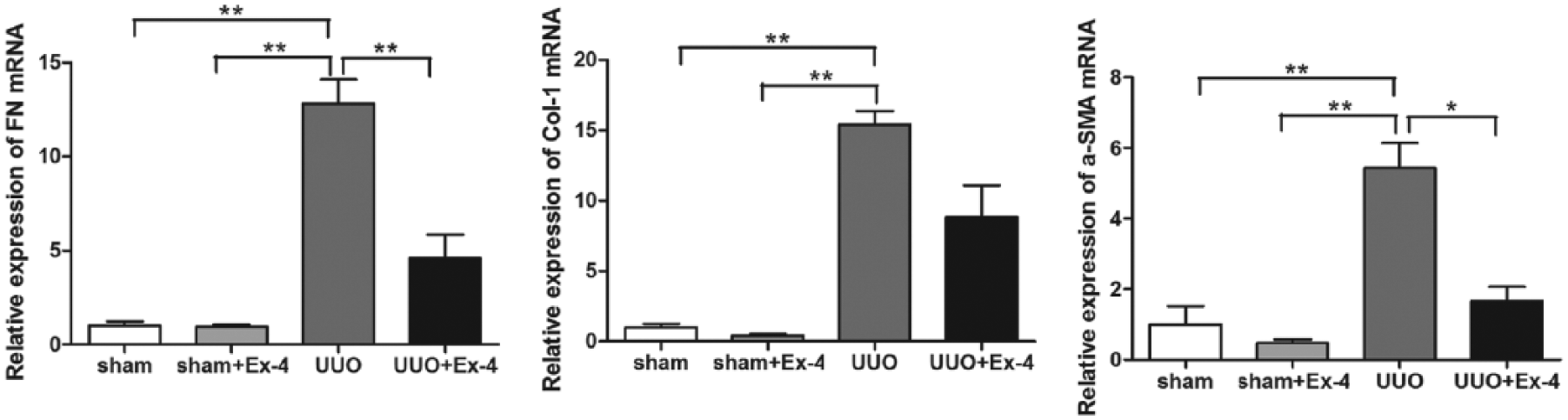
Effect of exendin-4 on mRNA expression of FN, Col-1 and α-SMA. sham+Ex-4: sham + exendin-4; UUO: unilateral ureteral obstruction; UUO + Ex-4: unilateral ureteral obstruction + exendin-4; FN: fibronectin; α-SMA: α-smooth muscle actin; mRNA: messenger RNA.
Effect of exendin-4 on protein expression of FN, Col-1 and α-SMA
Insets of Figure 3 show the protein expression of FN, Col-1 and α-SMA. Immunohistochemical staining results revealed that UUO caused a marked increase in kidney cortical expression of FN, Col-1 and α-SMA compared with sham mice. In contrast, mice treated with exendin-4 exhibited less ECM protein expression compared with the obstructed kidneys (Figure 3(a) to (d)). In accordance with the immunohistochemistry results, studies by Western blot analyses further confirmed the above results. The protein expression level of α-SMA was also reduced in the UUO treated with exendin-4 group compared with the UUO group, although the difference did not reach statistical significance (p > 0.05) (Figure 3(e) and (f)).

Effect of exendin-4 on protein expression of FN, Col-1 and α-SMA. sham+Ex-4: sham + exendin-4; UUO: unilateral ureteral obstruction; UUO + Ex-4: unilateral ureteral obstruction + exendin-4.
Effect of exendin-4 on mRNA expression of ACE and ACE2
Next, we evaluated the effect of exendin-4 on mRNA expression of ACE and ACE2. As shown in Figure 4, a significantly increased concentration of ACE mRNA was detected in the obstructed kidneys of UUO mice compared with the control sham kidneys (p < 0.05). Consistent with an increase in ACE mRNA level, the mRNA expression level of ACE2 was reduced in the obstructed kidneys, although this did not reach statistical significance compared with the control sham kidneys (p > 0.05). Notably, exendin-4 administration restored the imbalance between ACE and ACE2 mRNA expression, which exhibited a downregulation of ACE mRNA and upregulation of ACE2 mRNA in the UUO treated with exendin-4 group compared with mice in the UUO group (p > 0.01 and p < 0.05, respectively).

Effect of exendin-4 on mRNA expression of ACE and ACE2. sham+Ex-4: sham + exendin-4; UUO: unilateral ureteral obstruction; UUO + Ex-4: unilateral ureteral obstruction + exendin-4; FN: fibronectin; α-SMA: α-smooth muscle actin.
Effect of exendin-4 on protein expression of ACE and ACE2
We also investigated the effect of exendin-4 on protein expression of ACE and ACE2 as shown in Figure 5. Similarly, an imbalance between ACE and ACE2 protein expression was also detected by immunohistochemistry analyses in accordance with the real-time polymerase chain reaction (RT-PCR) results. However, these changes were significantly attenuated in the obstructed kidneys by treatment with exendin-4 (Figure 5(a) to (c)). The results of Western blot analyses were consistent with the above findings (Figure 5(d) and (e)).

Effect of exendin-4 on protein expression of ACE and ACE2. sham+Ex-4: sham + exendin-4; UUO: unilateral ureteral obstruction; UUO + Ex-4: unilateral ureteral obstruction + exendin-4; FN: fibronectin; α-SMA: α-smooth muscle actin; GAPDH: glyceraldehyde 3-phosphate dehydrogenase.
Effect of exendin-4 on intrarenal Ang II and Ang-(1–7) levels
Intrarenal Ang II and Ang-(1–7) levels were measured with the expectation that they would reflect the imbalance in ACE and ACE2 expression observed in the kidneys. Therefore, we explored the kidney levels of Ang II and Ang-(1–7) and then evaluated the effect of exendin-4 on intrarenal Ang II and Ang-(1–7) levels as shown in Figure 6. Compared with the control sham kidneys, significantly higher levels of intrarenal Ang II and lower levels of Ang-(1–7) (p < 0.01) were detected in obstructed kidneys of UUO mice. Interestingly, treatment of UUO mice with exendin-4 caused a significant reduction in intrarenal Ang II levels compared with mice in the UUO group (p < 0.05), whereas renal Ang-(1–7) levels were elevated in response to exendin-4 treatment, although this increase did not reach statistical significance (p > 0.05).

Effect of exendin-4 on intrarenal Ang II and Ang-(1-7) levels. sham+Ex-4: sham + exendin-4; UUO: unilateral ureteral obstruction; UUO + Ex-4: unilateral ureteral obstruction + exendin-4; GAPDH: glyceraldehyde 3-phosphate dehydrogenase; α-SMA: α-smooth muscle actin.
Effect of exendin-4 on TGF-β1/Smad3 signaling pathway in UUO mice kidneys
Subsequently, we explored whether enhanced interstitial fibrosis in the UUO kidneys was associated with an upregulation of TGF-β1 as well as the phosphorylation of Smad3 levels (p-Smad3) and whether exendin-4 could inhibit p-Smad3 in UUO kidneys. As shown in Figure 7, both mRNA and protein expression levels of TGF-β1 were significantly higher in obstructed kidneys than those in control mice (p < 0.05). Moreover, p- Smad3 in renal tissue was significantly increased in UUO mice compared with sham controls (p < 0.01). Treatment with exendin-4 significantly inhibited TGF-β1 as well as p-Smad3.

Effect of exendin-4 on TGF-β1/Smad3 signaling pathway. sham+Ex-4: sham + exendin-4; UUO: unilateral ureteral obstruction; UUO + Ex-4: unilateral ureteral obstruction + exendin-4; ACE: angiotensin-converting enzyme; mRNA: messenger RNA.
Discussion
Our data provide compelling evidence for the first time that exendin-4 attenuates interstitial fibrosis partially by regulating the balance between intrarenal ACE and ACE2 and then inhibiting the Ang II-mediated TGF-β1/Smad3 signaling pathway.
UUO is the most-used animal model to represent the main pathological feature of CKD. Concretely, it is characterized by tubular cell injury, recruitment of interstitial inflammatory cells, proliferation of interstitial fibroblasts and increased deposition of ECM proteins. Among them, tubular epithelial cells differentiate into myofibroblasts in the obstructed kidneys and the activated interstitial fibroblasts are responsible for the increased synthesis of α-SMA and ECM proteins.20,21 In our experiments, the levels of α-SMA and ECM proteins are effectively elevated in obstructed kidneys. Furthermore, we have observed severe tubular injury and remarkably increased collagen deposition in the interstitium of obstructed kidneys detected by H&E and Masson’s trichrome staining, respectively, suggesting that we have successfully induced the feature of interstitial fibrosis and created the experimental model of CKD.
Accumulating evidence indicates that discordance of ACE and ACE2 will lead to higher concentration of intrarenal Ang II, contributing to the development of renal injury. Ye et al. demonstrated that increased ACE with decreased ACE2 was found in the kidney tissues of diabetic mice, which fostered glomerular accumulation of Ang II and contributed to the development of kidney injury. 10 Another study showed that the consumption of a high-salt diet in rats significantly increased the glomerular ACE/ACE2 ratio, leading to oxidative stress and renal damage. 11 An in vivo study also detected increased intrarenal ACE and decreased ACE2 expression in patients with type 2 diabetic nephropathy, which might contribute to renal damage. 12 Here, we have observed that the decreased ACE2 as well as increased ACE at mRNA and protein levels is found in the renal cortical tissues from UUO mice. Additionally, this combination leads to excessive intrarenal Ang II accumulation detected by RIA, which may relate to the renal damage in UUO mice.
Ang II plays a pivotal role in renal fibrogenesis. It promotes the growth of renal mesangial and tubular cells, modulates epithelial-mesenchymal transition (EMT) and increases the expression and synthesis of ECM proteins depending on multiple fibrotic mediators such as TGF-β1, connective tissue growth factor, endothelin-1, matrix metalloproteinase-2 and plasminogen activator inhibitor-1. Among them, Ang II may foster renal fibrosis mainly by interactions with TGF-β1.5,9 In cultured mesangial cells, Ang II could directly stimulate TGF-β1 gene activation through protein kinase C (PKC)- and p38 mitogen-activated protein kinase (MAPK)-dependent pathways. 22 Further, TGF-β1 regulates kidney injury primarily by Smad3. Deletion of Smad3 from mice ameliorated renal fibrosis in rodent models of obstructive nephropathy. 23 Additionally, the use of Smad3 inhibitor to EMT and renal fibrosis in streptozotocin (STZ)-induced diabetes provided a novel strategy to retard the progression of kidney disease by targeting Smad3 signaling. 24 In our experiments, we find that significantly higher levels of intrarenal Ang II as well as the expression of TGF-β1 and p-Smad3 are detected in obstructed kidneys of UUO mice compared with the control sham kidneys, which is consistent with previous research.
Exendin-4 is a sound agent for use in the comprehensive treatment of patients with type 2 diabetes mellitus (T2DM) by high-affinity GLP-1 receptor (GLP-1R) in the pancreas. The first study to explore the renoprotective effects of exendin-4 was performed in 2007. It confirmed that eight weeks of exendin-4 treatment inhibited 8-hydroxy-deoxyguanosine excretion and decreased albuminuria in T2DM db/db mice. 14 Then studies were undertaken in STZ-induced T1DM models. Kodera et al. reported that exendin-4 attenuated kidney injury through anti-inflammatory actions without lowering blood glucose levels. 15 Mima et al. indicated that exendin-4 induced protective actions on the glomerular endothelial cells by inhibiting Ang II signaling on c-Raf (Ser259). 16 Ojima et al. investigated the renoprotective effect of exendin-4 by blocking the advanced glycation endproduct-receptor for advanced glycation endproduct (AGE-RAGE)-mediated asymmetric dimethylarginine (ADMA) generation. 17 Our group performed an experiment in vitro revealing that exendin-4 exerted an inhibitory effect on cell proliferation and FN secretion in rat mesangial cells partly through 5’ AMP-activated protein kinase (AMPK) activation. 18 Consistent with previous reports, our study demonstrates that exendin-4 effectively alleviates renal injury in a mouse model of UUO nephropathy.
However, the mechanisms responsible for improved renal injury after exendin-4 treatment are complex and not fully understood. Interestingly, to date, there have been many studies exploring the interactions between GLP-1 and the RAS. Skov et al. observed GLP-1 significantly decreased circulating concentration of Ang II. 25 Moreover, it has previously been reported that GLP-1 effectively ameliorates Ang II-induced hypertension as well as glomerular endothelial cell and mesangial cell damage.16,26-28 Based on all this research, we speculate that inhibition of the intrarenal RAS may be involved in the mechanisms of GLP-1-mediated renoprotection. Here, our study discovers that exendin-4 is very effective in restoring the balance of the intrarenal RAS in obstructed kidneys by decreasing the expression of ACE and increasing the expression of ACE2. Interestingly, treatment of UUO mice with exendin-4 causes a significant reduction in intrarenal Ang II levels and then inhibits the Ang II-mediated TGF-β1/Smad3 signaling pathway. To our knowledge, our study is the first to report the ability of exendin-4 to alleviate interstitial fibrosis resulting from UUO by regulating the balance between ACE and ACE2 and then inhibiting the Ang II-mediated TGF-β1/Smad3 signaling pathway.
In addition, another interesting finding of our study is that lower levels of Ang-(1–7) are detected in obstructed kidneys of UUO mice compared with the control sham kidneys, and intrarenal Ang-(1–7) levels are elevated in response to exendin-4 treatment, although this increase did not reach statistical significance. To date, it has been reported that Ang-(1–7) may have renoprotective properties.29,30 Whether Ang-(1–7) has influence on interstitial fibrosis in UUO mice needs to be confirmed in future research. Another limitation of the current study is that we have demonstrated mechanisms described only in animal models of UUO and are unable to provide direct evidence of exendin-4 on the intrarenal RAS.
In conclusion, using the UUO as a model of CKD, for the first time we demonstrate that the changes in the expression of ACE and ACE2 in obstructed kidneys, the alterations in the levels of Ang II and Ang-(1–7), and the onset of renal fibrosis can be reversed by exendin-4 administration. We present a novel strategy to balance the activities of ACE and ACE2, thus suppressing the activated Ang II-mediated TGF-β1/Smad3 signaling. Our results suggest that the intrarenal RAS has become a more promising antifibrotic target of GLP-1R agonists.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by research grants from the National Natural Science Foundation of China (grant number 81570724) and the Natural Science Foundation of Guangdong (grant number 2014A030313290).
