Abstract
Introduction:
The effects of renin–angiotensin system blockade with angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin II type 1 receptor blockers (ARBs) on cancer remain inconsistent.
Methods:
We searched existing databases from 1960 to August 2015, for randomised controlled trials and observational studies (case–control studies and cohort studies) of ARB/ACEI therapy with a minimal one year of follow-up. Outcomes were incidence and mortality of cancer.
Results:
We included 14 randomised controlled trials and 17 observational studies of 3,957,725 participants (350,329 ARB/ACEI users). The users had a lower incidence of cancer in the observational studies (RR 0.82, 95% CI 0.73–0.93) but not in the randomised controlled trials (RR 1.00, 95% CI 0.92–1.08). The protection persisted for lung cancer (RR 0.85, 95% CI 0.75–0.97) but not for other sites of cancer. The relative risk of cancer associated with renin–angiotensin system blockade was reduced along with time of follow-up. Mortality reduction with ARB/ACEI was marginally significant in the observational studies (RR 0.71, 95% CI 0.55–0.93) but not in the randomised controlled trials (RR 0.99, 95% CI 0.89–1.09).
Conclusions:
The significant benefits of renin–angiotensin system blockade observed in case–control studies and cohort studies might diminish in randomised controlled trials. Clinical design, site of cancer and duration of follow-up may affect the clinical outcomes.
Keywords
Introduction
The renin–angiotensin system (RAS) is a key therapeutic target for diabetes mellitus, chronic kidney disorders, hypertension, heart problems, chronic obstructive pulmonary disease and stroke. RAS blockers include angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin II type 1 receptor blockers (ARBs). The impacts of RAS blockade on the incidence and mortality of cancer remain debated. Some studies suggest that the use of ACEIs and ARBs may increase the risk of cancer. 1 A meta-analysis showed an increased risk of cancer by ARBs compared with controlled therapy. 2 Intriguingly, the US Food and Drug Administration claims no increase in the risk of cancer with ARBs. 3
Angiotensin II and angiotensin II type 1 receptors (AT1) play major roles in the development and progression of cancer.4,5 Angiotensin II acts on the AT1 receptor to promote cell proliferation and angiogenesis.6–8 The expression of AT1 receptors has been reported to be upregulated in hyperplasic and cancer tissues.9,10 ACEIs prevent the generation of angiotensin II by inhibiting angiotensin-converting enzymes (ACEs) while ARBs selectively block angiotensin II binding to the AT1 receptor. These actions may have important implications for cancer development. However, the existing clinical evidence is inconsistent.2,11,12 Therefore we conducted a systematic review and meta-analysis to evaluate the impact of an RAS blockade with ACEI/ARB therapy on the risk of cancer and death.
Materials and methods
Search strategy
Candidate studies were identified through electronic literature searches of PubMed, Cochrane Library databases, Chinese National Knowledge Infrastructure (CNKI) and Wanfang databases from 1960 to August 2015. We used the following MeSH terms and keywords: ‘cancer’, ‘carcinoma’, ‘sarcoma’, ‘neoplasia’ or ‘malignancy’ in combination with ‘renin–angiotensin system’, ‘RAS’ and ‘angiotensin-receptor blocker’, ‘ARB’ or ‘angiotensin-converting enzyme inhibitor’, ‘ACEI’. A manual search of reference lists from reports of review articles, meta-analyses and original studies was performed to identify additional relevant studies.
Selection criteria
Our inclusion criteria were as follows: (a) clinical trials, including randomised controlled trials (RCTs), cohort studies and case–control studies; (b) use of ACEIs and/or ARBs in the participants; (c) incidence and/or mortality due to cancer as an outcome with detailed description of relative risk ratios (RRs), corresponding 95% confidence intervals (CIs), size of the baseline samples and years of follow-up; and (d) each study should have enrolled at least 200 participants. Literature meeting any of the following criteria was excluded: non-clinical nature, non-human studies, duplication, unclear outcome evaluation and non-original studies including reviews, letters, editorials and commentaries.
Data extraction
The extracted data included first author name, study title, year of publication date, country of origin, disease, demographic characteristics of participants, details of intervention, outcome measurements, intervention durations, incidence and mortality of cancer and RR for cancer with the corresponding 95% CI. All articles were read by two independent reviewers (JS and XZ) who extracted data from the articles according to a standardised data extraction form. Disagreements were resolved in all cases by discussion among our team members.
Quality assessment
The methodological quality of studies was assessed by the Newcastle–Ottawa scale (NOS). Using the NOS, a study is judged on three broad perspectives: the selection of the study groups, the comparability of the groups and the ascertainment of the outcome of interest. 13 Studies with a score of less than 3 were considered as low quality, while scores of 4–6 were considered as moderate quality and 7–9 were considered as high quality. All studies were reviewed by two investigators (JS and X-NS). A third reviewer (H-LZ) served to resolve disputes.
Statistical analyses
This study is reported in accordance with the preferred reporting items for systematic reviews and meta-analyses (PRISMA) statement.
14
Dichotomous outcome data from individual trials were analysed by using RR and corresponding 95% CI. Data were pooled using the random effects model or fixed effect model according to the heterogeneity between studies. Heterogeneity was assessed using the chi-square test, with values greater than 50% regarded as being indicative of moderate to high heterogeneity. For studies of moderate to high heterogeneity, a random effects meta-analysis model was used;
15
otherwise, we used the fixed effects meta-analysis model.
16
The possibility of publication bias was quantified using the Begg’s and Egger’s test.17,18 A two-tailed
Results
Study description
Figure 1 shows the study selection process. A total of 31 studies met our inclusion criteria and involved 3,957,725 participants with hypertension, cardiomyopathy, vascular disease, breast cancer, colon cancer, lung cancer, melanoma, type 2 diabetes mellitus and glomerulonephritis. The age of the participants ranged from 18 to 80 years. All the studies were published between 1998 and 2014 (Table 1). There were 17 observational studies and 14 RCTs. Seven studies tested dual blockade with ARBs and ACEIs, 24 studies assessed ARB monotherapy and eight studies focused on ACEI monotherapy. The duration of follow-up ranged from 1.9 to 7.8 years. Sites (number) of the studies were as follows: America (13),19–31 England (5),11,32–35 Taiwan (4),12,36–38 Denmark (2),39,40 Sweden (2),41,42 Netherlands (1), 43 Japan (1), 44 Canada (1), 45 Norway (1) 46 and South Korea (1). 47 Table 1 shows the characteristics of the 31 studies included in the meta-analysis.
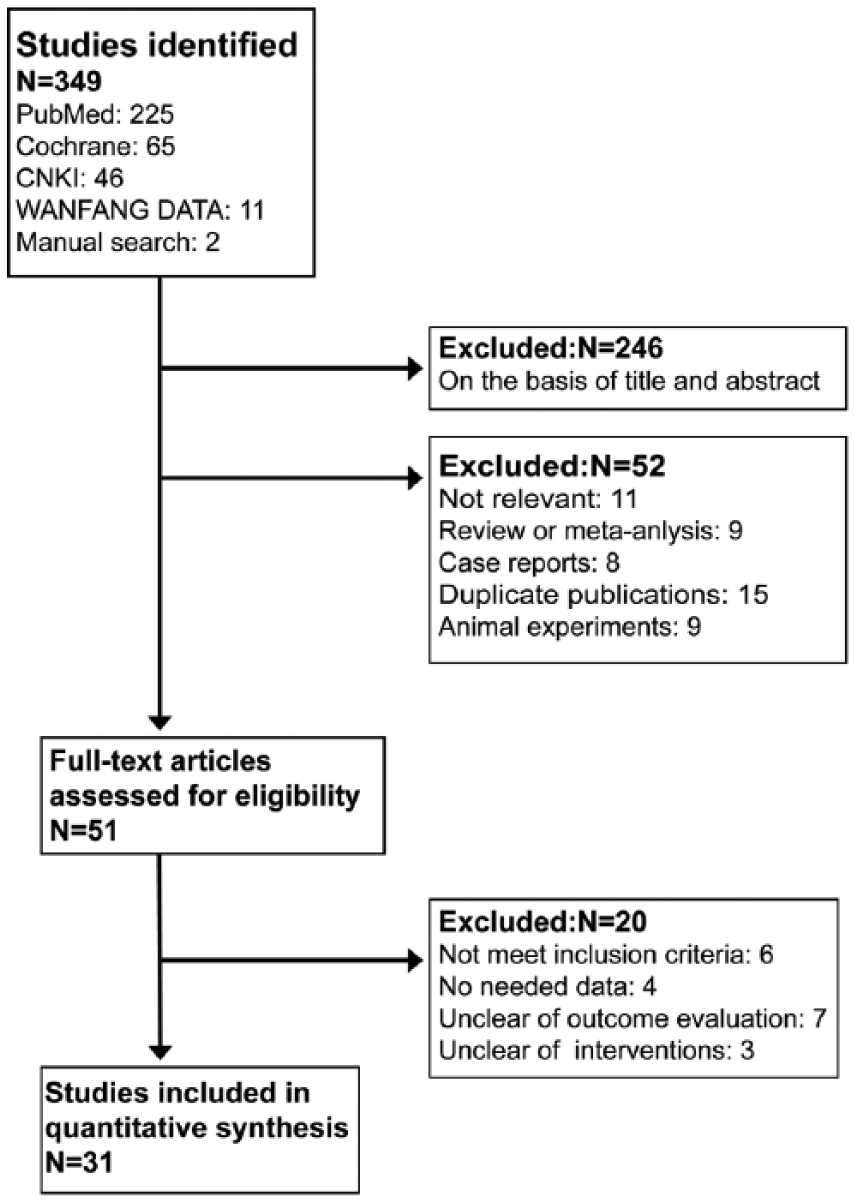
Flow chart of selection process in this study.
Summary of the characteristics of the included trials.

ARB: angiotensin II type 1 receptor blocker; ACEI: angiotensin-converting enzyme inhibitor.
Risk of bias within studies
As shown in Table 2, the range in the total NOS score for the 31 studies was 6 to 9 (theoretical range 0 to 9) and the mean (SD) was 7.7 (0.77). No publication bias was evident by Begg’s test (
Quality assessment of the 31 studies.

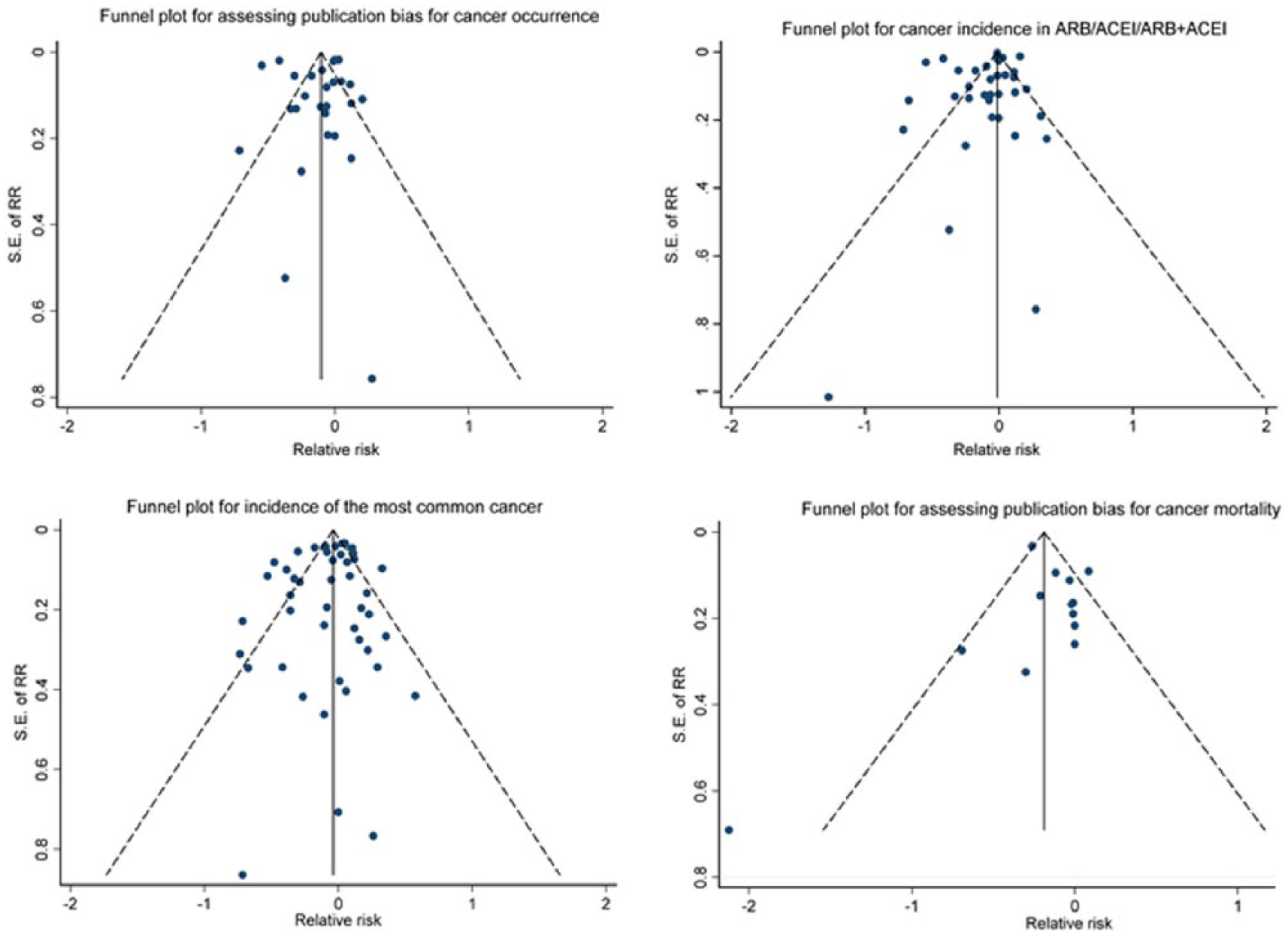
Funnel plots for assessing publication bias.
Outcomes
RAS blockade on incidence of cancer
There were 28 studies reporting the incidence of cancer (Table 1). Figure 3 shows incidence reduction with ARB/ACEI in the observational studies (RR 0.82, 95% CI 0.73–0.93,

Incidence reduction with angiotensin-converting enzyme inhibitor/angiotensin II type 1 receptor blocker therapy in randomised controlled trials and observational studies.
In subgroup analysis by RAS blockers, ACEIs (RR 0.84, 95% CI 0.72–0.99,

Subgroup analyses of monotherapy with angiotensin-converting enzyme inhibitor/angiotensin II type 1 receptor blocker therapy and dual renin–angiotensin system blockade.
Figure 5 illustrates the pooled data of the observational studies and RCTs stratified by sites of cancer. The observational studies revealed significant incidence reduction with ARB/ACEI in lung cancer (RR 0.85, 95% CI 0.75–0.97,

Incidence reduction with angiotensin-converting enzyme inhibitor/angiotensin II type 1 receptor blocker therapy in site-specific cancer.
Although the pooled data of 28 studies in total disclosed significant incidence reduction with the RAS blockade (Figure 6(a)), the protective effects were not statistically significant with the duration of follow-up stratified into less than 3, 3–5 and over 5 years. Figure 6(b) demonstrates a trend of reduced RR along with the time of follow-up.
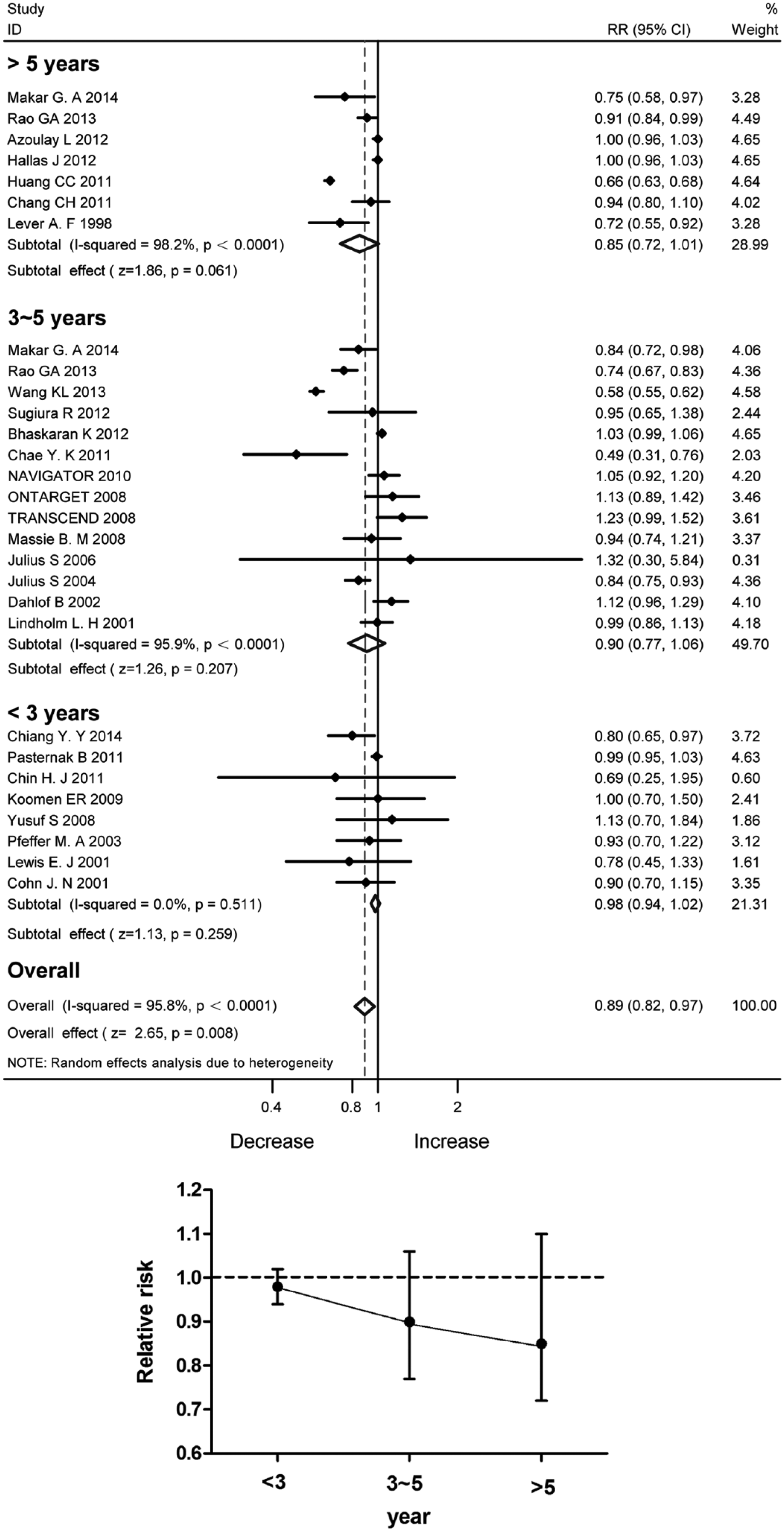
Incidence reduction with angiotensin-converting enzyme inhibitor/angiotensin II type 1 receptor blocker therapy stratified by the duration of follow-up. (a) Forest plot; (b) Relative risk ratios.
RAS blockade on mortality of cancer
Figure 7 shows mortality reduction with ARB/ACEI in the observational studies (RR 0.71, 95% CI 0.55–0.93,

Mortality reduction with angiotensin-converting enzyme inhibitor/angiotensin II type 1 receptor blocker therapy in observational studies and randomised controlled trials.
Differences in outcomes for cancer in observational studies and RCTs
Differences in the observational studies and RCTs included duration of follow-up and sample sizes (Table 3). The range of follow-up duration in observational studies was 2.4–7.8 years, compared to 1.9–5.0 years in RCTs. The mean duration of follow-up was 23% higher in observational studies than in RCTs. Moreover, the 17 observational studies had a sample size much larger than the 14 RCTs. The observational studies included 289,858 RAS blockade users and 3,833,261 participants, compared with 59,802 and 124,464, respectively, for the RCTs.
Differences in observational studies and randomised controlled trials (RCTs) included in this meta-analysis.

ARB: angiotensin II type 1 receptor blocker; ACEI: angiotensin-converting enzyme inhibitor.
Discussion
This meta-analysis reveals that the significant benefits of the RAS blockade observed in case–control studies and cohort studies might diminish in RCTs. Monotherapy with ACEI/ARB might have protective effects on the incidence and mortality of cancer in the pooled analysis of observational studies. The claimed therapeutic benefits in observational studies could not be validated in RCTs. The observed benefits of RAS blockade against the risk of cancer and death could largely result from non-randomised clinical design with a prolonged period of follow-up.
Observational studies have shown that ACEI/ARB may reduce the incidence and mortality of cancer. Angiotensin II receptors AT1 and AT2 are widely distributed in the cardiovascular system, brain, liver, kidney, adrenal cortex, muscle and connective tissue.48,49 Angiotensin II, the known key active peptide of RAS, binds the AT1 receptor to promote the initiation and progression of cancer by stimulating cell proliferation, angiogenesis and inflammation50–53 (Figure 8). The network regulation may explain the decreased risk of cancer incidence and mortality with RAS blockade.54–56 Cancer cells are angiogenesis dependent, and thus blockade of angiogenesis could limit tumour growth.57–60 Indeed, several studies have shown angiotensin II in the promotion of angiogenesis due to increased vascular endothelial growth factor expression by activation of the AT1.61–63 In contrast to angiotensin II, the angiotensin-(1-7) inhibits both angiogenesis and cell proliferation.64–66 It is well known that the ACE2–angiotensin-(1-7)–Mas axis serves as the principal counter-regulatory mechanism for the ACE–angiotensin II–AT1 axis. 67 An increased ACE/ACE2 activity ratio might lead to increased angiotensin II generation and increased catabolism of angiotensin (1-7). Monotherapy with ACEIs could upregulate ACE2 expression to lower the risk of cancer. 68 These findings might explain the consistent findings in the observational studies included in this meta-analysis.

Current view of the renin–angiotensin system cascade.
The protection with RAS blockade against cancer in observational studies diminished in the pooled analysis of the RCTs. The difference in outcomes for cancer between the two types of studies is attributed to sample size and duration of follow-up (Table 3). The 17 observational studies included in this meta-analysis provided a larger sample size than the 14 RCTs, and had a longer average duration of follow-up. Although observational studies have inherent limitations, which might compromise real associations between tested drugs and outcomes, it is difficult to implement RCTs in real clinical settings and lifetime treatment. In the present study, RCTs included in this analysis were not conducted to evaluate the effects of ARBs and ACEIs on the risk of cancer and cancer-related death as the primary endpoints. Concerning the differences in outcomes for cancer, here we have reported cancer incidence and mortality changes separately in RCTs and observational studies.
There are limitations in this study. First, the incidence and mortality of cancer were not the primary endpoints in some of the included case–control studies. Second, lack of the original data has prevented our direct evaluation of the effects of ARB/ACEI on different ethnic groups. Finally, the exact doses and dosages were inconsistent in the 31 studies included. All these limitations might affect the implication and interpretation of the findings from the present study.
In summary, the therapeutic benefits with ACEIs and ARBs reported in case–control studies and cohort studies might diminish in RCTs. The clinical design, site of cancer and duration of follow-up may affect the clinical outcomes.
Footnotes
Acknowledgements
The authors would like to thank Mr Vincent Von Bokern and Golden Wilson for his English proofing. All authors agreed on the final version of the manuscript
Authors’ contributions
The study was designed by JS, YMH, XNS, XZH, XZ and HLZ; YMH, JS, XNS were responsible for critical revision of the article for important intellectual content and draft the manuscript; YMH, MW, YHP, XZH conducted the collection and assembly of the data; MW, YMH and WL performed the statistical analyses; MHZ, XXZ, YS and HLZ interpreted the data; JS and YMH drafted the manuscript; HLZ had primary responsibility for the final content.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by grants from the Guilin Medical University (KY2011002), Program for Innovative Research Team of Guilin Medical University (PIRTGMU) and National Natural Science Foundation of China (81270934).
