Abstract
Background and objective:
All-trans retinoic acid (ATRA) exerts various effects on physiological processes such as cell growth, differentiation, apoptosis and inflammation. Prohibitins (PHB), including prohibitin 1 (PHB1) and prohibitin 2 (PHB2), are evolutionary conserved and pleiotropic proteins implicated in various cellular functions, including proliferation, tumor suppression, apoptosis, transcription, and mitochondrial protein folding. The renin–angiotensin–aldosterone system plays a pivotal role in the regulation of blood pressure and volume homeostasis. All these factors and systems have been implicated in renal interstitial fibrosis. Therefore, the objective of this study was to investigate the effect of ATRA treatment on the renin–angiotensin–aldosterone system and expression of prohibitins to further understand its role in the processes leading to renal interstitial fibrosis.
Methods:
The hypoxic and oxidative stress conditions in obstructive renal disease were simulated in a hypoxia/reoxygenation model with renal tubular epithelial cells (RTEC) as a model system. Subsequently, the effect of ATRA on mRNA and protein expression levels was determined and correlations were established between factors involved in the renin–angiotensin–aldosterone system, the prohibitins, cellular redox status, renal interstitial fibrosis and ATRA treatment.
Results:
Correlation analysis showed that both PHB1 and PHB2 protein levels were negatively correlated with angiotensin I, ACE1, angiotensin II, TGF-β1, Col-IV, FN, ROS, and MDA (PHB1: r = −0.792, –0.834, –0.805, –0.795, –0.778, –0.798, –0.751, –0.682; PHB2: r = −0.872, –0.799, –0.838, –0.773, –0.769, –0.841, –0.794, –0.826; each p < 0.05), but positively correlated with ACE2, SOD, and GSH (PHB1: r = 0.796, 0.879, 0.824; PHB2: r = 0.785, 0.914, 0.849; each p < 0.05). ACE1 was positively correlated with angiotensin I, angiotensin II, TGF-β1, Col-IV, FN, ROS, and MDA, and negatively correlated with ACE2, SOD, and GSH (each p < 0.05). ACE2 was negatively correlated with ACE1, angiotensin I, angiotensin II, TGF-β1, Col-IV, FN, ROS, and MDA, and positively correlated with SOD and GSH (each p < 0.05).
Conclusion:
The results suggest that ATRA acts as a positive regulator of PHB1, PHB2 and ACE2, and as a negative regulator of ACE1, angiotensin I, and angiotensin II in a RTEC model system under hypoxia/reoxygenation conditions.
Keywords
Introduction
All-trans retinoic acid (ATRA), an active metabolite of vitamin A from the retinoid family, has been shown to induce cell growth, differentiation and apoptosis, and has an emerging role in gene regulation. 1 ATRA has been implicated to play a significant role in a number of diseases, such as acute myeloid leukemia, hepatocellular carcinoma, glomerulosclerosis, renal interstitial fibrosis (RIF), etc.,2–7 either actively through changes in its levels or positively affecting some of the aforementioned disease via ATRA treatment. Histopathologically, RIF is characterized by distinct tubulo-interstitial changes, including interstitial cell infiltration, tubular degeneration, increased number of myofibroblasts, oxidative stress, and accumulation of profibrotic factors and extracellular matrix (ECM) components.8,9
Prohibitins (PHB) are ubiquitously expressed in a variety of cell types and multiple cellular compartments, including the mitochondria, nucleus, and the plasma membrane. 10 The family includes prohibitin 1 (PHB1) and prohibitin 2 (PHB2), and several other multifunctional intracellular proteins, which all play important roles in the regulation of cellular proliferation, apoptosis and differentiation. 11 Our previous investigations 6 showed that ATRA treatment was associated with an increased expression of PHB in RIF rats, including renal tubular epithelial cells (RTEC). RTEC are polarized cells that constitute the predominant cell type within the tubulointerstitium, and play a critical role in normal kidney function.12,13 Furthermore, injury to RTEC cells has recently been proposed to directly contribute to the accumulation of myofibroblasts in RIF. 14
The renin–angiotensin–aldosterone system (RAAS) plays a pivotal role in the regulation of blood pressure and volume homeostasis, 15 and the RAAS is also important in renal physiological and pathological processes. Increased vascular pressure has long been implicated to promote some of the factors that lead to renal vascular and glomerular fibrosis, particularly deposition of ECM components. We previously reported 7 that ATRA treatment was associated with RAAS expression in glomerulosclerosis rats. Since little is known of the effect of ATRA treatment on the RAAS with respect to RIF and no data are available on PHB and RAAS expression under hypoxia/reperfusion and ATRA treatment conditions, we studied the role of ATRA treatment on the RAAS, and tried to correlate the induced oxidative stress and the presence or absence of ATRA with the expression of RAAS components, RIF-associated factors, and PHB1 and 2 in the model environment of the RTEC cell line.
Materials and methods
RTEC cell culture, induction of oxidative stress, and gene interference
The rat renal proximal tubular epithelial cell line (RTEC), NRK-52E, was purchased from the Cell Bank and Type Culture Collection of the Chinese Academy of Sciences (Shanghai Institute of Cell Biology, Shanghai, China) and maintained in DMEM (2 mM glutamine, 1% non-essential amino acids) supplemented with 5% fetal bovine serum (FBS) in a humidified atmosphere (37°C; 5% CO2).
Oxidative stress and cell injury was induced by hypoxia/reoxygenation (H-R: 12/24 h) as described in more detail by Bhogal et al. 16 The cells were divided into four groups: (i) normal control (control), (ii) cell injury induced by H-R, (iii) H-R cells treated with ATRA (0.1 μM dissolved in dimethyl sulfoxide [DMSO]), and (iv) H-R cells treated with DMSO only, n = 6, respectively. The treated cells were harvested for further analysis 48 h after the treatment.
Real-time reverse transcription polymerase chain reaction to detect PHB1, PHB2, angiotensin-converting enzyme 1 (ACE1), angiotensin-converting enzyme 2 (ACE2) and transforming growth factor-β1 (TGF-β1) mRNA expression
Total RNA was extracted with TRIzol (Life Technologies-Invitrogen, Grand Island, NY, USA). Signals were measured with a Gel Doc XR+, UV/vis-Molecular Imager (Bio-Rad Laboratories, Hercules, CA, USA) and evaluation of the 18S and 28S RNA bands after agarose gel electrophoresis confirmed that there had been no RNA degradation. 17 Primers were designed according to standard primer design principles with Primer Premier 5.0 (Premier Biosoft, Palo Alto, CA, USA).
One microgram total RNA was reverse transcribed into cDNA with an ExScript RT reagent kit (Thermo Scientific-Fermentas, Waltham, MA, USA). PHB1, PHB2, ACE1, ACE2 and TGF-β1 were amplified with SYBR Premix Ex Taq (Roche Inc., Basel, Switzerland). Gene expression of the housekeeping β-actin (ACTB) was used as an internal loading control and to determine reverse transcription efficiency. The average threshold cycle (Ct; the cycles of template amplification to the threshold) was determined for each sample and the fold change in the data was analyzed according to Livak and Schmittgen. 18
Western blot analysis
Proteins were isolated from homogenized RTEC cells with radio immunoprecipitation assay (RIPA) lysis buffer (Sigma-Aldrich Corp., St. Louis, MO, USA) containing 0.25 nM of the protease inhibitor phenylmethanesulfonyl fluoride (PMSF; Sigma-Aldrich Corp., St. Louis, MO, USA). After protein concentration, quantization was performed with the modified Bradford assay (Bio-Rad Laboratories, Hercules, CA, USA) 19 and 40 mg total protein was subsequently used for Western blotting with primary antibodies against PHB1, PHB2, angiotensin I and II (Santa Cruz Biotechnology, Inc., Dallas, TX, USA), ACE1, ACE2, TGF-β1, collagen IV (Col-IV), and fibronectin (FN); all from Abcam (Cambridge, MA, USA) unless stated otherwise. Beta-actin was used as an internal loading control. Near-infra red fluorescence from manually selected bands of interest were imaged with an Odyssey Fc scanner (Li-Cor, Lincoln, NE, USA); Raw fluorescence intensities were background subtracted (intra-lane) using Li-Cor Odyssey 3.0 analytical software. 20
Reactive oxygen species, lipid peroxidation, and antioxidant measurements
Reactive oxygen species (ROS) measurements were essentially performed according to Hempel et al., 21 with some modifications. Cell supernatants were obtained by scraping cell monolayers, subsequent sonication and centrifugation (15000×g; 5 min) at 4°C, and 500 µl supernatant was incubated for 3 h at 37°C with 10 μL of a 10 μM DCF-DA (2,7-dichlorodihydro-fluorescein diacetate) solution (Life Technologies/Molecular probes, Eugene, OR, USA). The fluorescence signal was measured at 485/525 nm on a S-3100 spectrofluorometer (Scinco Co. Ltd., Seoul, Korea), equipped with a 1024 channel photodiode array detector and expressed as arbitrary units.
Malonyldialdehyde (MDA), glutathione (GSH), and superoxide dismutase (SOD) were determined as extensively described by Wu et al. 22 Cell supernatants were obtained by scraping cell monolayers and subsequent sonication and centrifugation (15000×g; 5 min) at 4°C. The supernatant was used for the determination of MDA and GSH levels, and SOD activity. Protein concentration of the samples was determined with a BCA (bicinchoninic acid) protein assay kit (Sigma-Aldrich Corp., St. Louis, MO, USA) with bovine serum albumin (BSA) as a standard. Absorbance was measured spectrophotometrically at 562 nm.
Statistical analysis
All data are shown as mean ± standard deviation (SD). To compare the groups in relation to parameters with normal distribution, one-way analysis of variation (ANOVA) with post-hoc Fisher’s LSD (least significant difference) was used. Conversely, for those parameters without normal distribution, Kruskal–Wallis with post-hoc Mann–Whitney (only for the weight parameter) was used. Pearson’s correlation coefficients were used to determine the relationships between the indicators for detection in the cell culture. A value of p < 0.05 was accepted as statistically significant. Statistical analysis was performed using the Statistical Package for Social Sciences–SPSS, version 13.0 (SPSS Inc., Chicago, IL, USA).
Results
PHB1, PHB2, ACE1, ACE2, and TGF-β1 mRNA expression in RTEC
In this study, H-R treatment was able to reduce the mRNA expression of PHB1, PHB2, ACE2, and increased the mRNA expression of ACE1 and TGF-β1 (Figure 1). Interestingly, ATRA treatment increased the mRNA expression of PHB1, PHB2, ACE2, and down-regulated the mRNA expression of ACE1 and TGF-β1 (Figure 1). No significant difference in PHB1, PHB2, ACE1, ACE2 and TGF-β1 mRNA expression was observed between the H-R and DMSO groups (each p > 0.05, Figure 1).
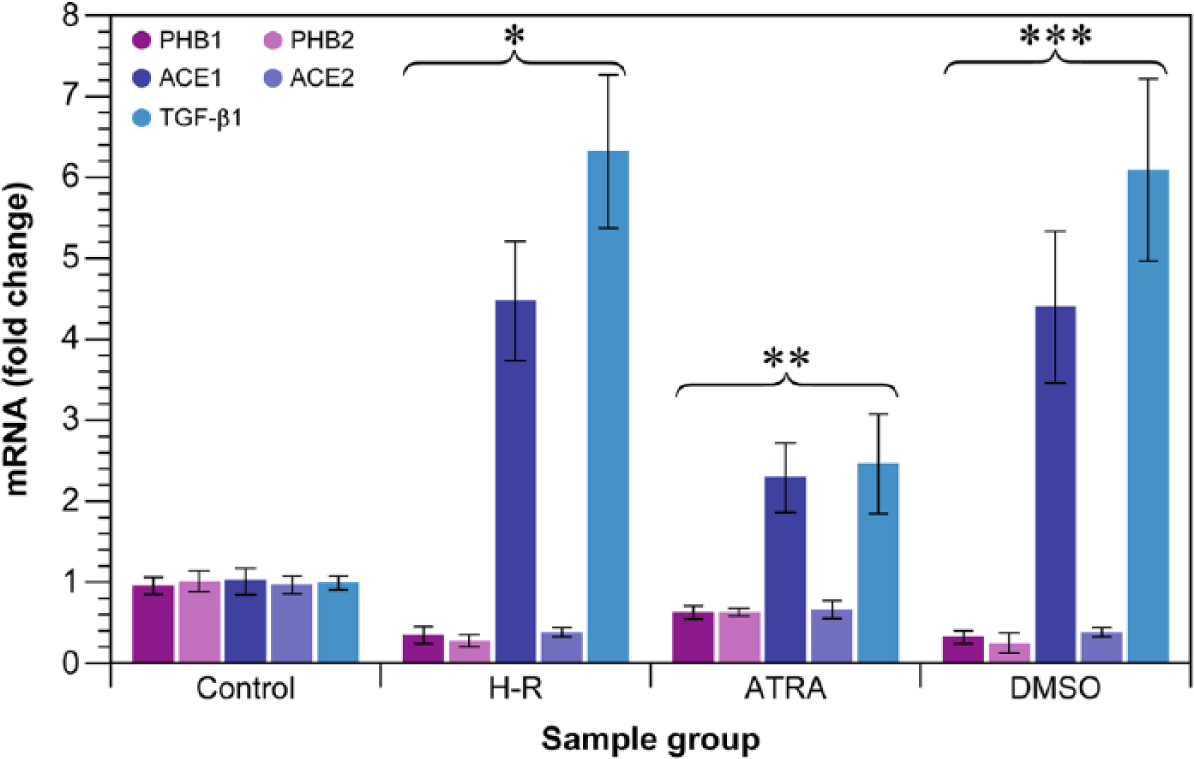
mRNA expression of prohibitin 1 (PHB1), prohibitin 2 (PHB2), angiotensin-converting enzyme 1 (ACE1), angiotensin-converting enzyme 2 (ACE2) and transforming growth factor-β1 (TGF-β1) in renal tubular epithelial cells. *: p < 0.01 compared with control group; **: p < 0.01 compared with hypoxia/reoxygenation (H-R) group (cell injury induced by H-R); ***: p > 0.05 compared with H-R group. ATRA: all-trans retinoic acid; DMSO: dimethyl sulfoxide.
PHB1, PHB2, ACE1, ACE2, angiotensin I, angiotensin II, TGF-β1, Col-IV, and FN protein expression in RTEC
On Western blot, protein expression levels closely followed the results of the mRNA experiments (Figures 2 and 3). H-R treatment resulted in a reduction of the protein expression of PHB1, PHB2, ACE2, and concomitantly increased the protein expression of ACE1, angiotensin I, angiotensin II, TGF-β1, Col-IV, and FN compared with the control group (each p < 0.01, Figures 2 and 3). In concordance with the mRNA measurements, ATRA treatment increased the protein expression of PHB1, PHB2, ACE2, and down-regulated ACE1, angiotensin I, angiotensin II, TGF-β1, Col-IV, and FN (each p < 0.01, Figures 2 and 3). No significant differences in PHB1, PHB2, ACE1, ACE2, angiotensin I, angiotensin II, TGF-β1, Col-IV, and FN protein expression were observed between the H-R and the DMSO groups (each p > 0.05, Figures 2 and 3), which indicated that DMSO did not interfere with the measurements.

Evaluation of protein expression (Western blot) of prohibitin 1 (PHB1), prohibitin 2 (PHB2), angiotensin-converting enzyme 1 (ACE1), and angiotensin-converting enzyme 2 (ACE2) in renal tubular epithelial cells.
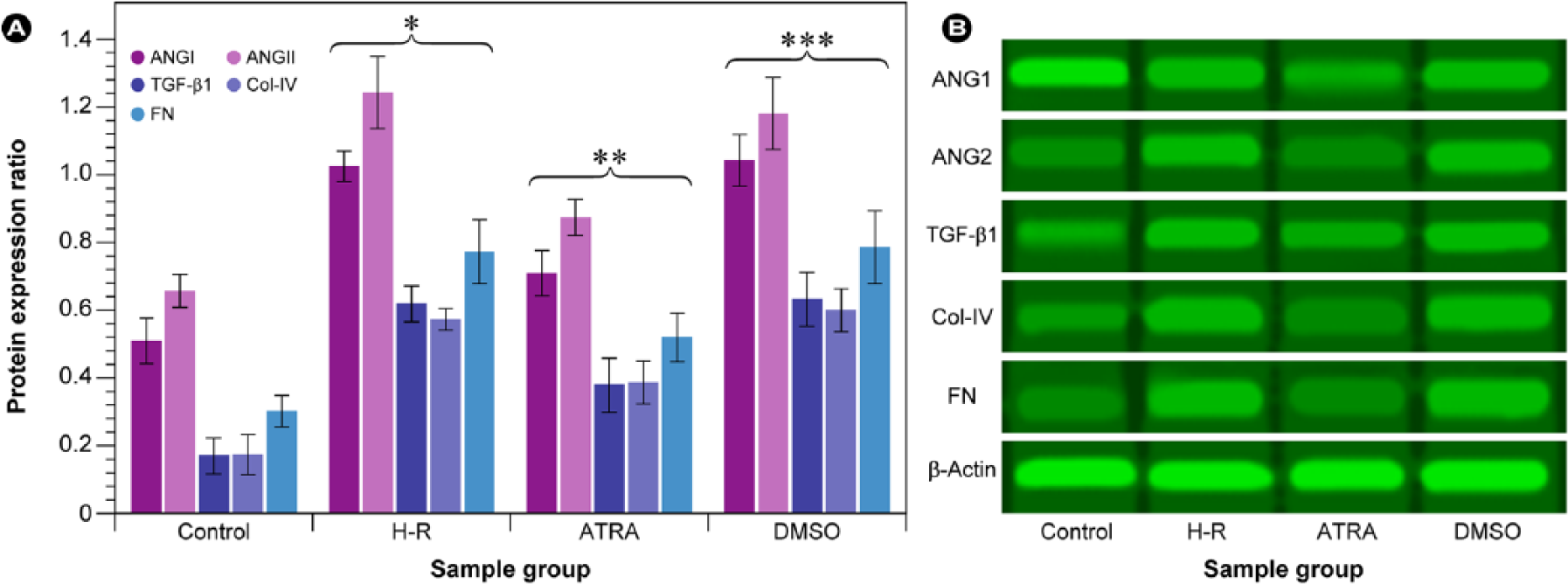
Evaluation of protein expression (Western blot) of angiotensin I (ANGII), angiotensin II (ANGII), transforming growth factor-β1 (TGF-β1), collagen IV (Col-IV), and fibronectin (FN) in renal tubular epithelial cells.
Redox status measurements
H-R treatment per se induced an increase in the formation of ROS and MDA, and concurrently reduced the expression of SOD and GSH levels (each p < 0.01, Figure 4). Due to its antioxidative activity, ATRA treatment attenuated ROS and MDA formation, and up-regulated the protein expression of SOD and increased GSH levels (Figure 4). No significant effect of DMSO was observed, since ROS, MDA, SOD, and GSH levels (each p > 0.05, Figure 4) in the H-R group were similar to the DMSO group; DMSO is a well-known radical scavenger.
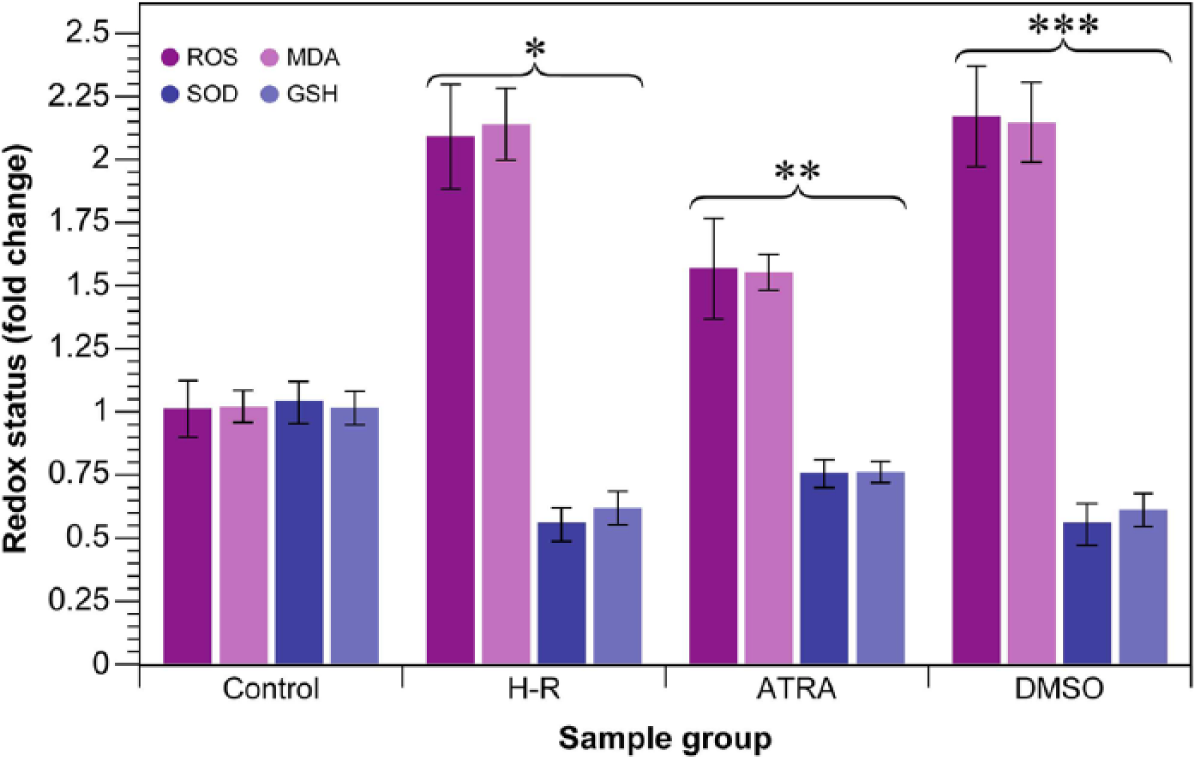
Assessment of oxidative stress and antioxidative capacity. Changes in general reactive oxygen species (ROS) levels and malondialdehyde (MDA) generation, a final degradation product of lipid peroxidation, and superoxide dismutase (SOD) and reduced glutathione (GSH) are shown. *: p < 0.01 compared with control group; **: p < 0.01 compared with hypoxia/reoxygenation (H-R) group (cell injury induced by H-R); ***: p > 0.05 compared with H-R group. ATRA: all-trans retinoic acid; DMSO: dimethyl sulfoxide.
Correlation analysis
Correlation analysis showed that both PHB1 and PHB2 protein levels were negatively correlated with angiotensin I, ACE1, angiotensin II, TGF-β1, Col-IV, FN, ROS, and MDA (PHB1: r = −0.792, –0.834, –0.805, –0.795, –0.778, –0.798, –0.751, –0.682; PHB2: r = −0.872, –0.799, –0.838, –0.773, –0.769, –0.841, –0.794, –0.826; each p < 0.05), but positively correlated with ACE2, SOD, and GSH (PHB1: r = 0.796, 0.879, 0.824; PHB2: r = 0.785, 0.914, 0.849; each p < 0.05). ACE1 was positively correlated with angiotensin I, angiotensin II, TGF-β1, Col-IV, FN, ROS and MDA and negatively correlated with ACE2, SOD, and GSH (each p < 0.05). ACE2 was negatively correlated with ACE1, angiotensin I, angiotensin II, TGF-β1, Col-IV, FN, ROS, and MDA and positively correlated with SOD and GSH (each p < 0.05).
Discussion
The effect of ATRA treatment on the expression of PHB1 and PHB2, and components of the RAAS in a RTEC injury model induced by H-R was studied in this investigation. The results showed that ATRA treatment was associated with increased PHB1 and PHB2 expression. Furthermore, we observed that ATRA treatment reduced the expression of TGF-β1, Col-IV, and FN; important components involved in extracellular matrix deposition and RIF. These results were similar to the results from our previous study in RIF rats. 6 RTEC injury plays a pivotal role in RIF and, therefore, the results suggest that ATRA attenuates the deposition of profibrotic components and might play a protective role against RIF lesion formation and progression. Currently, no reports from other research groups are available that assess the association of ATRA with RIF-associated renal disease.
Derangements in RAAS expression are distinctly associated with vascular lesions and implicated in various renal diseases. This study shows that ATRA treatment directly affects RAAS expression. ATRA treatment resulted in a decreased ACE1 expression and increased ACE2 expression. These results are in concordance with our previous study, in which we found that ATRA treatment was associated with reduced expression of ACE1 and increased expression of ACE2 in glomerulosclerosis rats. 7 Furthermore, Zhong et al. 23 reported that chronic ATRA treatment increased both gene and protein expression of ACE2, which resulted in the reduction of blood pressure and the attenuation of myocardial damage in spontaneously hypertensive rats. ATRA was associated with up-regulated expression of ACE2, and their results and conclusion were similar to ours, albeit in a different cell type.
To further investigate the effect of ATRA on the RAAS, the expression of angiotensin I and II was also studied in this investigation. Angiotensin I can be converted into the vasoactive and aldosterone-stimulating peptide angiotensin II, 24 and angiotensin II is the major effect messenger of the RAAS and exhibits a wide range of responses, such as induction of the accumulation and deposition of collagen.25,26 In this study, we found that ATRA treatment could reduce the expression of angiotensin I and II in RTEC under H-R conditions. Although no studies are available on the relationship between ATRA treatment and angiotensin I expression in RIF disease, several studies report positive effects of ATRA treatment on angiotensin II expression, predominantly in the cardiovascular system. Haxsen et al. 27 reported that ATRA could inhibit the action of angiotensin II on vascular smooth muscle cells. Choudhary and co-workers 28 showed that the pressure overload-induced production of angiotensin II was inhibited by retinoic acid via up-regulation of the expression of ACE2 in the development of cardiac remodeling in a pressure overload rat model. He et al. 29 reported that ATRA inhibited the angiotensin II-induced increase in cell growth and collagen secretion by cardiac fibroblasts in neonatal rats. The results on the effect of ATRA treatment on angiotensin II expression from the aforementioned reports generally reflect our results and considerations.
Evaluation of the effect on oxidative stress showed that ACE1 was positively correlated with ROS and MDA levels, and negatively correlated with SOD expression and GSH levels. Hung et al. 30 showed that RAAS blockers prevented excessive lipid accumulation and ROS generation in differentiating adipocytes. Chalghoum and colleagues 31 reported that statistical tests had shown negative correlations between the activities of ACE1 and SOD. No studies are available on the association of ACE1 with MDA expression and GSH levels. Our results indicated that ACE1 was associated with MDA and GSH levels in RTEC under H-R conditions. Furthermore, ACE2 was negatively correlated with ROS, MDA, but positively correlated with SOD and GSH.
In conclusion, we show that ATRA treatment was associated with increased expression of PHB1, PHB2, ACE2, SOD, and GSH, and concurrently associated with reduced expression of angiotensin I, ACE1, angiotensin II, ROS, and MDA in cellulo. Furthermore, expression of factors that affect RIF, i.e. TGF-βl, Col-IV, and FN, were down-regulated and consequently ECM deposition might be reduced by ATRA treatment. Overall, ATRA treatment affects multiple processes associated with RIF in parallel, including oxidative stress, expression of profibrotic components, and factors involved in regulating renal vascular blood flow and pressure, and might therefore be an ideal therapeutic compound in the treatment of RIF-associated renal disease. Nonetheless, further investigations are required to elucidate the exact mechanism(s) through which ATRA exerts its protective effects in RIF.
Footnotes
Conflict of interest
None declared.
Funding
This study was supported by the Nature Science Foundation of China (no. 81060061 and no. 81150017), and the sub-item of 985 Project Foundation of Sun Yat-Sen (The Hundred Talents Program Foundation; No. 88000-3311300).
