Abstract
Objective:
The objective of this paper is to study the effect of aliskiren on metabolic parameters and micro- and macrovascular reactivity in individuals diagnosed with or at high risk for developing type 2 diabetes mellitus (T2DM).
Research design and methods:
We studied 47 T2DM and 41 at-risk individuals in a randomized, double-blinded, placebo-controlled trial. All subjects were treated with 150 mg aliskiren or placebo daily for 12 weeks. Twenty-six (55%) of T2DM and four (8%) at-risk subjects were also treated with angiotensin-converting enzyme (ACE) inhibitors or angiotensin II receptor blockers.
Results:
Aliskiren treatment was associated with improvement in systolic and diastolic blood pressure and endothelium-independent vasodilation at the skin microcirculation in those with T2DM but not in those at risk. There were no incidences of hypotension and no significant changes in serum potassium or creatinine levels with aliskiren treatment in either study group.
Conclusions:
Aliskiren improves blood pressure and vascular smooth muscle function in the skin microcirculation of T2DM patients.
Introduction
Endothelium-dependent and -independent vasodilation, collectively referred to as vascular reactivity, is impaired in the skin microcirculation of patients with type 2 diabetes (T2DM) with or without evidence of long-term diabetes complications, in individuals with impaired glucose tolerance (IGT), and in offspring of individuals with T2DM.1,2 Endothelial function is also impaired in the macrocirculation of these individuals as evaluated at the brachial artery.2,3 Impairment of vascular reactivity in the microcirculation is associated with the development of peripheral neuropathy while impaired reactivity in the macrocirculation is associated with the development of foot ulceration.1,4 Additionally, endothelial dysfunction in the macrocirculation is associated with insulin resistance and the development of atherosclerosis. 5 Given the vast and increasing number of individuals diagnosed with type 2 diabetes or prediabetes (impaired fasting glucose or IGT), identifying pharmacotherapeutic agents that improve endothelial function in individuals with or at risk for type 2 diabetes addresses an important clinical need.
Aliskiren is a direct renin inhibitor that decreases plasma renin activity (PRA) and inhibits conversion of angiotensinogen to angiotensin I, the proximal rate-limiting step in the renin–angiotensin–aldosterone system (RAAS). Recent animal studies have shown that aliskiren has protective effects on endothelial function and atherosclerotic changes. 6 In humans, co-treatment with a direct renin inhibitor and an angiotensin II receptor blocker (ARB) confers additive protective effects on both endothelial function and atherosclerotic changes and improves albuminuria in patients with diabetic kidney disease. 7 Additionally, individuals with diabetes may derive particular benefit from the effect of renin inhibition on left ventricular (LV) mass and remodeling.8–10 The effects of aliskiren on the vascular reactivity in individuals with T2DM or at risk for developing the disease have not been previously studied. The main aim of this study was to examine this effect in a 12-week randomized, double-blind, placebo-controlled study.
Research design and methods
Subjects
We studied two groups of subjects: One group included individuals at risk for developing T2DM and a second group included individuals previously diagnosed with T2DM. Subjects at risk for developing diabetes had at least one first-degree relative with T2DM; a history of gestational diabetes; or a history of IGT or impaired fasting glucose. The diagnosis of T2DM diabetes and impaired glucose homeostasis was made according to the American Diabetes Association Guidelines. 11 Subjects with T2DM had an A1C > 6.5% and < 13% and did not have a history of severe proliferative retinopathy; severe gastroparesis requiring hospitalization; nonhealing foot ulceration due to peripheral diabetic neuropathy; and history of diabetic nephropathy manifested as macroalbuminuria. Concomitant treatment with sulfonylureas, metformin, dipeptidyl peptidase-IV (DPPIV) inhibitors, glucagon-like peptide-1 (GLP-1) agonists, or insulin was permitted.
To avoid confounding factors known to affect endothelial function, we excluded individuals who smoked any amount of cigarettes during the previous six months, subjects with cardiac arrhythmia, congestive heart failure (CHF), recent stroke, chronic renal disease, macroalbuminuria (albumin/creatinine ratio > 300 mcg/mg), severe dyslipidemia (triglycerides > 600 mg/dl or total cholesterol > 300 mg/dl), or any other serious chronic disease requiring active treatment. Controlled hypertension was not an exclusion criterion and subjects were maintained on their usual antihypertensive medications during the study. No class of antihypertensive medication was exclusionary.
Methods
We conducted a 12-week, randomized, double-blind, placebo-controlled study. Participants were allocated to one tablet of placebo or 150 mg aliskiren once daily. Interested subjects attended a screening visit, and eligible subjects completed an enrollment/baseline visit and an end-treatment/exit visit. The screening visit took place at the Beth Israel Deaconess Medical Center Clinical Research Center after subjects fasted overnight for at least eight hours. The visit included a medical history, physical examination, and review of the inclusion/exclusion criteria. Baseline and end-treatment visits included vascular reactivity measurements, which were performed in the Microcirculation Laboratory at the Beth Israel Deaconess Medical Center. The protocol was approved by the Institutional Review Board at the Beth Israel Deaconess Medical Center and all participants gave written informed consent. Volunteers for the study were recruited through local advertisements and physician offices (www.clinicaltrials.govNCT01165983).
Vascular reactivity tests of micro- and macrocirculation
Skin blood flow was measured on the ventral surface of the right forearm using laser Doppler flowmetry (Lisca PIM 2.0, Lisca Development AB, Linkoping, Sweden) as previously described. 2 Measurements were obtained before and after iontophoresis of ACh, and before and after iontophoresis of sodium nitroprusside (SNP) (endothelium-dependent and -independent microvascular reactivity, respectively) using an MIC1 iontophoresis system (Moor Instruments Ltd, Millwey, Devon, England). The percentage change with each study treatment was calculated.
Endothelium-dependent flow-mediated brachial dilation (FMD) and nitroglycerine-induced dilation (NID, endothelium-independent) were measured using a high-resolution ultrasound with a 10.0-MHz linear array transducer (Aloka Prosound α7, Hitachi Aloka Medical Ltd, Tokyo, Japan), according to standard guidelines. 12
Skin biopsies
Two-mm skin-punch biopsies were taken from the volar aspect of the forearm under local anesthesia (1% plain lidocaine) in the subset of participants who agreed to this procedure. The hematoxylin and eosin (H&E) staining analysis was performed by a trained pathologist with experience in dermatopathology (AK) who was unaware of the randomization status of each participant. We evaluated round cells around skin vessels that we have shown in previous studies correspond to lymphocytes and are strong indicators of inflammation. 4 We also looked for neutrophilic infiltrates, an indication of acute inflammatory response related to microbial infection.
Serum biomarkers
Standard biochemistries obtained at the screening visit were processed at LabCorp. Circulating markers of endothelial dysfunction inflammation and growth factors were measured at baseline and after 12 weeks of treatment using Luminex technology.
Statistical analysis
Our primary endpoint was the effect of aliskiren on endothelium-dependent and -independent vasodilation of the macro- and microcirculation. Additional endpoints included the effect of aliskiren on biochemical markers of endothelial function, inflammatory markers, and growth factors that are increased in diabetes and associated with diabetic complications. We used the Minitab statistical package (Minitab Inc, State College, PA, USA) and the Statistical Analysis System (SAS, Cary, NC, USA) for statistical analysis. Our primary analysis examined the difference in mean change between placebo- and aliskiren-treated groups of these measures. The change between baseline and exit visit in each group was evaluated using the paired t test for parametrically distributed data and the Wilcoxon matched-pair signed rank test for nonparametrically distributed data. The t test was also used to compare baseline characteristics between the T2DM and at-risk groups. Multiple regression analysis was used to investigate the effect of aliskiren on the endothelium-independent vasodilation after adjusting for co-treatment with angiotensin-converting enzyme (ACE) inhibitors and/or ARBs and beta blockers.
Results
Baseline characteristics
We screened 124 subjects and randomized 100 to aliskiren or placebo (Figure 1). Seven subjects (16%) at risk of T2DM (four treated with aliskiren and three treated with placebo) and five (10%) T2DM patients (three treated with aliskiren and two treated with placebo) did not complete the study. Among the 12 noncompleters, four were withdrawn by the investigators on recommendation from the sponsor to terminate the study prematurely based on interim analysis of the Aliskiren Trial in Type 2 Diabetes Using Cardio-Renal Endpoints (ALTITUDE) multicenter clinical trial. 13 The remaining eight subjects withdrew consent. There were no major differences in baseline characteristics between the subjects who did not complete the study and those who did.
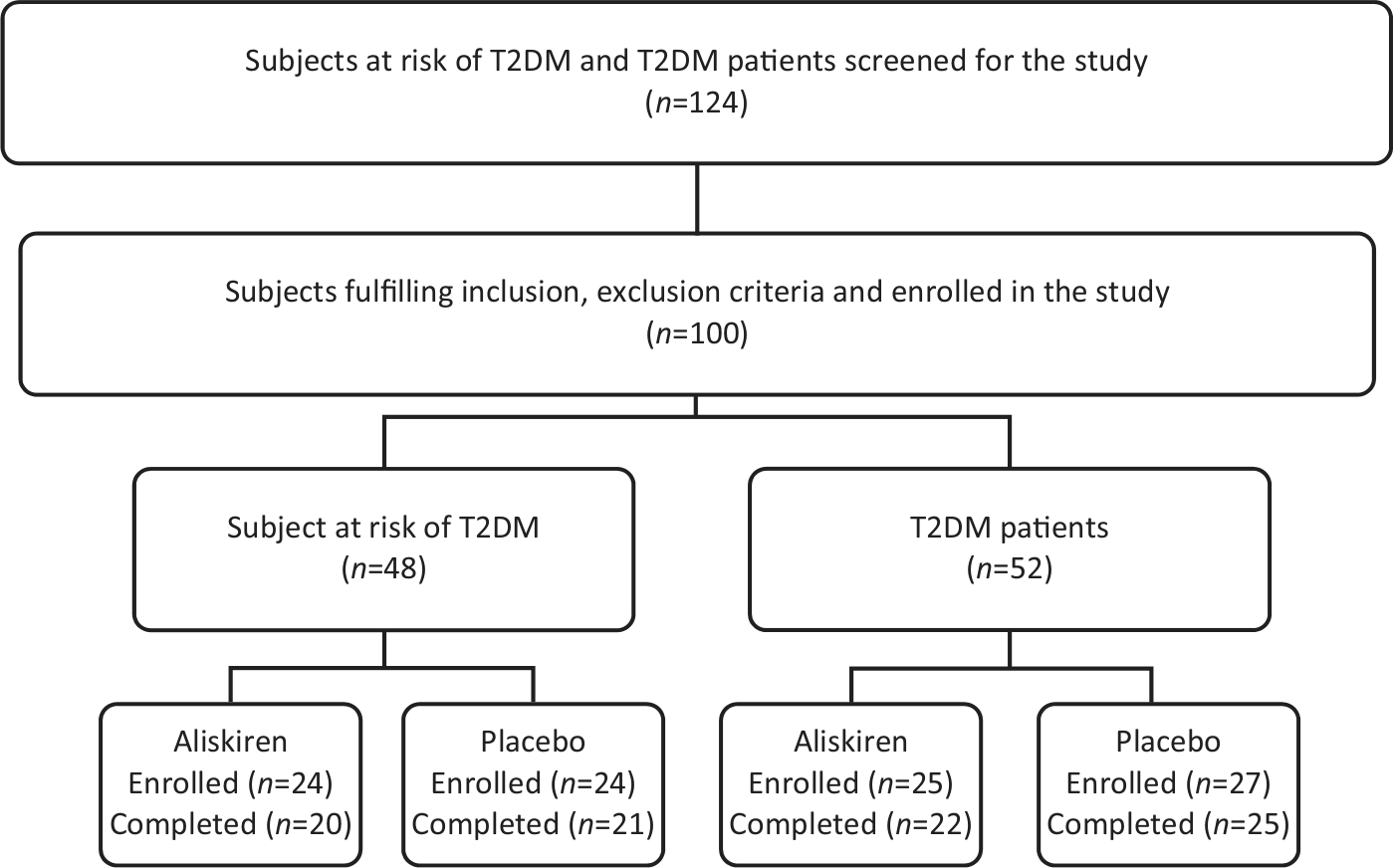
Scheme of the clinical trial.
Baseline clinical characteristics of the study population are presented in Table 1. Overall, subjects with T2DM were older but had very good glycemic control as indicated by their HbA1c level. Metformin was the most common antidiabetic agent used in the diabetic group (77% of subjects), followed by sulfonylureas (30%) and insulin (21%). Subjects with T2DM were more often treated with a statin or antihypertensive medication and had lower total cholesterol and low-density lipoprotein (LDL) levels than the at-risk group. Fifty-five percent of the T2DM subjects were treated with an ACE inhibitor or ARB at baseline compared to 10% in the at-risk group; both groups had ongoing treatment with these agents for the duration of the study. Baseline serum creatinine and potassium levels and baseline estimated glomerular filtration rate (eGFR) were within normal limits and were not different between subjects with or at risk for T2DM.
Baseline clinical characteristics of subjects.
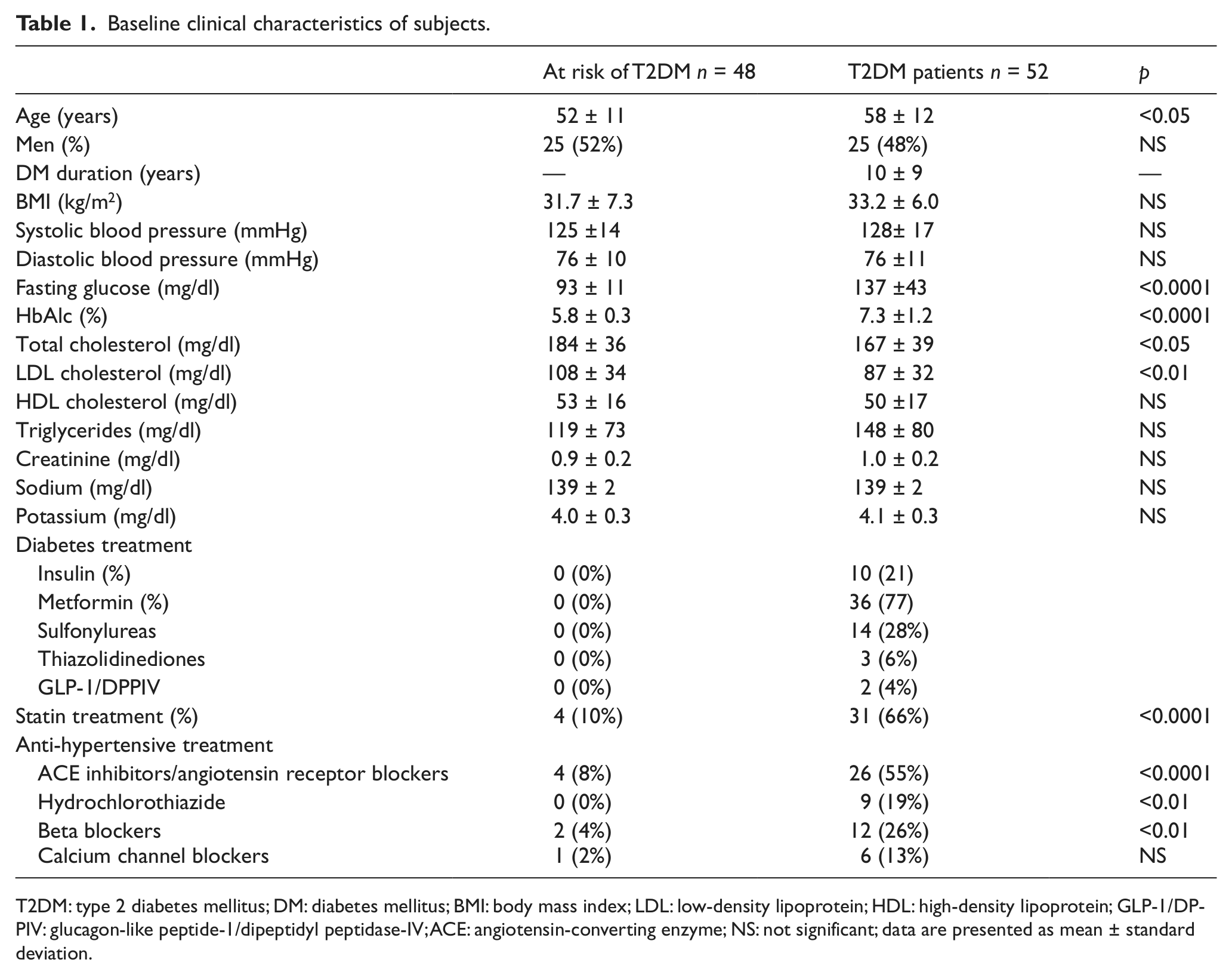
T2DM: type 2 diabetes mellitus; DM: diabetes mellitus; BMI: body mass index; LDL: low-density lipoprotein; HDL: high-density lipoprotein; GLP-1/DPPIV: glucagon-like peptide-1/dipeptidyl peptidase-IV; ACE: angiotensin-converting enzyme; NS: not significant; data are presented as mean ± standard deviation.
Baseline measurements of vascular reactivity and biochemical markers of endothelial function and inflammation are presented in Table 2. The macrophage inflammatory marker macrophage-derived chemokine (MDC) was slightly higher in the T2DM group than the at-risk group, but otherwise there were no significant differences in any other parameters between the diabetic and the at-risk groups.
Baseline measurements of vascular reactivity and biochemical markers of endothelial function.

T2DM: type 2 diabetes mellitus; NS: not significant; data are presented as mean ± standard deviation or median (25–75 percentiles).
Effect of aliskiren treatment in metabolic parameters, vascular reactivity, and inflammatory markers
Treatment with aliskiren was associated with a reduction in systolic (p < 0.05) and diastolic (p < 0.01) blood pressure compared to placebo among individuals with T2DM but not those at risk for the disease (Table 3). Skin endothelium-independent vasodilation (response to iontophoresis of sodium nitroprusside) was also significantly improved in the aliskiren-treated T2DM patients compared to placebo (p < 0.05) but not in the at-risk group (Figure 2). There was no change in endothelium-dependent vasodilation in either group. Soluble CD40 ligand, a biochemical marker of platelet activation and inflammation, was significantly decreased in aliskiren-treated T2DM patients compared to placebo (p < 0.01). Osteoprotegerin was significantly increased in the aliskiren-treated subjects at risk of T2DM when compared to placebo (p < 0.02). A similar trend was also present in T2DM patients but failed to reach statistical significance (p = 0.12). There were no other changes in serum markers of inflammation between aliskiren and placebo treatment in either study group.
Results of changes in demographic characteristics and in endothelial function in the two groups who completed a 12-week period treatment with aliskiren or placebo.

T2DM: type 2 diabetes mellitus; BMI: body mass index; LDL: low-density lipoprotein; HDL: high-density lipoprotein; NS: not significant.

Skin endothelium-independent vasodilation (response to iontophoresis of sodium nitroprusside) improvement in the aliskiren-treated T2DM patients compared to placebo.
Multivariate regression analysis showed that the effect of aliskiren on the endothelium-independent vasodilation was not influenced by co-treatment with ACE inhibitors and/or ARBs. However, co-treatment with beta blockers was negatively associated with the noticed improvement (p < 0.05).
Forearm skin biopsies
Eighteen at-risk and 32 T2DM subjects underwent forearm-skin biopsy at the baseline and exit visits. There were no major differences between the subjects who underwent a skin biopsy and those who did not. No baseline differences were observed between at-risk and T2DM patients in the total number of dermis cells or blood vessels per biopsy or the number of inflammatory cells around vessels, a strong indicator of chronic inflammation (Table 3). There were no neutrophilic infiltrates in any of the studied biopsies.
There were no changes in the above parameters with aliskiren or placebo treatment in either group (Table 3).
Adverse events
There were no episodes of hypotension or hyperkalemia and there were no significant changes in renal function as measured by eGFR during aliskiren treatment compared to placebo in either study group. Similarly, there was no significant change in serum creatinine or potassium levels in individuals with T2DM or in those at risk of developing T2DM compared to placebo (Table 3). There was a statistically significant but clinically modest decrease in serum sodium levels in the diabetic group treated with aliskiren versus placebo.
Discussion
We conducted a 12-week, double-blinded, placebo-controlled study examining the effect of aliskiren 150 mg daily versus placebo on metabolic parameters, serum markers of inflammation, and endothelial function in two study groups, one including individuals with T2DM and the other including those at high risk for developing the disease based on established risk factors. Importantly, our study population included individuals with normal renal function, normal electrolytes, and well-controlled hypertension at baseline. Our diabetic study group also had only modest hyperglycemia (average A1C 7.3%). Our results show that aliskiren reduces systolic and diastolic blood pressure and increases endothelium-independent vasodilation, which depends on vascular smooth muscle function, only in the T2DM study group. This improvement was not influenced by co-treatment with ACE inhibitors and/or ARBs and was negatively associated with co-treatment with beta blockers.
Activation of the RAAS leads to increased levels of angiotensin II, which acts on the vascular smooth muscle of the arterioles that express the angiotensin 1 (AT1) receptor and causes vasoconstriction. 14 Increased RAAS activity is known to be associated with the development of microvascular complications in diabetes. Increasing plasma levels of angiotensin II by peripheral infusion failed to suppress PRA.15,16 Increased renal perfusion in response to an angiotensin II antagonist in T2DM patients with suppressed baseline PRA has led to the hypothesis that there is increased intrarenal angiotensin II production in T2DM that could account for suppressed circulating renin. 17
Blockade of the RAAS through upstream direct renin inhibition may be particularly beneficial in individuals with T2DM. The recent Aliskiren in Left Ventricular Hypertrophy (ALLAY) study demonstrated that aliskiren treatment was associated with reduced LV mass in the diabetic subgroup. 10 Similarly, subjects with diabetes who were treated with aliskiren had reduced LV remodeling in the Aliskiren Study in Post-MI Patients to Reduce Remodeling (ASPIRE) study. 8 Of interest, no such benefits were observed in the nondiabetic participants in both studies.
The results of the present study are in agreement with the findings of the ALLAY and ASPIRE studies as they also demonstrate beneficial effects of aliskiren on systolic and diastolic blood pressure and the endothelium-independent vasodilation in T2DM patients. Of interest, no beneficial effects were observed in healthy subjects at risk of developing T2DM, a group that is known to have impaired vascular reactivity in both the micro- and macrocirculation. 2 These findings are compatible with animal studies in which aliskiren improved cardiovascular function in T2DM mouse models but not in nondiabetic animals. 18 The mechanisms underlying differential benefit among those with diabetes versus those subjects at risk for the disease remain unclear. The mean duration of diabetes in our subjects was 10 years, which may be long enough for individuals to have developed physiological changes in RAAS that partially explain our observed results. However, the exact mechanisms are not known at present and require further investigation.
A large portion (55%) of T2DM patients were being treated with an ACE inhibitor or ARB before enrollment in the study and continued the same treatment during the study. Treatment with these RAAS modulators was not a confounder in the observed results in multi-regression analysis. As previous aliskiren clinical trials in T2DM have included patients who were also receiving treatment with an ACE inhibitor or ARB, our results indicate that aliskiren exerted its beneficial effects regardless of further downstream inhibition of RAAS.
We did not observe significant hypotension, change in renal function, hyperkalemia or any other adverse events with aliskiren treatment compared to placebo in either study group. These results are in contrast with the recently published ALTITUDE trial that showed that aliskiren as an adjunct to treatment with an ACE inhibitor or ARB had no efficacy in preventing cardiovascular and renal events and was associated with hyperkalemia, renal impairment, and hypotension. 13 Differences in the dose of aliskiren and the study populations may, at least partially, account for the differences in adverse events in the present study compared to the ALTITUDE trial. We used a dose of 150 mg daily compared to 300 mg daily used in the ALTITUDE study. Additionally, we excluded subjects with cardiovascular and renal disease while the ALTITUDE study actually targeted this population. The results of the present study indicate that 150 mg of aliskiren daily may be more useful in the early stages of T2DM when severe cardiovascular and renal disease are not present.
Treatment with beta blockers was found to negatively affect the beneficial effects of aliskiren on endothelium-dependent vasodilation. This is not unexpected as beta blockers, especially nonselective ones, are known to cause vascular smooth muscle contraction and mild vasoconstriction in peripheral vessels. 19
In vitro and animal studies have shown that aliskiren has an anti-inflammatory effect in human mononuclear cells and in the kidney and retina of diabetic rats.20–22 In the present study, we examined the effect of aliskiren on forearm skin inflammation, as previous studies in our unit have indicated increased number of inflammatory cells in diabetic patients. 4 Our results indicate that aliskiren did not have any effect on skin inflammatory cells. Furthermore, no changes were observed between active and placebo treatment in the serum inflammatory cytokines in either study group. These results indicate that the observed changes in the skin microcirculation were not related to any anti-inflammatory function of aliskiren.
Aliskiren increased the serum levels of osteoprotegerin, a decoy receptor for the receptor activator of nuclear factor kappa B ligand (RANKL) in the group at risk of developing T2DM. Osteoprotegerin increases bone mineral density but has also been associated with LV hypertrophy and the development of coronary artery disease.23,24 In addition, aliskiren reduced the serum levels of the pro-inflammatory cytokine CD40 ligand (CD40L) in the T2DM group. CD40L has been associated with the development of atherosclerosis and is increased in both type 1 and 2 diabetes. CD40L can be reduced in diabetic patients by treatment with troglitazone, a thiazolidinedione. 25 To the best of our knowledge, this is the first time such effects of aliskiren are reported, and further studies will be required to investigate the mechanisms of these actions and their possible effects.
Aliskiren did not affect the resting skin blood flow in the present study. Previous studies in our unit have shown that valsartan, an ARB, increased the resting skin blood flow but had no effect on the endothelium-independent vasodilation. 26 This finding supports the concept that RAAS inhibition at different levels may have different effects on the microcirculation.
Our study has its limitations. First, the at-risk study group was younger than the T2DM group. However, in agreement with previous studies in our unit, there were no differences in the vascular reactivity measurements in these two groups, indicating that the difference in mean age between the groups did not present significant physiological differences affecting this measurement. 2 Second, the number of subjects who did not agree to have skin biopsies was considerable, which may have affected our ability to detect differences between groups and between treatments. In addition, our study population with T2DM had very good glycemic control and well-managed comorbidities, which does not necessarily reflect the larger population of people with T2DM in the United States and worldwide. Our findings must therefore be interpreted in the context of the population we studied. Finally, we did not measure PRA in this study. Renin inhibition has been shown to improve endothelial function and 24-hour blood pressure levels, 27 and PRA levels are useful for monitoring the effect of pharmacologic treatment of hypertension. 28 Several previous studies have extensively shown that aliskiren reduces PRA in a dose-dependent manner. 29 However, the magnitude of the reduction is affected by concomitant treatment with antihypertensives that increase PRA, including hydrochlorothiazide, ACE inhibitors, and ARBs. Given that the majority of our T2DM subjects but only a minority of at-risk subjects were treated with those medications, we do not believe that PRA measurements would have further elucidated aliskiren’s mechanism(s) of action on the vasculature in the current study.
In summary, this 12-week, double-blind, placebo-controlled study demonstrates that aliskiren improves blood pressure and vascular smooth muscle function in the skin microcirculation of T2DM patients. These effects were not seen in individuals at high risk for T2DM. Importantly, there were no adverse effects associated with the use of aliskiren in either of our groups.
Footnotes
Author contributions
AV was responsible for the study concept, design, and data analysis. JRD provided all medical care, researched data, and contributed to the discussion. FT performed all vascular reactivity evaluations and cytokine measurements and contributed to the discussion. MM was responsible for subject recruitment and follow-up. MA performed the skin biopsy immunostaining. AK performed the skin biopsy analysis. CG conducted the statistical analysis. TD performed the skin biopsies. JRD, FT, and AV wrote the manuscript. All authors participated in the data discussion, read the manuscript, and approved the final version.
Conflict of interest
None declared.
Funding
This was an investigator-initiated research study that was supported by a research grant from Novartis Pharmaceuticals Corporation (CSPP100AUS25T) to AV. The project was also supported by the Clinical Translational Science Award UL1RR025758 to Harvard University and Beth Israel Deaconess Medical Center from the National Center for Research Resources. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health. Clinical trial registration number: NCT01165983.
