Abstract
Background:
We compared the 2-year clinical outcomes between prediabetes and type 2 diabetes mellitus (T2DM) according to the pre-percutaneous coronary intervention (PCI) thrombolysis in myocardial infarction (TIMI) flow grade in patients with ST-segment elevation myocardial infarction.
Methods:
Overall, 6448 STEMI patients were divided into two groups: pre-PCI TIMI 0/1 group (n = 4854) and pre-PCI TIMI 2/3 group (n = 1594). They were further divided into patients with normoglycemia, prediabetes, and T2DM. The major endpoint was major adverse cardiac events (MACEs), defined as all-cause death, recurrent myocardial infarction, or any repeat revascularization.
Results:
In the pre-PCI TIMI 0/1 group, all-cause death rate was higher in both prediabetes (adjusted hazard ratio [aHR]: 1.633, p = 0.045) and T2DM (aHR: 2.064, p = 0.002) groups than in the normoglycemia group. In the pre-PCI TIMI 2/3 group, any repeat revascularization rate was also higher in both prediabetes (aHR: 2.511, p = 0.039) and T2DM (aHR: 3.156, p = 0.009) than normoglycemia. In each group (pre-PCI TIMI 0/1 or 2/3), the MACEs and all other clinical outcomes rates were similar between the prediabetes and T2DM groups.
Conclusions:
Prediabetes showed comparable worse clinical outcomes to those of T2DM regardless of the pre-PCI TIMI flow grade.
Introduction
Thrombolysis in myocardial infarction (TIMI) flow grade 2/3 before percutaneous coronary intervention (PCI) has shown better clinical outcomes in patients with ST-segment elevation myocardial infarction (STEMI) than TIMI flow grade 0/1.1,2 Patients with acute myocardial infarction (AMI) and diabetes mellitus (DM) show a more than two-fold higher risk for recurrent MI (Re-MI) and long-term mortality than patients without DM.3,4 According to recent reports, prediabetes is also associated with worse cardiovascular outcomes.5–7 However, studies6,7 comparing between prediabetes and diabetes did not investigate long-term clinical outcomes based on the TIMI flow grade. Hence, in this study, we aimed to compare the 2-year clinical outcomes between prediabetes and type 2 diabetes mellitus (T2DM) according to the pre-PCI TIMI flow grade of the infarct-related artery (IRA) in patients with ST-segment elevation myocardial infarction (STEMI) who underwent successful PCI with newer-generation drug-eluting stents (DESs).
Methods
Study population
Patients from the Korea AMI Registry (KAMIR) were included in this study. KAMIR 8 is a nationwide, prospective, multicenter registry in South Korea that was established in November 2005 to evaluate the current epidemiology, as well as short-term and long-term clinical outcomes of patients with AMI. The details of the registry can be found at the KAMIR website (http://www.kamir.or.kr). In this study, we tried to confine T2DM patients to diabetes cases. Therefore, we defined T2DM based on a previous study 9 that also included patients from the KAMIR. Hence, a total of 13,006 STEMI patients aged ⩾30 years at the onset of diabetes, who underwent successful PCI using newer-generation DESs from November 2005 to June 2015 were evaluated. Among them, patients with incomplete laboratory results including unidentified results of blood glycated hemoglobin (HbA1c) and blood glucose (n = 5895, 45.3%) and those who were lost to follow-up (n = 663, 5.1%) were excluded. Finally, a total of 6448 STEMI patients who underwent successful primary PCI with newer-generation DESs were included. These patients were divided into two groups: pre-PCI TIMI flow grade 0/1 group (n = 4854, 75.3%) and pre-PCI TIMI flow grade 2/3 group (n = 1594, 24.7%). Subsequently, these two groups were further divided into patients with normoglycemia (group A1 [n = 1303, 26.8%] and group A2 [n = 446, 28.0%]), prediabetes (group B1 [n = 1632, 33.6%] and group B2 [n = 471, 29.5%]), and T2DM (group C1 [n = 1919, 39.5%] and group C2 [n = 677, 42.5%]) (Figure 1). All patients provided written informed consent before enrollment. We followed up the enrolled patients through face-to-face interview, phone call, and chart review. A total of 6448 STEMI patients completed the scheduled follow-up. Additionally, all clinical events were evaluated by an independent event adjudicating committee. The process of event adjudication has been described in a previous publication of the KAMIR investigators. 8 The study protocol was approved by the institutional review board of each participating center, and the study was conducted in accordance with the 1975 Declaration of Helsinki.
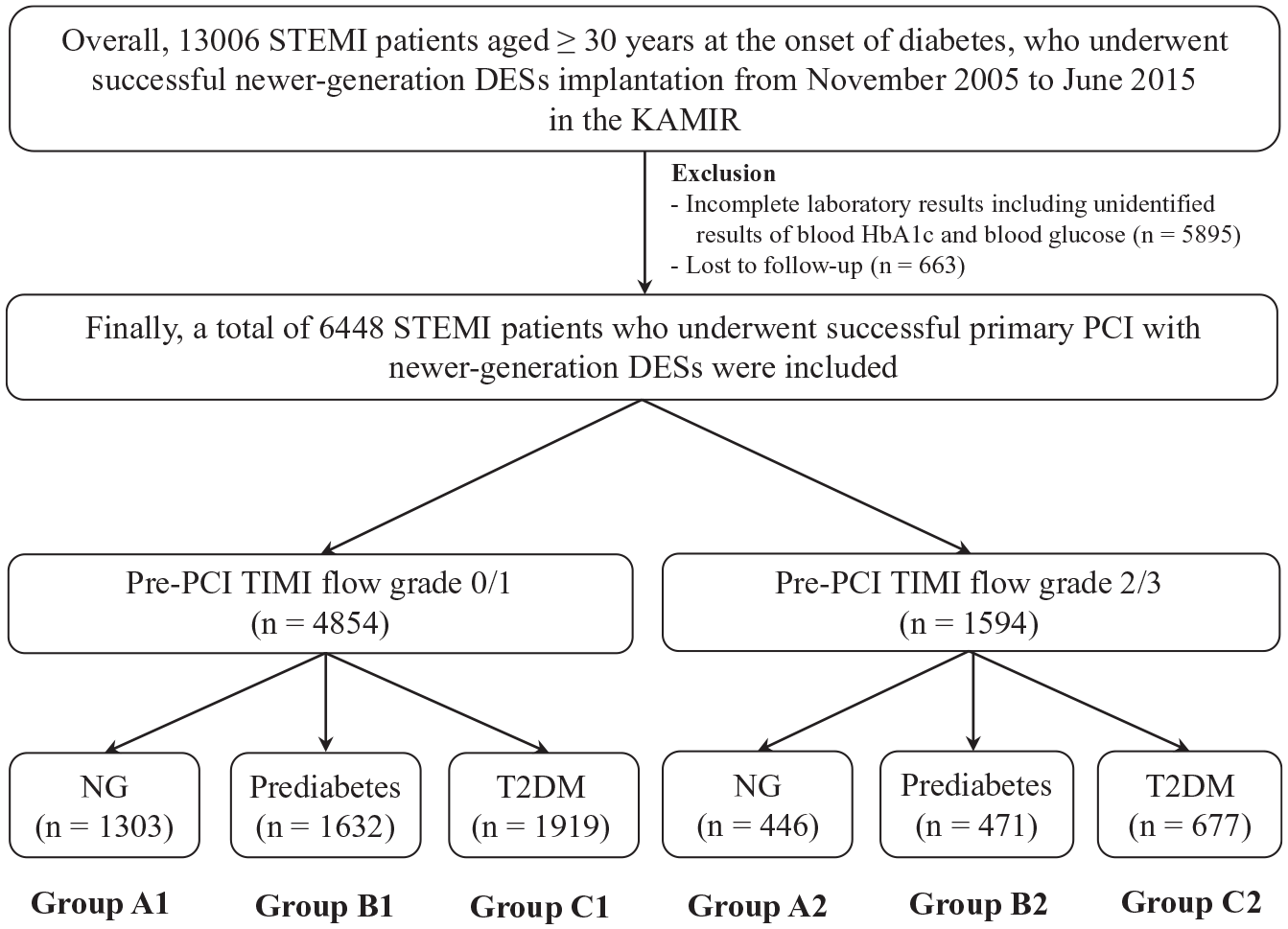
Flow chart.
PCI procedures and medical treatment
Diagnostic coronary angiography (CAG) and PCI were performed using standard techniques. 10 Unfractionated heparin at a dose of 50–100 IU/kg was administered during the procedure to maintain an activated clotting time of 250 to 300 s. Before PCI, all patients received loading doses of aspirin (200–300 mg) and other anti-platelet agents, including clopidogrel, ticagrelor, or prasugrel before PCI. After the index PCI, aspirin at a dose of 100 mg once daily was continued indefinitely, and clopidogrel (75 mg once daily) was recommended for at least 1 year and dual antiplatelet therapy (DAPT; a combination of aspirin 100 mg/day with clopidogrel 75 mg/day or ticagrelor 90 mg twice daily or prasugrel 5–10 mg/day) was recommended for at least 1 year. Optimal medications therapy, including β-blocker, renin-angiotensin system blockade, calcium channel blocker, and lipid-lowering agents were also recommended to all patients at the discretion of the responsible clinicians. Additionally, based on previous reports11,12 triple antiplatelet therapy (TAPT, aspirin + clopidogrel + cilostazol [100 mg twice daily]) has been used and the use of TAPT was left to the discretion of the individual operators.
Study definitions and clinical outcomes
The inclusion criteria of STEMI was defined according to the current guidelines.13,14 The degree of coronary flow before PCI was classified according to TIMI flow grade as assessed by the investigators. 15 Glycemic categories were determined according to medical history and HbA1c and fasting plasma glucose (FPG) or random plasma glucose (RPG) levels at the index hospitalization. 7 We classified glycemic categories according to the American Diabetes Association guidelines. 16 Normoglycemia was defined as HbA1c < 5.7% and FPG < 100 mg/dL (5.6 mmol/L), prediabetes as HbA1c 5.7%–6.4% and FPG 100–125 mg/dL (5.6–6.9 mmol/L), and T2DM as known T2DM for which patients receive medical treatment (insulin or antidiabetics) or as newly diagnosed T2DM defined as HbA1c ⩾ 6.5%, FPG ⩾ 126 mg/dL (7.0 mmol/L), or RPG ⩾ 200 mg/dL (11.1 mmol/L). 7 If there were some discrepancy between the level of HbA1c and those FPG or RPG, we made the level of HbA1c a priority. 17 The major clinical endpoint of this study was the occurrence of major adverse cardiac events (MACEs), defined as all-cause death, Re-MI, or any coronary repeat revascularization. All-cause death was classified as cardiac death (CD) or non-CD. Any repeat revascularization included target lesion revascularization (TLR), target vessel revascularization (TVR), and non-TVR. The definitions of successful PCI, complete or incomplete revascularization, Re-MI, TLR, TVR, and non-TVR were also previously reported.18,19
Statistical analysis
Baseline characteristics were described by using counts and percentage for categorical data and the differences between the groups were analyzed using the chi-square test or Fisher’s exact test. In case of continuous variables, data are expressed as mean ± standard deviation, or as median (quartile 1–quartile 3). The differences among the three groups were evaluated using analysis of variance or the Jonckheere-Terpstra test. Moreover, post-hoc analysis between two groups was performed using the Hochberg test or Dunnett T3 test. Various clinical outcomes were estimated using Kaplan-Meier curve analysis, and differences between the groups were compared using the log-rank test. A two-tailed p-value of <0.05 was considered statistically significant. In order to adjust for possible confounders as much as possible, the Cox proportional hazard regression analysis was performed. Variables with a p value of <0.001 in the univariate analysis were entered into the multivariate analysis. The statistical analysis was performed with Statistical Package for the Social Sciences (SPSS) version 20 (IBM, Armonk, NY, USA).
Results
Baseline characteristics
Tables 1 and 2 and Supplemental Material 1 show the baseline characteristics of the study population. In both the pre-PCI TIMI flow grade 0/1 and 2/3 groups, the normoglycemia group (groups A1 and A2) included the highest proportion of men, highest prescription rates of ticagrelor, and angiotensin-converting enzyme inhibitors, highest number of patients with single-vessel disease, and highest mean diameter of deployed stents. The prediabetes group (groups B1 and B2) included the greatest number of smokers, patients with the highest mean value of total cholesterol and low-density lipoprotein-cholesterol, and patients with the highest prescription rates of clopidogrel. The T2DM group (groups C1 and C2) included the oldest mean patient age; the highest number of patients with hypertension, dyslipidemia, and a previous history of PCI; the greatest proportion of patients who received the highest number of stents; the highest mean values of N-terminal pro-brain natriuretic peptide and triglyceride; the highest prescription rates of angiotensin receptor blockers; the highest number of patients who had the right coronary artery as the IRA and treated vessel; the highest number of patients with three-vessel disease; and the lowest mean estimated glomerular filtration rates.
Baseline clinical, laboratory, and discharge medication characteristics.
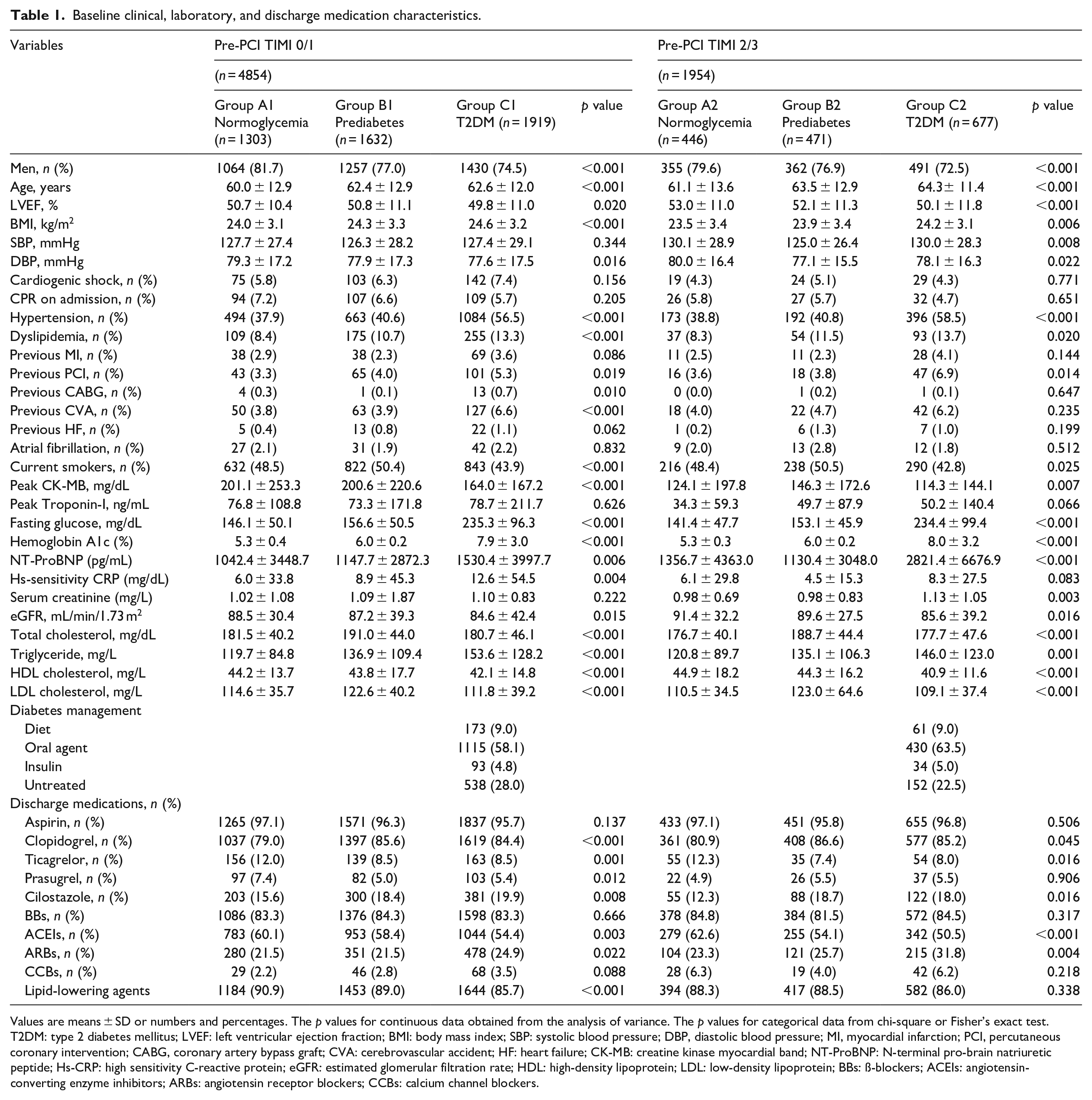
Values are means ± SD or numbers and percentages. The p values for continuous data obtained from the analysis of variance. The p values for categorical data from chi-square or Fisher’s exact test.
T2DM: type 2 diabetes mellitus; LVEF: left ventricular ejection fraction; BMI: body mass index; SBP: systolic blood pressure; DBP, diastolic blood pressure; MI, myocardial infarction; PCI, percutaneous coronary intervention; CABG, coronary artery bypass graft; CVA: cerebrovascular accident; HF: heart failure; CK-MB: creatine kinase myocardial band; NT-ProBNP: N-terminal pro-brain natriuretic peptide; Hs-CRP: high sensitivity C-reactive protein; eGFR: estimated glomerular filtration rate; HDL: high-density lipoprotein; LDL: low-density lipoprotein; BBs: ß-blockers; ACEIs: angiotensin-converting enzyme inhibitors; ARBs: angiotensin receptor blockers; CCBs: calcium channel blockers.
Angiographic characteristics.
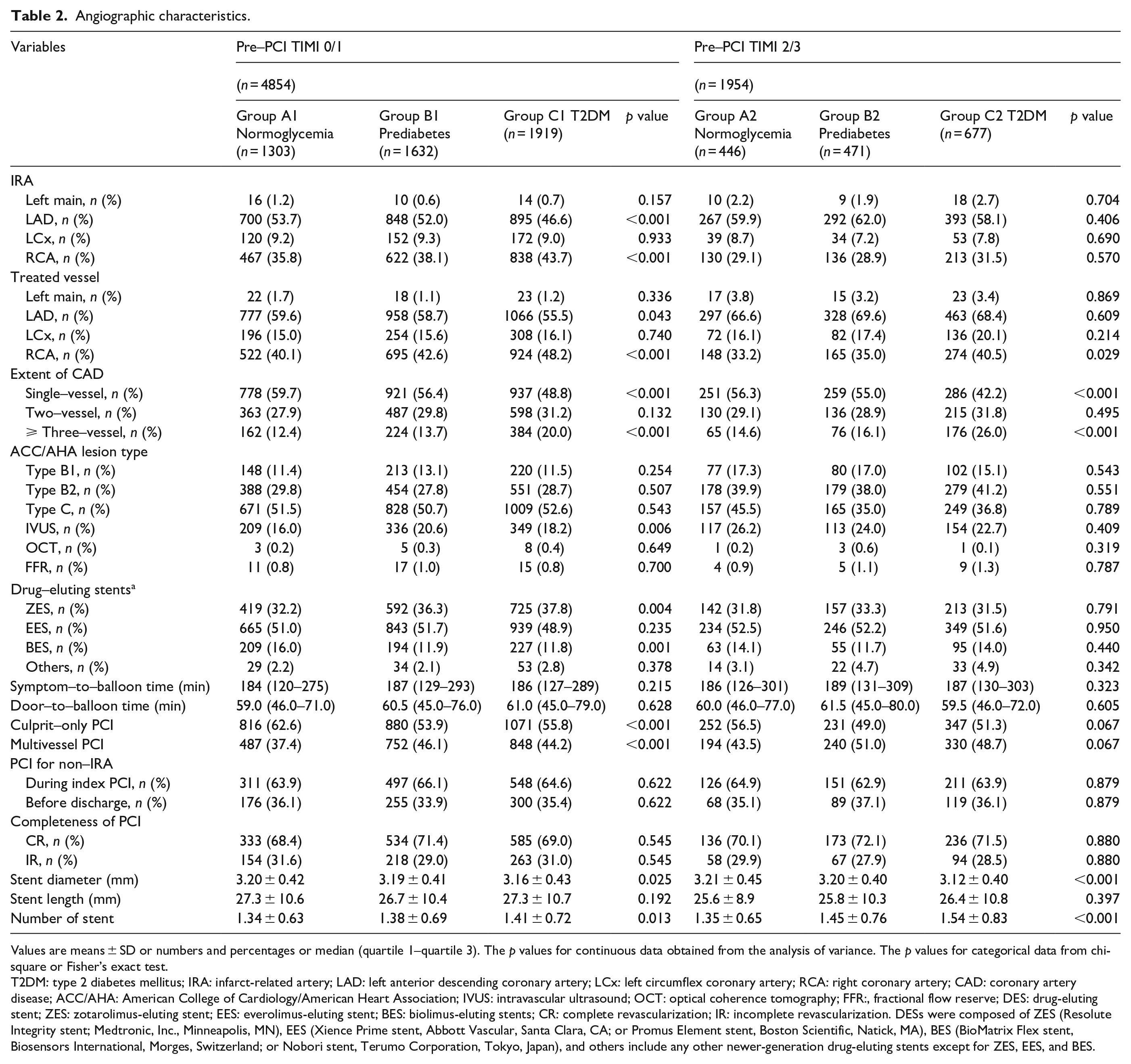
Values are means ± SD or numbers and percentages or median (quartile 1–quartile 3). The p values for continuous data obtained from the analysis of variance. The p values for categorical data from chi-square or Fisher’s exact test.
T2DM: type 2 diabetes mellitus; IRA: infarct-related artery; LAD: left anterior descending coronary artery; LCx: left circumflex coronary artery; RCA: right coronary artery; CAD: coronary artery disease; ACC/AHA: American College of Cardiology/American Heart Association; IVUS: intravascular ultrasound; OCT: optical coherence tomography; FFR:, fractional flow reserve; DES: drug-eluting stent; ZES: zotarolimus-eluting stent; EES: everolimus-eluting stent; BES: biolimus-eluting stents; CR: complete revascularization; IR: incomplete revascularization. DESs were composed of ZES (Resolute Integrity stent; Medtronic, Inc., Minneapolis, MN), EES (Xience Prime stent, Abbott Vascular, Santa Clara, CA; or Promus Element stent, Boston Scientific, Natick, MA), BES (BioMatrix Flex stent, Biosensors International, Morges, Switzerland; or Nobori stent, Terumo Corporation, Tokyo, Japan), and others include any other newer-generation drug-eluting stents except for ZES, EES, and BES.
Clinical outcomes
The cumulative incidences of major clinical outcomes during the 2-year follow-up period are summarized in Table 3 and Figure 2. In the total study population, after adjustment, the cumulative incidences of MACEs (adjusted hazard ratio [aHR]: 1.380; 95% confidence interval [CI]: 1.073–1.775; p = 0.012), all-cause death (aHR: 1.602; 95% CI: 1.092–2.350; p = 0.016), CD (aHR: 1.993; 95% CI: 1.262–3.149; p = 0.003), and Re-MI (aHR: 1.717; 95% CI: 1.030–2.860; p = 0.038) were significantly higher in the pre-PCI TIMI flow grade 0/1 group than in the pre-PCI TIMI flow grade 2/3 group. The cumulative incidences of MACEs (aHR: 1.402; 95% CI: 1.069–1.839; p = 0.010), all-cause death (aHR: 1.530; 95% CI: 1.009–2.319; p = 0.035), and CD (aHR: 1.630; 95% CI: 1.011–2.628; p = 0.032) were significantly higher in the prediabetes group (groups B1 and B2) than in the normoglycemia group (groups A1 and A2). The cumulative incidences of MACEs (aHR: 1.716; 95% CI: 1.332–2.211; p < 0.001), all-cause death (aHR: 1.879; 95% CI: 1.265–2.791; p = 0.002), CD (aHR: 2.117; 95% CI: 1.345–3.333; p = 0.001), and any repeat revascularization (aHR: 1.597; 95% CI: 1.087–2.348; p = 0.017) were significantly higher in the T2DM group (groups C1 and C2) than in the normoglycemia group. Moreover, the cumulative incidence MACEs (aHR: 1.277; 95% CI: 1.029–1.584; p = 0.026) was significantly higher in the T2DM group (groups C1 and C2) than in the prediabetes group (groups B1 and B2). However, the cumulative incidences of all-cause death, CD, Re-MI, and any repeat revascularization were similar between these two groups (group B1 and B2 vs group C1 and C2) (Table 3).
Clinical outcomes by Kaplan-Meier analysis and Cox-proportional hazard ratio analysis at 2 years.
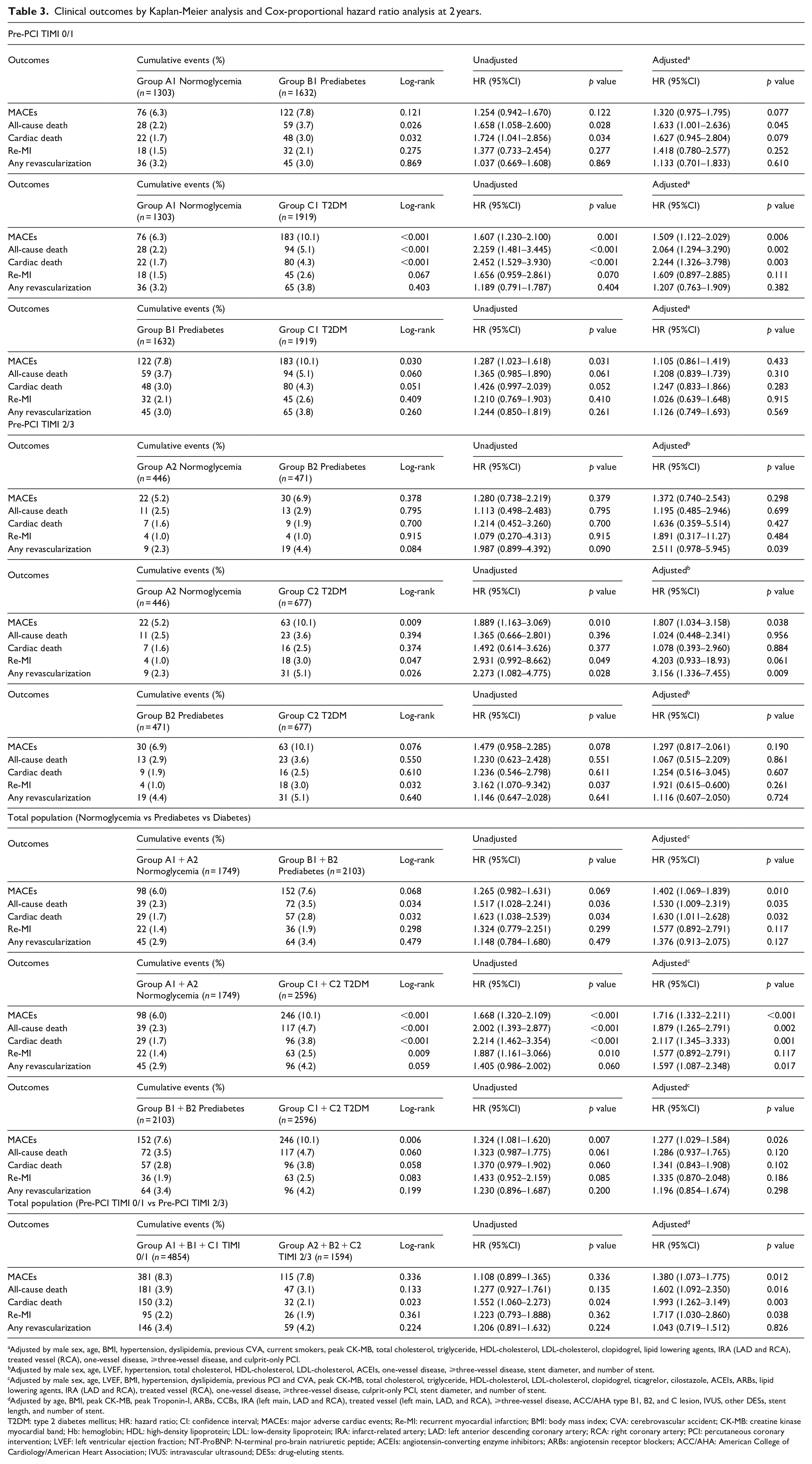
Adjusted by male sex, age, BMI, hypertension, dyslipidemia, previous CVA, current smokers, peak CK-MB, total cholesterol, triglyceride, HDL-cholesterol, LDL-cholesterol, clopidogrel, lipid lowering agents, IRA (LAD and RCA), treated vessel (RCA), one-vessel disease, ⩾three-vessel disease, and culprit-only PCI.
Adjusted by male sex, age, LVEF, hypertension, total cholesterol, HDL-cholesterol, LDL-cholesterol, ACEIs, one-vessel disease, ⩾three-vessel disease, stent diameter, and number of stent.
Adjusted by male sex, age, LVEF, BMI, hypertension, dyslipidemia, previous PCI and CVA, peak CK-MB, total cholesterol, triglyceride, HDL-cholesterol, LDL-cholesterol, clopidogrel, ticagrelor, cilostazole, ACEIs, ARBs, lipid lowering agents, IRA (LAD and RCA), treated vessel (RCA), one-vessel disease, ⩾three-vessel disease, culprit-only PCI, stent diameter, and number of stent.
Adjusted by age, BMI, peak CK-MB, peak Troponin-I, ARBs, CCBs, IRA (left main, LAD and RCA), treated vessel (left main, LAD, and RCA), ⩾three-vessel disease, ACC/AHA type B1, B2, and C lesion, IVUS, other DESs, stent length, and number of stent.
T2DM: type 2 diabetes mellitus; HR: hazard ratio; CI: confidence interval; MACEs: major adverse cardiac events; Re-MI: recurrent myocardial infarction; BMI: body mass index; CVA: cerebrovascular accident; CK-MB: creatine kinase myocardial band; Hb: hemoglobin; HDL: high-density lipoprotein; LDL: low-density lipoprotein; IRA: infarct-related artery; LAD: left anterior descending coronary artery; RCA: right coronary artery; PCI: percutaneous coronary intervention; LVEF: left ventricular ejection fraction; NT-ProBNP: N-terminal pro-brain natriuretic peptide; ACEIs: angiotensin-converting enzyme inhibitors; ARBs: angiotensin receptor blockers; ACC/AHA: American College of Cardiology/American Heart Association; IVUS: intravascular ultrasound; DESs: drug-eluting stents.
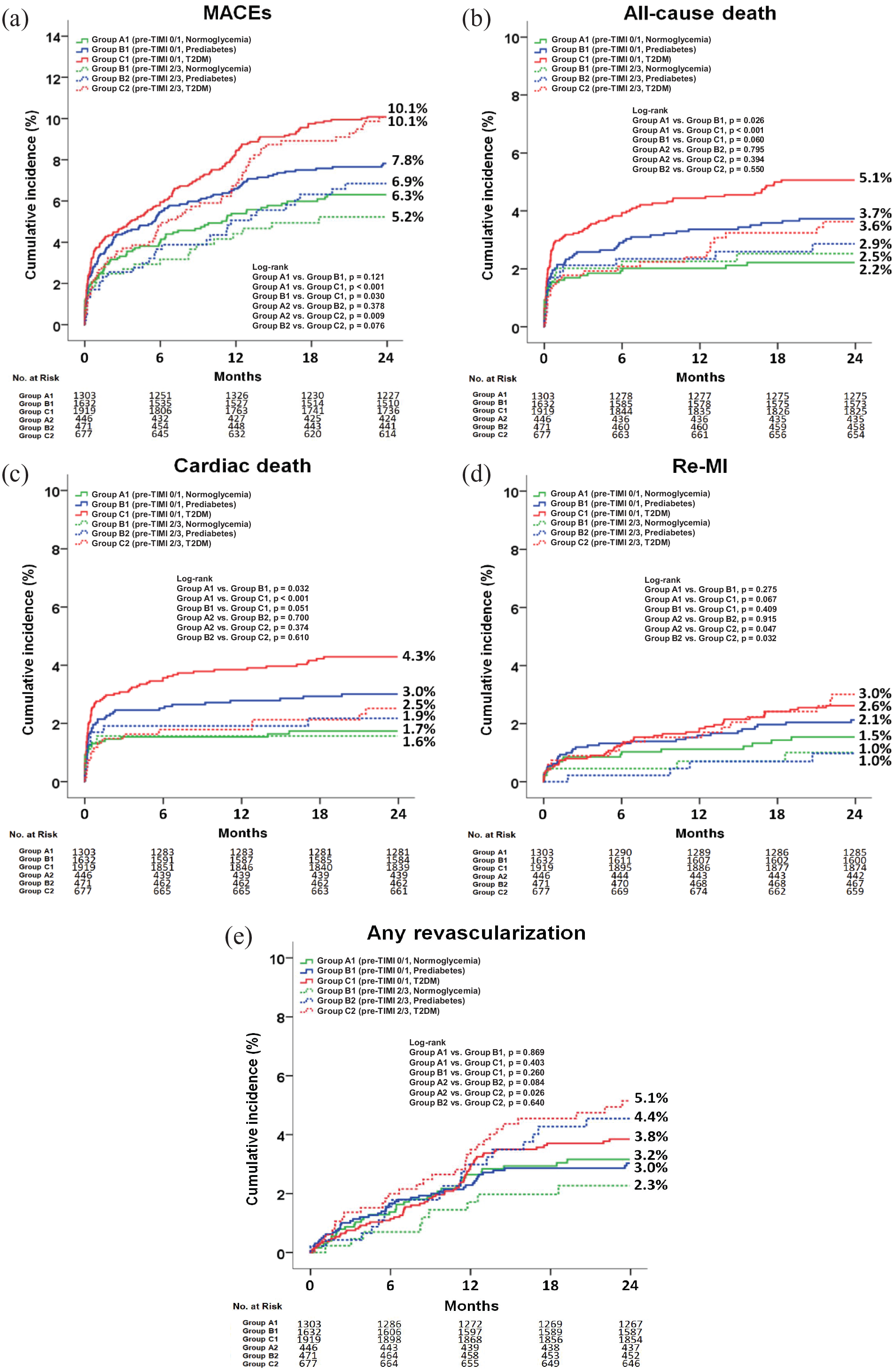
Kaplan-Meier analysis for MACEs (a), all-cause death (b), cardiac death (c), Re-MI (d), and any repeat revascularization (e) at 2 years.
In the pre-PCI TIMI flow grade 0/1 group, after adjustment, the cumulative incidence of all-cause death (aHR: 1.633; 95% CI: 1.001–2.636; p = 0.045) was significantly higher in group B1 than in group A1. The cumulative incidences of MACEs (aHR: 1.509; 95% CI: 1.122–2.029; p = 0.006), all-cause death (aHR: 2.064; 95% CI: 1.294–3.290; p = 0.002), and CD (aHR: 2.244; 95% CI: 1.326–3.798; p = 0.003) were also significantly higher in group C1 than in group A1. However, the cumulative incidences of MACEs, all-cause death, CD, Re-MI, and any repeat revascularization were similar between groups B1 and C1.
In the pre-PCI TIMI flow grade 2/3 group, after adjustment, the cumulative incidence of any repeat revascularization (aHR: 2.511; 95% CI: 0.978–5.945; p = 0.039) was significantly higher in group B2 than in group A2. The cumulative incidences of MACEs (aHR: 1.807; 95% CI: 1.034–3.158; p = 0.038) and any repeat revascularization (aHR: 3.156; 95% CI: 1.336–7.455; p = 0.009) were also significantly higher in group C2 than in group A2. However, the cumulative incidences of MACEs, all-cause death, CD, Re-MI, and any repeat revascularization were similar between groups B2 and C2.
Table 4 shows the independent predictors for all-cause death and any repeat revascularization in the pre-PCI TIMI flow grade 0/1 group at 2 years. Old age (⩾65 years) decreased left ventricular ejection fraction (LVEF, <40%), dyslipidemia, use of lipid-lowering agents, more than three-vessel disease, and a >28 mm length of the deployed stent were independent predictors of MACEs. More than three-vessel disease and culprit-only PCI were independent predictors of any repeat revascularization. Table 5 shows the independent predictors of all-cause death and any repeat revascularization in the pre-PCI TIMI flow grade 2/3 group at 2 years. Decreased LVEF, use of lipid-lowering agents, and culprit-only PCI were independent predictors of all-cause death. More than three-vessel disease, culprit-only PCI, a <3.0 mm diameter of deployed stents, and a >28 mm length of deployed stents were independent predictors of any repeat revascularization in this study.
Independent predictors for all-cause death and any revascularization in pre-PCI TIMI grade 0/1 group at 2 years.
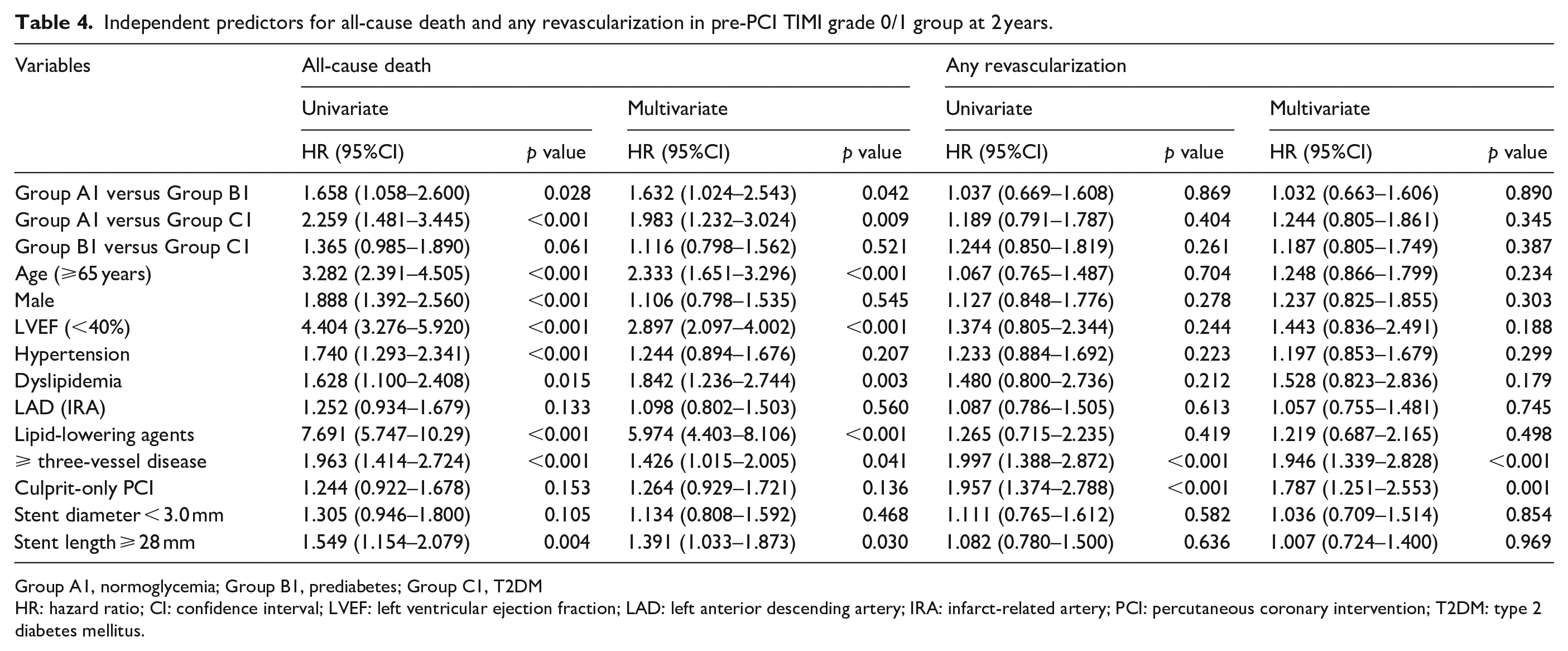
Group A1, normoglycemia; Group B1, prediabetes; Group C1, T2DM
HR: hazard ratio; CI: confidence interval; LVEF: left ventricular ejection fraction; LAD: left anterior descending artery; IRA: infarct-related artery; PCI: percutaneous coronary intervention; T2DM: type 2 diabetes mellitus.
Independent predictors for all-cause death and any revascularization in pre-PCI TIMI grade 2/3 group at 2 years.
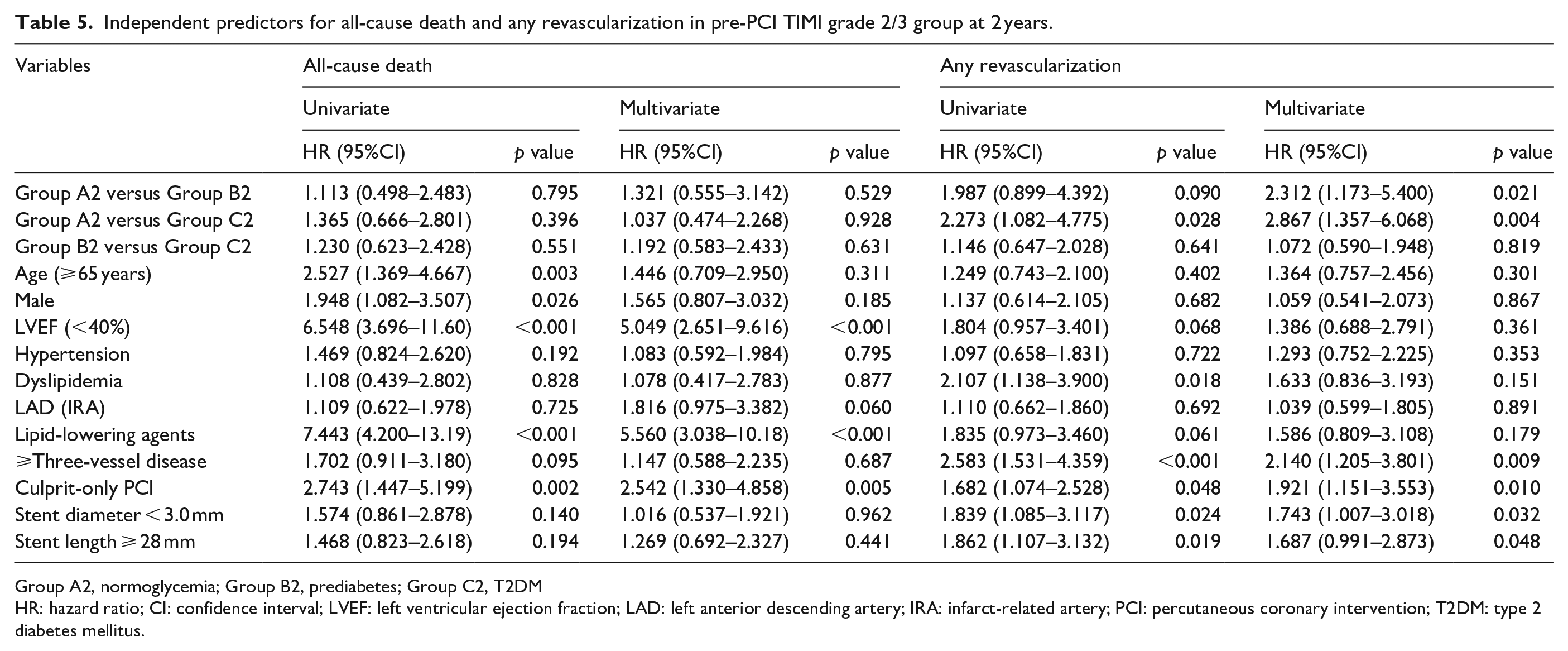
Group A2, normoglycemia; Group B2, prediabetes; Group C2, T2DM
HR: hazard ratio; CI: confidence interval; LVEF: left ventricular ejection fraction; LAD: left anterior descending artery; IRA: infarct-related artery; PCI: percutaneous coronary intervention; T2DM: type 2 diabetes mellitus.
Discussion
The main findings of this study were as follows: (1) In the total study population, the cumulative incidences of MACEs, all-cause death, CD, and Re-MI were significantly higher in the pre-PCI TIMI flow grade 0/1 group (groups A1, B1, and C1) than in the pre-PCI TIMI flow grade 2/3 group (groups A2, B2, and C2), and the cumulative incidence of MACEs was significantly higher in the T2DM group (groups C1 and C2) than in the prediabetes group (groups B1 and B2). (2) However, in each group (pre-PCI TIMI flow grade 0/1 or 2/3), the cumulative incidences of all major clinical outcomes were not significantly different between the prediabetes and T2DM groups (group B1 vs C1 or group B2 vs C2). (3) In the pre-PCI TIMI flow grade 0/1 group, the cumulative incidence of all-cause death was higher in both the prediabetes and T2DM groups (groups B1 and C1) than in the normoglycemia group (group A1). (4) In the pre-PCI TIMI flow grade 2/3 group, the cumulative incidence of any repeat revascularization was higher in both the prediabetes and T2DM groups (groups B2 and C2) than in the normoglycemia group (group A2).
The main focus of this study was comparing the major clinical outcomes between prediabetes and T2DM in patients with STEMI based on the TIMI flow grade of the IRA, especially after the implantation of newer-generation DESs. As previously mentioned, TIMI flow grade 2/3 at the initial CAG has been reported to show better clinical outcomes than TIMI flow grade 0/1 in patients with STEMI.1,2 The suggested explanations for this result include the observation that compared with “a lack of patency (TIMI flow grade 0/1),” “patency (TIMI flow 2/3)” 20 had lower in-hospital rates for new-onset heart failure and hypotension and were less likely to require intubation for respiratory failure, which are all surrogates of improved myocardial function. 1 Moreover, patients with TIMI flow grade 3 at the initial CAG were more likely to have TIMI flow grade 3 at the end of the procedure.1,21 Recent reports22,23 also showed that the patency of the IRA in patients with STEMI was related to procedural success and decreased enzymatic infarct size, fatal arrhythmic events, and in-hospital mortality. Because recent studies5–7 did not use TIMI flow grade as a variable in estimating the clinical outcomes between these two groups, we considered that the TIMI flow grade can be used as a meaningful variable for the comparison of major clinical outcomes between prediabetes and T2DM.
In this study, the pre-PCI TIMI flow 2/3 group showed better major clinical outcomes, including the cumulative incidences of MACEs, all-cause death, CD, and Re-MI, in the total study population (Table 3). These results were consistent with previous findings.1,2 In each group (pre-PCI TIMI flow 0/1 or 2/3), the major clinical outcomes were similar between prediabetes and T2DM. Despite these comparable results, in the total study population, the cumulative incidence of MACEs (aHR: 1.277; 95% CI: 1.029–1.584; p = 0.026, Table 3) was significantly higher in the T2DM group than in the prediabetes group. This difference of MACE rates may be related to the summation of numerical differences in each group (pre-PCI TIMI flow 0/1 or 2/3) between prediabetes and T2DM. Hence, we can speculate that prediabetes was a worse condition similar to T2DM regardless of pre-PCI TIMI flow grade in this study.
The suggested underlying pathological mechanisms related to the adverse clinical outcomes in hyperglycemia include thrombus formation, vascular inflammation, and platelet aggregation.24,25 Previous data suggesting that even in a population with diabetes and treated according to present guidelines post-MI, the risk for recurrent cardiovascular events has decreased to some extent but still is higher compared to a population without diabetes. 26 Moreover, prediabetes is associated with worse clinical outcomes.5,26 However, these harmful effects of prediabetes on long-term clinical outcomes in patients with AMI are relatively less well understood than those of diabetes. Published reports17,27,28 demonstrated that the comparative clinical outcomes were similar between prediabetes and diabetes. In contrast, other report 29 suggested that prediabetes is not associated with long-term adverse cardiovascular outcomes in patients with coronary artery disease and PCI. However, these studies27–29 were not confined to STEMI patients and newer-generation DES. Therefore, we believe that our results could be more suitable for reflecting the contemporary real-world practice of primary PCI.
Based on our results, we suggest that much more attention and careful treatment strategies similar to those for T2DM are required for prediabetes population. Furthermore, we believe that our study can provide meaningful information about the significance of prediabetes to interventional cardiologists who perform PCI with contemporary newer-generation DESs in patients with STEMI.
Limitations
This study had several limitations. First, although KAMIR is prospective registry, 8 this is a retrospective study. Therefore, some data might have usual limitations and biases inherent to retrospective study designs.7,30 Second, we defined T2DM according to medical history and adult-onset diabetes (after the age of 30). However, <5% of the diabetes cases that occur in adulthood are type 1 DM. 31 Hence these might have affected the outcomes. Third, compared with an oral glucose test, measurement of HbA1c may not be ideal. 7 Moreover, it has been proposed that impaired glucose tolerance (IGT) prediabetes may be associated with a higher cardiovascular risk compared to the impaired fasting glucose (IFG) subtype. 32 However, in this KAMIR data, prediabetes was not classified according to these subtypes (isolated IGT, isolated FGT, and combined IGT and IFG). Therefore, these were other important bias in this study. Fourth, we could not provide complete information about the degree of glycemic control in the enrolled patients during the follow-up period because detailed information on this variable was not included in KAMIR. 30 Fifth, we could not consider the duration of diabetes as a major determinant factor for long-term clinical outcomes in this study because lack of information of this variable. Sixth, because this study was based on discharge medications, we could not precisely know the adherence or non-adherence to antidiabetic drugs of the enrolled patients.7,17,30 Seventh, although multivariate analysis was performed to account for baseline differences, the effect of unmeasured baseline confounders or variables not included in KAMIR may have affected the study outcomes. 7 Finally, because the information regarding number of patients who received warfarin or new oral anticoagulants (NOACs) were not included in this registry, we could not provide the results of combination therapy of DAPT and warfarin or NOACs.
Conclusion
In the era of contemporary newer-generation DESs, prediabetes was found to show worse clinical outcomes similar to those of diabetes regardless of pre-PCI TIMI flow grade in this study. However, larger randomized controlled studies are warranted to confirm these results in the future.
Supplemental Material
sj-pdf-1-dvr-10.1177_1479164121991505 – Supplemental material for Prediabetes versus type 2 diabetes mellitus based on pre-percutaneous coronary intervention thrombolysis in myocardial infarction flow grade in patients with ST-segment elevation myocardial infarction after successful newer-generation drug-eluting stent implantation
Supplemental material, sj-pdf-1-dvr-10.1177_1479164121991505 for Prediabetes versus type 2 diabetes mellitus based on pre-percutaneous coronary intervention thrombolysis in myocardial infarction flow grade in patients with ST-segment elevation myocardial infarction after successful newer-generation drug-eluting stent implantation by Yong Hoon Kim, Ae-Young Her, Myung Ho Jeong, Byeong-Keuk Kim, Sung-Jin Hong, Seunghwan Kim, Chul-Min Ahn, Jung-Sun Kim, Young-Guk Ko, Donghoon Choi, Myeong-Ki Hong and Yangsoo Jang in Diabetes & Vascular Disease Research
Footnotes
Acknowledgements
The authors thank all of the clinical investigators who contributed time and effort to this study, as well as the Korea Acute Myocardial Infarction (KAMIR) Investigators. Korea Acute Myocardial infarction Registry (KAMIR) investigators. Myung Ho Jeong, MD, Youngkeun Ahn, MD, Sung Chul Chae, MD, Jong Hyun Kim, MD, Seung-Ho Hur, MD, Young Jo Kim, MD, In Whan Seong, MD, Donghoon Choi, MD, Jei Keon Chae, MD, Taek Jong Hong, MD, Jae Young Rhew, MD, Doo-Il Kim, MD, In-Ho Chae, MD, Junghan Yoon, MD, Bon-Kwon Koo, MD, Byung-Ok Kim, MD, Myoung Yong Lee, MD, Kee-Sik Kim, MD, Jin-Yong Hwang, MD, Myeong Chan Cho, MD, Seok Kyu Oh, MD, Nae-Hee Lee, MD, Kyoung Tae Jeong, MD, Seung-Jea Tahk, MD, Jang-Ho Bae, MD, Seung-Woon Rha, MD, Keum-Soo Park, MD, Chong Jin Kim, MD, Kyoo-Rok Han, MD, Tae Hoon Ahn, MD, Moo-Hyun Kim, MD, Ki Bae Seung, MD, Wook Sung Chung, MD, Ju-Young Yang, MD, Chong Yun Rhim, MD, Hyeon-Cheol Gwon, MD, Seong-Wook Park, MD, Young-Youp Koh, MD, Seung Jae Joo, MD, Soo-Joong Kim, MD, Dong Kyu Jin, MD, Jin Man Cho, MD, Sang-Wook Kim, MD, Jeong Kyung Kim, MD, Tae Ik Kim, MD, Deug Young Nah, MD, Si Hoon Park, MD, Sang Hyun Lee, MD, Seung Uk Lee, MD, Hang-Jae Chung, MD, Jang-Hyun Cho, MD, Seung Won Jin, MD, Myeong-Ki Hong, MD, Yangsoo Jang, MD, Jeong Gwan Cho, MD, Hyo-Soo Kim, MD and Seung-Jung Park, MD.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a fund (2016-ER6304-02) by Research of Korea Centers for Disease Control and Prevention.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
