Abstract
Introduction:
The objective of this article is to investigate the combination of telmisartan with vildagliptin therapy versus monotherapy of vildagliptin and telmisartan on diabetic nephropathy in type 2 diabetes mellitus rats.
Materials and methods:
In adult rats streptozotocin (65 mg/kg) and nicotinamide (110 mg/kg) were injected intraperitoneally to produce diabetic nephropathy. Rats of either sex allotted to the following groups: (i) triple therapy: metformin (120 mg/kg, o.d.) + pioglitazone (1.25 mg/kg, o.d.) + glimepiride (0.7 mg/kg, o.d.); (ii) dual therapy: vildagliptin (8.76 mg/kg, o.d.) + telmisartan (6.48 mg/kg, o.d.); (iii) vildagliptin (8.76 mg/kg, o.d.); and (iv) telmisartan (6.48 mg/kg, o.d.); therapy was carried out for 35 days orally. Weekly at days 7, 14, 21, 28 and 35, blood pressure, blood glucose level, body weight, blood serum creatinine level, protein albumin level in urine, and blood urea nitrogen (BUN) were estimated. Renal structural changes were observed.
Results:
Blood pressure, blood glucose level, blood serum creatinine level, protein albumin level in urine, BUN and renal deterioration increased significantly in diabetic rats compared with normal control rats. The vildagliptin + telmisartan treatment group showed no weight gain and controlled blood pressure, renovascular structural and biochemical parameters in diabetic neuropathy rats.
Conclusions:
The addition of telmisartan to vildagliptin demonstrated the best control over blood pressure, glycemia and diabetic nephropathy markers, renal structural changes and improvement of renal function as opposed to monotherapy with either drug, possibly because of the dual inhibitory effect on the renin–angiotensin system.
Keywords
Introduction
Diabetic nephropathy (DN) is the leading cause of renal failure. It is defined by proteinuria > 500 mg in 24 hours in the setting of diabetes, but this is preceded by lower degrees of proteinuria, or “microalbuminuria.” Microalbuminuria is defined as albumin excretion of 30–299 mg/24 hours. Without intervention, diabetic patients with microalbuminuria typically progress to proteinuria and overt DN. This progression occurs in types 1 and 2 diabetes. The pathological changes to the kidney include increased glomerular basement membrane (GBM) thickness, microaneurysm formation, mesangial nodule formation (Kimmelsteil-Wilson bodies), and other changes. As many as 7% of patients with type 2 diabetes may already have microalbuminuria at the time they are diagnosed with diabetes. 1
DN, or nephropatia diabetica, also known as Kimmelstiel-Wilson syndrome, or nodular diabetic glomerulosclerosis and intercapillary glomerulonephritis, is a progressive kidney disease caused by angiopathy of capillaries in the kidney glomeruli. It is characterized by nephrotic syndrome and diffuse glomerulosclerosis. It is due to longstanding diabetes mellitus and is a prime indication for dialysis in many Western countries. DN is a common complication of diabetes mellitus in which there is long-term damage to the kidneys as a result of long-term poorly controlled diabetes. The renal vessels and the glomerulus are the main areas affected. DN is characterized by the presence of a protein called albumin in the urine, hypertension (high blood pressure), edema (swelling) and progressive renal insufficiency. 2
Vildagliptin is an antihyperglycemic agent that selectively inhibits the dipeptidyl peptidase-4 (DPP-4) enzyme. Such inhibition prevents the degradation of the incretin hormones glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). This results in improved glycemic control as determined by glycated hemoglobin (HbA1c) and fasting plasma glucose (FPG) levels and, in addition, an enhancement of pancreatic α- and β-cell function. Vildagliptin is indicated in the European Union and elsewhere in the world for the management of type 2 diabetes mellitus in combination with metformin, a sulfonylurea or a thiazolidinedione in patients with inadequate glycemic control following monotherapy. Vildagliptin is also available as a fixed-dose formulation with metformin. 3
One study has demonstrated that treatment with telmisartan significantly decreased fasting blood glucose (FBG). The antidiabetic activity of telmisartan may be due to improved glucose metabolism via the blocking of the inhibitory effect of angiotensin II (Ang II) on insulin signal transmission and telmisartan’s function as a moderately potent partial peroxisome proliferator-activated receptor gamma (PPAR-γ) agonist activating the receptor to 25–30% of the levels of the full agonists, such as the thiazolidinedione agents. There are structural resemblances between the angiotensin II receptor blocker (ARB) agents and the full PPAR-γ agonists, such as rosiglitazone and pioglitazone. 4
Oral vildagliptin in combination with metformin, a sulfonylurea or a thiazolidinedione, improved glycemic control in adults with type 2 diabetes and appeared to slow the progression of β-cell degeneration in trials of 24–52 weeks’ duration. In trials in patients with diabetes inadequately controlled with metformin, vildagliptin provided an additional reduction of HbA1c levels of 1.1% and was shown to be as effective as pioglitazone as add-on therapy in a noninferiority trial. Vildagliptin had a low risk of hypoglycemia, was weight-neutral overall and was generally well tolerated. 3
Further investigation is required to accurately position vildagliptin relative to other drugs. This further investigation may be based on a combination of vildagliptin and several angiotensin-converting enzyme inhibitors /ARBs. Randomized studies have demonstrated that ARBs, controlling systemic blood pressure, delay progression of proteinuria, reduce microalbuminuria, prevent progression to macroalbuminuria and maintain renal function in patients with DN. Telmisartan is a member of the ARB class and is being investigated in the most ambitious and far-reaching research program ever conducted with an ARB. Telmisartan has a number of features that may make it particularly suitable for the treatment of DN. In addition to its long duration of action and almost exclusive fecal excretion, its high lipophilicity should assist in tissue penetration. 5
So, the present study focuses on dual therapy of vildagliptin and telmisartan on DN in experimentally induced type-2 diabetic mellitus rats.
Materials and methods
Animals
Adult Wistar Albino Rats weighing 150 to 250 g bred in the Animal House, Gyan Vihar School of Pharmacy, Suresh Gyan Vihar University, were used. All animal experiments were approved by the Institutional Animal Ethical Committee (Gyan Vihar School of Pharmacy), Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA) No. 1234/a/08/CPCSEA. The animals were housed in polycarbonate cages in a room with a 12-hour (h) day-night cycle, temperature of 24 ± 2°C and humidity of 45 to 64%. During the whole experimental period, animals were fed with a balanced commercial diet and water ad libitum.
Induction of diabetic neuropathy
Nicotinamide-streptozotocin (STZ)-induced type 2 diabetes in the rat6–9
Noninsulin-dependent diabetes mellitus was induced in overnight fasted rats by a single intraperitonial injection of 65 mg/kg body weight STZ, 15 minutes (min) after the intraperitonial administration of 110 mg/kg body weight of nicotinamide. STZ was dissolved in citrate buffer (0.1 M, pH 4.5) and nicotinamide was dissolved in normal saline. The injection site was the dorsal midpoint between the pelvis and ribs close to the right side of the spine. Hyperglycemia was confirmed by the elevated glucose levels in plasma, determined at 72 h and then on day 7 after injection. Animals with blood glucose concentration more than 200 mg/dl were used for the study.
Drugs and chemicals
Ketamine (Themis Medicare Ltd, Mumbai, India), STZ (Sigma Aldrich), nicotinamide (Lobal Chemie), vildagliptin (Novartis Pharma) and telmisartan (Aristo Pharma Ltd. Mumbai, India) were used in the current study.
Experimental protocol
Animals used included insulin-resistant (IR) type 2 diabetic rats (induced by intraperitoneal STZ and nicotinamide). IR rats of either sex were randomly allotted into different experimental groups, each containing six animals. Drug treatment (i) triple therapy: metformin (120 mg/kg, o.d.) + pioglitazone (1.25 mg/kg, o.d.) + glimepiride (0.7 mg/kg, o.d.); (ii) dual therapy: vildagliptin (8.76 mg/kg) + telmisartan (6.48 mg/kg); (iii) vildagliptin (8.76 mg/kg); and (iv) telmisartan (6.48 mg/kg) was carried out for 35 days with the help of an oral catheter every morning. Weekly at day 7, 14, 21, 28 and 35, blood pressure, blood glucose level, body weight, blood serum creatinine level, protein albumin level in urine, blood urea nitrogen (BUN) was estimated in STZ-nicotinamide-induced DN in rats (Table 1).
List of experimental groups used.

STZ: streptozotocin.
Collection of blood samples
At the end of drug treatment, all the animals were fasted overnight but allowed free access to water. The next morning, a blood sample was withdrawn by retro orbital sinus under mild ether anesthesia. The blood samples were collected into vacutainers precoated with ethylenediaminetetraacetic acid (EDTA) as anticoagulant. Blood samples were centrifuged at 3000 rpm for 10 min in a refrigerated centrifuge (i.e. temperature 4°C). The plasma separated as straw-colored supernatant was used for various biochemical parameters. It was stored at −20°C until the completion of the analysis.
Collection of urine sample
At the end of drug treatment, all the animals were kept in metabolic cages for 24 h. Animals were fasted but allowed free access to water. Urine samples were collected after 24 h in urine-collecting bottles.
Biochemical estimations
Measurement of renal function and biochemical parameters
Blood glucose was measured by Accu-Chek Active glucose strips. The blood glucose estimation was performed weekly after administration of the test compound.
Albumin excretion rate and total protein excretion rate in urine were measured using Span and Ranbaxy diagnostic kits by autoanalyzer (Echo, Logotech Pvt. Ltd, India).
BUN values were measured by the BUN GLDH kit (Bhat Bio-tech Pvt. Ltd, Bangalore, India) technique as per instructions of the kit manufacturers.
A BUN test measures the amount of nitrogen in blood that comes from the waste product urea. Urea is made when protein is broken down in the body. Urea is made in the liver and passed out of the body in urine.
A BUN test is performed to see how well the kidneys are working. If the kidneys are not able to remove urea from the blood normally, the BUN level rises. Heart failure, dehydration, or a diet high in protein can also make the BUN level higher. Liver disease or damage can lower the BUN level.
Serum creatinine rate was measured using CREATININE KIT by Mod. Jaffe’s Kinetic Method (Coral Clinical System, Goa, India).
Creatinine is the catabolic product of creatinine phosphate, which is used by the skeletal muscle. The daily production depends on muscular mass, and it is excreted out of the body entirely by the kidneys. Elevated levels are found in renal dysfunction, reduced renal blood flow (shock, dehydration, congestive heart failure) and diabetes acromegaly. Decreased levels are found in muscular dystrophy.
Blood pressure was monitored using the tail-cuff method for all of the groups.
Histopathological examination
At the end of the experiments, all rats were sacrificed and pathological analysis of the kidney was performed. The kidney tissues were preserved in buffered neutral formalin and stored at −20 ºC until processed for histopathology. Tissues were preserved in 1% w/v glutaraldehyde and 4% w/v formaldehyde in phosphate buffer, pH −7.2 at 4ºC until processed for electron microscopy. Tissues were processed for histopathology at room temperature and involved the following steps: (a) fixation, (b) processing of tissues — dehydrating, clearing and embedding, (c) preparation and cutting of sections, and (d) attaching sections to slides. After processing, sections were stained with hematoxylin-eosin stain using Harris’s alum hematoxylin and stock 1% w/v alcohol eosin solution. The stained sections were finally mounted in DPX Mountant.
Statistical analysis
Results were shown as mean ± SEM. The paired Student’s t test was used to analyze differences in variables before and after treatment. Comparison between different groups was performed using one-way analysis of variance (ANOVA) followed by a Student-Newman-Keuls method. P values <0.05 were considered statistically significant. Statistical analysis was performed by using Sigma Stat 3.5.
Results
Glycemic control
After the completion of the study protocol, glycemic control improved in both treatment groups during the study; a similar proportion of animals in each treatment group achieved a decrease in FBG, vildagliptin + telmisartan (181.55 ± to 120 ± mg/dl, p < 0.001), and vildagliptin alone (169.16 ± to 131 ± mg/dl, p < 0.01) as compared to the diabetic group (Table 2; Figure 1).
Effect of drugs on fasting blood glucose level in diabetic nephropathy rats.
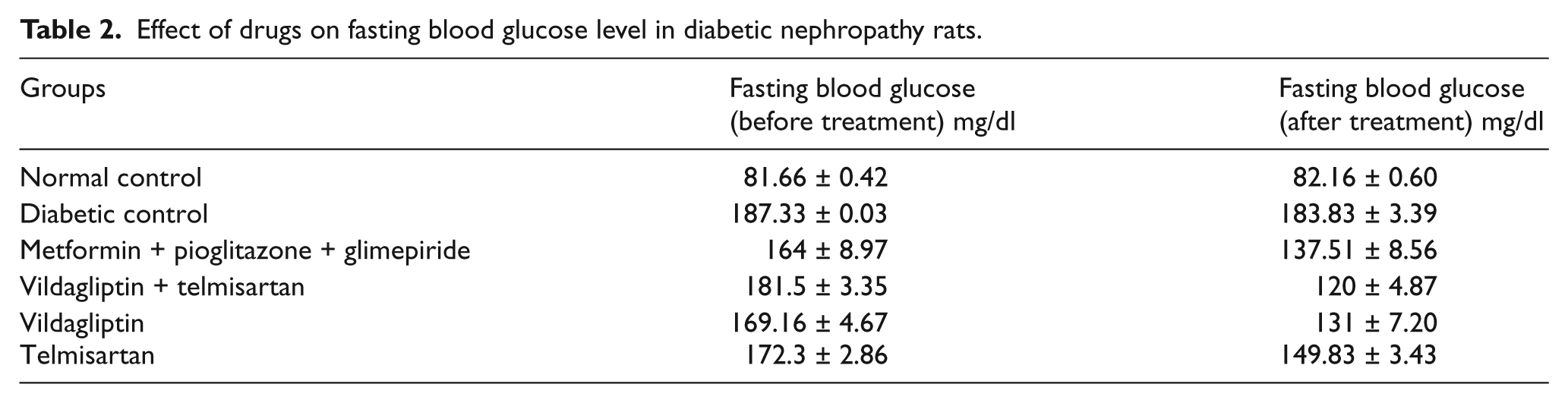

Fasting blood glucose (mg/dl) of different groups analyzed before and after drug treatment. Metformin + pioglitazone + glimepiride; vildagliptin + telmisartan; vildagliptin only; telmisartan only, values are mean ± SEM; n = 6; ***p < 0.001; **p < 0.01; *p < 0.05; p > 0.05 is considered nonsignificant (ns) as compared before and after drug treatment (t test).
Albumin excretion rate in urine
The vildagliptin + telmisartan treatment group showed a significant decrease in urine albumin level (UAL) (96.02 ± 4.13 to 56.52 ± 2.45 mg/day, p < 0.001), while vildagliptin alone caused a decrease in UAL (96.02 ± 4.13 to 69.66 ± 3.48 mg/dl, p < 0.001) as compared to the diabetic group (Table 3; Figure 2).
Effect of drugs on albumin excretion rate (mg/dl) in diabetic nephropathy rats.


Urine albumin level (mg/dl) of different groups analyzed before and after drug treatment. Metformin + pioglitazone + glimepiride; vildagliptin + telmisartan; vildagliptin only; telmisartan only, values are mean ± SEM; n = 6; ***p < 0.001; **p < 0.01; *p < 0.05; p > 0.05 is considered nonsignificant (ns) as compared to diabetic control (t test).
Serum creatinine level
The vildagliptin + telmisartan treatment group showed a significant decrease in serum creatinine level (206.69 ± 7.36 to 126.06 ± 7.51 µmol/l, p<0.001), while vildagliptin alone caused a decrease in serum creatinine level (206.69 ± 7.36 to 141.23 ± µmol/l, p<0.01) as compared to the diabetic group (Table 4; Figure 3).
Effect of drugs on serum creatinine level in diabetic nephropathy rats.

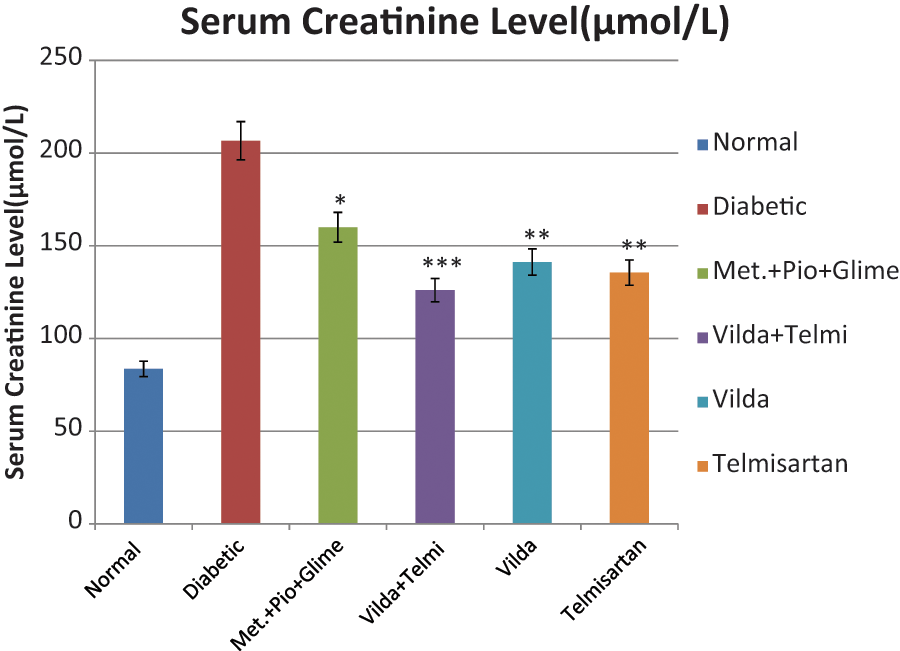
Serum creatinine level (µmol/l) of different groups analyzed before and after drug treatment. Metformin + pioglitazone + glimepiride; vildagliptin + telmisartan; vildagliptin only; temisartan only, values are mean ± SEM; n = 6; ***p < 0.001; **p < 0.01; *p < 0.05; p > 0.05 is considered nonsignificant (ns) as compared the diabetic control (t test).
Body weight
During the study a significant increase in body weight was observed in the metformin + glimepiride + pioglitazone (control) group, while in the vildagliptin + telmisartan and vildagliptin-only groups, no weight gain was seen as compared to the diabetic group (Table 5; Figure 4).
Effect of drugs on body weight in diabetic nephropathy rats.


Body weight variation (grams) of different groups analyzed before and after drug treatment. Metformin + pioglitazone + glimepiride; vildagliptin + telmisartan; vildagliptin only; telmisartan only; values are mean ± SEM; n = 6; ***p < 0.001; **p < 0.01; *p < 0.05; p > 0.05 is considered nonsignificant (ns) as compared to diabetic control (t test).
BUN
BUN was significantly increased in the diabetic group (20.91 ± 0.32 mg/dl to 105.66 ± 4.11 mg/dl, p<0.001) as compared with the normal control group. There was a significant decrease in BUN in the vildagliptin + telmisartan-treated group (105.66 ± 4.11 mg/dl to 39.16 ± 3.93 mg/dl, p < 0.001) compared with the untreated diabetic group (Table 6; Figure 5).
Effect of drugs on blood urea nitrogen in diabetic nephropathy rats.


Blood urea nitrogen (mg/dl) of different groups analyzed before and after drug treatment. Metformin + pioglitazone + glimepiride; vildagliptin + telmisartan; vildagliptin only; telmisartan only; values are mean ± SEM; n = 6; ***p < 0.001; **p < 0.01; *p < 0.05; p > 0.05 is considered nonsignificant (ns) as compared to diabetic control (t test).
Blood pressure measurement of different groups
Blood pressure was significantly increased in the diabetic group (105.62 ± 2.72 mmHg diastolic; 145.85 ± 4.02 mmHg systolic, p < 0.001) as compared with normal control group (66.91 ± 2.91 mmHg diastolic; 112.37 ± 3.82 mmHg systolic). There was a significant decrease in blood pressure in the vildagliptin + telmisartan-treated group (67.23 ± 2.73 mmHg diastolic; 104.41 ± 3.07 mmHg systolic, p < 0.001) compared with the untreated diabetic group and triple therapy group (98.16 ± 4.01 mmHg diastolic; 110.09 ± 3.14 mmHg systolic) (Table 7; Figure 6).
Effect of drugs on blood pressure in diabetic nephropathy rats.


Blood pressure (mmHg) diastolic/systolic of different groups analyzed before and after drug treatment. Metformin + pioglitazone + glimepiride; vildagliptin + telmisartan; vildagliptin only; telmisartan only, values are mean ± SEM; n = 6; ***p < 0.001; **p < 0.01; *p < 0.05; p > 0.05 is considered nonsignificant (ns) as compared to diabetic control (t test).
Histopathology study in different groups
Histopathological examination of kidney tissues was performed at the end of the experiment. After 35 days of examination, the rats were sacrificed by a single intraperitonial injection of kitamine. Light microscopy study in kidney tissue sections stained with hematoxylin-eosin revealed glomerulosclerosis and interstitial fibrosis, tubular vacuolization (TV), thickening of the GBM, mesangial matrix expansion (ME) and nodular lesion (NL). In the diabetic group (Figure 8) maximum renovascular degeneration was found, but in the treatment groups these changes were attenuated. The vildagliptin plus telmisartan-treated group (Figure 10) showed maximum renoprotection as compared to vildagliptin alone (Figure 10) and the standard (pioglitazone + glemipiride + metformin)-treated group (Figure 9) because of the absence of glomerulosclerosis and interstitial fibrosis, NL and improvement of GBM thickness and mesangial ME. In the diabetic group (Figure 8), increased glomerulosclerosis and interstitial fibrosis, TV, thickening of the GBM, mesangial ME and NL was observed as compared to the normal group (Figure 7), but in the treatment groups these changes were attenuated, possibly because of the different drugs’ renoprotective mechanisms.

Photomicrograph a of section of the normal control group (after eight weeks of treatment with purified water). There is no degree of deterioration by glomerulosclerosis (GS) or interstitial fibrosis (IF), tubular vacuolization (TV), thickening of the glomerular basement membrane, mesangial matrix expansion (ME) or nodular lesion (NL).

Photomicrograph of a section of the diabetic control group showing nephropathy (after eight weeks of treatment with purified water). There is a high degree of deterioration by glomerulosclerosis (GS) and interstitial fibrosis (IF), tubular vacuolization (TV), thickening of the glomerular basement membrane, mesangial matrix expansion (ME) and nodular lesion (NL).

Photomicrograph of a section of the standard group treated with the combination of metformin, pioglitazone and glimepiride, showing nephropathy (after eight weeks of treatment). There is a lesser degree of deterioration by glomerulosclerosis (GS) and interstitial fibrosis (IF), tubular vacuolization (TV), thickening of the glomerular basement membrane, mesangial matrix expansion (ME) and nodular lesion (NL), but not as significant as with the dual therapy.

Photomicrograph of a section of the treatment group treated with the combination of vildagliptin and telmisartan showing nephropathy (after eight weeks of treatment). There is a lesser degree of deterioration by glomerulosclerosis (GS) and interstitial fibrosis (IF), tubular vacuolization (TV), thickening of the glomerular basement membrane, mesangial matrix expansion (ME) and nodular lesion (NL).
Discussion
Captopril, lisinopril, quinapril and fosinopril are currently used ACE inhibitors to treat DN. ACE inhibitors increase the bioavailability of nitric oxide (NO) and activate nitric oxide synthase (eNOS) by inhibiting synthesis of Ang II. Lisinopril inhibits the formation of transforming growth factor-beta (TGF-ß) and tubulointerstitial fibrosis in DN patients. Proteinuria, glomerular hypertrophy and tubulointerstitial fibrosis were reduced by fosinopril in experimental DN. Quinapril reduces proteinuria, cholesterol levels, glomerular lesions and podocyte damage in DN. AT1 ARBs like candesartan and telmisartan have been noted to attenuate DN by reducing proteinuria. ABT-62 and endothelin (ETA) receptor antagonists have been noted to reduce proteinuria in experimental DN.10,11
Palosuran, a novel and selective urotensin-II receptor blocker, was noted to reduce albuminuria in renal disease. Fenofibrate activates the peroxisome proliferator-activated receptor-α (PPAR-α) and produces a renoprotective effect by suppressing renal plasminogen activator inhibitor type 1 (PAI-1) in experimental DN. 12
PPAR-γ agonists such as pioglitazone and rosiglitazone significantly reduced glomerulosclerosis and tubulointerstitial fibrosis in patients with DN. In addition, pioglitazone markedly reduced glomerular hypertrophy, mesangial expansion and urinary albumin excretion in patients with DN. 13
Recently it has been suggested that suppression of the Rho-kinase pathway by fasudil, a selective Rho kinase inhibitor, could be a novel strategy to treat DN by downregulating TGF-ß and reducing reactive oxygen species (ROS) formation. Inhibition of 3-hydroxy-3-methyl coenzyme A (HMG-CoA)-reductase by statins like atorvastatin, pravastatin and cerivastatin was noted to activate eNOS, maintain glomerular filtration rate (GFR) and renal cortical blood flow and consequently reduce glomerular lesions. Resveratrol, a polyphenolic phytoalexin and potent antioxidant present in red wine, attenuated renal dysfunction by reducing proteinuria and ROS formation in rats with DN. On the basis of this discussion, it may be suggested that the above-mentioned drugs may have improved renal function in nephropathy because of their properties of protecting the function of vascular endothelium. 14
Our study has demonstrated that treatment with vildagliptin and telmisartan significantly decreased FBG. The antidiabetic activity of vildagliptin is due to improved insulin secretion and peripheral insulin sensitivity. The DPP-4 inhibitor vildagliptin is a novel class of antidiabetic medication. Inhibition of DPP-4 by vildagliptin prevents degradation of GLP-1 and reduces glycemia in patients with type 2 diabetes mellitus, with a low risk for hypoglycemia and no weight gain. Vildagliptin binds covalently to the catalytic site of DPP-4, eliciting prolonged enzyme inhibition. This raises intact GLP-1 levels, both after meal ingestion and in the fasting state. 14
The combination of vildagliptin and telmisartan has shown to significantly decrease symptoms of DN. Diabetic rats treated with vildagliptin and telmisartan showed a reduction in albumin excretion rate, serum creatinine rate, BUN, FBG and renal structural changes. There were also reported markedly changes in albuminuria, protenuria that is a marker and potential contributor to renal injury that accompanies DN. Interventions that have ameliorated the progression of DN have been associated with a reduction in urinary protein excretion.
Recent large landmark clinical studies have shown that intensive glucose control reduces the risk of the development and progression of DN, and the blockade RAS is also an important target for both metabolic and hemodynamic derangements in DN. It has been demonstrated that treatment with telmisartan significantly decreases FBG. The antidiabetic activity of telmisartan may be due to improved glucose metabolism via blocking of the inhibitory effect of Ang II on insulin signal transmission and telmisartan’s function as a moderately potent partial PPAR-γ agonist activating the receptor to 25–30% of the levels of the full agonists, such as the thiazolidinedione agents. There are structural resemblances between the ARB agents and the full PPAR-γ agonists, such as rosiglitazone and pioglitazone. 4
Renoprotective therapy should aim to achieve the maximal antialbuminuric effect. There are several mechanisms whereby increased microalbuminuria and activation of the RAS has been shown to play a role in the pathogenesis of renal diseases. Hyperglycemia as an observed symptom in diabetes greatly activates the RAS. Activation of RAS leads to Ang II formation. AT-1 receptor stimulation by Ang II may directly induce synthesis of TGF-β1. Ang II, the effector molecule of RAS, stimulates extracellular matrix protein synthesis through induction of TGF-β1 in mesangial cells, renal interstitial fibroblasts and proximal tubular epithelial cells. Thus, the reduction in plasma TGF-β1 concentration with telmisartan, demonstrated in the present study, may reflect attenuated TGF-β1 synthesis by all of these renal cell types. Pro-inflammatory cytokines may play a significant role in the development of renal injury in type 2 diabetes. Therefore, results from experimental studies indicate that inhibition of Ang II receptor activity is associated with beneficial renal effects, suggesting that modulation of this cytokine may have a real clinical application for the treatment of DN.8,9
Blood pressure is the most important clinical driver/progression factor of DN. Careful monitoring is thus a must in any DN study. In the present study, blood pressure in DN rats was monitored. Vildagliptin has a poor blood pressure effect, whereas the telmisartan group had better systolic and diastolic blood pressure control. However, the combination of vildagliptin and telmisartan acts synergistically and has the best control over blood pressure, glycemia and DN markers. We have shown that the dual therapy of vildagliptin and telmisartan resulted in a greater decrease in blood glucose, albumin excretion rate, serum creatinine rate, BUN and renal structural changes than treatment with vildagliptin alone. The result of the present work was that the combination of vildagliptin with telmisartan could be more proficient in preventing STZ-induced type 2 DN than an individual drug therapy or triple therapy, and our results support our hypothesis. Histopathological findings also have demonstrated recovery in glomerulosclerosis, interstitial fibrosis, NL, TV, mesangial ME and thickening of GBM by the combination of vildagliptin and telmisartan.
Conclusion
The hypothesis of the present work was to compare the dual therapy of vildagliptin with telmisartan versus vildagliptin alone on STZ-induced type 2 DN in rats. Our results show that the combination of vildagliptin with telmisartan is more effective in ameliorating DN than vildagliptin or telmisartan single-drug therapy or triple therapy, by suppressing blood pressure, albumin excretion rate in urine, total serum creatinine rate and BUN during the development of STZ-induced type 2 DN. Finally, the significant effect of combined therapy (vildagliptin plus telmisartan) could be a result of synergistic/potentiative action on the RAS, and together they may be able to target multiple mechanisms involved in the pathophysiology of DN.
Footnotes
Conflict of interest
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
