Abstract
Introduction
Within society, it is now widely accepted that mobile phones are a ubiquitous commodity.1–3 As a result, there has been a noticeable rise in the prevalence of mobile health (mHealth) focused interventions, with almost 83% of clinicians now using smart devices or medical apps. 4 The proliferation of mHealth technology has aided the development of smart apps as an effective medium for the delivery of digital health in several long-term health conditions such as asthma, 5 mental health conditions, 6 obesity, 7 and cancer. 8 mHealth, and more specifically digital health provisions, are continuing to gain traction as a contextually appropriate and accessible way to improve health-related behaviours for young people living with chronic diseases.9–11
Moreover, the emerging role of mHealth within cancer treatment is notable 12 with adherence and access to preoperative rehabilitation and rehabilitation considered the largest barriers for people suffering from the disease. 13 Paediatric oncology is also emerging as a focus within mHealth research,14,15 often highlighting that paediatric populations undergoing cancer treatment have significantly lower health-related quality of life outcomes than other long-term health conditions populations. 16 Exercise has been shown to increase health related quality of life, strength and aerobic fitness and decrease fatigue. 17 However, paediatric oncology patients report additional barriers to exercise when compared to healthy age matched peers, such as fatigue, fear of injury, overprotective attitudes and lack of provisions in clinical and community settings.18,19 A reduction in physical activity (PA) levels are almost always observed in childhood cancer patients,20–22 with safety concerns surrounding PA persisting into survivorship. 23 These challenges have been exacerbated in recent years by the COVID-19 pandemic. In many cases, by immunocompromised children have faced stricter levels of precaution and constraints including longer periods of isolation, a greater lack of physical education resulting from school closures and the sudden termination of community-based exercise provisions. 14 Due to these particular barriers it is increasingly important to consider different mediums to deliver health-related behaviour change interventions.
Current literature has explored different tools for mHealth within paediatric rehabilitation 24 and paediatric oncology rehabilitation, 25 both with limited evidence of effectiveness. The main driver for digital health within the field of paediatric oncology exercise rehabilitation is cited as a lack of training and knowledge among healthcare professionals, such as nurses 26 to promote exercise participation. The development of a digital health that uses algorithmic prescription methodology could present a significant cost saving to health services. 27
Previous work has highlighted that children and young people spend much of their time on screen-based activities,28,29 with increases in total and leisure screen time observed during the COVID-19 pandemic. 30 However, user adherence and motivation to engage with mHealth has still been found to be low. 3 Existing suggestions for reducing the lack of adherence within oncology include elements of gamification to digital health solutions. 31 These have included, but not limited to; virtual reality 32 as well as tracking and progression charts 33 also implemented within mobile devices, such as in the form of smartphone applications (i.e. apps). Other literature has suggested the development of avatars to be an effective gamification technique to improve young people user adherence to a digital tool.34,35 Literature has also suggested that Augmented Reality (AR) can be an effective mode of delivery for mHealth interventions 36 such as when implemented within smartphone apps.
Challenges while developing technology within child health (i.e., the present AR app and its exercise prescription algorithm) need solutions. 37 It is imperative to explore functionality and usability prior to assessing contextual user experiences and performance when implemented among paediatric oncology patients. Evidence has highlighted the difficulty of comparing healthy children with clinical immunocompromised peers who face stricter levels of precaution and constraints. 38 Nevertheless, it has been shown that healthy age-compatible peers have been successfully compared, for example, in several studies of social functioning,39,40 and healthy peers are expected to provide comparable and meaningful insights into general user experience and functionality.
Therefore, the current study aims to explore healthy user’s feedback of a smartphone app using an AR avatar to develop more effective technologies aiming to prescribe a structured exercise rehabilitation programme for paediatric oncology patients undergoing treatment.
Methods
The AR app to be tested in the described pilot study is novel and has been developed specifically for this research, as no other comparable apps are currently commercially available. Therefore, a comprehensive overview of the app development process as well as the design and functionality of the app is provided below. This work was supported by Horizon 2020, the European Union's research and innovation programme, as part of grant agreement no. 945153. This reflects only the author’s view, and the European Commission is not responsible for any use that may be made of the information it contains.
For the reporting of methods to be applied within the pilot study, the 32-item Consolidated Criteria for Reporting Qualitative Research (COREQ) checklist will be used. 41
AR app development
Development process
The AR app was specifically developed for children and young people aged 9-21 years undergoing cancer treatment. However, for the previous AR app evaluation the present study will be carried out in healthy children and young people. The app’s main functionality is to support the performance of exercise sessions to promote physical and mental health via an algorithm prescribing exercise. During the development process, an interdisciplinary team of software developers, academics specialising in digital health and exercise professionals in paediatric oncology collaborated to design a child-friendly and user-centred app. Due to previous concerns with data protection and privacy within digital health tools, 42 the app was designed to operate offline.
Initially, the development process consisted of an exploration of existing mobile apps supporting exercise performance for children and young people, followed by an assessment of user needs within the target population. This included the following factors for consideration within the exercise prescription algorithm: user mental and physical health parameters, including fatigue and physical capability. Following this, an initial algorithm structure was generated and initial theoretical testing performed (i.e. confirmation of logical flow within the algorithm). As the app was planned to be used both during intensive treatment and during aftercare/maintenance treatment phases, the algorithm was then split into two separate functions. This process allowed for the specific parameters of the respective treatment phases, such as treatment schedule, clinician support and predicted physical and mental capabilities of users, to be considered. Several personas were designed to test the suitability of the algorithm for the two treatment phases 43 to ensure user input appropriately informed algorithm functionality and the respective exercise prescription was deemed safe and effective by the paediatric oncology exercise professionals.
App design and functionality
The AR app enables the user to perform strength-based exercise sessions with animated avatars in an AR setting. The virtual avatar performs the respective exercises on the screen of the device whilst the participant follows the prescribed workout. The app uses the above-mentioned algorithm to select or exclude exercises as well as generate the exercise prescription (volume and intensity) based on the input provided by a trained exercise professional and study participant. To enable the algorithm to accurately prescribe exercise, it was programmed to include initial on-boarding questionnaires to be completed by an exercise professional and the app user. This process also involves a low-level functional test performed by the user. The algorithm is programmed to adapt the exercise prescription based on the user’s input, with rest days recommended should participation not be deemed safe or appropriate (e.g. due to recent hospital treatment, new infection or pain symptoms). Figure 1 demonstrates an overview of the on-boarding process for the aftercare/maintenance treatment functionality within the app, which was used during testing as part of this current study. App onboarding process for aftercare/maintenance treatment functionality.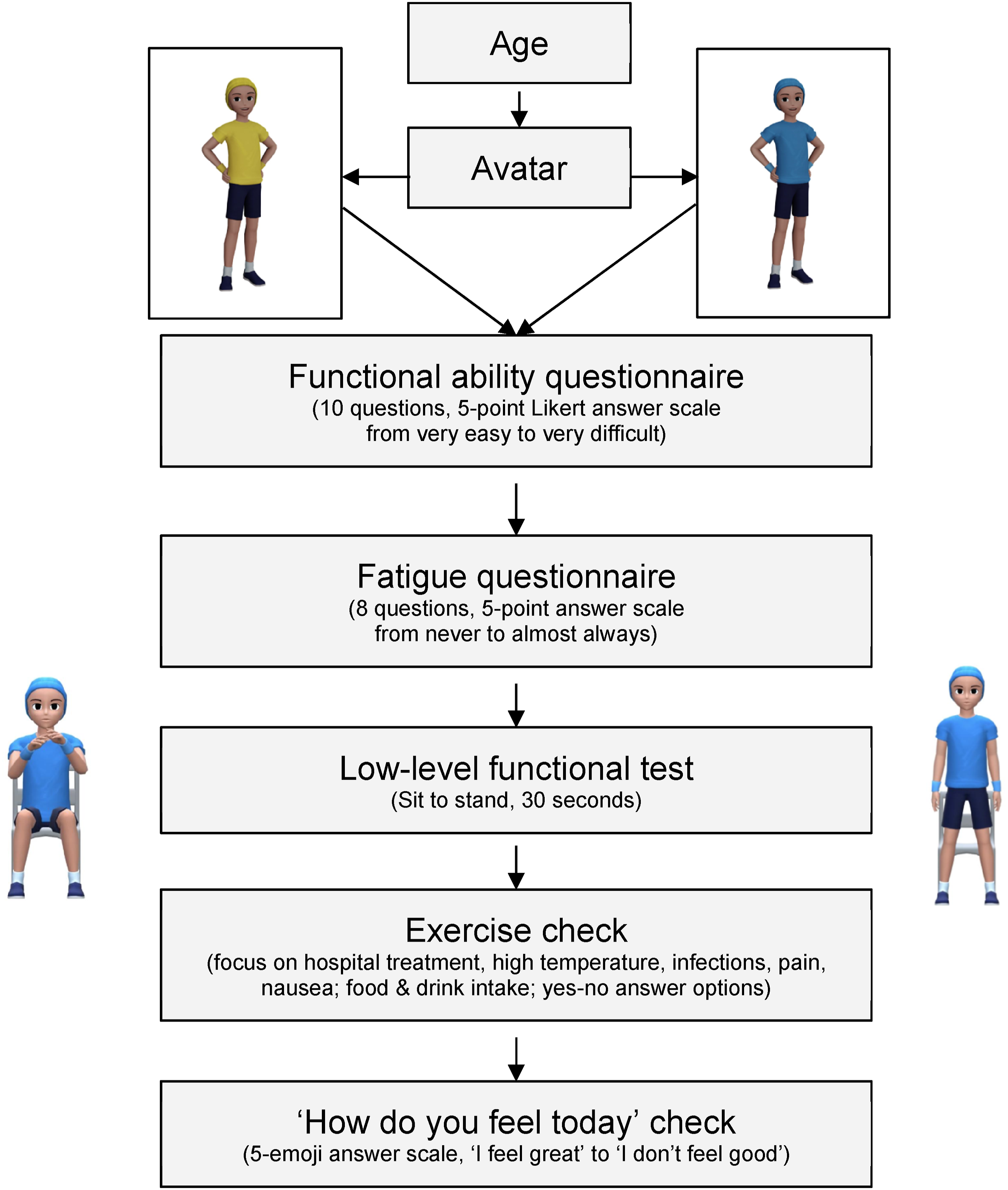
As the app was designed to be operated independently by users as well as those with potentially limited experience of engaging with digital health tools, a clean layout and clear colour, font and image options were chosen. Figure 2 contains a set of example screens from the app’s maintenance/aftercare treatment functionality. App onboarding example screens. (a) Welcome screen, (b) age selection, (c) avatar selection, (d) questionnaire example screen, (e) pre-exercise checklist, (f) fitness test.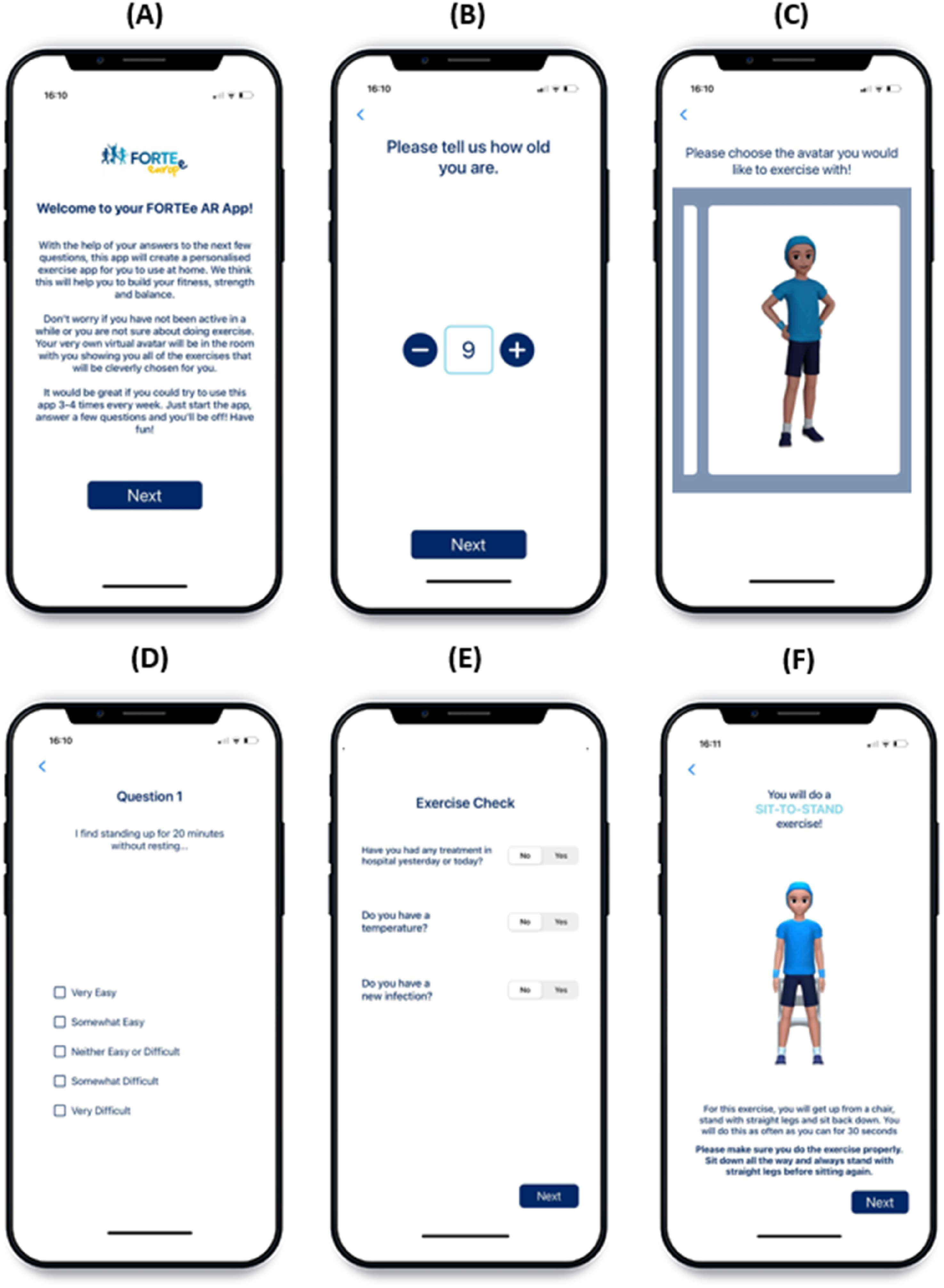
Recruitment and setting
Eligibility criteria.

The primary and secondary school have had previous contact with the research team, however had not previously been involved with any research projects. The University that will be used for the recruitment of young adults was the employing university of staff conducting this study. None of the recruited participants will have had any previous engagement with or experience of the augmented reality app. During the recruitment process for primary school age participants, the team will be in contact with the relevant school and teachers providing potential participants with study information. For secondary school age participants, visits to the school will take place in order to speak to potential participants about the study aims and process of taking part in the workshops and focus groups. During the recruitment of university students, the team will attend a sample of lectures from modules in which the identified cohort will be attending to provide study information. It will be reiterated that participation is voluntary and not a requirement of their academic course. One member of the team has been specifically selected for this role as they are not involved in any assessments, supervision or academic advising of students within the relevant cohort. Modules that are not currently led by any member of staff within the research team will be selected for student recruitment.
Potential participants will be handed information sheets, consent forms and data privacy notes to support their consideration of taking part in the study. For participants under the age of 16, participant assent and parent/guardian consent will be sought, with only direct participant consent sought for participants aged 16 and over. All participants will have at least 48 h to consider their participation and will be given contact details of one of the researchers in case they have any questions or concerns. Only participants/guardians that provided valid written consent participated in the study.
Target participant numbers per institution.

N = target number of participants.
Data collection
To obtain user feedback on the functionality and design of the app as well as suggested improvements, workshops and focus group discussions will be conducted. During the workshops, participants will use the intensive phase functionality of the app. For the purpose of this explorative study of a novel app, healthy participants will be used. Research Assistants with comprehensive experience of working with children and young people and skilled in qualitative data collection, will facilitate all workshops and focus groups. For the workshops, both research assistants will complete the non-user facing onboarding process to ensure participants can engage with the main features of the app, regardless of their lack of cancer diagnosis. This will include pre-selecting the following: all body parts can be exercised; users will not exclusively exercise sitting down; users do not have a prosthetic limb; users do not suffer from peripheral neuropathy. The phone will then be passed on to the respective participants, who will click through the user-facing on-boarding process shown in Figure 1. Following this, participants will engage with the prescribed exercise session and subsequent follow-up questionnaire which includes a Rating of Perceived Exertion scale.
Semi-structured interview guide for focus groups.
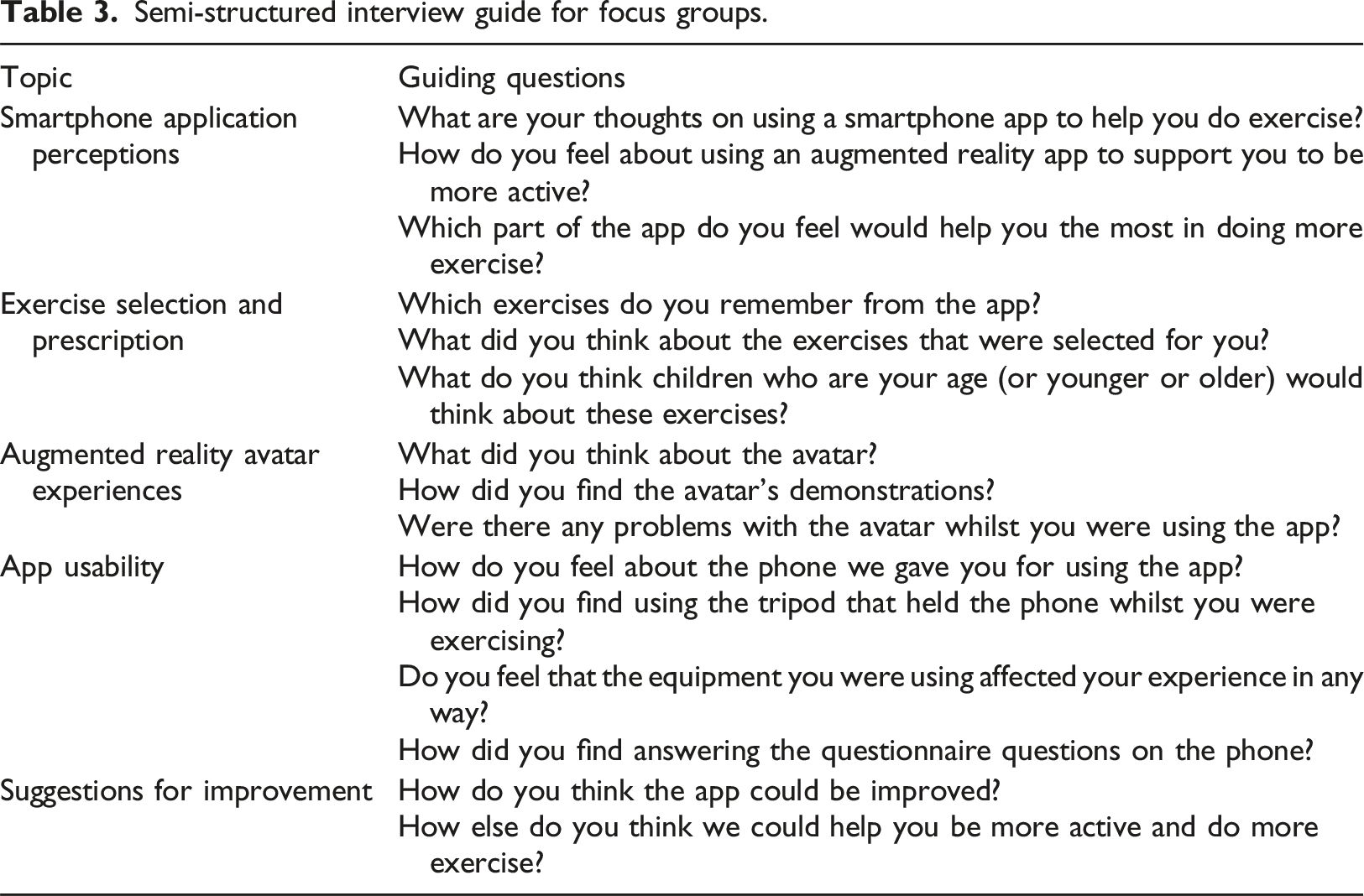
At the beginning of the individual workshops, participants will be receiving information about what the app is designed to do and who the target group is as well as informed that both research assistants had contributed to the development of the AR app. Participants will be actively encouraged to provide honest reflections on their impressions of the AR app to reduce social desirability and acquiescence bias. Participants will be reassured by the researchers that all feedback is valuable and will be taken on board during the subsequent development of app updates. During the recruitment and data collection process, a rapport will be established with potential participants to facilitate their involvement. As the aims of this research rely upon honest and open feedback on the AR app, whilst limiting pressure to respond favourably, the study team will focus on an informal and collegiate report between both research assistants and all potential participants.
Data analysis
As methodological orientation, a descriptive approach will be applied, with a focus on data collection that fosters an open environment allowing participants to honestly and authentically share their perceptions of using an app for PA, exercise selection and prescription, experiences of using an AR avatar, usability of the app, and suggestions for improvement. Firstly, audio recordings of all focus groups will be transcribed using edited transcription. Due to the participants’ age, it is expected that some may re-start sentences multiple times as well as frequently use filler words and non-verbal communication. Non-verbal communication, gestures and facial expressions are not expected to significantly contribute to or influence the perception of the participants’ responses in this study. Thus, edited transcription is expected to be most suitable to maintain full meaning, whilst simultaneously editing the transcripts to a degree that facilitates subsequent analysis. To ensure familiarisation with the content, all focus groups will be transcribed manually, without the use of an external transcription service or software. Transcripts will be read and re-read multiple times during the analysis process.
Edited transcriptions will be printed into paper format, with the researchers collaboratively conducting the coding process. In the present study, data will be explored and coded using qualitative content analysis with a combination of techniques adapted from two approaches46–48: (1) the deductive category application, which uses a prior formulated coding framework based on theory or empirical literature for analysis; and (2) the inductive category formation, which focuses on developing codes coming from the data material itself. Initially, data will be coded deductively deriving from the semi-structured interview guide: hardware user experience, app interface, app design, AR functionality, avatar design, exercise prescription, questionnaire completion, perceived benefits. In a second step, all remaining quotes will be reviewed and coded inductively. Inter-coder conversations will be held to discuss potential development of new categories from inductively coded comments or how these may fit within existing categories through establishing their explicit meaning.
For ensuring trustworthiness of the data collection and analysis processes, the study team will consider credibility, transferability, dependability and confirmability. 49 To ensure credibility, the present study will employ well-established methods for data collection and analysis, with specific protocols in place such as the use of consistent semi-structured interview guides, as well as use random sampling of participants from the respective institutions to ensure complete voluntary and un-coerced participation. Moreover, continued feedback and collaborative discussions will occur within the research team. Regarding transferability and dependability of data, the focus will be on providing thick descriptions and a comprehensive account of participant characteristics important for contextualising any resulting findings. This will allow other researchers to interpret data within its given context and transfer it to their own if applicable. To enhance confirmability of data, detailed descriptions of the applied methodology will be provided, its limitations, and their potential impact acknowledged. 49
Discussion
This study will be among the first globally to explore user experiences, preferences and suggested improvements of a mobile AR app that uses algorithmic technology to prescribe exercise for childhood cancer patients and survivors. As healthcare systems are increasingly burdened, the use of mHealth to support digital health within clinical practice presents a unique opportunity for increasing access and cost savings, 50 while overcoming clinician time restraints and geographic barriers. 51 It is worth highlighting that the digitalisation of exercise interventions for long-term health conditions populations is a subject still little explored, however, digital interventions may provide an opportunity to supplement in-person interventions and act as a valuable adjunct to clinical care. 52 Digital interventions are scalable interventions that may increase accessibility for more individuals when compared to face-to-face interventions that are often limited by a reduced instructor to participant ratio. 53 The use of mHealth interventions may be of particular importance for a UK model of care as there is currently an overwhelming lack of PA provisions both within clinical and community settings for families affected by cancer.1819 The development and implementation of such technologies is a promising prospect, however, there are significant complexities and challenges with regard to recruitment and engagement of patient groups and effective implementation. 54 A user-centred approach is recommended to ensure that preferences and needs are incorporated into mHealth tools. 16
The availability of commercial mHealth apps has increased over recent years, with a peak during the COVID-19 pandemic in early 2021. Currently, there are 52,406 mHealth apps available in the Apple App Store 55 with 54,603 available in the Google Play Store. 56 Research has also increasingly focused on evaluating such apps, with 1712 papers published since 2000 focusing on mHealth and eHealth interventions related to PA, sedentary behaviour and diet. 57 Specifically, over 500 have been published exploring the use of gamification/games or mobile apps/smartphones. 57 Despite such apps being widely available and widely researched, gaps in the literature remain. It has been established that when evaluating mHealth tools such as apps, appropriate methodologies need to be chosen in line with the respective evaluation aims. 58 Existing research evaluating app interventions has mainly focused on assessing usage logs or employing questionnaires. 59 Few studies used interviews or focus groups, which are vital for obtaining in-depth qualitative data on user experiences and suggested improvements, especially during the early stages of development. 59 Therefore, the current study will provide vital evidence to progress current trends in app development for clinical populations, such as cancer patients.
As the proposed study involves the evaluation of a novel app, it is crucial to perform usability testing in controlled laboratory conditions before assessing its commercial use and efficacy on a wider scale. 58 As part of this methodological approach, supervised use of the app (e.g., in the form of workshops) followed by gathering data exploring user experiences is deemed appropriate. It has been recognised that a variety of users from different population groups should be involved in the evaluation of user-centred mHealth tools. 60 It has also been recommended that applying methodologies focused on more controlled environments (i.e. testing an app in a laboratory setting) is a useful process, prior to allowing users to test the app in daily life. 60
As this study involves children and young people, using qualitative methods to explore their views and preferences on the app interface and its functions will be crucial to obtain in-depth data as well as leave room for unintended or unexpected results to be discussed. Considering the young age of primary school children and their potentially limited experience of using a smartphone app, the current study methodology allows for some variation and flexibility that prioritises the collection of meaningful data over standardisation across the different age groups. 60 Data gathered as part of this study will be used to directly inform future development of the app and is also expected to contribute to advancing the empirical evidence base used for the design of future health-related apps, within both adult and paediatric care.
Limitations
In this formative study, age-matched, healthy participants will be used. This is, in part, due to the complexity of recruiting immunocompromised individuals in a group setting. While feedback on the app’s functionality is expected to be comparable between cancer patients and healthy age matched peers, there may be differences in the usability of the app between these two populations. It is acknowledged that paediatric cancer patients will have a unique intention and motivation in regard to being physically active, whilst also having specific physical and cognitive performance needs and preferences. Whilst the app’s onboarding process (see Figures 1 and 2) is specific to cancer patients, and its components thus more familiar to this population, the main aims of this study were to explore the functionality and usability of the app with a focus on the user experience of navigation, layout, instructions, and AR exercise demonstrations. It is expected that informing participants in this study of the discrepancy between their own experiences and the relevance of certain features for paediatric oncology patients will allow for meaningful insights into user experiences of children and young people. It has been suggested that it is particularly important to ensure current developments in digital health are relevant to the target user groups, such as cancer patients. Users who see the relevance of such tools within their treatment pathways are expected to be more likely to engage with these technologies, for example for self-management of their condition. Moreover, mHealth tools need to be applicable and relevant also within the daily life of its users, 59 further underlining the importance of future user testing with paediatric oncology patients in a real-world setting. 59
Further, the app is designed for users to engage multiple times per week over the course of weeks or months, depending on their cancer treatment. Whilst the current study will not investigate the use of the app over a number of weeks and instead during a one-off workshop, it will provide foundational data to inform future work focusing on longer term use. Such studies will also allow exploring longer term adherence and barriers for regular participation, currently not included in the present study. To better understand the long-term effectiveness of AR apps, changes in behaviour (including exercise participation) and health outcomes (including cancer-related fatigue) should be more closely investigated.
Furthermore, it is also important to consider that user-centred design should extend to other interest groups, such as families, healthcare professionals and industry partners. 61 It is crucial to include those involved in the multidisciplinary approach within oncology care for optimal integration of technologies within clinical settings. It is for this reason that the involvement of wider interest groups should be strongly considered in the early stages of future research and technology development.
Another limitation is that the onboarding questions are not validated questionnaires, but are designed specifically for the app; their purpose is not to measure a specific outcome in a validated way, but rather to elicit answers to a limited number of basic 'check-up' questions. This enables the algorithm embedded within the application to automatically prescribe tailored exercises.
Conclusions
The findings of this study will provide invaluable information about user experiences, preferences and suggestions of healthy children and young people to improve an AR app prescribing exercise. User feedback gathered as part of the current research is important for informing the development of future mHealth tools such as apps and provides vital insights that can enhance the usability of and adherence to such tools. It is crucial to conduct user-centred, qualitative studies and use their findings to strengthen the current empirical evidence base on digital technology used within healthcare settings. The current study findings will influence future development of the AR app before this can be scaled up and employed during a longer-term randomised controlled trial involving childhood and young cancer patients.
Supplemental Material
Supplemental Material - The development of an augmented reality application for exercise prescription within paediatric oncology: App design and protocol of a pilot study
Supplemental Material for The development of an augmented reality application for exercise prescription within paediatric oncology: App design and protocol of a pilot study by Kim Straun, Hayley Marriott, Alba Solera-Sanchez, Stan Windsor, Marie Neu, Elias Dreismickenbecker, Jorg Faber, Peter Wright, on behalf of the FORTEe Consortium in Health Informatics Journal
Footnotes
Acknowledgements
We are grateful for the organisational support from Oxford Brookes University. Written informed consent and assent were required to participate in the project in accordance with the project approvals granted by the University Research Ethics Committee (registration number 211547).
Author contributions
All authors have made a substantial contribution to the article, read and approved the final version of the manuscript, and agree with the author's order of presentation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project has received funding from the European Union's Horizon 2020 research and innovation programme under grant agreement No 945153. This publication reflects only the author's views and the European Union is not liable for any use that may be made of the information contained therein.
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
