Abstract
Objective
Many cancer survivors do not meet recommended levels of exercise, despite the benefits physical activity offers. This study aimed to understand experiences of insufficiently active overweight/obese breast or colorectal cancer survivors, in efforts to (1) examine regular physical activity barriers, and (2) determine perceptions and acceptability of a remotely delivered physical activity intervention utilizing wearable sensors and personalized feedback messages.
Methods
In-person and virtual small group interviews were conducted engaging overweight/obese cancer survivors (n = 16, 94% female, 94% breast cancer survivors) in discussions resulting in 314 pages of transcribed data analyzed by multiple coders.
Results
All participants expressed needing to increase physical activity, identifying lack of motivation centering on survivorship experiences and symptom management as the most salient barrier. They indicated familiarity with activity trackers (i.e., Fitbit) and expressed interest in biosensors (i.e., continuous glucose monitors [CGMs]) as CGMs show biological metrics in real-time. Participants reported (1) personalized feedback messages can improve motivation and accountability; (2) CGM acceptability is high given survivors’ medical history; and (3) glucose data is a relevant health indicator and they appreciated integrated messages (between Fitbit and CGM) in demonstrating how behaviors immediately affect one's body.
Conclusions
This study supports the use of wearable biosensors and m-health interventions to promote physical activity in cancer survivors. Glucose-based biofeedback provides relevant and motivating information for cancer survivors regarding their daily activity levels by demonstrating the immediate effects of physical activity. Integrating biofeedback into physical activity interventions could be an effective behavioral change strategy to promote a healthy lifestyle in cancer survivors.
Introduction
Cancer diagnosis rates continue to increase, with almost 17 million cancer survivors in the U.S.1,2 The most common cancers are female breast cancer and colorectal cancer (combined 32%). 2 Cancer survivors have increased risks of mortality and morbidity even after receiving treatment, along with reduced quality of life. 3 As the number of cancer survivors increases, there is a need to foster a sense of normalcy and a healthy lifestyle for patients to improve their health outcomes. 4 At the forefront of this healthy lifestyle is engaging in physical activity. 5 Increased levels of physical activity among cancer survivors are associated with improved long-term health outcomes such as survival and prevention of cancer recurrence, as well as positive short-term outcomes such as improved mood, anxiety and depressive symptoms, physical functioning, health-related quality of life, and decreased fatigue.6–9 The strongest evidence of an association between physical activity and reduced cancer-specific mortality is for breast cancer (28%-38% risk reduction) and colorectal cancer (39% risk reduction).6,10 Further, increased levels of sedentary behavior (defined as low amounts of energy expenditure), separate from lack of physical activity, elevates risks for both cancer incidence and mortality.10,11
In late 2019, the American College of Sports Medicine updated their exercise recommendations for cancer patients and survivors. The new guidelines recognized that cancer survivors can benefit from engaging in at least 90 min of aerobic activity per week and should minimize time spent in sedentary activity. 8 Similar to the general population, many cancer survivors do not exercise regularly to achieve the many health benefits from being active,, 12 especially survivors who are overweight or obese. 13 Cancer survivors may face additional barriers to physical activity that healthy adults do not have to contend with, such as physical symptoms like fatigue, pain, and psychological stress and anxiety.14,15 Therefore, there is a need to identify approaches that may help overweight and obese cancer survivors to adopt and maintain an active lifestyle through physical activity interventions.
With the increased adoption of wearable activity trackers, one of the promising behavior change techniques for physical activity interventions is to provide personalized and timely feedback based on the wearable sensor data. 16 Providing performance feedback is easier and more efficient for individuals using activity trackers (i.e., comparing physical activity performance with a set goal). 17 However, providing performance-based feedback alone might not be effective enough to promote regular physical activity. In previous studies utilizing wearable activity trackers (i.e., Fitbit), usage (even when combined with daily prompts to increase physical activity) does not increase physical activity levels, particularly long-term or enhance health-related outcomes such as weight loss.18,19 More recently, there is an increased interest in providing biological feedback as a behavior change technique.20,21
Biological feedback or biofeedback is defined as providing feedback about the body (e.g., physiological or biochemical state) using monitoring device as part of a behavior change strategy. 17 Unlike wearable activity trackers, biosensors are wearable devices that either continuously or frequently measure individuals’ physiological parameters in free-living environments.22,23 It is possible that data from biosensors can be used to demonstrate the acute benefits (or consequences) of behaviors on dynamic indicators of health in real-time to motivate health behavior change. For example, continuous glucose monitors (CGMs) can provide real-time indications of biological responses (i.e., acute changes in glucose level) to physical activity, which activity trackers such as Fitbit cannot do. 21 CGMs have traditionally been used to support diabetes care management, with recent technology advances allowing for usage in other contexts. 24
Social cognitive theory (SCT) 25,26 and self-determination theory (SDT) 27 provide the theoretical framework for this study. Both have been used in studies testing physical activity interventions. Studies using SCT show how human behavior (e.g., physical activity) is associated with one's environment and personal characteristics, like physical characteristics, as well as self-efficacy. 26 Recent research calls for using SCT qualitatively to improve physical activity among cancer patients. 28 As wearable devices may be new or (due to the self-insertion process) frightening, understanding these personal characteristics and self-efficacy are particularly important for furthering research in this area. Studies using SDT demonstrate how an individual is motivated to perform specific behaviors based on their basic competence, relatedness, and autonomy. The motivation one has to perform a behavior is on a continuum, ranging from extrinsic (e.g., a power figure is involved, or perhaps rewards) to intrinsic (there is an inherent pleasure or interest of the individual to perform the behavior). Understanding the motivation (and the range of extrinsic to intrinsic) one has to begin using wearable sensors, and eventually maintaining usage, along with how social support (through information sharing, perhaps is another step forward in developing theory-based interventions.
Although previous research has demonstrated excellent feasibility and acceptability in using wearable activity trackers such as Fitbit in cancer survivors,29,30 little is known regarding cancer survivors’ perceptions about using biosensors, the relevance of data obtained from them, and the extent to which biofeedback can motivate health behavior change. Therefore, the aims of this study were to understand the experiences of insufficiently active overweight and obese cancer survivors regarding: (1) barriers and challenges related to engaging in regular physical activity, (2) their perceptions of using wearable trackers and biosensors to provide physical activity-related feedback, and (3) their opinions of sample biofeedback messages that utilizing data from the wearable sensors to motivate physical activity.
Methods
Study design
We used an exploratory, qualitative focus group research approach to investigate cancer survivors’ experiences with and perceptions of exercise and wearable sensors. The study protocol (PA19-0317) was approved by The University of Texas MD Anderson Cancer Center. Signed informed consent was obtained from all participants.
Recruitment and eligibility
Adult male and female patients with breast or colorectal cancer (stage 0-III) were identified from MD Anderson's tumor registry. Because the study was designed to be in the format of in-person group meetings, we selected patients who lived in Harris County where MD Anderson's main campus is located. Patients were contacted by email with a brief study description. Those who expressed an interest in participating were invited to fill out an eligibility screener either through an online survey or over the phone.
The goal of this focus group study was to inform the future development of a physical activity intervention that utilizes personalized biofeedback, therefore, we aimed to recruit cancer survivors who would benefit the most from of such intervention (i.e., insufficiently active and being overweight or obese)Therefore, the study inclusion criteria were: (1) being over 18 years of age, (2) having completed adjuvant therapy (i.e., chemo and/or radiation therapy), (3) having a body mass index ≥ 25 kg/m2 based on self-reported height and weight), (4) being insufficiently active (i.e., engage in less than 150 min of moderate-intensity physical activity per week in the past month based on self-report), (5) having the ability to walk one block without pain or discomfort, and (6) being fluent in English. Patients were excluded if they have self-reported health issues that limit physical activity or if they are on dialysis.
A total of 16 survivors (aged 50–74, M = 57.87 years) participated in this study across five groups. Table 1 details their characteristics. Three small group interviews were conducted in-person prior to the advent of the COVID-19 pandemic in March 2020 and two occurred virtually after March 2020. The group sizes ranged from two people to five people.
Participant characteristics (n = 16).

Note: All data are self-reported.
Procedure
Eligible patients were scheduled for an in-person group meeting at MD Anderson. All participants signed an informed consent prior to the start of the group discussion. One moderator guided the group discussions using a guide based on the social cognitive theory 31 and self-determination theory. 32 The questions were designed to have participants think on elements of motivation, self-efficacy, environment, behaviors, and other SCT and SDT constructs. These two theories are the most widely used behavioral change theories for physical activity research. Specifically, SCT was used to guide the questions related to barriers and facilitators, while SDT focuses on motivation. The discussion started with a needs assessment (i.e., discussions about patients’ current physical activity levels and whether they feel they need to become more active, as well as general biosensor familiarity), perceived benefits and expectations from being more physically active, barriers to regular physical activity, and identifying resources that could help them maintain regular physical activity. Then, the moderator presented an educational handout that lists the various health benefits of physical activity. The moderator highlighted one of the immediate impacts of physical activity using a simulated website that demonstrates the different daily glucose patterns in response to exercise.21,33 Next, the moderator presented the group with two types of wearable sensors, activity trackers (i.e., a Fitbit wristband) and continuous glucose monitors (i.e., a Dexcom model inserted around the belly area and an Abbott Freestyle Libre model inserted on the back of upper arm). Along with the sleep and step measurements, we also considered and discussed heart rate measurements from Fitbit during the groups. The sensors and their features (including each of the sensor's companion smartphone app and sample visual outputs) were described by the moderator who then asked the group to discuss their general experiences, perceptions, preferences, and opinions on using the sensors. Lastly, the moderator presented sample text messages that aimed to motivate physical activity using data from these two wearable sensors and asked for participants’ impression and feedback regarding the tone, content, and delivery frequency of the text messages. These sample messages included a mixture of wearable sensor data summaries, physical activity reminders and tips, and links between physical activity (Fitbit data) and glucose pattern (CGM data). The first message presented data with specifics on glucose levels obtained from the CGM, followed by a recommendation of what to do to help the glucose levels. The second message linked physical activity (e.g., steps walked) obtained from a Fitbit, followed with tips to continue engaging in physical activity. The third message summarized active times as well as provided average glucose levels (i.e., data only). The fourth message specifically linked the CGM and Fitbit data together with a behavior. Each session lasted approximately 60 to 90 min. All sessions were audio-recorded and transcribed verbatim by a professional transcription company.
The small group interviews began November 2019. In March 2020, the in-person groups were halted due to the COVID-19 pandemic. Shortly after, the study protocols were amended to enable virtual group meetings via an institutional supported Zoom videoconferencing platform. All study procedures remained the same for the virtual meetings. Participants were compensated for their time ($15 gift card). We stopped conducting further group interviews when we found data adequacy was reached.
Analysis
Braun and Clarke's six-step process was used to manage data collection, processing, and analysis. 34 First, the transcripts were read in their entirety during fall 2020 and spring 2021 by the principal investigator, a co-investigator, and a graduate research assistant to understand the depth of the data, alongside the original interview guides. Initial coding categories were then identified, followed by subcategories. Differences regarding coding or interpretation were resolved by discussion. Each of the categories and subcategories were grouped into themes, which were then reviewed with pertinent examples for each identified. The transcripts were read again by the coders on Microsoft Word to ensure that the data was fully analyzed. Once responses became similar, with no new or emergent themes, data adequacy marked the end of data collection.35,36
The following section describes the identified themes, grounded by participants’ answers. Using quotes from the participants, the findings of the present study are further clarified.
Results
Sample characteristics
Focus group discussions
Results of our qualitative analysis have been organized into the three key topic areas from the small group interviews based on the interview guides: physical activity (expectations of physical activity and barriers to physical activity), using wearable sensors to motivate physical activity (familiarity and experience with wearable sensors, Fitbit and CGMs specifically), and feedback messaging preferences (delivery and content, sample message feedback). In general, participants wanted more active lifestyles and stated specific goals of obtaining the goal lifestyles. They faced several challenges in meeting these goals, however, such as perceiving exercise as repetitive and boring. Messaging (intervention-related) were perceived as motivating, and graphs were mentioned as useful. Participants also indicated they would like positive summaries and plan of action (“it tells you that something was noticed, why it was noticed, and then what to do about it”). Regarding wearable sensors, most were curious, and as long as they did not perceive pain, were open to wearing them. In each table, the key topic areas and the unique themes that correspond with that overarching topic are included.
Topic area 1: physical activity
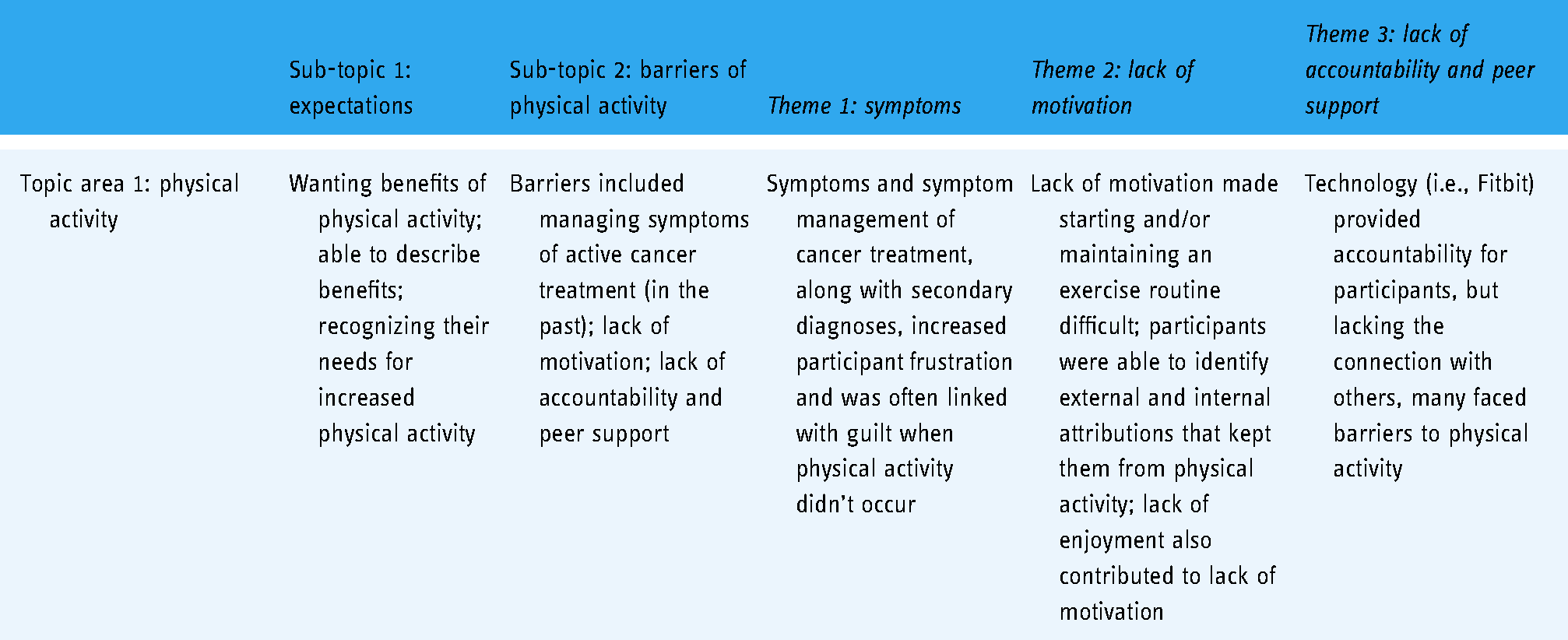
All participants expressed that they would want to be more active than their current activity levels. They discussed their expectations of physical activity and identified major barriers to being more active. Themes related to expectations and barriers are described below. See Table 2 for a summary.
Summary of results for topic area 1: physical activity.
Sub-topic 1: expectations
Participants identified several expectations in response to the questions, “Why do you want to be more active?” and “What do you want to get out of being more active?” Several participants mentioned the typical physical activity benefits, such as P2's comment of, “More energy; you sleep better; your attitude is better; you feel healthier.” Almost every participant also indicated that they wanted to do more physical activity, often describing specific benefits such as a more positive attitude, increased energy levels, improved bone density, improved sleep patterns, increased endurance, reduce medication dosages, “wanting to get my sexy back,” lessen stiffness, and improve mobility. The survivors’ comments were quite clear, “I need to be more active,” recognizing that they were not meeting specific levels of physical activity that either their health care team had set as their goal or their own personal goals. Following their explanations related to their goals and expectations of physical activity, participants identified barriers inhibiting their physical activities.
Sub-topic 2: barriers of physical activity
When asked, “What are the challenges or barriers that you face for exercising?” during the discussions, participants identified barriers to exercise within the context of either pre-pandemic or during the pandemic. These barriers included: managing symptoms of active cancer treatment (in the past), lack of motivation, lack of accountability and peer support.
Theme 1: symptoms
A complicating factor related to regular exercise was that of symptoms and symptom management based on cancer treatment. Some participants, like P9, described how her cancer treatment (e.g., surgery) complicated her regular physical activity routine. P10 felt similarly, “[after cancer treatment] you get kind of afraid, you get very, very cautious of what you think you want to do now.” She also explained how her routine pre-diagnosis included weightlifting, but “all that had to go away” and because of her health issues, her physical activity was limited. Some participants, like P16 and P15, talked about how their cancer treatments led to other health diagnoses, such as type 2 diabetes mellitus, or high blood pressure, and these diagnoses inhibited physical activity. These simultaneous medical complications (either pre-existing or stemming from cancer treatment) are understandably frustrating, particularly as steps towards recovery may be happening related to one diagnosis, steps backwards regarding another diagnosis may be occurring simultaneously creating feelings of being stuck or increasing feelings of depression, as P15 implied. P2 also described how her lack of energy affected her willingness to engage in physical activity, yet also could not push through because of her lack of endurance. Each of these participants, along with others, indicated some frustration or guilt that they could not maintain their former physical activity routines, even while knowing that they had undergone a life-saving medical treatment.
Theme 2: lack of motivation
Participants also described lack of motivation as a barrier to exercise that manifested itself in several ways. First, most participants described a lack of motivation as one of the difficulties of starting a physical activity routine. “Finding time” (P2) and “just getting up” (P3) were other areas where the survivors faced barriers to physical activity. P15 said that while there was access to equipment and resources, there just wasn't the extra “push to get it done.” Each of these participants, among others, could very easily and quickly identify why they weren't engaging in physical activity, with others mentioning the weather, or other demotivators, such as pain as other issues they faced when attempting to self-motivate. Foot pain while on the treadmill was one such issue P1 described.
Second, participants described that continuing an established workout routine was difficult to maintain. Specifically, survivors indicated that it was often so easy to take a day off and then not return to their activities. P1 simply stated, “It's hard to find that constant motivation,” which was echoed across groups. Some participants felt that if they did not engage in physical activity every day, that they were not adhering to their goals. These goals, when not met, created consistency issues for the participants. Others described experiences where they were attending an exercise class but started feeling unwell and then became disappointed that they couldn't engage at the level they wanted to, resulting in frustration and in some cases cessation of the workout routine. For some survivors these experiences were particularly disheartening, creating a comparison of pre-diagnosis life and post-diagnosis reality.
Third, a lack of enjoyment related to physical activities also was reported by several participants. P2 stated, “I feel that I do it [physical activity] just for quality of life, but I don't like it.” These types of statements indicate that the participants know how vital physical activity is, yet still felt tensions related to engaging in physical activity. Several other participants also described how they didn't find enjoyment in physical activities at bigger facilities (e.g., gyms), or even when recalling their sports experiences in school. P16 described, You just can't do something just because it's good for you: you’ve got to be able to find something you enjoy on top of--just because someone says it's good for you to go walk, if it miserable, I just can't do it. It's 100 degrees outside, I just can't do it.
These responses are particularly interesting as participants don't simply recognize that they are facing barriers, but that these barriers are in direct contrast with the knowledge they have about the benefits of physical exercise.
Theme 3: lack of accountability and peer support
Lack of accountability from peer support made maintaining physical activity routines difficult for participants. In fact, P9 described needing accountability to have any motivation for engaging in physical activity, “I didn't have anybody holding me accountable, and I think that was the first thing that came to my mind, “Oh, it’d be good if somebody was holding me accountable."“ Others reported using technology (e.g., Fitbit activity trackers) to facilitate friendly competitions. P3 stated that, along with her friends, “We’d have challenges to each other: who can get the most number of steps, that kind of stuff. And that seemed to be quite motivational.” Yet others, like P11, described a lack of accountability as a barrier to maintaining regular activity, “I think I’m missing an accountability factor,” and further described Fitbits as one option that wouldn't work for increasing accountability because of other medical issues. For some survivors, fitness group classes helped with maintaining the routine (pre-COVID-19, at least). For others, they relied upon family or friends.
Topic area 2: wearable sensors
Participants reported several varied perspectives based on using wearable sensors for physical activity purposes. See Table 3 for a summary.
Summary of results for topic area 2: wearable sensors.

Sub-topic 3: familiarity and experience with wearable sensors
Participants discussed their previous experience and familiarity with wearable activity trackers, like the Fitbit or Apple Watch, and CGMs (e.g., Freestyle Libre and Dexcom G5/6 as presented by the moderator during the discussion). Themes specific to the use of activity trackers and opinions towards biosensors such as CGM emerged.
Theme 4: activity tracker experiences
Much of the discussion around activity trackers specifically referenced Fitbit. The discussion highlighted that the use of wearable activity trackers for physical activity is highly individual in this population. P13 discussed how using Fitbit increases personal accountability, something many participants identified as a barrier to exercise previously, “I do use Fitbit. It's kind of a way to, you know, I record all of my exercise time, and I like to see that I haven't broken my commitment.” P13 also described how using the Fitbit helped her stay active and committed to her personal exercise goal after her cancer treatment. P10 mentioned that using her Fitbit helped keep her motivated, by providing accountability through someone else tracking her reported activities, “I don't know if I just don't want to disappoint people or what, but I love having the accountability of somebody saying, “You know you didn't hit your goals this week.” Yet, she also explained that she preferred her family not have access to that information, saying that her husband and son were both triathletes and she felt more comfortable talking “with other people in this situation.” Other participants, like P4, described her Fitbit, saying, “Mine's a glorified timekeeper. A glorified watch” and indicated that she did not use her Fitbit as intended.
Theme 5: continuous glucose monitor (CGM) experiences
Most survivors knew about CGM through advertisements or commercials and several knowing family members who currently wore one. Having this informational feedback, particularly when shared with close relational others, may improve motivation and accountability as it provides talking points with one's friends and family members, particularly if there is the social element of an activity challenge present. Participants also indicated their knowledge and their comfort levels with CGMs. The depth and breadth of this subtheme was focused on: (1) perceptions related to technology acceptability, contrasted with technology invasiveness, and (2) the relevance of glucose data. First, the general CGM perceptions will be presented.
Participants with diabetes indicated that they would be willing to at least try the CGM to compare with their glucometer experiences. P1 compared the CGM with a Fitbit, Yeah. To me, that [the CGM] has--because the Fitbit is good. It's a nanny device and I see how corrupted it can be when you’re watching TV and shaking your arm so it doesn't get mad at you, so I know it's easily defeated. But this, obviously, would be a system where you can't cheat it.
P16, who has diabetes, thought her Fitbit was useful, and indicated that a CGM would provide added benefits, But I do like the activity tracker on my watch, I pay attention to it because it’ll say something like, “You need to stand up” after an hour… But I think being able to maybe see the benefit of exercise while you’re doing it, like with a continuous blood sugar tracker, that might work for me.
Another participant, P2, without diabetes, described that she had mixed feelings about using a CGM based upon her Fitbit experiences, Just like with a Fitbit, when you get a Fitbit the first time--like I’ve been in other research, right--and I get the Fitbit because I’m competitive. So now I know that I’m in this group, I have this Fitbit, and we link up with everyone and stuff. And so, I want to walk as much as you do, because I know that that stuff is recorded. So in the initial phase, yes, stick me. Just go on and put it in and stuff. But then after that and you get the information and you see how bad or good I exercise or eat, then it's like, okay, so now what?
These comparisons, drawing from previous experiences with other wearable devices and smartphone apps, were common across groups.
Sub-topic 4: technology invasiveness and acceptability
Some concerns related to the invasiveness of the subcutaneous wearable sensor as well as placement were mentioned. For example, P2 mentioned that she was somewhat concerned that she might not want to take the sensor off after the 10–14 day wear period and then reinsert a new sensor; both P2 and P6 also initially thought that the device was solely surgically implanted rather than easily inserted without needing anesthesia. Like P13, P12r felt that there was no issue with the subcutaneous insertion of a CGM device. Other participants recalled a subcutaneous device, the Neulasta, that provided medication for their cancer treatment and was considerably larger than CGMs. Given these experiences as comparisons, participants indicated they were in support of a CGM even with the insertion. After getting clarification from the moderator the mechanisms of how a CGM works, most participants expressed that they would be willing to wear the CGM, at least for a 14-day period. When presented with options for sensor placement area preference, many participants also indicated that they were open to either the belly area or on the back of the arm, with almost all participants being willing to place the CGM on their belly. Reasons included the CGM being more subtle, concerns with knocking the sensor off of one's arm if you bumped into a door, or skin irritation. However, even with their concerns, many participants felt that the information obtained from the CGM could help them improve their health behaviors before obtaining another, or a more severe, health diagnosis.
Theme 6: relevance of glucose information for those with diabetes
For participants who had diagnoses of diabetes or pre-diabetes, they were somewhat able to connect their glucose levels with diet. However, while these participants did make the connection between diet and glucose levels, they were less likely to recognize the link between exercise and glucose levels. P15 discussed that the data garnered from a CGM could help improve her setting small physical activity goals, such as taking a walk after eating a meal. She added “I think all of that [the data from the CGM] would help me to get myself back on track [of engaging in physical activity]. Yeah, so it would be good to know it.” P14 felt that, with her recent prediabetes diagnosis, the CGM would be motivating for both managing diabetes directly (e.g., via blood glucose levels) as well as indirectly through physical activity. P8 thought that receiving glucose alerts would be helpful as they would provide the possibility of taking action “at the moment” of the alert (if a glucose threshold was reached).
Theme 7: relevance of glucose information for those without diabetes
Participants without a diabetes or pre-diabetes diagnosis were not able to make the connection between their daily behaviors and glucose without guidance. Because CGMs aren't easily accessed by people without diabetes in the U.S. (often used primarily by people with type 1 diabetes mellitus), the participants spent quite a bit of time discussing reasons why they might be interested in knowing their glucose data.
P11 described the example that her father used a CGM, but felt that he relied upon it “as a crutch” and that is was used “in a negative way rather than the way that it's intended” by using the glucose readings to further dose more insulin rather than engage in physical activity. Therefore, P11felt reticent to use a CGM based on that experience. Importantly, after hearing the explanation by the moderator, participants without a diabetes or pre-diabetes diagnosis were particularly intrigued by the idea of using glucose-related information as a medium-term solution to understand more about biological effects of physical activity. P12 asked clarifying questions during the group interview, as she didn't quite understand the connections between the wearable sensor and physical activity. P12 further asked how this particular information may be a potential motivator, specifying that she didn't have diabetes and felt if she did, it would likely be more useful of a device. Others asked similar clarifying questions, such as if the CGM can be worn in the shower. These types of questions indicated that participants were engaging with the idea of themselves wearing the CGMs and attempting to find potential barriers. Of course, some participants, having limited knowledge about CGMs and glucose readings, discussed that in order to adopt these technologies, they would need to know the thresholds of a “good” or “normal” glucose reading, “or we’re going to do like [participant name] and Google.”
After allowing the participants space to examine their thoughts related to the CGM-motivation for engaging in physical activity relationship, the moderator provided some background information on the relationship between CGMs and physical activity. After hearing the explanation, survivors felt that having the real-time (approximately every five minutes) data from CGMs might help them achieve their physical activity goals by providing them additional actionable information. For example, P4, P8, and P9 felt the data from a CGM would help improve their physical activity levels by providing glucose levels. Specifically, some participants felt they were high-risk for developing diabetes, so they thought having CGM data reporting glucose level ranges would be helpful for their physical activity engagement.
Topic area 3: feedback messaging preferences
Participants discussed their preferences related to remote interventions and related communication (such as frequency, tone, and content of the messages) based on data from the wearable sensors. See Table 4 for a summary.
Summary of results for topic area 3: feedback messaging preferences.

Theme 8: general delivery methods, timing, and frequency
Participants agreed that texts were preferred over other methods of communication (such as emails or app notifications). Specifically, participants reported the need for accountability or peer support, agreeing that personalized feedback was particularly important, and that the text message reminders to exercise may help them feel a sense of community regarding their physical activity goals. P12 and P16, in separate groups, talked about enjoying when apps were integrated and shared daily activity levels information across themselves. When asked about preferences related to message timing, some participants liked the reminders that their Fitbits or other smartwatches used to remind them to move or take steps while others indicated preferences for congratulatory text messages after a workout rather than a reminder. P10 gave the example of, I think what I would like to see, what motivates me is, “Oh, you’re this close to your goal this week.” Because I get that with the apps. “Do 20 more pushups and you’ll have hit your goal this week,” or it kind of tracks where you’re supposed to be and where you’re actually at, because sometimes it's like I can knock that out. I just want to see that little gold star or whatever that says, “Aha, you hit the goal this week.”
Most participants preferred minimal contact, no more than once a day.
Theme 9: message content and tone
Survivors were asked to view four sample messages and provide feedback. Participants provided feedback on each message individually and then considered the four messages as a whole, with many participants finding the messages linking the two types of data particularly pertinent. For example, P16 stated, “Well, I think it's nice to have the daily information (messages 1-3) and the weekly information (message 4), because the cumulative--you may not no-tice a trend. But if the data is there to support something, then it's kind of nice to have someone tell you that.” The data from the first three messages, without having the linkage made clear between the physical activity and glucose data, was not enough on its own. P12 said, “it's sort of like, it's lower than the past three days, but so what? What does that mean?” P11 agreed, stating that the “big picture” needed to be clear, with P9 echoing “I don't even know what's good.” P14 described more in depth though how referencing glucose levels alone, which could indicate risk of developing type 2 diabetes, was frightening, particularly as these types of comorbidities “can also negatively impact cancer-related outcomes. So, whenever I see that ‘C’ word in anything, I take heed, because I’ve been there, done that.” Essentially, the physical activity data on its own (reporting steps taken, along with recommendations) made sense to participants. However, the addition of glucose data along with physical activity data needed to be explicitly clear, as the participants did not understand the connection between the two or what constituted “good” levels. Yet, even with this caveat, participants were open to receiving these types of messages linking the physical activity and glucose data together, as long as context was provided, and even found these messages suitable for potential physical activity motivators.
Some participants talked about the need for occasional messages that take more of a “check in” tone (e.g., one participant gave an example of “Hey, did you exercise today?” as a preferred message) rather than a reminder to exercise (e.g., as P8 said, “as long as it's not bossy about it”). The primary reports were that negative tones were unwanted, and that numerical data alone was uninspiring. In fact, most participants described that they would greatly prefer the numerical data with a qualifier – such as a “congratulations” or “great/good job” as the data alone was hard to interpret qualitatively. P13 described it as, “Because it is very factual, indeed, but you don't know, is that okay? Is that great, or it's just good enough? So I think maybe a qualification of how good that was.”
Discussion
To our knowledge, this is the first study to explore in-depth how sedentary cancer survivors who were overweight and obese perceive the acceptability of biosensors (i.e., a CGM) in addition to more commonly used activity trackers (such as Fitbit), and their preferences of personalized feedback messages based on these wearable sensors. Previous research has examined specific physical activity barriers for cancer survivors 37 ; however, there is a paucity of literature, particularly from the qualitative perspective. 38 Similar to previous studies,,36,39 data adequacy was reached after interviewing 15 participants across five groups. Three major findings emerged from our study: (1) personalized feedback messages are perceived as a useful strategy to address motivation and accountability, the two major barriers to physical activity identified by cancer survivors; (2) perceived acceptability of wearable biosensor (such as CGM) is in general high among cancer survivors given their medical history; and (3) glucose data is a relevant and important health indicator for cancer survivors and they appreciated integrated messages (between a Fitbit and CGM) in demonstrating how one's behaviors immediately affect their body.
First, the present study echoes previous research findings that cancer survivors face many barriers related to physical activity.12,40 Importantly, although participants identified symptoms as one of the barriers, participants discussed that in the context of their experience (i.e., during cancer treatment), and how that experience negatively impacted their current motivation to exercise. Throughout the discussion, participants have identified lack of motivation and lack of accountability as the biggest barriers to adopting and maintaining their desired physical activity levels. Previous mHealth research among cancer survivors, found that reminders and personalized feedback based on wearable sensors are effective methods for increasing motivation and accountability. 41 Our findings may support this research, as the participants were generally open to reminders and personalized feedback based on wearable sensor data, indicating that this feedback may increase their motivation for engaging in physical activity.
Second, this study extends previous research on wearable activity monitors (such as Fitbits) to examine cancer survivors’ perceptions on usefulness of wearable biosensors (such as CGM) as a tool for physical activity interventions. Recent research demonstrates the increasingly cost-effectiveness of using CGM for patients with diabetes, with other research expanding the health context beyond diabetes patients.24,42,43 As using data from CGM represents an exciting new way to provide personalized feedback to assist with behavioral changes, it would be important to understand how cancer survivors might perceive the usage of such wearable devices, especially given the generally older age of cancer survivors when compared to healthy populations. For example, one systematic review of literature found that age of the patient is one of the top barriers for patients in adopting telemedicine. 44 This study's participants, aged 50 to 74 and majority without diabetes (69%), were generally positive about using CGMs for future physical activity interventions. Participants varied in their preexisting knowledge levels about CGMs, yet they reported that they felt CGMs were a viable and acceptable option for providing a new type of information (i.e., glucose readings) to motivate physical activity. Participants in general felt comfortable with subcutaneous device insertions partly due to their previous cancer treatment experiences and frequent bloodwork draws. Therefore, the perceived invasiveness of biosensors (such as CGMs in its current form) might not be a huge barrier for adoption among cancer survivors.
Third, this study found that cancer survivors perceive glucose data as a relevant and important health indicator for them. In general, they appreciate information regarding their biological status, again, possibly due to their frequent encounters with blood tests. However, participants were not always able to make connection between their glucose number and their behaviors. We found that, for participants with diabetes, they were somewhat able to make the connection between diet and glucose levels; and for all participants, the link between exercise and glucose levels was less aware. Thus, in order to use CGM as a tool to provide biofeedback to motivate physical activity, educating the links between their physical activity behaviors and glucose pattern, especially for the non-diabetic population is needed. Based on participants’ reactions from our study, they were able to grasp the link quickly after a brief explanation by the moderator. Previous study also demonstrated that adults without diabetes were able to understand the acute impact of physical activity on glucose pattern after a brief education session. 21 Further, participants expressed that the sample biofeedback messages that link their physical activity behavior (i.e., using data from Fitbit) with their glucose pattern (i.e., using data from CGM) were very personalized and motivating. And they appreciated the feedback messages that can help them make the connection between a biological data with their own behaviors compared to if they were have to figure out the connection on their own by just looking at the numbers from the two devices.
Limitations
Most of the cancer survivors interviewed in the groups were female breast cancer survivors, so the experiences of survivors of other cancer types as well as males are not well-represented. Specifically, recruitment criteria and demographic questions did not specify differences based on type of cancer or cancer treatments. While the study was open to men and colorectal cancer survivors, the demographic was underrepresented. Given that one male and one colorectal cancer survivor did participate in a group interview and contribute to the discussion, the potential influence of those responses must be acknowledged; therefore, they are included as study participants. The relatively small sample size also limits interpretation of the results; a larger sample size could produce a wider breadth of narratives. Nevertheless, our sample size is like previous studies that examine barriers and facilitators of physical activity among overweight and obese people; future research should recruit larger participant pools. Size of the interview groups may have also affected the participants’ interactions, with the largest group in our study showing similar opinions. Specifically, while research on group size shows 4-8 individuals are ideal (3-5 for small groups), there are many other influencing factors (including personality type, length of time with condition, personal experiences, etc.) that might be at play. Virtual group participants seemed to have more equal opportunity to speak than face-to-face group participants, as the virtual group participants took turns muting their mics and speaking. All study participants were patients from a comprehensive cancer center in a metropolitan area, and thus might have better access to care and survivorship-related resources compared to survivors from other areas. Their opinions about wearable sensors and mobile technologies could be different from survivors from the rural areas or the underserved populations. Further, in order to help study participants to better understand the link between glucose and physical activity, we briefly presented some standard physical activity educational materials that discuss the benefits of physical activity during the focus group. This informational presentation may have affected the participants’ responses by altering their understanding and positive appraisal of the immediate impacts of PA and the glucose's response to physical activity compared to if that was not the procedure. Future studies can investigate and compare the acceptability of wearable sensors and mobile technologies in survivors from rural underserved populations, particularly given the rising issue of the “digital divide,” and strategize how to address this issue. The questions also prompted many participants to compare their pre-diagnosis physical activity experiences with their current experiences, some of whom indicated there was some time between those experiences, so recall bias is possible.
Implications and conclusions
This study contributes valuable information to the scientific literature about the current state of cancer survivors’ perceptions related to wearable sensors and physical activity, as well as the acceptable uses of CGMs in physical activity interventions to increase motivation. Our study indicated that, among cancer survivors, the acceptability of both wearable activity tracker and biosensors is high. The wearable technology industry (i.e., Apple and Samsung) recently has pushed for noninvasive glucose monitoring through watch functions, which will likely increase accessibility to CGM-like devices if commercialized. 45 The cost of CGMs is rapidly decreasing as well, 46 making them more accessible and attractive to companies that target “the worried well”. 47 However, the increased availability of such devices should be taken with caution as having the data available does not directly correspond with understanding of applicability. Essentially, if people are provided a device but had limited knowledge about how to interpret the data itself, the devices and subsequent data will likely have minimal lasting impacts on one's health. Overall, results from this small group interview study provides preliminary evidence regarding the acceptability and relevance of using biosensors such as CGMs to provide personalized feedback in cancer survivors to motivate physical activity. Further, integrating Fitbit (an already highly accepted wearable activity tracker) and CGM data is a particularly promising avenue to help cancer survivors make the connection between their daily behaviors and biological status, a topic that they are highly interested in. Thus, future physical activity intervention that targets cancer survivors could consider utilizing personalized biofeedback message as a behavioral change strategy to motivate the adoption and maintenance of an active lifestyle.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Duncan Family Institute Integrative Health Initiative Jason's Deli Funding Program at the University of Texas MD Anderson. The authors would also like to acknowledge support from MD Anderson's Center for Energy Balance in Cancer Prevention and Survivorship.
Ethical approval
The study protocol (PA19-0317) was approved by The University of Texas MD Anderson Cancer Center.
Guarantor
YL
Contributorship
GEB conducted formal data analysis, interpreted the data, and drafted the manuscript. MAR conducted formal data analysis and interpreted the data. PHC and MLB recruited study participants and conducted the interviews. MSB and TBB contributed to the discussion of clinical implications and practice. SMS and KBE contributed to the design of the study and the discussion of future intervention applications. YL conceptualized the project, designed the study, conducted the interviews, and was a major contributor in writing the manuscript. All authors read and approved the final manuscript.
