Abstract
A CDSS generates a high number of inappropriate alerts that interrupt the clinical workflow. As a result, clinicians silence, disable, or ignore alerts, thereby undermining patient safety. Therefore, the effectiveness and appropriateness of CDSS alerts need to be evaluated. A systematic review was carried out to identify the factors that affect CDSS alert appropriateness in supporting clinical workflow. Seven electronic databases (PubMed, Scopus, ACM, Science Direct, IEEE, Ovid Medline, and Ebscohost) were searched for English language articles published between 1997 and 2018. Seventy six papers met the inclusion criteria, of which 26, 24, 15, and 11 papers are retrospective cohort, qualitative, quantitative, and mixed-method studies, respectively. The review highlights various factors influencing the appropriateness and efficiencies of CDSS alerts. These factors are categorized into technology, human, organization, and process aspects using a combination of approaches, including socio-technical framework, five rights of CDSS, and Lean. Most CDSS alerts were not properly designed based on human factor methods and principles, explaining high alert overrides in clinical practices. The identified factors and recommendations from the review may offer valuable insights into how CDSS alerts can be designed appropriately to support clinical workflow.
Introduction
The Institute of Medicine advocates that health information technologies (HITs), including electronic clinical decision support systems (CDSSs), can improve and enhance the quality of care. 1 A CDSS, with its alert function, can be integrated into other health information systems (HISs), such as electronic medical records (EMRs) and computerized provider order entry (CPOE), to enhance the decision making of clinicians by providing relevant information at the point of care. 2 A CDSS uses different notification methods, such as alerts, pop-ups, reminders, and screen flashes, to inform clinicians about dangerous drug–drug interactions (DDIs), drug allergies, drug duplication, and drug doses that may lead to adverse events and medication errors.2–4 Medication alerts remain an important component of CDSSs because they provide evidence-based recommendations to users. 5 However, clinicians encounter too many medication alerts when prescribing, reviewing, dispensing, and administering medications, which disrupt their workflow.6,7
Factors contributing to inappropriate alerts include lack of data granularity in CDSS knowledge base, poorly categorized DDI alerts, low alert specificity/sensitivity, and perceived risk of legal liability.4,5,8,9 Most alerts triggered during medication order entry are overridden or disregarded because they display inappropriate information and unclear warnings.9,10 Clinicians should respond to alerts appropriately to realize the full potential of CDSS in improving the quality of care and patient safety.4,11 Appropriate CDSS alerts are alerts that are presented to users in the right context and at the right time, less intrusively displayed on the screen, and display clear, correct, and useful patient information.11,12 Therefore, the appropriateness of CDSS alerts should be evaluated to ensure proper design, implementation, and configuration to support clinical processes.
Theoretical background
A CDSS generates many inappropriate alerts, which explains why most clinicians override alerts. 10 Increasing rates of alert override denote that alerts impair the judgments of clinicians and do not assist clinicians in making timely, informed, and high-quality decisions.9,13 The five rights of CDSS state that CDSS interventions such as alerts should be designed to provide the right information to the right person in the right format through the right channel at the right time during task execution. 14 Achieving the five rights for CDSSs is challenging because of several socio-technical aspects, including poor CDSS design, unreliable hardware and software, interface usability, and data integrity, accessibility, and security.15–17
According to Wasylewicz et al., 18 understanding how to alert clinicians at the point of care is a complex socio-technical issue that requires consideration of technological, human, process, and organizational aspects. We proposed socio-technical dimensions and measures for CDSS alert appropriateness based on a critical review of selected models, namely, human–organization—technology–fit, 19 human-automation and interaction, 20 business process management (BPM) life cycle, 21 systems engineering initiative for patient safety, 22 and previous works on CDSS alert and workflow problems (Table 1). The models were selected based on their inclusion of socio-technical interactions among human agents, tools, technology, and their environment.23,24 We argue that considering technology, human, organization, and process and the fit among them can increase alert effectiveness and appropriateness, reducing alert overrides (Figure 1) and subsequent compliance to the five CDSS rights.
Example of evaluation measures for CDSS alert appropriateness.


Considering and aligning socio-technical aspect for CDSS alert.
Workflow disruption largely causes clinicians to ignore, misinterpret, and mishandle drug alerts.16,25,26 Alerts that support clinical workflow are likely to be used.4,5,8,9,26 The assessment of workflow and its fit with CDSS alert should be carried throughout CDSS development and post implementation. 26 Workflow assessment can be carried out using established process-oriented methods, such as BPM and Lean, to identify barriers to the fit of CDSS alert and workflow. BPM is the art and science of overseeing how work is performed in an organization to ensure consistent outcomes and to take advantage of improvement opportunities. 21 Typical examples of improvement include reducing cost, execution time, error rates, and waiting times. BPM encompasses various tools and methods that are used to analyze, model, simulate, monitor, and execute end-to-end business processes. 27 BPM can be combined with other process improvement methods, such as Lean, to improve or enhance business processes. 21
Lean methods are designed to improve processes by eliminating non-value added (NVA) activities known as waste.12,28,29 Waste in HIS can be defined as NVA activities in the work process that hinders useful information to its users. 30 Lean categorizes eight types of waste as defects, overproduction, transportation, waiting, inventory, motion, over processing, and underutilized human potential.12,28,29 The waste model has been applied and extended to identify different types of waste in healthcare, including overtreatment; failures in the coordination and execution of care processes; administrative complexity; and pricing failure, fraud, and abuse.31,32 This review used the waste model to analyze workflow inefficiencies related to alerts. The wide application of Lean and BPM in healthcare is attributed to its ability to analyze and redesign clinical process and address the challenges of complying to the five rights of CDSS. This review aims to identify factors affecting the appropriateness of CDSS alerts in supporting clinical workflow based on the proposed evaluation measures for CDSS alerts to illustrate the factors in compliance to the five rights, inefficiencies, and complexity, which are also featured in the measures.
Methodology
We followed the guidelines of Preferred Reporting Items for Systematic Reviews and meta-analyses in reporting our systematic review (Figure 2). We registered the review on the Prospero database; however, the review slightly deviated from the Prospero protocol by applying the five-right, socio-technical, and Lean perspective. The search was conducted on 11 October 2018 to 28 December 2018 by using the following keywords: computerized provider order entry, CPOE, alert, reminder, pharmacy information systems, PIS, clinical decision support system, CDSS, alert fatigue, alert override, electronic health record, EHR, electronic medical record, EMR, health information system, HIS, prompt, notification, health information technology, HIT, clinical workflow, clinical process, patient safety, medical errors, adverse events, business process management, BPM, process mapping, and process re-engineering (Table 2). The electronic search was performed on seven bibliographic databases, namely, PubMed, Scopus, ACM, Science Direct, IEEE, Ovid Medline, and Ebscohost, for relevant literature from 1997 to 2018.
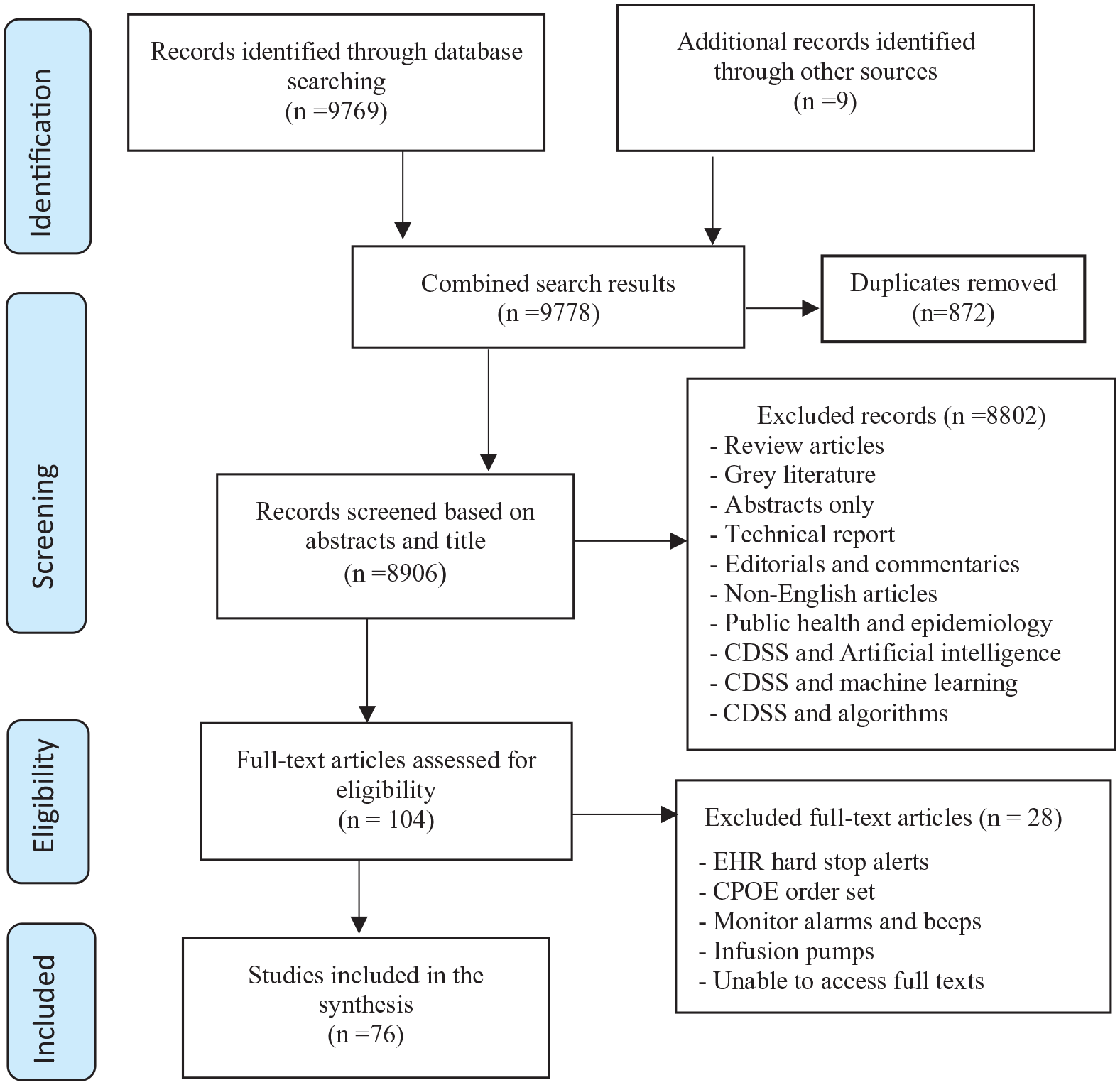
Study selection.
Search terms used for bibliographic database.
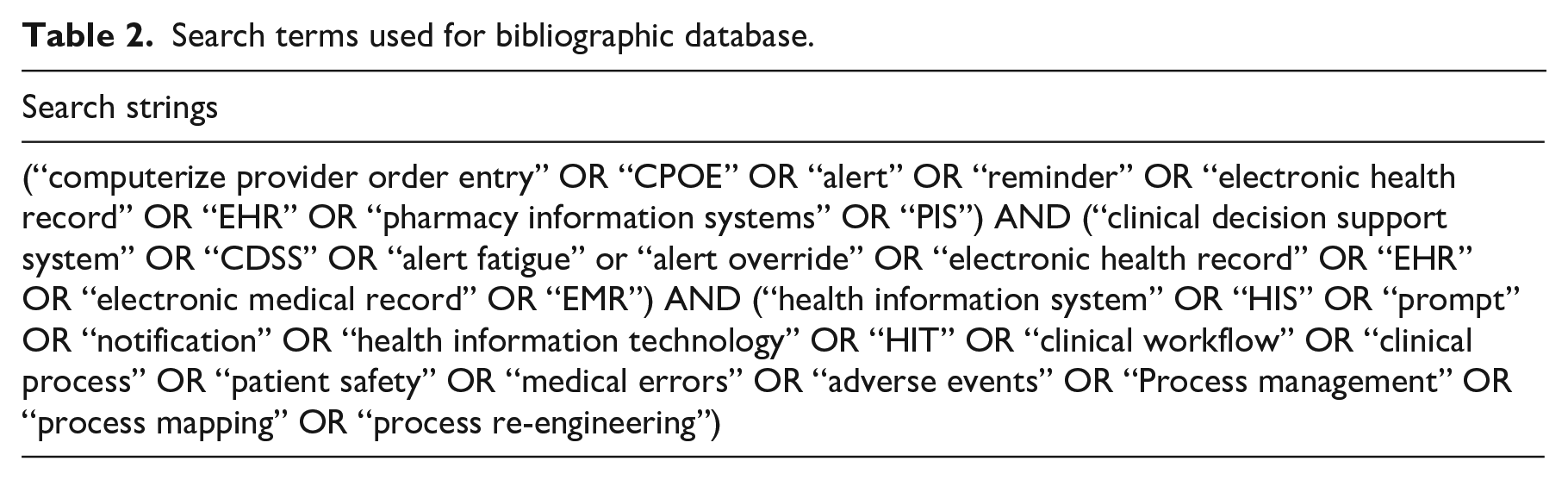
Article screening and quality assessment were carried out by one reviewer (OO). We seek to include comprehensive studies, regardless of their methods, that are related to CDSS appropriateness to identify and understand their influencing factors. Articles were included if the title and abstracts indicated that they are primary research studies that employ qualitative, quantitative, retrospective cohort, or mixed methods focusing on basic and advance medication alerts 33 embedded in CDSSs. Furthermore, full texts of relevant articles reporting on features and functions pertinent to CDSS alert appropriateness were identified by including articles reporting on one or more rights in the five rights principles, 34 evaluation measures for CDSS alert appropriateness (Table 1), and medication alert- and workflow-related problems in healthcare settings. Studies on medical device alarms, 35 such as physiological monitors, ultrasonic humidifiers, infusion pumps, ventilators, and defibrillators, were excluded. Reports, reviews, editorials, commentaries, and non-English articles were also excluded. Citation chaining of included articles was performed to obtain additional relevant publications.
The bibliographic databases returned 9778 publications. The numbers of articles retrieved from each database are as follows: Scopus (
One reviewer (OO) used data extraction forms to extract specific information regarding the study location, objectives, types of CDSS, country, methods, and key findings. Each paper went through the iterative process of interpreting and coding. Deductive reasoning was conducted based on the identified themes of the proposed evaluation measures for CDSS alert appropriateness, whereas inductive reasoning was applied to new, emerging themes. Findings were synthesized narratively and tabulated based on the overall themes. We employed two quality assessment tools, namely, Critical Appraisal Skill Program (CASP) 36 and Mixed Methods Appraisal Tool (MMAT). 37 Both tools can be used for systematic reviews that include qualitative, quantitative, and mixed methods studies,36,37 but they slightly differ in terms of quantitative design types. Systematic reviews of mixed studies, such as ours, can benefit from these hybrid appraisal tools. We used a yes or no checklist to assess the quality of selected papers on the basis of the questions in Table 3 for qualitative and retrospective studies. 37 The assessment shows that majority of the selected papers are of high quality. In addition, MMAT is used for assessing the quality of quantitative (survey) and mixed method studies (Table 3). 37 The quality assessment reveals that over 73 of the papers are of high study quality, and the remaining three papers have acceptable study quality.
Critical appraisal for selected studies.

Results
In summary, 76 papers were obtained (64 main papers and 14 additional papers from citation chaining) (Appendix A), of which 26 papers are retrospective cohort studies, 24 are qualitative, 15 are quantitative, and 11 are mixed-method studies. These studies were conducted in different settings of inpatient and outpatient care (
Factors that affect CDSS alert appropriateness in supporting clinical workflow.

Technology
Fourteen studies reported that an overwhelming number of inappropriate DDI and drug-allergy alerts are received through different communication channels during prescription, resulting in increased override rates (Table 5).6,7,38–46,95,107 In a previous study, 2700 alerts in an ambulatory care were triggered to prevent a single adverse drug event, and 90% of the alerts were overridden. 7 Five studies reported that alert overload increases the mental workload of clinicians and decreases situation awareness in the workplace, causing clinicians to miss important information.2,6,7,45,107 Although alert overload can overwhelm clinicians, limited information can also pose serious harm to patients.43,44,46 Hence, alerts should be configured to provide relevant and sufficient information to its users without overwhelming them with unnecessary alerts.7,42
Key themes, specific issues, and recommendations.

Nine studies conducted in primary care, inpatient, and outpatient settings indicated that stand-alone CDSS alerts do not provide clinicians with sufficient information needed to make prescribing decisions.3,49,59–67 Two surveys reported that the information displayed by drug alerts during medication order entry is difficult to interpret, unclear, and unconcise, thereby slowing down clinicians’ work.3,59,60 Drug alerts are also redundant and display information that does not match the needs of individual or different users (e.g. general practitioner, pharmacist).50,61,62 Another study carried out in outpatient setting showed that the quality of alert information, such as signal word (e.g. caution) and hazards (e.g. seriousness), determines whether or not the clinicians cancel a drug with DDI risk or continue with the prescription.63,65 Clinicians emphasized that drug alerts should be concise, informative (about the patient’s condition, i.e. diagnoses, allergies, interactions, and medical history), and provide options for alternative medications.3,60,62,65 Clinicians also claimed that having useful information at the point of care saves them from going through the tedious tasks of searching the system for additional information (e.g. vitals, dosage, weight, and height).60,61
Nine studies conducted in inpatient and outpatient settings reported that poor specificity and sensitivity of CDSS alerts are the major reasons for alert overload.5,13,45,52,68–73 The CDSS alerts support drug prescription, administration, and are integrated into CPOE or EHR systems. Five studies showed that alerts with high specificity and low sensitivity generate a high number of inappropriate alerts, which result in alert overload.45,68–71 In a pediatric center, nearly two-thirds of drug dose alerts are overridden, raising questions about alert specificity and the accuracy of the displayed information. 68 In this regard, clinicians emphasized that alerts should be highly specific, sensitive, and contain warnings indicating the severity level (e.g. critical, danger, or high risk).52,68,71,72 Three studies suggested that alert specificity/sensitivity can be improved by modifying alerting rules in the knowledge base, default settings, codes for specific patient groups, and tailoring drug safety alerts to the clinical context.5,13,52 In one outpatient setting, alert specificity and sensitivity are increased by re-categorizing the knowledge base of the DDI alerts and suppressing inappropriate alerts that are overridden by clinicians. 5
Eight studies conducted in primary care,47,48,50,55,57 outpatient setting, 74 and community hospitals 75 reported that alerts integrated into CPOE or EHR systems cause clinicians to experience difficulties in distinguishing between serious and less serious DDI, drug duplication, and drug allergy alerts during prescription because of poorly designed interface.47,48,50,55,57,74–76 Some of these studies showed that poorly designed alert attributes, such as font, icon, label, and drop-down menu, lead to wrong order selection and juxtaposition errors.47,48,50 Clinicians have to scroll through medication lists that are counter-intuitively arranged.47,48,50 Medication lists grouped with similar drug names increase the chances of clinicians selecting the wrong drugs.50,76 Clinicians prefer the basic principle of user interface design, such as minimized scrolling, mouse clicks, and steps, to reduce their chances of prescribing wrongly.48,57,74 Five studies conducted in inpatient pharmacies reported that communication problems persist because clinicians face difficulty in understanding and interpreting alert messages that prevent them from using the alerting functions optimally.61,77–80 Clinicians often ask for help, such as calling a pharmacist, to clarify why an alert is triggered and request to a have real-time or face-to-face communication with a pharmacist to interpret and resolve an alert that may endanger patients.77–79
Human
Eleven studies conducted in primary care and ambulatory care settings showed that asynchronous alerts embedded in EHR increase the workload of clinicians. The asynchronous alerts inform clinicians about test results and medication refill requests via email, resulting in alert-related workload.41,43,81 On average, clinicians in a primary care setting receive over 56 alerts per day and spend 49 min responding to asynchronous alerts. 42 These alerts burden clinicians in terms of physical fatigue and cognitive weariness.81,82 Providing clinicians with protected time to respond to alerts reduces alert-related workload and improves patient safety.43,81,82
Other instances of workload involve clinicians documenting clinical data into EHR (e.g. dose route, frequency, and duration) before an alert can be triggered and selecting reasons for bypassing medication alerts during order entry.52,54,57,82–85 Workload of clinicians can be reduced by minimizing data entry tasks, decreasing the overall number of reminders, and eliminating work duplication. 54 CDSS alerts should be configured according to the respective user. For instance, an alert notifies a nurse when a patient shows early signs of sepsis, enabling a clinician to place an order using a sepsis order set. 86 Clinicians recommend options to accept or decline important alerts that may pose a threat to patient or normal alerts that provide known information.54,57
Four studies conducted in primary care and inpatient pharmacies reported inadequate training, knowledge, and skills as a barrier to effective use of CPOE alerts during medication order entry.49,50,54,78 Clinicians could not view, remove inapplicable, or activate reminders because of insufficient training, which lead to difficulty in managing alerts.49,54 A study found that the lack of knowledge of providers on how to use CPOE for reviewing and verifying medication orders increases pharmacist workload. 78 Another study showed that educating clinicians through e-mail or monthly clinic meetings is ineffective because e-mails are often unread, and meetings are missed because of the heavy workload of clinicians. 50 These clinicians prefer hands-on training provided by local physicians and pharmacists. 50
Organization
Three studies in primary care and in outpatient setting showed that deficiencies in organizational rules and security policies impede alert use, contribute to HIT failure, and disrupt workflow.36,59,87 According to Zazove et al., 55 institutional rules prohibit clinicians from modifying the best practice alert (BPA) fonts, colors, and word placement to suit their personal needs. Another study reported that alerts display information that conflicts with the hospital policy and standard medication practices. 64 Furthermore, security policies or measures such as providing justification to override password before prescription completion and enforcing role-based access control for clinicians hinder the effective use of CDSS alert. 87
Process
Using the Lean method, we identified various wastes that affect CDSS appropriateness (Table 6). Clinician’s preferences regarding alert presentation format (i.e. interruptive or non-interruptive) during prescription and medication order entry varied across 11 studies.4,48,53,88–95 Interruptive or active alerts require clinicians to respond to the alerts before proceeding with other tasks, whereas non-interruptive alerts are passive alerts that clinicians choose to review.4,48,91 Scheepers et al. 4 compared four alert presentation methods (i.e. pharmacy intervention, physician alert list, EHR section, and pop-up alerts) to determine the clinician’s preferred method of alerting. The findings showed that active alerts, such as pop-ups, are more effective than passive alerts because they automatically appear during the workflow, reminding clinicians of items they might have overlooked. 4
Waste in Lean IT and CDSS alert.

Conversely, other clinicians indicated that they prefer alerts to be displayed in a non-interruptive manner because they can be accessed at any time.19,46 A survey conducted by Kucher et al. 92 showed that the use of non-interruptive alerts increases clinician’s compliance with care measures by preventing venous thromboembolism (VTE) prophylaxis among hospitalized patients. These studies have shown that careful consideration is needed to determine alerts that should be designed as either interruptive or non-interruptive.4,48,53,92 Phansalkar et al. 93 used a pragmatic approach to identify 33 low-priority DDI alerts that can be safely presented in an interruptive manner during workflow. Alert tiering by risk severity has been suggested as a strategy for presenting urgent and less urgent information to clinicians.88,89,94,95 For example, level 1 alerts (drug contraindication) indicate a very serious interaction that requires clinicians to discontinue the drugs or cancel an order. Level 2 alerts (moderate or severe risk) refer to a less serious DDI but also require clinicians to provide valid reasons before completing a drug prescription. Level 3 alerts (mild or moderate risk) are the least serious interactions that appear in a non-intrusive manner at the bottom of the computer screen. 90
Seven studies reported that drug allergy and dosing alerts triggered after a medication are prescribed or dispensed.53,59,78,98–100 With CPOE use, determining the appropriate time for administering medication becomes difficult for clinicians because of wrongly configured default timing settings. 78 Another study 99 found that an inappropriate medication timing or medication timing error occurs when medication doses are not administered according to the standard administration time. These studies showed that pharmacists must use their own experience and judgment to ascertain the appropriate timing for administering medication.78,99 Whether or not alerts should be triggered at different times is unclear, such as (1) when reviewing medication, (2) entering patient information, (3) when selecting an order, or (4) after an ordering session has been completed.58,91,98 Therefore, the appropriate timing for alerting user needs to be determined to improve their effectiveness and increase their use.51,101
Five studies identified unnecessary delays during clinical care processes in inpatient and outpatient clinics that were caused by hardware and software malfunctions.8,56,96–98 Examples include clinicians waiting for an alert before proceeding to other tasks because of slow processing time, computer crashes, and Internet connectivity problems.96,97 Saleem et al. 57 found that when the computer system crashes, clinicians could do little until the system is resumed because all nursing and provider functions are performed through the computerized provider record system (CPRS). Similarly, McMullen et al. 78 found that the safety features in a CPOE delay medication ordering. The study further revealed that clinicians experience system lockout every 10 min because the CPOE does not provide sufficient time to enter and verify orders. 78 Hence, hardware and software should be properly configured so that alerts can be activated in a timely manner. 8
Outcomes
Researchers are yet to conclude whether or not the use of CDSS alert has improved clinical outcomes. Six studies conducted in ambulatory care, inpatient, and outpatient settings showed that poorly designed CDSS alerts reduce physician productivity and consequently affect healthcare quality and clinical outcomes.3,38,42,45,86,99 For instance, usability problems such as the number of clicks required to prescribe a drug or create referrals obscure the productivity of clinicians. The use of CDSS alerts and reminders in various care settings and clinical specialties to support care processes during personal interaction with patients contributes to productivity loss.3,38,42,45,86,99 Therefore, CDSS alerts must be designed to provide clinicians with patient-specific assessment and recommendation (i.e. appropriate alert) during task execution to improve the quality of care and clinical outcomes.3,44,49
Discussion
This systematic review identifies factors affecting CDSS alert appropriateness in supporting clinical workflow from a socio-technical perspective, five rights principles, and Lean. Clinician’s response to medication alerts decreases as the total number of inappropriate alerts increases.25,94 Managing alert overload burdens healthcare institutions because it requires a careful analysis of institution-specific practice and alert data.103,105 Alert overload can be addressed by using data from reported alert usage, such as alert type (e.g. interaction, allergy, and duplicate drug), trigger (e.g. medication, dose, route, and schedule), result (e.g. alert override and order abandonment), and reason for overrides, to suppress inappropriate alerts for specific users. 105 Previous studies suggested many strategies to counteract alert overload, such as turning off frequently overridden alerts, 104 prioritizing alerts, 42 tiering alert by severity,52,104 and implementing highly specific algorithms. 69 Increasing the sensitivity and specificity of CDSS alerts in different care settings can positively influence system use and ultimately increase alert acceptance.32,52 The specificity of a CDSS is a measure of its ability to distinguish between events that put an individual at risk of harm and non-events that will not: the more false positives, the lower the specificity. 52 Sensitivity is the ability of the system to alert prescribers correctly when patients are at risk of experiencing drug-induced harm, such as from a DDI or drug allergy. 52 Alert overload also occurs when knowledge base rules are wrongly edited or configured. 106 A complex system such as CPOE is usually integrated in one or more knowledge bases of drug dosing rules for reference purposes.53,103 Therefore, knowledge bases for CDSS should be designed to display alerts judiciously by maintaining the right balance between inappropriate and relevant or useful alerts with important interactions that can undermine patient safety.53,105
Alert appropriateness is also attributed to information content and display. Although incomplete or outdated allergy information in EHR leads to the proliferation of inappropriate alerts, incomplete allergy documentation still occurs because clinicians lack understanding on how to document information and what information needs to be documented (e.g. self-reported allergy, confirmed allergy, and nature of the reaction).105,107 This scenario results in fragmented allergy documentations in patient records and subsequent delays in treatment. 107 To facilitate appropriate alert triggers, an updated and accurate allergy information must be documented and captured in standard location within the EHR.33,108 Additional research is needed to ascertain how to best record this information and present clinicians with useful drug alerts. 107 Alerts should be designed or configured to provide sufficient information (e.g. frequency of interaction occurrences and alternative medications) that can enhance the decision making of clinicians.
Few studies focused on how alerts should be designed and display information to support the workflow of clinicians.25,26 Careful design consideration of when to present alerts to prescribers or where they are displayed in the workflow is crucial. The conventional method of presenting information in the user interface is via interruptive and non-interruptive model dialogs (e.g. pop-up alerts).16,25,109 Interruptive alerts are effective in obtaining the attention of clinicians, but these alerts have several usability problems, such as scrolling to view information and missing information at the point of care.16,109 Presenting alerts based on its severity effect (i.e. the order of importance) allows prescribers to quickly identify important alerts and subsequently reduce alert fatigue and override. 25 Alert timing and its execution point are key factors in ensuring clinicians response. 110 Clinicians have different preferences regarding alert timing across different care settings.111,112 Some clinicians in inpatient and outpatient settings prefer receiving an interaction alert early in the decision-making process, particularly when selecting medication. 111 However, clinicians in primary care settings prefer viewing or receiving an alert at the end of the patient visit. 112
Overall, our review findings across care settings and clinical specialties show that alerts are frequently ignored because they are inconsequential, too frequent, ineffective in design, implementation, and poor fit with workflow, thereby violating the five rights of CDSS. Other contributing factors include rigid organization structure and policies, and legal and ethical issues.103,113 For instance, EHR developers are reluctant to remove alerts because of the fear of being held accountable if patients are harmed in the absence of a warning. 113 According to Reidmann et al. 73 , the five rights may be better achieved when alerts are tailored and filtered to consider the characteristics of (1) an organization and its users and (2) a patient or a case. Thus, socio-technical factors, such as workload, communication, usability, and organizational policies, are crucial for the appropriate design and implementation of CDSS alerts. Insufficient knowledge and skills regarding basic CPOE function (e.g. alerts) hinder the effective use of CDSS alerts. Education and training should provide clinicians with an understanding of the benefits of CDSS and how clinicians can respond appropriately to alerts. 105 Such knowledge would help clinicians have a better understanding on how CDSSs function. 105
Care settings and clinical specialties play an important role in managing alert. Presenting the same drug alerts to clinicians in different care settings and clinical specialties may contribute to inappropriate alert overrides. For instance, increase in override rates for various alert types, such as duplicate drugs, DDI, drug allergy, and formulary substitution, ranges from 50% to 90% in inpatients and 33% to 90% in outpatients.114–116 These overrides are appropriate when patients had previously tolerated the medication and the provider would monitor for the recommended DDI and alert duplication.114–116 In primary care settings, reasons for overriding alert include lack of relevance to specific clinical situation, lack of timeliness, and “benefit outweigh risk.”101,107 Therefore, fine tuning drug alerts to match the needs of clinicians in different care settings and clinical specialties may improve the response of clinicians to alerts.2,107
Analysis of CDSS inefficiencies using the waste category revealed the specific limitation of CDSS in supporting clinical workflow. Clinicians spend time performing NVA activities, such as data entry task, switching between screens, and logging into multiple systems. Thus, standardized process improvement methods, such as Lean and BPM, can be used to gain a deep understanding of care processes performed by clinicians, such as the information they use and user–computer interaction. Process analysis can identify process efficiency, problems, and automation potential to improve the process. These detailed insights may inform the design of CDSS alerts. The combination of approaches, namely, socio-technical model, five rights of CDSS, and Lean, can be adopted to provide a holistic view in analyzing, improving, and redesigning CDSS alerts. Our socio-technical approach guides us in interpreting the review findings. However, some measures in the framework could be categorized under multiple dimensions. For example, within the process dimension, measures such as alert timing and alert presentation may also be categorized under the technology dimension. Further application of the proposed evaluation dimensions and measures may lead to additional or specific new measures.
A systematic review ideally involves a group of reviewers to collaboratively implement several steps to avoid bias in the review process. The steps include paper screening, data extraction, and evaluation of study quality. Our review involves researcher OO, a PhD candidate, as a sole reviewer; but this limitation is addressed through a series of consultations between researcher OO and supervisor MMY, who has experienced in conducting systematic review. The discussions include designing the overall study and systematic review method; developing a protocol on the basis of the PRISMA method; adhering to the protocol; defining clear inclusion and exclusion criteria for the referral of different paper screening stages; designing and mapping the data extraction form and extracting data on the basis of the study objectives; and assessing the quality of the selected studies. In addition, the review process is documented in detail to enable readers assess the thoroughness of the study. 117 However, we realized that as an untrained reviewer, the conduct of the review process, including the criteria assessment, is prone to error, 118 subjectivity, bias, and lack of rigor, all of which may affect the generalizability and applicability of the findings.119,120
Conclusion
CDSSs can improve patient safety and quality of care, but poorly designed CDSSs may undermine patient safety. This review shows that human factor methods and principles have not been properly applied to CDSS design. Our review identified factors affecting CDSS alert appropriateness in supporting clinical workflow, including technology (alert content, usability, information overload), human (alert flexibility, workload and training), organization (internal rules and policies), and process (alert presentation, alert timing, delays). These factors can inform practitioners on how a CDSS alert can be designed to fit into clinical workflow. Future research should focus on how human factor engineering principles, socio-technical framework, and process improvement methods, such as BPM and Lean, can be used to inform the design of CDSS alerts.
Footnotes
Appendix
Characteristics of selected studies.

| Study (author) | CDSS tool type | Design | Country | Settings | Number of sites | Study target | Types of alerts |
|---|---|---|---|---|---|---|---|
| Kassakian et al. 13 | Integrated in EHR | Retrospective cohort study | USA | Inpatient | Single | Clinicians | Medication alerts |
| Ancker et al. 83 | Integrated in EHR | Retrospective cohort study | USA | Primary care | Single | Clinicians | Drug and clinical reminders |
| Austrian et al. 87 | EHR | Retrospective cohort study | USA | Primary care | Single | Clinicians | Sepsis alert |
| Seidling et al. 70 | Standalone CDSS | Retrospective cohort study | Germany | Outpatient | Single | Clinicians | DDI alerts |
| Cuéllar Monreal et al. (2017) | AEPS | Mixed method | Spain | Inpatient | Single | Clinicians | DDI and allergy alerts |
| Baseman et al. 42 | e-prescribing | Qualitative study | USA | Public health agencies | Single | Health care professionals (HCP) | E-mail notifications |
| Murphy et al. 44 | Integrated in EHR | Retrospective study | USA | Primary care | Single | Primary care practitioners (PCP) | Asynchronous alerts |
| Wright et al. 8 | Integrated in EHR | Mixed method | USA | Outpatient settings | Single | Chief medical informatics officers, CDS developers | DDI alerts |
| Bauer et al. 76 | Standalone CDSS | Retrospective cohort study | USA | Not stated | Single | Clinicians and patients | CHICA Prompts |
| Carroll et al. 96 | Standalone CDSS | Retrospective cohort study | USA | Not stated | Single | Clinicians | CHICA Prompts |
| McEvoy et al. 95 | Integrated in EHR | Retrospective study | USA | Inpatient and outpatient | Multi | Clinicians | Dosing alerts |
| Stultz et al. 69 | CDSS | Retrospective study | USA | Inpatient | Single | Clinicians | Dosing alert |
| Van der Sijs, et al. 72 | Integrated in CDSS | Retrospective study | Netherland | Inpatient | Single | Clinicians | TTDI alerts |
| Blecker et al. 54 | CDSS | Qualitative | USA | Inpatient | Single | Clinicians | ACE alerts |
| Jani et al. 78 | e-prescribing | Retrospective cohort study | UK | Tertiary care and Paediatric center | Single | Clinicians | Drug allergy |
| Chused et al. 80 | CPOE with CDSS | Retrospective cohort study | USA | Not stated | Single | Clinicians | Drug alerts |
| Bryant et al. 46 | CDSS | Retrospective cohort study | USA | Inpatient | Single | Clinicians | DDI alerts |
| Idemoto et al. 100 | CPOE | Retrospective cohort study | USA | Primary care | Single | Clinicians | Drug alerts |
| Russ et al. 65 | CPOE with CPRS | Observation (qualitative study) | USA | Outpatient | Single | Clinicians | DDI |
| Jaensch et al. 102 | CPOE with CDSS | Qualitative study | Australia | Emergency department | Single | Junior doctors | DDI |
| Sweidan et al. 61 | CDSS | Comparative study | Australia | Primary care | Single | Clinicians | DDI |
| Seidling et al. 64 | CDSS | Retrospective cohort study | USA | Inpatient | Single | Clinicians | DDI and drug duplicate |
| Nasuhara et al. 88 | DAIS | Retrospective cohort study | Japan | Outpatient | Single | Physicians | DDI and drug duplicate |
| Smithburger et al. 89 | CDSS | Expert opinion | USA | Not stated | Single | Clinicians | DDI |
| Fischer et al. 90 | e-prescribing | Retrospective cohort study | USA | Inpatient | Single | Clinicians | Drug formulary |
| Patterno et al. 91 | CPOE | Retrospective cohort study | USA | Inpatient | Multi | Adult inpatients and providers | DDI |
| Trinkley et al. 92 | CDS | Qualitative (focus group) | USA | Primary care | Single | Clinicians | Drug allergy |
| McDaniel et al. 102 | CDSS with CPOE | Retrospective cohort study | USA | Inpatient | Single | Clinicians | DDI |
| Zenziper et al. 71 | CDSS | Retrospective cohort study | Israel | Multiple |
Single | Clinicians | DDI, drug duplicate, and dose |
| Weingart et al. 104 | e- prescribing system | Quantitative (survey) | USA | Ambulatory care | Single | Clinicians | DDI |
| Ash et al. (2004) | PCIS with CDSS | Qualitative (interviews) | Cross country | Inpatient and Outpatient | Single | Pharmacists, Physician, |
Warning alert |
| Koskela et al. 84 | CDSS | Qualitative (focus group) | Finland | Primary care | Single | Physicians and nurses | DDI and drug dosing |
| Birmingham et al. 48 | Integrated in EHR | Qualitative (focus group, interview) | USA | Primary care | Single | Clinicians | Vaccination alerts |
| Coleman et al. 53 | CDSS with CPOE | Expert Participant discussions | Cross country | Workshop | Not stated | Not stated | Medication alerts |
| Krall & Sittig 49 | EMR | Qualitative study (focus group) | USA | Primary care | Single | Clinicians | Drug alerts and reminders |
| Feldstein et al. 51 | Integrated in CPOE | Qualitative study (interviews) | USA | Primary care | Single | Prescribers | Drug safety alerts |
| Saleem et al. 58 | Integrated in EMR | Qualitative study (observation, interviews) | USA | Outpatient | Multi | Clinicians and nurses | Clinical reminders |
| Lugtenberg et al. 50 | CDSS | Qualitative study (focus group) | Netherlands | Primary care | Single | Primary care practitioners (PCP) | Drug alerts |
| Dixon et al. 67 | CDSS | Qualitative study (interviews) | USA | Outpatient | Multi | Clinicians and nurses | HPV reminders |
| Baysari et al. 59 | CDSS with CPOE | Qualitative study (observations, interviews) | Australia | Ward rounds | Single | Clinicians | Drug allergy and therapeutic duplication |
| Russ et al. 62 | CPRS with CPOE | Qualitative study (observations, interviews) | USA | Primary care | Single | Prescribers | Drug alerts |
| Zazove et al. 56 | EHR | Qualitative study (interviews) | USA | Primary care | Multi | Clinicians | Best practice alerts (BPA) |
| Trivedi et al. 98 | CDSS | Observations | USA | Outpatient | Multi | Clinicians and nurses | Reminders for guidelines |
| Kucher et al. 93 | Electronic prescribing system | Retrospective cohort study | Switzerland | Outpatient | Single | Clinicians | VTE alerts |
| Stone 66 | Integrated in EHR | Case study | USA | Inpatient and outpatient | Multi | Clinicians | Drug safety alerts |
| Embi and Leonard 52 | EHR | Randomized cohort study | USA | Not stated | Single | Clinicians | Clinical trial alerts |
| Phansalkar et al. 94 | EHR | Retrospective cohort study | USA | Outpatient | Single | Clinical informaticists and clinicians | DDI alerts |
| Cornu et al. 44 | CDSS | Quantitative (survey) | Belgium | Inpatient | Single | Clinicians | DDI and drug allergy |
| St Sauver et al. 63 | CDSS | Quantitative (survey) | USA | Primary care | Single | Clinicians | PGx-CDS alerts |
| Long et al. 3 | CDSS | Quantitative (survey) | USA | General practice | Single | Nurses | Sepsis alert |
| Glassman et al. 40 | CPRS | Quantitative (survey) | USA | Inpatient and Outpatient | Single | Clinicians | DDI alert |
| Magnus et al. 41 | EMIS | Quantitative (survey) | United Kingdom | Primary care | Multi | General practitioners (GP) | Drug interaction alerts |
| Jung et al. 43 | CPOE with CDSS | Quantitative (survey) | Cross country | Cross hospitals | Multi | Physicians | DDI and dosing alerts |
| Campmans et al. 77 | PIS | Quantitative (survey) | Netherlands | Community pharmacies | Single | Pharmacists and pharmacy technicians | Drug name and drug confusion alert |
| Scheepers et al. 4 | CDSS | Retrospective cohort study | Netherlands | Inpatient | Clinicians | Single | Pop-up, pharmacy interventions, physician alert list |
| Patterson et al. 55 | CPRS | Mixed method | USA | Outpatient | Clinicians | Single | HIV clinical reminders |
| Lapane et al. 6 | e-prescribing | Mixed method | USA | Primary care | Clinicians | Multi | DDI |
| Gregory et al. 82 | EHR with CPRS | Mixed methods | USA | Primary care | Clinicians | Single | Asynchronous alerts |
| McMullen et al. 79 | CPOE | Mixed method | USA | Inpatient pharmacies | Pharmacist | Multi | Drug allergy |
| Hayward et al. 99 | CDSS | Mixed method | UK | Primary care | Single | General practitioners and patients | DDI |
| Stultz and Nahata 9 | CDSS | Retrospective analysis | USA | Inpatient | Single | Patients | Dosing alerts |
| Weingart et al. 103 | e-prescribing | Retrospective cohort study | USA | Ambulatory care | Single | Clinicians | Medication safety alerts |
| Saiyed et al. 39 | CPOE | Retrospective cohort study | USA | outpatient | Single | Clinicians | Drug dose alerts |
| Hsieh et al. 47 | Integrated in CPOE | Retrospective cohort study | USA | Inpatient | Single | Clinicians | Drug allergy alerts |
| Varonen et al. 85 | CDSS | Qualitative study (focus group) | Finland | Outpatient | Multi | Clinicians | DDI |
| Schnipper et al. 86 | Integrated in EHR | Mixed method (survey and interviews) | USA | Ambulatory care | Single | Clinicians | Smart forms and reminders |
| Noblin et al. 81 | EHR | Qualitative study (interviews) | USA | Not stated | Single | Clinicians | Medication safety alerts |
| Ash et al. 68 | CDSS | Qualitative study (interviews) | USA | Inpatient and outpatient | Single | Clinicians | DDI and drug allergy |
| Saxena et al. 73 | Integrated in CPOE | Retrospective cohort study | USA | Outpatient | Single | Clinicians | Drug duplication and contradictions |
| Steele et al. 60 | Integrated in CPOE | Retrospective cohort study | USA | Primary care | Single | Clinicians | Drug laboratory interactions |
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We gratefully acknowledge the funding received from the Universiti Kebangsaan Malaysia (DIP 2016-033) and Ministry of Higher Learning Malaysia (FRGS/1/2018/ICT04/UKM/02/5).
